Abstract
Introduction
Primary immune thrombocytopenia (ITP), originally known as idiopathic thrombocytopenic purpura, is an autoimmune bleeding disorder that primarily manifests as megakaryocyte maturation disorder in the bone marrow and increased platelet destruction in the peripheral blood. ITP was thought to occur primarily due to antibody-mediated autoimmune diseases. Current studies1,2 have reported that cellular immunity also plays a role in the pathogenesis of ITP. T cells especially play an important role in pathogenesis. However, the precise interaction among platelet destruction, megakaryocyte dysfunction, and humoral and cell-mediated immune disorders in the pathogenesis of ITP has not been elucidated completely. Due to the lack of clear biomarkers for clinical diagnosis and prognosis, the diagnosis of ITP is difficult. There have been many prior reports on the role of detection of autoantibodies to platelet glycoprotein in the diagnosis and treatment of ITP, but the main drawback to this approach is their low sensitivity so that some patients are still undiagnosed due to lack of diagnostic evidence. Therefore, the research group designed a research idea to conduct bone marrow examinations in patients with thrombocytopenia, analyze platelet parameters, and observe the number and morphological characteristics of megakaryocytes under the microscope. According to previously published literature, there are few studies on the value of bone marrow megakaryocyte number, morphology, and platelet parameters in the diagnosis of ITP. Therefore, in this study, we assessed the value of combined detection of megakaryocyte and platelet parameters for the diagnosis of primary ITP and aimed to enrich laboratory diagnostic information about ITP. We found that megakaryocyte count together with platelet parameters has potential diagnostic value for ITP.
Data and Methods
General Data
A total of 62 patients newly diagnosed with ITP who had not received previous treatment and nonsplenectomized patients at the Fuyang People's Hospital between January 2019 and October 2021 were included in the study. The patient cohort consisted of 25 males and 37 females aged from 14 to 83 years, with an average age of (45.6 ± 19.2) years. All ITP patients met the initial diagnosis and treatment criteria. 3 ITP diagnostic criteria were defined as a platelet count (PLT) ≤ 100 × 109/L, the blood cell morphology was normal and the spleen was generally not large, the bone marrow examination showed that the megakaryocyte count was increased or normal and with maturation disorders, platelets were rare, and other lineages such as erythrocyte system, granulocytes system, monocyte system, and the lymphatic system showed no obvious morphological changes. The purpose of bone marrow examination in this study is to exclude other secondary thrombocytopenia, such as malignant blood diseases, myelodysplastic diseases, lymphatic system proliferative diseases, thyroid diseases, autoimmune diseases, and drug-induced thrombocytopenia and gestational thrombocytopenia, etc The criteria for response were defined as follows: (i) complete response: PLT ≥100 × 109/L and absence of bleeding, (ii) partial response: PLT ≥30 × 109/L and at least 2-fold increase of the baseline PLT and absence of bleeding, (iii) no response: PLT <30 × 109/L or <2-fold increase of the baseline PLT or bleeding. A total of 40 non-ITP patients with normal bone marrow in the same period were selected as the control group, including 16 males and 24 females aged from 12 to 79 years, with an average age of (44.6 ± 22.7) years. The control group of non-ITP hospitalized patients was hypothyroidism, hypertension, iron deficiency anemia, upper respiratory tract infection, chronic non-atrophic gastritis, and other non-platelet system diseases. Roughly normal bone marrow image is judged by the proportion of granulocyte system, erythrocyte system, lymphocyte system, monocyte system, and plasma cell system within the normal range, no increase or decrease in the number of megakaryocytes, easy to see platelets, and no pathological morphological changes in each system. There were no statistically significant differences in age composition and sex ratio in the general data for the two groups (P > 0.05). Informed consent forms were signed by all subjects participating in this study. This study was reviewed and approved by the Medical Ethics Committee of the Fuyang People's Hospital (approval number 2020-11).
Methods
Sample collection
A total of 1–2 mL of venous blood was collected from the fasting subjects before ITP treatment and thoroughly mixed in the EDTA-K2 anticoagulant tube. Bone marrow aspiration was performed by clinicians following standard procedures. Bone marrow smear samples were sent out for examination.
Detection methods
PLT, PCT, MPV, PDW, and P-LCR% were performed using the Mindray BC-6800 automatic blood cell analyzer with a supporting series of reagents (Shenzhen Mindray Bio-Medical Electronics Co., China). The bone marrow megakaryocyte examination method was used to select a good-quality bone marrow smear sample stained with Wright's stain which was visible marrow granules, containing marrow-specific cells, and a greater marrow neutrophil or neutrophil granulocyte ratio than that of peripheral blood and dried naturally. The total number of megakaryocytes in the whole smear was determined using an Olympus CX31 optical microscope (Tokyo, Japan). The homogeneous parts of the smear were selected to classify the megakaryocytes (Meg) and calculate the proportion of promegakaryocytes (ProMeg), granular megakaryocytes (GMeg), platelet-producing megakaryocytes (PMeg), and naked megakaryocytes (NMeg) at each stage. The general morphological characteristics of promegakaryocytes are 30-50 µm in diameter, with abundant cytoplasm, dark blue color, and a few tiny light purple-red granules near the nucleus; GMeg are 40-70 µm in diameter, with extremely abundant cytoplasm, light blue color and filled with a large number of tiny light purple-red granules; platelet producing megakaryocytes are 40-70 µm in diameter, sometimes up to 100 µm, with abundant cytoplasm, light blue color, abundant granules and clusters of platelets, incomplete cytosol and adherent platelets; NMeg have no cytoplasm, only the nucleus, nucleated megakaryocytes have no cytoplasm and only nuclei. Cell classification and counting are highly subjective, and the above counts and classifications were reviewed by two senior experts in blood cell morphology in a double-blind manner and were checked by the technical supervisor. All data were expressed as the mean value or mean value ± standard deviation (SD) and the deviation between the results and the mean was CV% <5%.
Statistical Methods
All data processing and analysis were completed using the SPSS16.0 statistical software. Mean ± standard deviation (
Results
Comparison of the Total Number of Megakaryocytes Megakaryocyte Proportion at Each Stage Between the ITP and Control Groups
The total number of megakaryocytes in the ITP group ranged from 35 to 476 with a mean of 158.5 ± 104.2, while that in the control group ranged from 20 to 219 with a mean of 64.5 ± 48.9. The difference between the two groups was statistically significant (P<0.05; see Table 1). Compared with the control group, the proportion of promegakaryocytes, GMeg, and NMeg in the ITP group increased, while the proportion of PMeg decreased in the ITP group. All differences were statistically significant (P<0.05; Table 1 and Figure 1A–D).
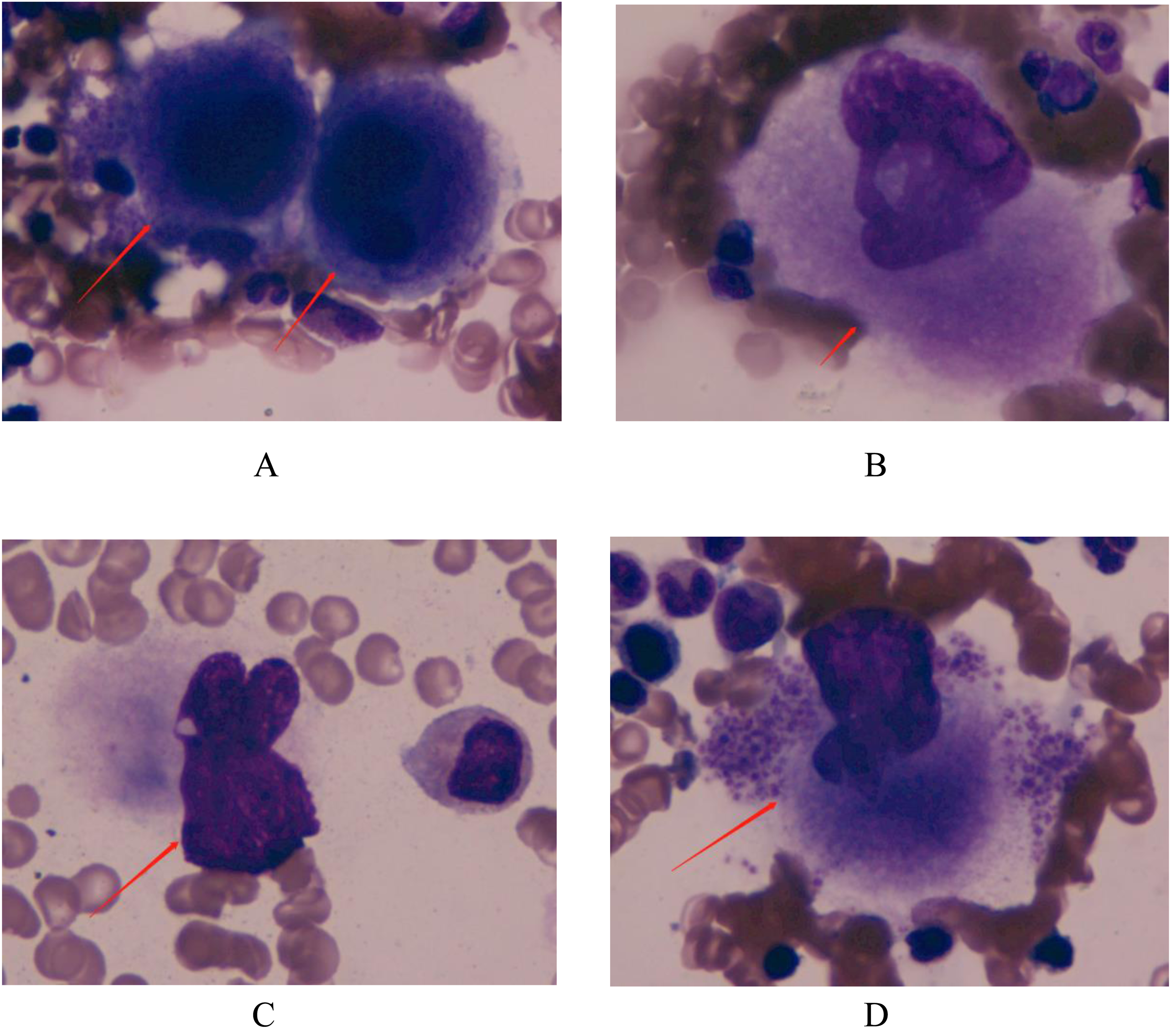
megakaryocytes at each stage (A) promegakaryocytes (B) granular megakaryocytes (C) naked megakaryocytes (D) platelet-producing megakaryocytes.
Comparison of the Total Number of Megakaryocytes and Megakaryocyte Proportion at Each Stage Between Itp and Control Groups.

Comparison of Platelet Parameters
PLT, PCT, MPV, PDW, and P-LCR% between ITP and control groups compared to the control group, PLT and PCT decreased, while MPV and P-LCR% increased in the ITP group, and all differences were statistically significant (P<0.01). In addition, PDW increased in the ITP group, and the difference was not statistically significant (P > 0.05; Table 2).
Comparison of Platelet Parameters PLT, PCT, MPV, PDW, and P-LCR% Between ITP and Control Groups.

Linear Correlation Analysis and Correlation Matrix Table Between Platelet Parameters and Megakaryocyte Proportion in ITP Groups
Linear correlation analysis among platelet parameters indicated PLT was strongly positively correlated with PCT (r = 0.921, p<0.01). PCT was weakly positively correlated with MPV (r = 0.309, p<0.05). MPV was positively correlated with P-LCR (r = 0.856, p<0.01). PDW was weakly positively correlated with P-LCR (r = 0.296, p<0.05). (Figure 2, Table 3). And the Pearson r coefficient reveals that PDW was weakly positively correlated with P-LCR (r = 0.296, p<0.05) and Meg (r = 0.301, p<0.05), and negatively correlated with PMeg (r = -0.336, p<0.05). Meg (number) was positively correlated with ProMeg (r = 0.394, p<0.01) and negatively correlated with PMeg (r = -0.458, p<0.01). ProMeg (%) was negatively correlated with GMeg (r = -0.873, p<0.01) and PMeg (r = -0.350, p<0.05). PMeg (%) is negatively correlated with NMeg (r = -0.372, p<0.01). A complete correlation matrix for these parameters is illustrated in Table 3.
Pearson Correlation Matrix Table of Correlation Between Platelet Parameters and Megakaryocyte Proportion.

* p < 0.05 ** p < 0.01

Linear correlation analysis among platelet parameters (PLT, PCT, MPV, PDW, and P-LCR%).
Diagnostic Performance of PLT, PCT, MPV, PDW, and P-LCR% in ITP
To estimate the clinical performance of the platelet indices in ITP, ROC curves were applied to determine whether the sensitivity and specificity of the parameters were sufficient to diagnose ITP. The sensitivities and specificities of PLT, PCT, MPV, PDW, and P-LCR% were listed in Table 4. The areas under the ROC curve (AUC) of PLT, PCT, MPV, PDW, and P-LCR% were 1.000, 0.906, 0.801, 0.605, and 0.871, respectively (Figure 3). ROC curves of PLT, PCT, MPV and P-LCR% were shifted to the upper left of the graph, indicating that PLT, PCT, MPV and P-LCR% had significance value [P = 0.000 for both, 95% confidence interval (CI) of AUC: 1.000-1.000 for PLT, and 95% CI of AUC: 0.844-0.967 for PCT, 95% CI of AUC: 0.710-0.892 for MPV, 95% CI of AUC:0.795-0.946 for P-LCR% ], but PDW had no predictive significance in ITP diagnosis (P = 0.073, and 95% CI: 0.497, 0.713 including 0.5).](Figure 3). These results reveal that PLT, PCT, MPV, and P-LCR% are reliable and sufficient to diagnose ITP.
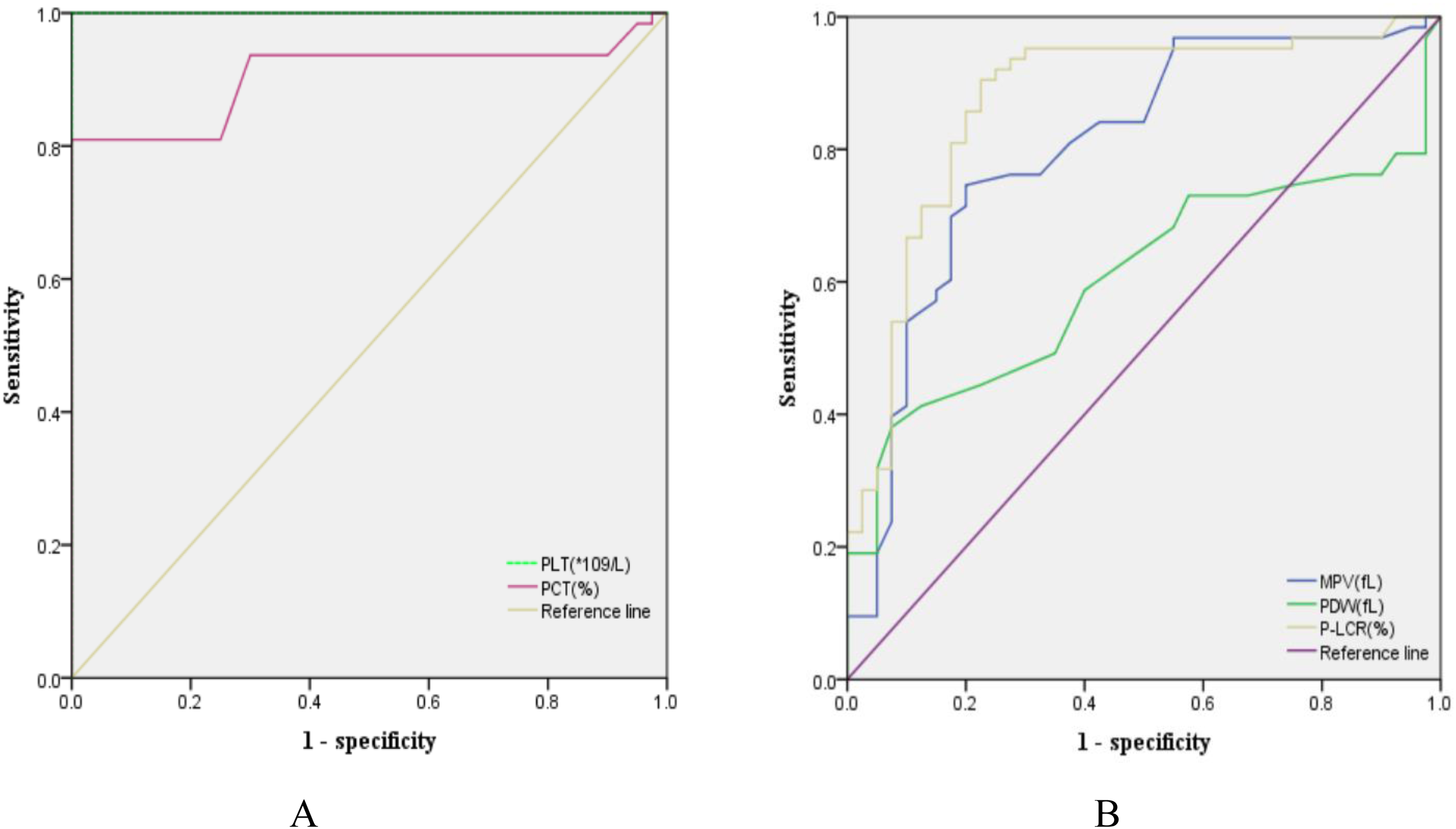
Diagnostic performance of PLT, PCT, MPV, PDW, and P-LCR% in ITP. (A) Receiver-operating characteristic curve of platelet count (PLT), platelet pressure product (PCT) (B) Receiver-operating characteristic curve of mean platelet volume (MPV), platelet distribution width (PDW), platelet large cell ratio (P-LCR%).
Receiver-Operating Characteristic Results of Immune Thrombocytopenic Purpura.

Discussion
ITP is a common hemorrhagic autoimmune disease with an incidence of 2–4/100,000 in adults. 4 ITP can cause asymptomatic thrombocytopenia in mild cases, petechiae and ecchymosis in skin mucosa in severe cases, and even life-threatening visceral bleeding and intracranial hemorrhage, resulting in decreased quality of life. The diagnosis of ITP is made based on the absence of other causes of thrombocytopenia. Some treatment options for ITP are costly and require long-term treatment, others are less expensive but have significant side effects. Splenectomy is considered to be the effective treatment for ITP, however prospective studies 5 have shown that with the advent of newer second-line therapy drugs such as thrombopoietin receptor agonist (TPO-RA), the clinical use rate of splenectomy for ITP is gradually decreased. 6 Nazy et al 7 have found that TPO and TPO receptor (C-MPL) autoantibodies were detectable in 44% of patients with ITP and other thrombocytopenic diseases. Decreased TPO is an important reason for a megakaryocyte maturation disorder in the bone marrow. 8 Antiplatelet autoantibodies are the main cause of ITP platelet destruction. 9 Approximately 60%-80% of ITP patients can produce antibodies predominantly targeting platelet glycoprotein (GP) platelet membrane components, mainly platelet glycoprotein Iba (GPIba) and platelet glycoprotein Ib/IV (GPIb/IX) complex. 10 Prior data in the literature suggest that platelet-associated immunoglobulin G (PAIgG) or antibody detection in plasma or serum is an insufficient diagnostic test and data for the performance of these assays are currently limited and their use is not currently recommended. 11 GP-specific tests are highly specific but lack sensitivity, missing roughly half of all patients with ITP. 12 Therefore, the detection of laboratory indicators will help overcome incorrect diagnoses and inadequate drug treatment.
In this study, we discussed the value of combined detection of megakaryocyte and platelet parameters for the diagnosis of primary ITP. The results showed that the total number of megakaryocytes and megakaryocyte proportion at each stage have potential diagnostic value for ITP. The present study is consistent with prior literature reports.13,14 ITP patients with PLT < 50*109/L had no abnormal morphological changes in the erythrocyte system and granulocyte system and megakaryocytes ≥ 35 with maturation disorder, which is consistent with the morphological characteristics of ITP bone marrow with 50*109/L ≤PLT < 100*109/L. The morphological characteristics of the ITP bone marrow were consistent. In this study, we found that the total number of megakaryocytes was correlated with classification of megakaryocytes. Therefore, the number and classification of megakaryocytes are of great value in the diagnosis of ITP.
Platelet parameters include PLT, PCT, PDW, MPV, and P-LCR%. PLT is the best indicator used to directly reflect the number of platelets. PCT reflects the pressure ratio of platelets to whole blood, which is consistent with the change in PLT. The present study results showed that PLT and PCT in the ITP group were significantly reduced compared to the control group (P<0.01), which was consistent with prior literature reports. 15 PDW is an index reflecting the degree of platelet volume dispersion. There was no significant difference in PDW between the ITP and control groups (P > 0.05), indicating that the size of platelet volume in ITP patients was not obvious. It has been reported in the literature 16 that PDW in ITP patients was significantly higher than that in the healthy control group (P<0.01). The possible reasons for this inconsistency might be related to the study subject's age and the number of included cases. The subjects of the study were adults and the number of cases was relatively small, which can be improved by expanding the number of cases. MPV is a parameter that indicates platelet volume. P-LCR% indicates the ratio of large platelets. The results of the present study showed that MPV and P-LCR% in the ITP group were higher than those in the control group (P<0.01). It can be concluded that self-destruction of platelets in the ITP patients was increased, while megakaryocytes in the bone marrow compensate for proliferative platelets by generating larger ones. Platelet volume can be used as an indirect indicator of platelet maturation. Prior studies 17 have reported that MPV in ITP patients was significantly higher than that in healthy people. Another literature study 18 showed that impaired megakaryopoiesis in aplastic anemia (AA) and acute leukemia (AL) with thrombocytopenia had no change in MPV levels and that MPV was higher in ITP patients than in AA and AL patients. The results of Akkuş et al 19 pointed out that MPV can be used as a predictor of early response to first-line treatment in newly diagnosed adult ITP patients. The above analysis shows that MPV and P-LCR% can be used as clinically valuable biological parameters for the diagnosis, differential diagnosis, and efficacy evaluation of ITP.
In this paper, linear correlation analysis, the Pearson correlation analysis indicated the correlation between megakaryocyte and platelet parameters. This study further analyzed and evaluated the performance of platelet parameters in diagnosing ITP by ROC curve analysis. ROC curve analysis indicated that PLT, PCT, MPV, and P-LCR% showed a promising diagnostic value. PDW has not predictive significance in ITP diagnosis (P = 0.073, 95% CI:0.497-0.713, including 0.5). The result of the 95% CI of AUC of PLT: 1.000-1.000 may be due to the small sample size of this study, and further evaluation is needed to increase the sample size. These results are consistent with the study byYang et al 20 .
At an early stage of research design and after considering the difficulty of sampling a single disease, pure disease selection was not considered. Instead, ITP was used as a co-morbidity and the object of the present study, and the combination of ITP with other diseases and non-platelet system diseases in the control group may introduce the influencing factors with varying degrees into the research results, which requires additional case data for further analysis and discussion needs to be further analyzed and discussed. In the control group, the bone marrow image was roughly normal, the number of megakaryocytes did not increase or decrease, and the proportion of megakaryocytes at each stage was within the allowable normal range. There is no mature disorder phenomenon, platelets are easy to see, and there are no obvious abnormal morbid morphological changes in each system. Since the collection of bone marrow from healthy people is not ethically allowed, the reference values of platelet parameters, bone marrow megakaryocyte counts, and proportions at each stage are all derived from healthy people. In addition, the previous literature reported that the platelet parameters, bone marrow megakaryocyte number, and the proportion of each stage of healthy people were within the normal range. Therefore, this study did not include the healthy population as the research object and the ITP group for comparative analysis.
In this study, the number of ITP cases at the present study was limited, so the ITP group was not subdivided into acute type, persistent type, and chronic type. And it is necessary to add more cases and use a reasonable research design and better statistical methods to evaluate the value of the above platelet parameters in the diagnosis of various types of ITP to more accurately predict and evaluate ITP progress.
Conclusion
In conclusion, the platelet parameter detection and bone marrow examination were performed in patients with suspected ITP, analyzed platelet parameters, counted the number of megakaryocytes under a biological microscope, and observed its morphological characteristics to classify, which had important clinical application value for the exclusion and diagnosis of ITP. The testing method used in the present study was simple and the data were easy to obtain. In addition, the low examination cost can reduce the economic burden on patients and has significant social benefits, which is suitable for widespread implementation in primary hospitals.
Footnotes
Acknowledgments
We thank the leader and colleagues for supporting this work. We are also grateful to the Department of Clinical Laboratory of Fuyang People's Hospital for providing technical support. Special thanks for the financial support provided by the Key Research and Development Program of Anhui Province, China(201904a07020049).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Weiwei Wang, Key Research and Development Program of Anhui Province, China (grant number 201904a07020049).
