Abstract
Aim
Our study's objectives were to study the clinical and laboratory characteristics that may serve as biomarkers for predicting disease severity, IL-10 levels, and frequencies of different T cell subsets in comorbid COVID-19 patients.
Methods
Sixty-two hospitalized COVID-19 patients with comorbidities were assessed clinically and radiologically. Blood samples were collected to assess the T lymphocyte subsets by flow cytometry and IL-10 levels by ELISA.
Results
The most common comorbidities observed in COVID-19 patients were diabetes mellitus (DM), hypertension, and malignancies. Common symptoms and signs included fever, cough, dyspnea, fatigue, myalgia, and sore throat. CRP, ferritin, D dimer, LDH, urea, creatinine, and direct bilirubin were significantly increased in patients than controls. Lymphocyte count and CD4+ and CD8+ T-cells were significantly decreased in comorbid COVID-19 patients, and CD25 and CD45RA expression were increased. CD4+ and CD8+ regulatory T cells (Tregs) and IL-10 levels were significantly decreased in patients.
Conclusions
Many parameters were found to be predictive of severity in the comorbid patients in our study. Significant reductions in the levels and activation of CD4+ and CD8+ T-cells were found. In addition, CD4+ and CD8+ Tregs were significant decreased in patients, probably pointing to a prominent role of CD8+ Tregs in dampening CD4+ T-cell activation.
Introduction
Coronavirus disease 2019 (COVID-19) is caused by a novel coronavirus called severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The outbreak of this disease was declared a global pandemic by the World Health Organization in March 2020. 1 The immunopathology of viral pneumonia reflects a complex interplay between the direct impact of virus replication on the lung tissue and both innate and adaptive immune response mechanisms. However, in most cases, the degree and type of lung injury are chiefly caused by the immune response to the viral infection rather than the direct impact of the virus itself. 2
T-cells play a crucial role in the immune response to SARS-CoV-2 infection. However, it remains unclear whether T-cell responses are suboptimal and dysfunctional or excessive; it is unknown whether these responses are helpful or harmful in COVID-19, as there is evidence for both of these possibilities. 3 Four types of T-cells have been shown to be important in responses to SARS-CoV-2 infection: CD8+ cytotoxic T-cells, CD4+ T helper (Th) cells, Th17 cells, and regulatory T-cells (Tregs). The latter inhibit inflammatory responses via their anti-inflammatory cytokines, namely TGF-β and IL-10, thereby minimizing collateral damage and allowing for viral clearance and tissue repair. 4
Understanding the complex functions of IL-10 remains an exceptional challenge for immunologists. Although IL-10 acts as an immune brake on inflammation, it can also have immunostimulatory effects. Thus, more studies are needed to elucidate whether stimulating activation or inhibition of IL-10 could be helpful in the treatment of severe COVID-19. 5
COVID-19 patients with comorbidities are at increased risk of infection. In addition, they tend to have more outcomes associated with deterioration, such as cytokine storm, resulting in increased rates of acute respiratory distress syndrome, multiorgan failure, and death compared with those without comorbidities. 6 The effects of these comorbidities on T-cell responses during COVID-19 are poorly understood, 3 creating a barrier for effective treatment of comorbid COVID-19 patients. Thus, clinical and laboratory biomarkers to predict the severity of COVID-19 among this vulnerable group are urgently needed for respiratory support readiness during this pandemic. The objectives of our research were to study the clinical and laboratory characteristics that may serve as biomarkers for predicting disease severity, as well as IL-10 levels and T-cell subsets, in COVID-19 patients with comorbidities to better understand the effects of the disease on the immune system.
Materials and Methods
This study was approved by Assiut University ethics committee (IRB no. 17300436). Informed consents were obtained from all participants in the study.
A total of 62 hospitalized patients with COVID-19 (confirmed by reverse-transcription polymerase chain reaction (RT-PCR) on nasopharyngeal swab specimens) were included in this case-control study conducted in January 2021. Patients were evaluated for the presence of the following comorbid diseases: diabetes mellitus (DM), hypertension, malignancies, ischemic heart disease, hepatitis C virus (HCV) infection, respiratory diseases, chronic renal failure, central nervous system (CNS) disease, and hypothyroidism. Patients were recruited from Assiut University Hospitals, Egypt. Twenty healthy individuals were also included as controls.
Demographic data and medical history were collected from the participants. Patients were evaluated for the presence of fever, cough, dyspnea, anosmia, sore throat, diarrhea, headache, myalgia, fatigue, and anorexia. Baseline laboratory tests included COVID-19 biomarkers [total leukocyte count (TLC); neutrophil and lymphocyte counts; and serum levels of C-reactive protein (CRP), D-dimer, ferritin, and lactate dehydrogenase (LDH)], fasting blood glucose (FBG), liver functions, and renal functions. Arterial blood gases were also measured. COVID-19 pneumonia was diagnosed based on chest computed tomography (CT). Patients were categorized into severe and non-severe cases based on the WHO severity classification. 7
Blood samples (2 ml) were collected in EDTA vacutainer tubes from all participants. Peripheral blood mononuclear cells (PBMCs) were separated by Ficoll density gradient centrifugation (Biowest, Riverside, MO) to assess the T-lymphocyte subsets by flow cytometry (South Egypt Cancer Institute, Assiut University, Egypt). In addition, 3 ml of blood was left to clot to assess the IL-10 level by enzyme-linked immunosorbent assay (ELISA).
T-cell subsets were enumerated using fluoroisothiocyanate (FITC)-conjugated anti-Foxp3 (eBioscience, USA) [FL-1], peridinin-chlorophyll-protein (PerCP)-conjugated anti-CD25 (IQ Products, The Netherlands) [FL-3], phycoerythrin-cyanine 7 (PE-CY7)-conjugated anti-CD8 [FL-4], APC-conjugated anti-CD45RA (Becton Dickinson Biosciences, USA) [FL-5], and allophycocyanin (APC)-H7-conjugated anti-CD4 [FL-6]. Approximately 1 × 106 cells in 100 µl phosphate-buffered saline (PBS) were placed in 12 × 75 FACS tubes for staining and incubated with 10 µl of fluorochrome-labeled anti-CD4, anti-CD8, anti-CD25, and anti-CD45RA monoclonal antibodies for 20 min in dark, at 4◦C. Cells were then washed with 2 ml PBS by centrifugation at 2500 r.p.m for 2 min. This was followed by the addition of 100 μl of fixation solution and incubation for 10 min. Next, cells were washed again with PBS and a 50 μl of the permeabilizing solution (IntraSureTM kit, BD CA, USA) together with 10 µl of anti-Foxp3 were added, and the cells were incubated for 30 min in dark, at room temperature. After one wash, the cells were resuspended in PBS. Anti-human IgG was used as an isotype-matched negative control for each sample. Flow cytometric analysis was performed by a FACSCanto flow cytometer using FACS DIVA 7.0 software (Becton Dickinson Biosciences, USA). The analysis strategy is shown in Figure 1.

Flow cytometric detection of T cell subsets
Serum IL-10 levels were quantified using a commercial ELISA kit (OriGene Technologies, Inc., USA) based on a standard sandwich ELISA technique per the manufacturer's instructions.
Statistical Analysis
Statistical analysis was done by IBM Statistical Package for the Social Sciences (IBM SPSS statistics, USA), version 20. Numerical data were presented as mean ± standard error, and categorical data were presented as numbers and percentages. Levene's test was used to evaluate the homogeneity of variances. Significance in differences between two groups was tested by the Mann-Whitney U test and Independent t-test. Spearman correlation coefficient was applied to evaluate the relations among different variables. The Receiver Operating Characteristics (ROC) curve in the MedCalc 10.2.0.0 Statistical Program (MedCalc Software, Belgium) was used to compare the performance of tested variables in predicting the severity level. A p-value <0.05 was considered to be significant.
Results
The study included 82 participants; 62 COVID-19 patients with a male to female ratio of 34:28, and a mean age of 58 ± 2 years, in addition to 20 age- and sex-matched healthy volunteers as controls. Among the COVID-19 patients, 22 were severe cases (35.5%), and none were critical cases. The main clinical characteristics and presenting comorbidities of COVID-19 patients are shown in Table 1.
Clinical And Radiological Characteristics And Presenting Comorbidities Of COVID-19 Patients.

CT computed tomography, DM diabetes mellitus, HCV hepatitis C virus, COPD chronic obstructive pulmonary disease, CNS central nervous system
Data were presented as numbers (percentage from the total number of patients).
The most common comorbidities observed in the enrolled COVID-19 patients were type 2 DM (46.8%), hypertension (45.2%), and malignancies (29%). Eight patients (12.9%) each had ischemic heart disease and HCV infection. Moreover, five had a respiratory disease(s) (8%), four had a chronic renal disease (6.5%), three had a CNS disorder (4.8%), and one had hypothyroidism (1.6%). Most patients had one comorbidity (59.7%), while the remaining patients had two or more comorbidities (40.3%).
Table 2 displays the main laboratory findings and blood gas measurements in COVID-19 patients. Numerous parameters showed statistically significant differences between COVID-19 patients and controls, as well as between the severe and non-severe patient groups. D-dimer, LDH, direct bilirubin and total bilirubin, prothrombin time (PT), and respiratory rate were significantly higher in severe cases. In contrast, lymphocyte counts, FBG, and PaCO2 were significantly lower in patients with severe COVID-19 infections.
Sociodemographic Data, Baseline Laboratory Characteristics, And Blood Gases Of COVID-19 Infected Patients.
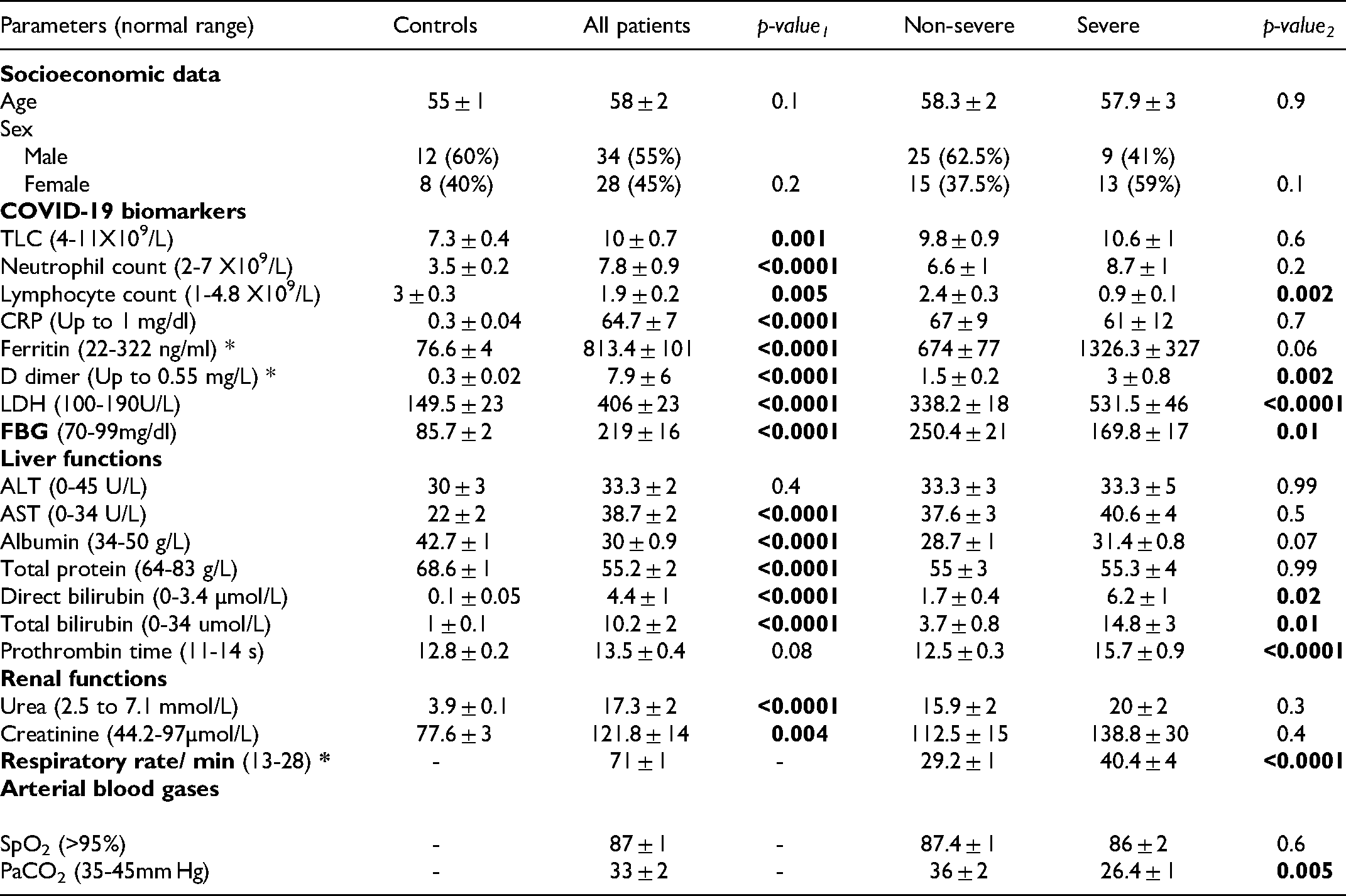
Figure (2) and Supplementary table (S1) show that the COVID-19 patient group had significantly lower percentages of CD4+ T-lymphocytes, CD8+ T-lymphocytes, CD4+CD25highFoxP3+ Tregs, and CD8+CD25high FoxP3+ Tregs than the controls. On the other hand, higher levels of CD25 expression were observed on the CD4+ and CD8+ cells from patients, resulting in significantly higher percentages of CD4
Percentages of the different lymphocyte subsets in COVID-19 patients and the controls. The percentages of CD4+ and CD8+ lymphocytes were assessed in the lymphocyte population, and the percentages of CD25+, CD25high, CD45RA+ and regulatory T cells were calculated among the CD4+ and CD8+ lymphocytes.
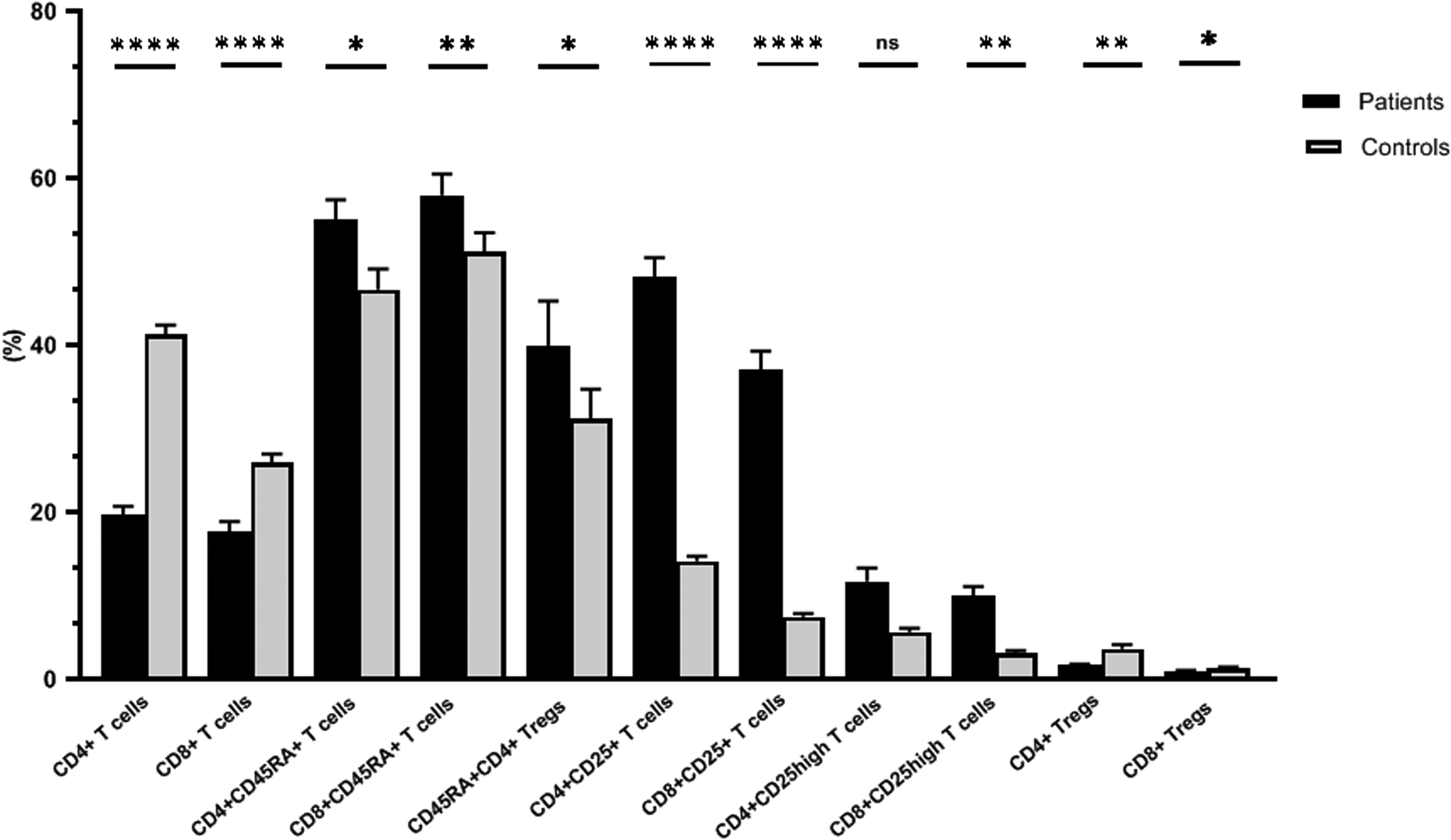
As shown in Figure (3) and Supplementary table (S2), while the level of CD4+ cells was significantly lower in the group of patients with severe COVID-19, the level of CD4+CD25high T-lymphocytes was significantly higher in this group

Relations of the different lymphocyte subsets with COVID-19 severity. The percentages of CD4+ and CD8+ lymphocytes were assessed in the lymphocyte population, and the percentages of CD25+, CD25high and regulatory T cells were calculated among the CD4+ and CD8+ lymphocytes.
In COVID-19 patients, CD4+CD25+ and CD4+CD25high T-lymphocytes were positively correlated with age, D-dimer, total and direct bilirubin. Additionally, CD4+CD25high T-lymphocytes were also directly correlated with the number of affected lung lobes and PT.
Likewise, in the comorbid COVID-19 patients, both the CD8+CD25+ and CD8+CD25high T-lymphocytes were negatively correlated with LDH, PT, respiratory rate, SpO2 percentage, and the presence of patchy opacities. In contrast, these T-cell subsets were positively correlated with the PaCO2.
CD8+CD25high T-lymphocytes were also negatively correlated with the levels of ferritin, total bilirubin, and direct bilirubin. The CD8+CD25+ T-lymphocytes were negatively correlated with the number of affected lung lobes and serum urea. Moreover, CD8+ Tregs were inversely correlated with the levels of D-dimer, total bilirubin, and direct bilirubin.
Receiver operating characteristic (ROC) curve analysis revealed that, among the different biomarkers and immune cell subsets, the lymphocyte count, CD4+ T-lymphocytes, CD4+CD25high T-lymphocytes, CD8+CD25+ T-lymphocytes, CD8+CD25high T-lymphocytes, LDH, direct bilirubin, and PT demonstrated good accuracy in predicting severity level in comorbid COVID-19 patients (Table 3 and Figure 4). The largest area under the curve (AUC) was observed for CD4+ T-lymphocytes, as this demonstrated the highest specificity (100%) and accuracy (88%), although the sensitivity was moderate (68%).

Receiver operating characteristics (ROC) curve comparing the performance of (A) LDH, direct bilirubin, prothrombin time (PT), and (B) some immune cells in the prediction of the severity level in COVID-19 patients.
Performance Of Some Biomarkers And Immune Cells In The Prediction Of The Severity Level In COVID-19 Patients.

p-value is significant if <0.05.
No significant changes in T-cell subsets were observed in COVID-19 patients with diabetes and/or hypertension compared to patients without diabetes or hypertension (data not shown). Conversely, several statistically significant differences were detected when comparing COVID patients with cancer and those with other comorbidities, Figure (5) and Supplementary table (S3). Despite the lack of statistical significance of the changes observed in the CD4
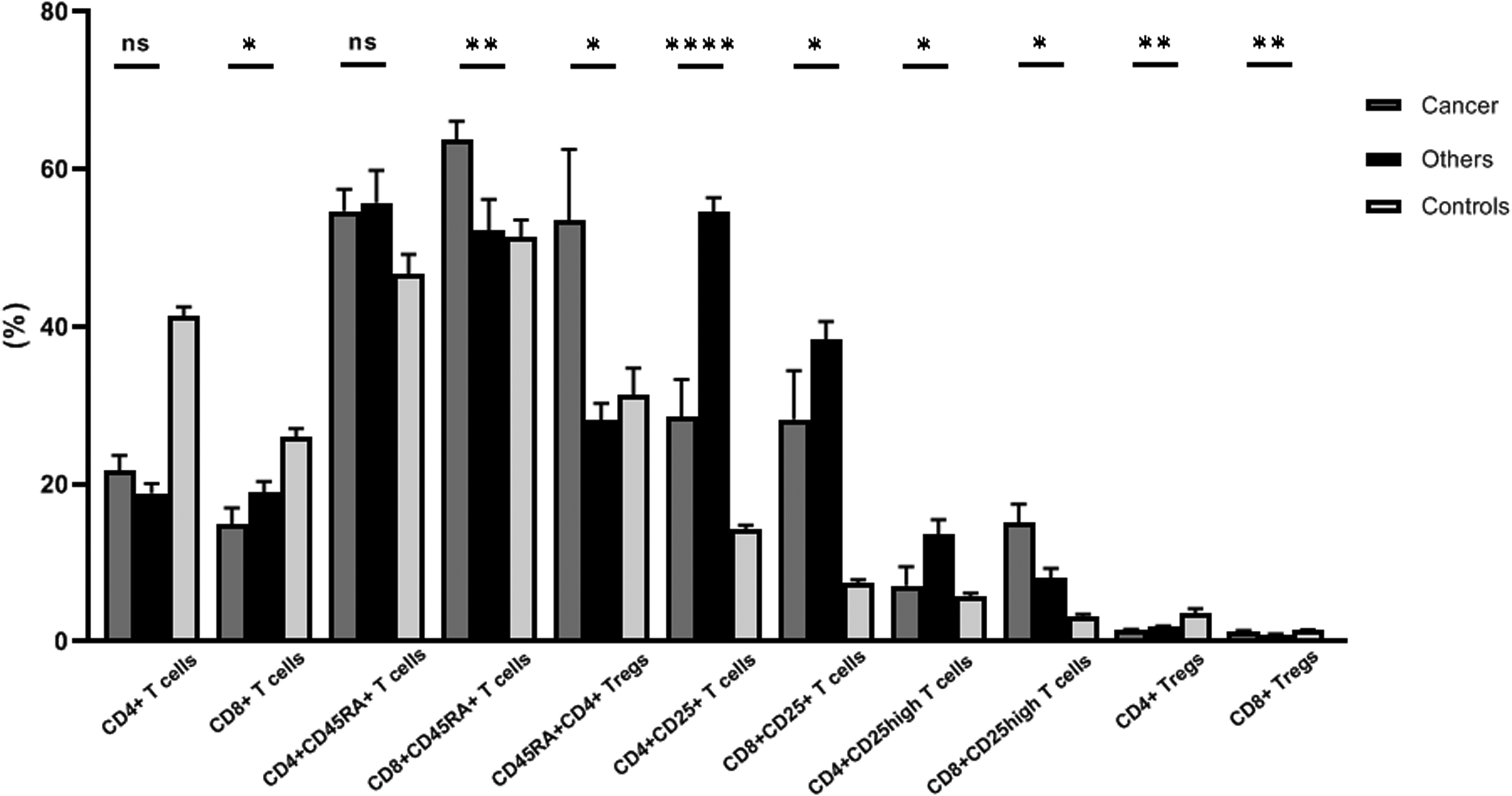
Percentages of the different lymphocyte subsets in COVID-19 patients having cancer and those with other comorbidities. The percentages of CD4+ and CD8+ lymphocytes were assessed in the lymphocyte population, and the percentages of CD25+, CD25high, CD45RA+ and regulatory T cells were calculated among the CD4+ and CD8+ lymphocytes.
CD8+ T-lymphocytes were significantly decreased in COVID-19 patients with cancer compared to patients without cancer. The overall expression levels of CD25 on CD8+ cells were higher in patients without cancer, yet the CD8+CD25high T-cells were significantly higher in the patients with cancer. Although the percentage of CD8+ Tregs in both cancer and non-cancer patients was not noticeably different than that in the control group, their level was significantly lower in the non-cancer patients than in those with cancer.
Furthermore, significantly higher percentages of CD8+CD45RA+ T-lymphocytes and CD45RA+ CD4+ Tregs were found in COVID patients with cancer. No significant difference in serum IL-10 levels was observed between patients with and without cancer.
Discussion
Patients with comorbidities are among the vulnerable groups susceptible to COVID-19–associated morbidity and mortality; thus, these patients were the target group of this study. In agreement with the results of a prior study, the most common symptoms among the comorbid COVID-19 patients in this study were fever, cough, dyspnea, and fatigue, although the percentages of patients with these symptoms were variable. 8 In addition, in line with a previously report, 9 DM, hypertension, and malignancies were also the most common comorbidities observed in COVID-19 patients in the present study.
A number of biomarker abnormalities have been identified in COVID-19 patients, especially those with severe disease. 10 Our results agree with those of previous studies reporting that the COVID-19 biomarkers CRP, ferritin, D-dimer (an indicator of coagulation and fibrinolysis), and LDH (triggered by necrosis of the cell membrane, suggesting lung damage) were increased in patients compared to controls. All these markers, except for CRP, were significantly higher in severe COVID-19 patients with comorbidities than in non-severe cases with comorbidities.11,13
Compared to other viral infections, elevated serum urea, creatinine, and direct bilirubin seem unique to patients infected with SARS-CoV-2. 13 Although the levels of all these three markers were significantly higher in COVID-19 patients with comorbidities than controls, only direct bilirubin showed a statistically significant increase in severe cases compared to non-severe cases.
Assessing biomarkers in a patient with SARS-CoV-2 infection can improve prognosis and outcomes, especially those with comorbidities. Among the biomarkers for severity assessed in this study, only a few (PT, LDH, and direct bilirubin) had significant predictive values for severity in comorbid COVID-19 patients, which is in accordance with other studies.14,15
In line with an earlier study, 16 neutrophil count was significantly increased in COVID-19 patients compared to controls, with no differences observed in patients with severe versus non-severe disease. In contrast, the lymphocyte count and CD4+ and CD8+ T-cells decreased significantly in comorbid COVID-19 patients compared to controls, with a significantly lower lymphocyte counts and CD4+ T-cells in severe cases compared to non-severe cases. The difference in CD8+ T-cells between severe and non-severe cases was not significant. These findings were in accordance with those of a previous study. 17
The lymphocyte count was found to be a significant predictive marker for disease severity in this study, as had been previously reported. 18 CD4+ T-cells, CD4+CD25high, CD8+CD25+, and CD8+CD25high T-lymphocytes were also significant predictive biomarkers in the comorbid COVID-19 patients in this study. Zhang and Wu found that CD4+ and CD8+ T-cell counts were important biomarkers for predicting the severity of COVID-19.
COVID-19–associated lymphopenia may be more severe than lymphopenia occurring in other viral infections and seems to be more selective for T-cell lineages. 3 There are many hypotheses for why this reduction in lymphocytes occurs, including lymphocyte infiltration and sequestration in the target organs; the expression of ACE2 receptors on lymphocytes, making them direct targets of SARS-CoV-2; the increase of pro-inflammatory cytokines in COVID-19; and the hyperactivation of T-cells with high levels of expression of pro-apoptotic molecules. 19
CD25, the alpha chain of the IL-2 receptor, is considered to be the most prominent cellular activation marker. 20 An alarming increase in CD25 expression was detected on both CD4+ and CD8+ T-cells in our comorbid COVID-19 patients. Additionally, expression of CD25 on CD4+ T-cells was significantly higher in severe cases compared to non-severe cases. These data are somewhat in agreement with previously reported findings. 21 The increase in CD25 expression may indicate the ability of SARS-CoV-2 to provoke an immense activation of immune responses in some infected patients. 22
Hyperactivated T-cells with increased production of inflammatory cytokines may enhance the immunopathological damage observed in COVID-19 patients. 23 This was underlined by the positive correlation observed between the frequency of CD4+CD25+ and CD4+CD25high T-lymphocytes and D-dimer, bilirubin, number of affected lung lobes, and PT. In addition, their positive correlations with age suggested that these cells could play a role in disease progression in elderly patients. Moreover, Mathew et al reported that the increased number of activated CD4+ and CD8+ T-cells was associated with an exhausted phenotype in persistent COVID-19, with these cells characterized by suppressed functions and cytotoxicity. 24
It is worth mentioning that, unlike the expression on CD4+ cells, CD25 expression by CD8+ cells were lower in severe cases. Both CD25low and CD25high CD8+ cells are potent killer cells and are thus crucial for viral clearance. 25 This may explain the inverse relationships observed between these cells and LDH, ferritin, bilirubin, respiratory rate, SpO2 percentage, number of affected lung lobes, and the presence of patchy opacities, as well as their positive correlation with the PaCO2.
The hyperactivation of T-cells observed in our comorbid COVID-19 patients could be attributed to several factors, including the significantly lower levels of CD4+ and CD8+ Tregs found in these patients compared to controls. This was highlighted by the negative correlations observed between the frequency of CD8+ Tregs and those of CD4
Elucidating the complex functions of the pleiotropic cytokine IL-10 remains an outstanding challenge for immunologists. 26 Although IL-10 can constrain inflammation by inhibiting the production of proinflammatory cytokines, it can also have immunostimulatory effects. Thus, its overall effects on antiviral immune responses are complex and depend on many parameters. 27
In a previous experimental study of coronavirus-induced encephalitis, it was reported that IL-10 was effective in promoting virus clearance by enhancing the activity of CD8+ cells while limiting antigen-presenting cell–driven inflammation. 28 In addition, findings from the previous SARS infections indicate that the levels of anti-inflammatory cytokines such as IL-10 were reduced. 29 Our results support these findings, as we reported significantly lower IL-10 levels in hospitalized comorbid COVID-19 patients than in controls. The phase of infection appears to be an important determinant of the level of IL-10, which cannot be standardized across studies.30,32 IL-10 production is maximal at the height of the adaptive inflammatory response, being expressed largely by virus-specific CD4+ and CD8+ T-cells. 33 This explains the significant decrease in IL-10 levels that accompanied the significant decrease in CD4+ and CD8+ T-cell counts in patients compared to controls in the current study. These cell counts anywhere not significantly different between severe and non-severe cases, and IL-10 levels followed the same pattern. This is in accordance with the study by Chen et al who did not find significant differences in patients with different levels of severity. In the contrast, others reported IL-10 levels were significantly higher in patients with severe infections. 17 Another important determinant of IL-10 level is T-cell exhaustion and consequent loss of cytokine production, including IL-10. 34 Thus, more experimental studies are needed to clarify whether overactivation or ablation of IL-10 could be helpful in the treatment of severe COVID-19. 5 The specific comorbidity may also affect the level of IL-10. For example, type 2 DM, the most common comorbidity observed among our patients, was accompanied by a lower level of IL-10 than measured in controls in earlier studies.35,36
Naive T-lymphocytes express the high molecular weight isoform (CD45RA), which is lost after activation and replaced by the low molecular weight isoform CD45RO. However, T-cells can revert from CD45RO+ to CD45RA+ due to antigen activation or homeostatic cytokine stimulation (IL-7 and IL-15), forming effector memory T-cells re-expressing CD45RA (TEMRA). 37 These cells lie within both the CD4+ and CD8+ subsets and increase considerably in certain situations, including aging and persistent viral infections.38,39 These cells have been reported to be characterized by many features, among which is having many signatures of senescence and being highly susceptible to apoptosis.40,41
Significant increases in the frequencies of CD4+CD45RA+ and CD8+CD45RA+ T-lymphocytes and CD45RA+CD4+ Tregs were found in our patients. Likewise, Qin et al observed an increase in the percentages of naïve CD4+ T-cells and naïve regulatory T-cells. 17 The frequency of CD45RA-expressing cells followed the same pattern as that of CD25-expressing cells in the CD4+ and CD8+ compartments: Both were significantly increased in COVID-19 patients compared to controls, indicating T-cell proliferation provoked by SARS-CoV-2. These findings may suggest that these CD45RA-expressing cells are mostly TEMRA rather than naïve T-cells, in line with the presence of a viral infection and the general theme of immune stimulation in these hospitalized comorbid COVID-19 patients. The analysis of additional markers is needed to confirm this suggestion.
The only comorbidity affecting the T-cell subsets tested in this study was cancer, as several statistically significant findings were detected among cancer patients. It is known that Tregs compromise the anti-tumor response by dampening the efficiency of T effector cells. In the current study, the percentage of CD8+CD25+FoxP3+ Tregs was significantly higher in COVID-19 patients with cancer than in patients with other comorbidities, in line with the important role of these cells for cancer development via immune evasion. These findings are in agreement with those of a previous study that reported an increase in CD8+ Treg subsets in patients with ovarian cancer compared to patients with benign ovarian tumors and healthy controls. 42 The significant increase in the CD8+ Tregs in the group of patients with cancer was accompanied by decreased CD4+CD25+ and CD4+CD25high T-cells, emphasizing the crucial role of CD8+ Tregs in impeding the excessive activation of CD4+ T-cells. In addition, the frequency of CD8+CD45R+ cells that possess regulatory suppressor functions 43 was higher in patients with cancer than those without cancer, in line with their role in the evasion of immune surveillance that promotes tumor growth.
The study has some potential limitations. Defining memory/effector T cell subsets including naïve T cells (TNs), stem cell-like memory T cells (TSCMs), central memory T cells (TCMs), effector memory T cells (TEMs), and TEMRAs would have given a broader insight into the differentiation and activation status of T cells. Also, using additional markers such as CD127 may further improve Tregs phenotypic characterization. Furthermore, analyzing the expression of exhaustion markers as PD-1 and CTLA-4 would have given a full-blown picture of the impact of the virus on T cell activity.
Conclusion
Many parameters were found to be predictive of severity in the comorbid patients in our study. Significant reductions in the levels and activation of CD4+ and CD8+ T-cells were found. In addition, CD4+ and CD8+ Tregs were significant decreased in patients, probably pointing to a prominent role of CD8+ Tregs in dampening CD4+ T-cell activation.
Supplemental Material
sj-docx-1-cat-10.1177_10760296221107889 - Supplemental material for COVID-19 Infection in Patients with Comorbidities: Clinical and Immunological Insight
Supplemental material, sj-docx-1-cat-10.1177_10760296221107889 for COVID-19 Infection in Patients with Comorbidities: Clinical and Immunological Insight by Omnia El-Badawy, Nahla M. Elsherbiny, Doaa Abdeltawab, Doaa M. Magdy, Lamees M. Bakkar, Shimaa A. Hassan, Elham A. Hassan, Ahmed M. Thabet, Ahmed M. Ashmawy, Ehab F. Moustafa, Wael A. Abbas, Ahmad Bahieldeen Ahmad, Amal Rayan, Khaled Saad, Amira Elhoufey, Hosni A. M. Hussein, Ali A. Thabet and Asmaa M. Zahran in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgement
The authors would like to express their special gratitude to Dr Mohamed Abdelghani, assistant lecturer at the Department of Gastroenterology and Tropical Medicine, Faculty of Medicine, Assiut University, Assiut, Egypt, for his kind help in sample collection.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The study was conducted as per the Declaration of Helsinki, and the Ethics Committee of the Faculty of Medicine, Assiut University, reviewed and approved the research (IRB NO. 17300436).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
