Abstract
Human monocytes are heterogeneous and play an important role in autoimmune diseases. However, the distribution and function of monocyte subsets remain unclear in primary immune thrombocytopenia (ITP). In this study, we determined the frequencies of monocyte subsets in 71 untreated patients with active ITP and 49 healthy controls by flow cytometry. Compared with controls, the frequency of nonclassical monocytes was significantly increased in patients with active ITP but decreased after complete remission. The intermediate subset was also increased in patients with active ITP and produced the highest levels of tumor necrosis factor α and interleukin 1β. Both the nonclassical and intermediate subsets were negatively correlated with the platelet counts. We further determined the correlation between monocyte subsets and the proliferation of platelet-autoreactive T cells. The purified monocyte subsets were cocultured with CD4+ T cells and autologous platelets. The nonclassical subset showed the highest capability of promoting platelet reactive T-cell proliferation and significantly promoted the secretion of interferon γ among the 3 subsets. In conclusion, the nonclassical and intermediate monocyte subsets are both expanded and play different roles in the pathogenesis of ITP.
Keywords
Introduction
Primary immune thrombocytopenia (ITP) is a common autoimmune disease that is characterized by the immune destruction of platelets and impaired platelet production with complicated immune dysfunctions. 1 Recent evidence emphasizes that the imbalance of cellular immunity plays a central role in the pathogenesis of ITP. 2 Importantly, T lymphocytes are abnormally polarized toward Th1 and Tc1. 3 –5 The subsequently activated platelet-reactive T cells mediate circulating B cells to produce antiplatelet autoantibodies. 6 Moreover, the antigen-presenting cells of patients with ITP are more efficient in stimulating the proliferation of platelet reactive T cells. 6,7
Monocytes are key components of both the innate and adaptive immune systems and recognized as the precursor of dendritic cells (DCs). 8 –10 Human blood monocytes have been segregated into 2 subsets based on CD16 expression for nearly 2 decades. The CD16+ monocyte subset was first identified by Ziegler-Heitbrock 11 and found to expand in numerous infected and autoimmune diseases. 12 –15 Labeled proinflammatory, the CD16+ monocyte subset highly expresses proinflammatory cytokines and possesses higher potency in antigen presentation. 8,16 –18 Recent evidence supports that the minor CD16+ monocyte subset could be further subdivided into 2 smaller subpopulations. According to the new nomenclature, 19 human monocytes can be subdivided into 3 subsets as classical (CD14++CD16−), intermediate (CD14++CD16+), and nonclassical (CD14+CD16++) subsets, and this was further supported by recently published data. 20 –22
Although the CD16+ monocytes are elevated in a host of autoimmune diseases, such as in rheumatoid arthritis (RA) 14,23 and Crohn disease (CD), 13,24 the exact increase in the intermediate (CD14++CD16+) or nonclassical (CD14+CD16++) subsets remains to be defined. Baeten et al 25 reported that the monocytes that were positively stained for the RA autoantigen human cartilage (HC) gp-39 were expanded significantly in both the blood and synovial tissues of patients with RA 25 and that these cells were further characterized as the nonclassical monocytes. In contrast, another group showed that these cells were the intermediate subset. 26
In patients with ITP, the total CD16+ monocytes are increased and polarize Th1 cells, which, in turn, negatively regulate interleukin (IL) 17 and Treg induction. 27 However, the distribution of intermediate (CD14++CD16+) and nonclassical (CD14+CD16++) monocytes has yet not been identified. In the present study, we aim to investigate the distribution and function of the 3 monocyte subsets in patients with ITP.
Materials and Methods
Patients and Controls
Seventy-one untreated patients with active ITP were enrolled in this study. Forty-nine sex and age-matched healthy persons were studied as controls. The diagnosis of ITP was based on the criteria reported previously. 28,29 Active ITP was defined by a platelet count below 100 × 109/L with or without bleeding episodes. Complete remission (CR) was defined as the disappearance of bleeding symptoms accompanied with platelet counts ≥100 × 109/L. 28 All participants were recruited to measure the frequency of peripheral monocyte subsets. Samples from 15 patients with active ITP were used for further functional analysis. Informed consent was obtained from all patients and controls. The study was approved by the hospital-based ethics committee. The clinical characteristics of all of the participants are summarized in Table 1.
Characteristics of Patients and Controls.
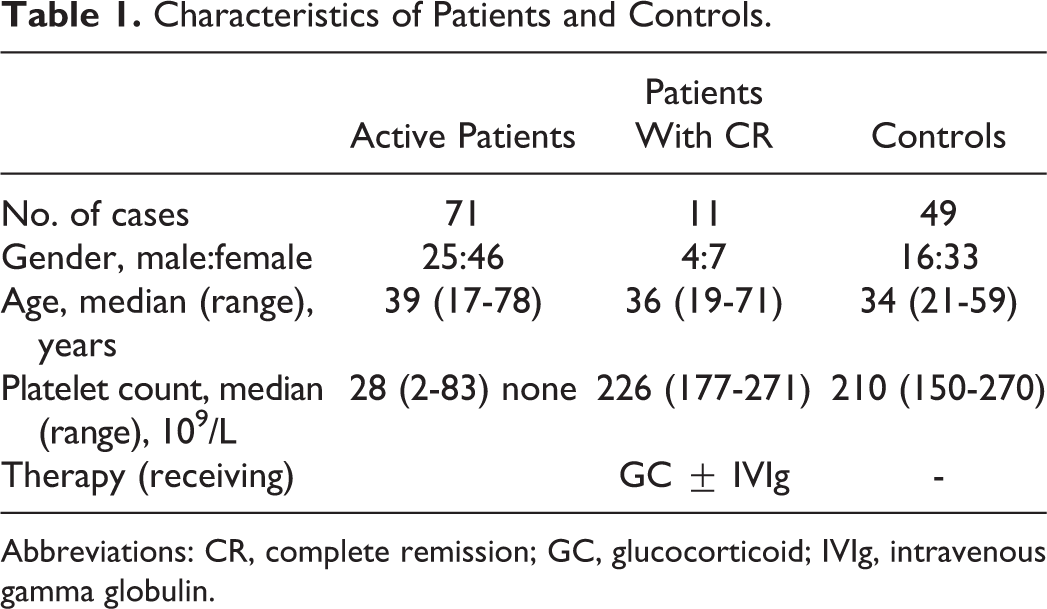
Abbreviations: CR, complete remission; GC, glucocorticoid; IVIg, intravenous gamma globulin.
Plasma Preparation and Isolation of Peripheral Blood Mononuclear Cells
Plasma was obtained from all of the study participants by centrifugation of EDTA anticoagulant venous blood and stored at −80°C until examination. Peripheral blood mononuclear cells were isolated by density gradient centrifugation over Ficoll-Hypaque gradients.
Flow Cytometric Quantification of Monocyte Subsets
EDTA anticoagulant peripheral blood samples were used to analyze the distribution of monocyte subsets on a BD FACSCanto II flow cytometer (BD Biosciences, San Jose, CA, USA). Monocyte subsets were defined as classical (CD14++CD16−), intermediate (CD14++CD16+), and nonclassical (CD14+CD16++) subsets. Whole blood (100 μL) was incubated with R-phycoerythrin (PE)-conjugated antihuman CD14, fluorescein isothiocyanate (FITC)-conjugated antihuman CD45, Allophycocyanin (APC)-conjugated anti-human CD16, and isotype controls, following the manufacturer’s instructions (Biolegend, San Diego, CA, USA). The distribution of monocyte subsets was analyzed using the Flowjo7.6.1 software by the same operator according to the gating strategy described by Wong et al. 30 Isotype controls were used as recommended previously. 19 Total leukocyte count was measured using the automated hematology analyzer Sysmex XE-2100 (Sysmex Corporation, Japan). An absolute count of monocytes was calculated by multiplying the respective percentages by the total leukocyte count. Monocyte subsets were expressed as a percentage of the total monocyte count.
Tumor Necrosis Factor α, IL-1β, and Interferon γ Enzyme-Linked Immunosorbent Assay
The plasma and/or supernatants levels of tumor necrosis factor (TNF) α, IL-1β, and interferon (IFN) γ were measured using enzyme-linked immunosorbent assay (ELISA) kits, according to the manufacturer’s instructions (NeoBioscience Technology, Shenzhen, P.R.China). The minimum detectable levels of TNF-α, IL-1β, and IFN-γ were 7, 3, and 1 pg/mL, respectively. The intra-assay and interassay precision of variation was <9.5%, <10%, and <9%, respectively.
Flow Cytometry Isolation of Monocyte Subsets and CD4+ T Cells
Peripheral blood mononuclear cells isolated in a previous preparation were incubated with PE-conjugated antihuman CD14, FITC-conjugated antihuman CD45, APC-conjugated anti-human CD16, and Peridinin-chlorophyll proteins (PerCP)-conjugated antihuman CD4 (Biolegend), washed twice with 2 mL phosphate-buffered saline (PBS), and centrifuged at 1200 rpm for 8 minutes. Monocyte subsets and CD4+ T cells were sorted immediately according to the gating strategy described previously 19,30 using FACS Aria III flow cytometry system (BD Biosciences). To ascertain the purity of the separated cell populations, a consecutive 3 samples were selected for quality control. Briefly, cells of 1 × 105 were reanalyzed using BD FACSCanto II, which showed that the purity of each isolated population was no less than 95%. The purity standard was referred from previous literature. 26,27
Cell Culture and Treatment
Sorted monocyte subsets (5 × 104/100 μL) were incubated separately in RPMI 1640 supplemented with 10% heat-inactivated human AB serum, 2 mmol/L
Effect of Monocyte Subsets on Platelet-Autoreactive T-Cell Proliferation In Vitro
Autologous platelets were isolated from patients with ITP and healthy controls. The platelet-rich plasma was obtained by centrifugation at 800 rpm for 15 minutes at 20°C. Platelet-rich plasma was centrifuged at 3000 rpm for 10 minutes, and the platelet pellets were then washed once in PBS and adjusted to 1 × 109 platelets/L.
Sorted monocyte subsets were incubated with 10 μg/mL mitomycin C at 37°C with 5% CO2 for 2 hours to inactivate their ability to undergo mitosis and were then washed in PBS twice and resuspended.
Each of the 3 sorted monocyte subsets (5 × 104/well) and CD4+ T cells from the same person were cocultured at a ratio of 1:10 together with 1 × 106 autologous platelets/well in 1 mL final volume of complete medium in a 24-well plate, and the CD4+ T cells without monocytes and platelets were treated as blank controls at 37°C with 5% CO2.
After 72 hours, coculture supernatants were separated by centrifugation, and cells from each well were separated equally into 3 wells in a 96-well plate. The platelet-autoreactive T-cell proliferation was measured using Cell Proliferation ELISA, Bromodeoxyuridine (BrdU) (colorimetric) kits, according to the manufacturer’s instructions (Roche Molecular Biochemicals, Mannheim, Germany).
Statistical Analysis
For statistical analysis, the SPSS version 16.0 software (SPSS) was used. Normally distributed data were described by mean and standard deviation and tested by an independent samples t test. Abnormally distributed data were described by median and range. The plasma cytokine levels and monocyte subset percentages were analyzed by nonparametric statistical analyses. In particular, the Mann-Whitney U or Wilcoxon test for independent or related 2-group analysis, respectively, was used. The correlation between subset frequency and platelet counts was determined by Spearman correlation coefficient. P < .05 was considered statistically significant.
Results
The Distribution of Monocyte Subsets in Patients With ITP and Normal Controls
The total counts of monocytes calculated by multiplying the respective percentages by the total leukocyte count were analyzed. Both the frequency in leukocyte (active ITP: 7.65% ± 3.65% vs controls: 5.12% ± 1.41%, P < .001) and the absolute count of total monocytes (active ITP: 0.5 ± 0.28 × 109/L vs controls: 0.32 ± 0.45 × 109/L, P < .001) were higher in untreated patients with active ITP than in normal controls.
Figure 1A to C showed examples of the gating strategy for the distribution of monocyte subsets. Peripheral blood neutrophils, monocytes, and lymphocytes were gated on the basis of side scatter and CD45 expression (Figure 1A). Then, the expression of CD14 and CD16 on peripheral blood monocytes was examined. A representative FCS manifestation of monocyte subsets in an patient with active ITP and a patient with CR was shown in Figure 1B and C, separately. As shown in Figure 1D, the frequency of the nonclassical monocytes was significantly elevated in patients with active ITP compared with controls (active ITP: 5.44% [0.36%-22.29%] vs controls: 1.83% [0.12%-7.11%], P < .001). At the same time, the frequency of the intermediate monocytes was also increased in active patients (active ITP: 10.54% [5.01%-45.15%] vs controls: 7.6% [1.6%-23.99%], P < .001; Figure 1E). On the contrary, the frequency of classical monocytes decreased in active patients (active ITP: 82.6% [51.23%-92.38%] vs controls: 90.75% [66.83%-95.92%], P = .001; Figure 1F).
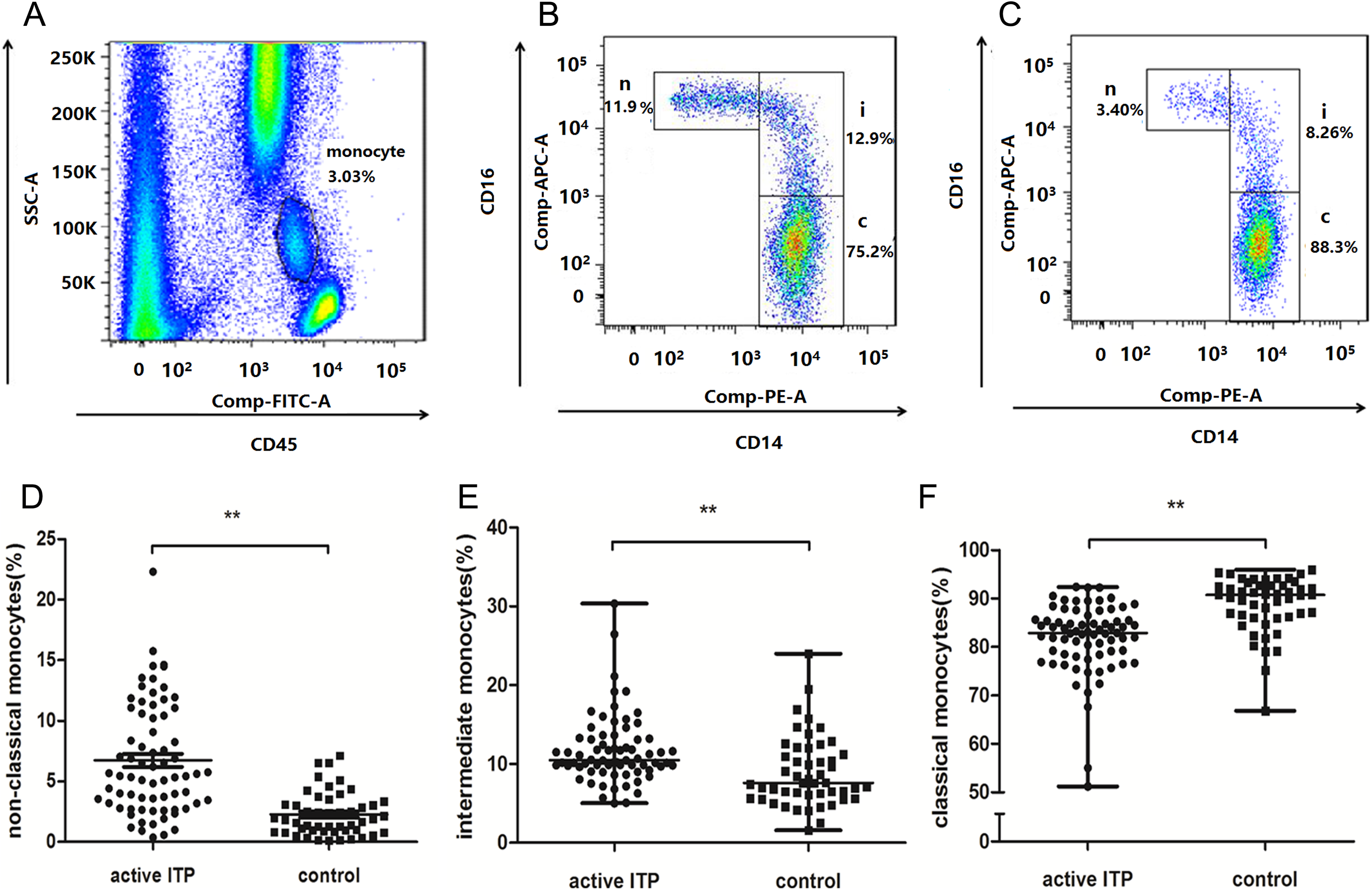
Distribution of peripheral blood monocyte subsets in patients and controls. A, Neutrophils, monocytes, and lymphocytes were gated based on SSC and CD45-FITC. Gated monocytes are shown with a circle. The expression of CD14 and CD16 on gated peripheral blood monocytes from a typical patient with ITP at active status (B) and in remission (C) is shown. D, The frequency of the nonclassical monocytes was significantly increased in patients with active ITP compared with that in controls. E, The frequency of the intermediate monocytes was increased in patients with active ITP compared with that in controls. F, The frequency of classical monocytes was decreased in active patients compared with that in controls. **P < .01. c indicates classical monocyte subset; i, intermediate monocyte subset; ITP, immune thrombocytopenia; n, nonclassical monocyte subset; SSC, side scatter.
In 11 patients, the monocyte subsets were retested after the patients achieved CR. As shown in Figure 2A, the frequency of nonclassical subsets was decreased substantially after CR (active: 6.32% [0.57%-11.85%] vs CR: 2.18% [0%-8.78%], P = .006), but the frequency of classical and intermediate subsets was stable (Figure 2B and C).
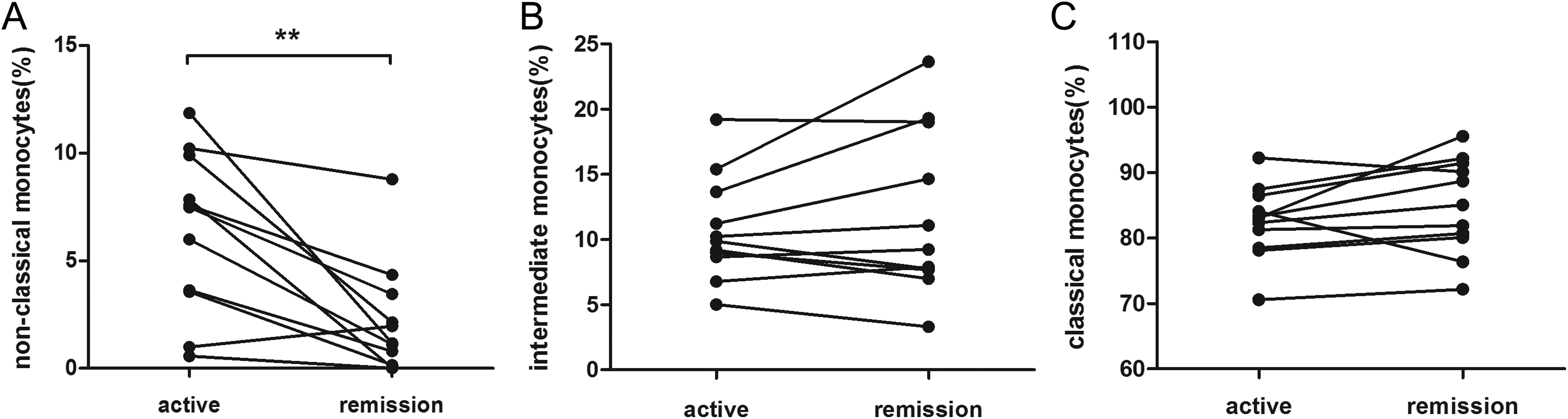
Differences in monocyte subset distribution before and after CR (n = 11). A, The frequency of nonclassical monocytes decreased substantially after CR. B and C, No significant changes in the intermediate or classical monocyte frequency were found between patients with active ITP and patients in remission. **P < .01. CR, complete remission; ITP, immune thrombocytopenia.
Correlation Between the Frequency of Monocyte Subsets and Platelet Counts in Patients With ITP
Spearman correlation coefficient was determined to assess the correlation between the frequency of monocyte subsets and the platelet counts. In patients with active ITP, the frequency of nonclassical and intermediate monocyte subsets negatively correlated with platelet counts (Figure 3B and C), and the frequency of classical subsets positively correlated with platelet counts (Figure 3A).
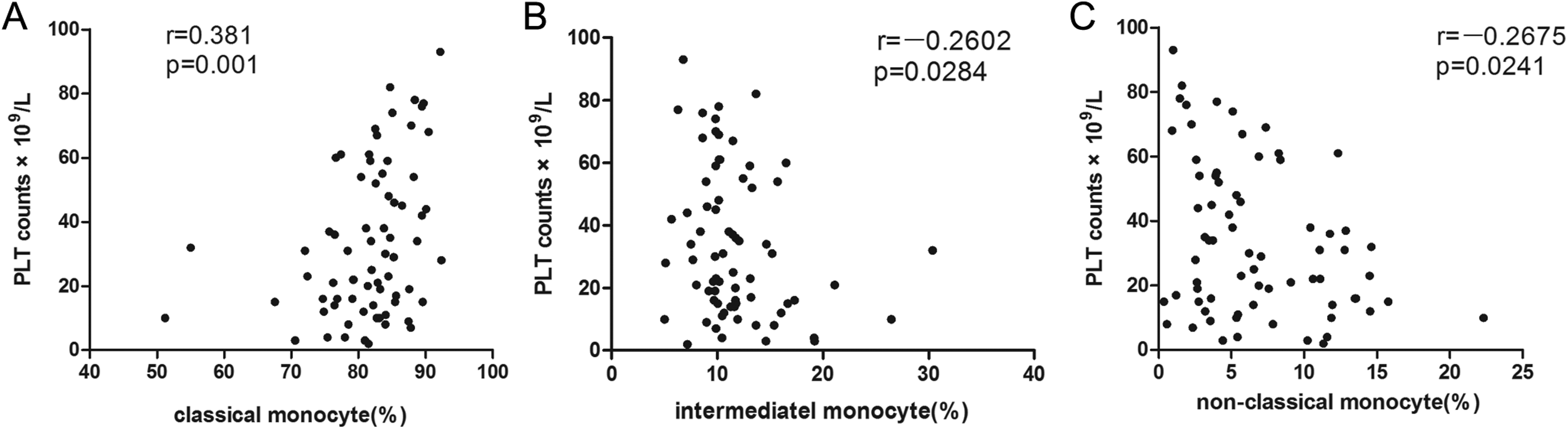
Monocyte subsets are associated with platelet counts. A, Classical monocytes showed a positive correlation with platelet counts. Intermediate (B) and nonclassical (C) monocytes showed a negative correlation with platelet counts. r indicates Spearman correlation coefficient.
The Secretion Function of Different Monocyte Subsets
We measured the cytokines primarily secreted by monocytes such as TNF-α and IL-1β in plasma. Compared with normal controls, the plasma TNF-α and IL-1β levels in patients with active ITP were elevated dramatically (n = 20). To further determine which monocyte subset produces the most TNF-α and IL-1β, different subsets were cultured separately in vitro. After stimulation by LPS for 24 hours, the intermediate subset showed the most capability of cytokine secretion both in patients with ITP and controls. As shown in Table 2, the intermediate subset produced the highest levels of TNF-α and IL-1β in patients and controls. We retested the cytokine levels after the patients obtained CR and found that the TNF-α secretion of intermediate subsets decreased significantly. No difference in the level of TNF-α was found in the classical and nonclassical subsets. There was also no difference in the IL-1β levels between patients in the active stage and those in CR. The cytokine detection values are shown in Table 2.
Cytokine Levels in Plasma and Culture Supernatant.a
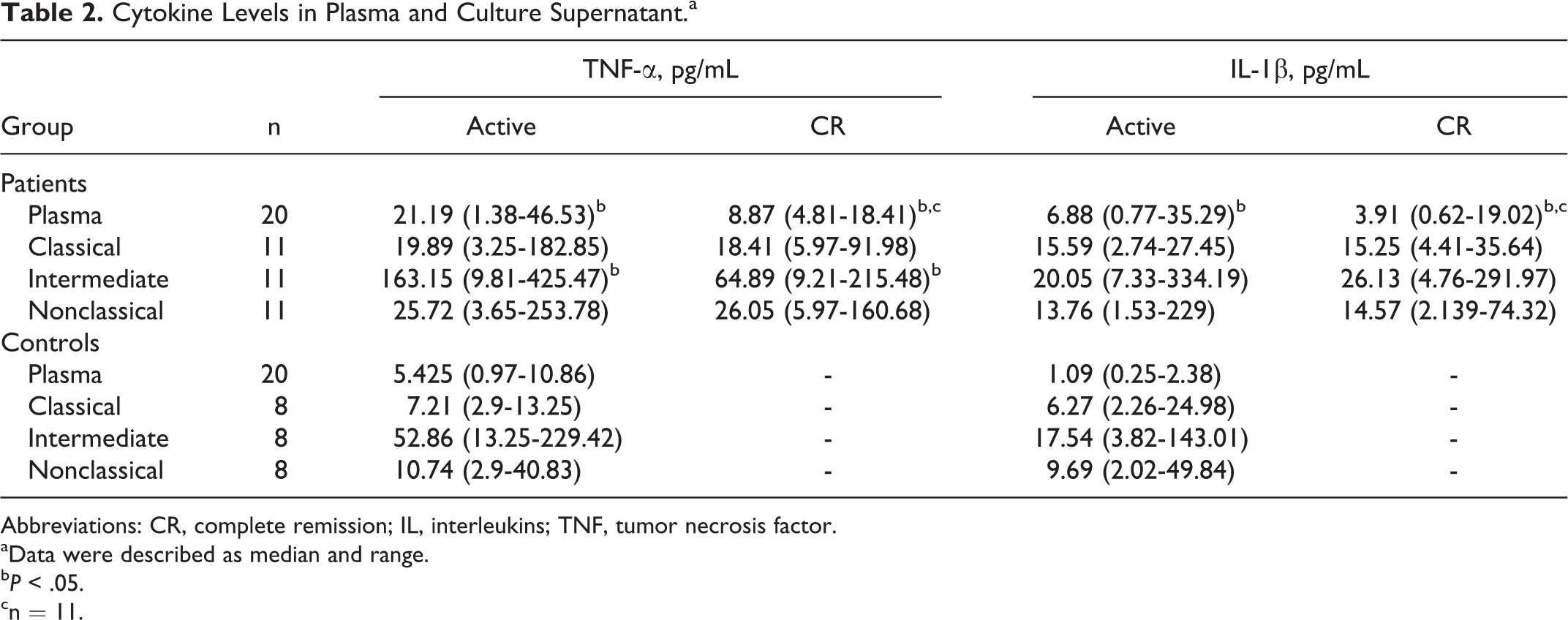
Abbreviations: CR, complete remission; IL, interleukins; TNF, tumor necrosis factor.
aData were described as median and range.
b P < .05.
cn = 11.
Effect of Monocyte Subsets on Platelet-Autoreactive T-Cell Proliferation and Th1 Polarization In Vitro
As shown in Figure 4A, the nonclassical subset presented the highest capability of promoting proliferation of platelet-autoreactive T cells (P = .037). To illustrate the effect of monocyte subsets on Th1 polarization, we determined the IFN-γ levels from CD4+ T cells. As shown in Figure 4B, the nonclassical subset significantly promoted secretion of IFN-γ among the 3 subsets.
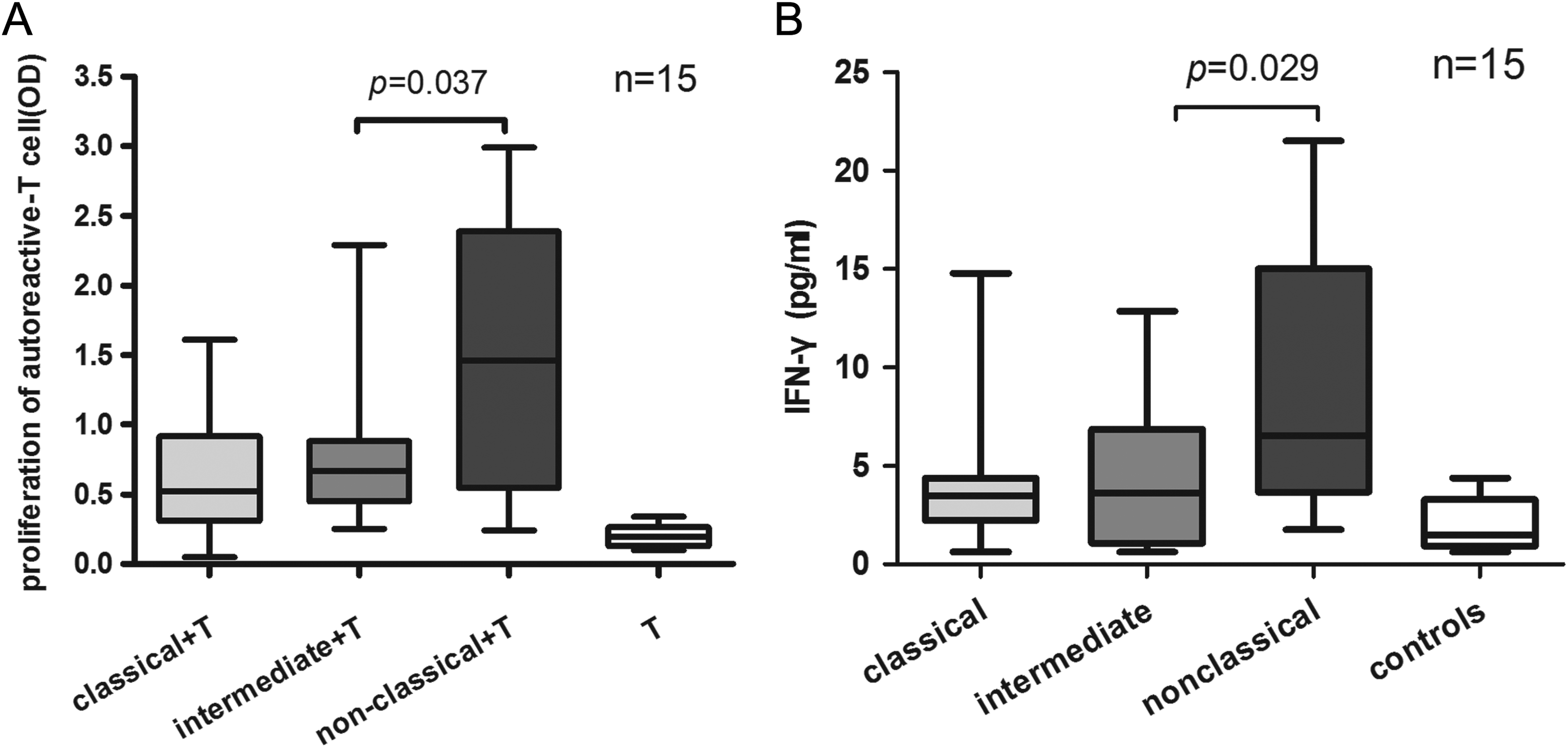
Nonclassical monocytes promote the proliferation of platelet-autoreactive T cells and Th1 polarization. A, Each purified monocyte subset of 11 patients was cocultured with autologous CD4+ T cells and platelets for 72 hours, and then the T-cell proliferation was determined by BrdU ELISA. The CD4+ T cells cocultured with the nonclassical monocyte subset showed the highest capacity of proliferation. B, After 72 hours, the IFN-γ levels of the coculture supernatant were measured by ELISA. The IFN-γ levels in the supernatant of the nonclassical monocyte subset group were higher than those in the other 2 subset groups. ELISA indicates enzyme-linked immunosorbent assay; IFN, interferon.
Discussion
The total CD16+ monocyte subset has been demonstrated to expand and promote Th1 development and negatively regulates IL-17 and Treg induction in patients with ITP. 27 Further studies showed that the CD16+ monocytes include intermediate and nonclassical subsets, and their functions in ITP remain to be defined. To the best of our knowledge, this is the first report describing that nonclassical monocyte were dramatically expanded and negatively correlated with platelet counts in patients with active ITP. In patients with CR, the nonclassical monocyte decreased correspondingly, which suggests that this subset may be a biomarker for active patients and may be involved in the pathogenesis of ITP. In contrast, the intermediate subset remains stable in active and remission stage, despite its expansion compared with controls.
We detected the elevated plasma levels of monocyte cytokines including TNF-α and IL-1β in patients with active ITP compared with controls. Tumor necrosis factor α is an inflammatory cytokine elevated in several autoimmune diseases. Anti-TNF agents have been implicated in the treatment of autoimmune diseases such as RA. 31 Additionally, TNF blockers may be effective in the treatment of patients with ITP by increasing Treg proliferation. 32 Consistent with the results in RA reported by Cros et al 33 and Rossol et al, 26 we identified that the intermediate subset produces the highest level of TNF-α and IL-1β among the 3 subsets in patients with active ITP. After CR, the TNF-α secretion decreased rapidly, which suggests that the intermediate subset participates in the pathogenesis of ITP through an increased capability for cytokine secretion. However, it contradicts the data related to the preferential production of cytokines from the 3 subsets. Wong et al 21 showed that nonclassical monocytes produced the highest levels of TNF-α and IL-1β. We propose that this discrepancy may be a result of the different disease stages tested and the methods used to isolate the subsets.
To date, the research on monocyte subsets in patients with ITP has mainly focused on Treg and Th development. Zhong et al found that CD16+ monocytes inhibited the development of Treg and IFN-γ+ CD4+ responses mediated by Th1-polarizing cytokines secreted by direct cell contact between CD16+ monocytes and T cells. 27 However, this study did not investigate the effect of monocyte subsets on T-effector cells, especially the platelet-autoreactive T cells. It is evident that in patients with ITP, CD14-derived peripheral DCs showed an increased capability of presenting platelets to T lymphocytes. 7 Because the intermediate and nonclassical subsets expanded significantly in patients with ITP, we further investigated which subset primarily promotes the proliferation of platelet-autoreactive T cells. In our study, we found that the nonclassical monocyte promoted CD4+ T-cell proliferation when cultured with autologous platelets, suggesting that the expansion of this subset may be the critical step of the antiplatelet antibody production. It is evident that IFN-γ+ autoantigen-specific CD4+ T cell can help B cells in autoantibody production. 6 Blood monocytes were found to be recruited into lymph nodes in response to LPS and gram-negative bacteria and differentiated into DCs as active classical DCs. 10 Cros et al found that the CD14dim subset can promote autologous T-cell proliferation in a pathogen-induced manner. 33 It should be noted that this report was published before the introduction of the new nomenclature for monocyte subsets and that the CD14dim subset is equal to the nonclassical one. Many patients with ITP are susceptible to pathogen infection. We postulate that the nonclassical monocytes could be activated by pathogens and promote platelet-reactive T-cell proliferation in patients with ITP. In fact, there appears to be a developmental relationship between these 3 subsets (from classical by intermediates to nonclassical). 19 It has been reported that the intermediate subset is a transitional status between the classical and nonclassical monocyte subsets. Th1 polarization presents important roles in the occurrence of ITP, and we found that the nonclassical subset plays a major role in promoting Th1 polarization. Previous studies have demonstrated that the CD16+ monocytes increased Th1 expansion via IL-12 secretion. 27 Further studies should investigate whether the nonclassical monocyte subset provides the major resources of IL-12.
In conclusion, we found that the nonclassical and intermediate monocyte subsets were both expanded and play different roles in the pathogenesis of ITP. The nonclassical monocyte subset is responsible for the stimulation of platelet-reactive T cells, and the intermediate one is responsible for the secretion of inflammatory cytokines. Further studies should focus on interfering with the expansion and adjusting the function of the 2 CD16+ subsets, which might be a new strategy for the treatment of ITP.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants of National Natural Science Foundation of China (81300385 and 81470286), Tianjin Municipal Science and Technology Commission (14JCZDJC35100).
