Abstract
Local anesthetics have been reported to have anticoagulant properties, but the mechanisms responsible for this action are poorly understood. Here, we evaluated the in vitro effects of 3 local anesthetics—lidocaine, ropivacaine, and bupivacaine—on the tissue factor expression by monocytes. Monocytes from peripheral blood were stimulated with lipopolysaccharide (LPS) in the presence or absence of local anesthetics. All 3 local anesthetics inhibited the expression of tissue factor antigen and tissue factor activity in LPS-stimulated monocytes in a dose- and time-dependent manner and reduced tissue factor messenger RNA (mRNA) expression in endothelial cells and a monocytic cell line. None of the 3 drugs induced apoptosis or affected the viability of monocytes. Our findings that local anesthetics inhibited the tissue factor induction in activated monocytes by inhibiting tissue factor mRNA level may demonstrate the feasibility of using local anesthetics in hypercoagulable and inflammatory conditions.
Introduction
Major surgery can induce a hypercoagulable and proinflammatory state,1,2 which predominantly reflects a stress response to surgical intervention. 3 It has been suggested that regional anesthetics and analgesics might be powerful tools for reducing perioperative stress, 4 with several studies5,6 reporting that these agents reduce the incidences of thromboembolic complications and decrease perioperative coagulability. Direct evidence of the anticoagulant properties of local anesthetics has been reported. For instance, amide local anesthetics were found to inhibit platelet function 7 and lidocaine significantly reduced coagulation time as measured by thromboelastography.8,9 However, the mechanism responsible for the anticoagulant effect of lidocaine has not been extensively studied.
Tissue factor (TF) is a receptor of activated factor VII. Factor VII binds to TF and they form a complex that initiates the blood-clotting cascade. Tissue factor is abundantly expressed in cell types that surround the vasculature, such as adventitial cells, where it is mainly found on the cell surface, but it is usually absent in blood cells that normally circulate in the vasculature. In intravascular cells, including monocytes and endothelial cells, TF expression can be transiently induced by inflammatory stimuli 10 including lipopolysaccharide (LPS) and inflammatory cytokines, such as tumor necrosis factor α (TNF-α), interleukin 1 (IL-1), and IL-6. 5 Because TF is responsible for triggering the cloting cascade in a variety of thrombotic disorders,11,12 an effective strategy for inhibiting TF expression in monocytes and endothelial cells would likely have significant therapeutic potential against hypercoagulable conditions, including those that occur during surgery and in the perioperative period.
In this study, we evaluated the in vitro effects of 3 local anesthetics—lidocaine, ropivacaine, and bupivacaine—on the TF expression in primary monocytes and a monocytic cell line and in endothelial cells. Our results demonstrate that all 3 local anesthetics inhibited the expression of TF antigen and activity in activated monocytes through inhibition of TF messenger RNA (mRNA) synthesis.
Materials and Methods
Cell Culture
Peripheral blood samples were collected from healthy volunteers into tubes containing ethylenediamine tetra-acetic acid and centrifuged over a Ficoll-Paque (1.077 g/mL; Pharmacia Biotech, Uppsala, Sweden) density gradient to obtain peripheral blood mononuclear cells (PBMCs). Peripheral blood mononuclear cells (2 × 106 cells/mL) were incubated in RPMI (Roswell Park Memorial Institute medium) 1640 medium containing 10% heat-inactivated fetal bovine serum (FBS; Invitrogen, Grand Island, New York). Monocytes were purified from PBMCs using CD14 microbeads according to the manufacturer’s instructions (Miltenyi Biotec Inc, Auburn, California, USA) and incubated in RPMI 1640 medium containing 5% heat-inactivated FBS. The THP-1 human monocytic cell line (ATCC) was maintained as a suspension in RPMI 1640 medium supplemented with 10% heat-inactivated FBS. This study was approved by the Institutional Review Board, and all patients provided written informed consent.
Human umbilical vein endothelial cells (HUVECs) were routinely maintained in endothelial cell basal media supplemented with a set of EGM-2 SingleQuots; HUVECs and associated culture reagent were obtained from Clonetics (San Diego, California). Human umbilical vein endothelial cells were grown to 90% confluence in a complete medium, and then the medium was replaced with a starvation medium (EBM (Endothelial Cell Basal Medium) supplemented with 1% FBS, heparin, and ascorbic acid) for 2 hours. The cells were incubated with or without local anesthetics and then stimulated with TNF-α (Sigma Aldrich, St Louis, Missouri) for 5 hours at 37°C.
Flow Cytometric Analysis
Peripheral blood mononuclear cells were stained with a phycoerythrin (PE)-conjugated monoclonal antibody (mAb) directed against TF (BD Biosciences, San Jose, California). Isotype-matched PE-conjugated immunoglobulin served as a control. Apoptotic and dead cells were determined quantitatively using PE-conjugated annexin V (BD Biosciences) and 7-amino-actinomycin D (7-AAD) viability dye (Beckman Coulter, Fullerton, California), respectively. Cell suspensions were analyzed using a FACS (Fluorescence-Activated Cell Sorting) Calibur cytofluorometer (BD Biosciences). Data were collected from at least 5 × 103 viable cells and analyzed with WinMDI 2.7 software. The monocyte population of PBMCs was separated from lymphocytes by gating based on their larger size (higher forward scatter) and more complex internal structure (higher side scatter; Figure 1A). Using an anti-CD14 antibody (Beckman Coulter), >99% of the gated monocyte population was confirmed to express surface CD14.
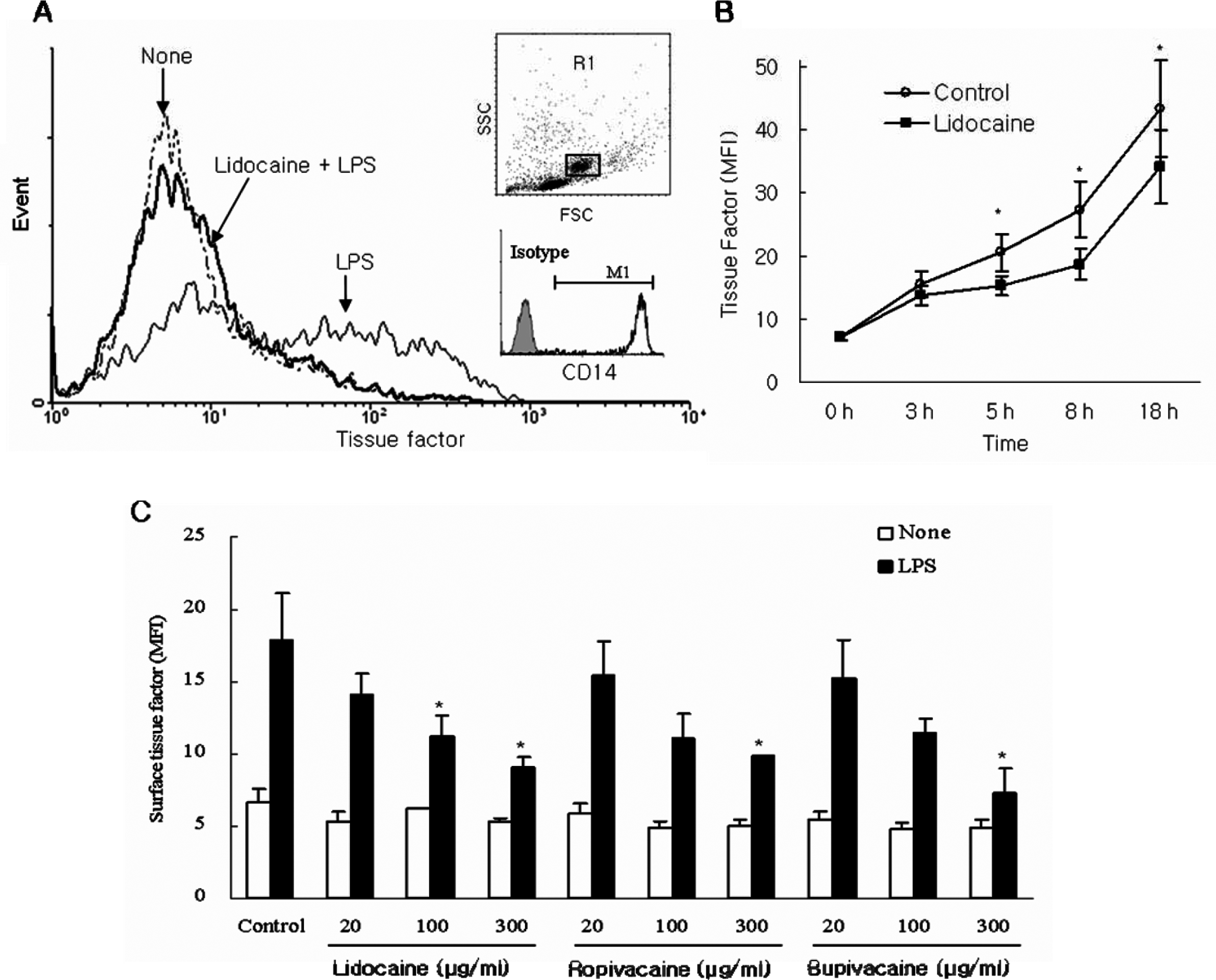
Effect of local anesthetics on surface expression of tissue factor antigen in LPS-stimulated peripheral monocytes A. Peripheral blood mononuclear cells (PBMCs) were pretreated for 1 hour with 100 µg/mL lidocaine. After a 5-hour incubation with 20 ng/mL lipopolysaccharide (LPS), the surface expression of tissue factor on monocytes (CD14-positive population) was determined by flow cytometry. The gated monocyte population (R1) was confirmed to be >99% positive for surface CD14 antigen. The histogram shows surface tissue factor expression on monocytes with LPS (solid line), with lidocaine and LPS (bold line), and without LPS and lidocaine (dotted line). B. Time-dependent inhibition of tissue factor expression in LPS-stimulated monocytes by lidocaine. C. Dose-dependent inhibition of tissue factor antigen expression in LPS-stimulated monocytes by lidocaine, ropibacaine, and bupivacaine. The results are expressed as the mean fluorescence intensity (MFI) of tissue factor (mean ± SEM of 4 separate experiments). *P < .05 vs LPS-stimulated control.
Measurement of Surface TF Activity
Tissue factor activity was determined using a commercial chromogenic assay kit (Actichrome TF, American Diagnostica, Standford, Connecticut). Peripheral blood mononuclear cells were resuspended in Tris-buffered saline (50 mmol/L Tris, 100 mmol/L NaCl, pH 7.4). One hundred microliters of the cell suspension (4 × 105 cells) was incubated with human factor VIIa and factor X for 15 min at 37°C. The amount of factor Xa generated is measured by a specific chromogenic substrate, Spectrozyme Xa. Tissue factor activity was calculated by comparing the measured absorbance with a standard curve established using serial dilutions of commercial TF standard.
Quantitative Reverse Transcription–Polymerase Chain Reaction Analysis
Total RNA was extracted from PBMC using TRIzol reagent according to manufacturer’s instructions (Life Technologies, Gaithersburg, Maryland). Complementary DNA (cDNA) was synthesized from 1 μg total RNA using TaqMan reverse transcription reagents (Applied Biosystems, Branchburgh, New Jersey). Real-time PCR amplification was used for quantitative analysis of gene expression, using the ABI Prism 7000 Sequence Detection system (Applied Biosystems, Foster City, California), TaqMan Universal PCR Master Mix, TF-specific primer and FAM-labeled probe sets (Applied Biosystems). The expression level of the target genes was normalized to that of an internal glyceraldehyde 3-phosphate dehydrogenase (GAPDH) primer/probe pair (VIC MGB probe, primer limited) and presented as relative expression.
Statistical Analysis
Statistical analyses were carried out using the SPSS package. All data are presented as means ± standard error (SE). Comparisons between 2 groups were done using t tests. Values of P < .05 were considered significant.
Result
Effect of Local Anesthetics on the Surface Expression of TF Antigen in Primary Monocytes, a Monocytic Cell Line and in Endothelial Cells
Peripheral blood mononuclear cells incubated at 37°C for 1 hour with or without different concentrations of lidocaine, ropivacaine, or bupivacaine were stimulated with LPS (20 ng/mL) for 5 hours and then assayed for surface expression of TF on monocytes by flow cytometry. Stimulation with LPS, which is known to promote TF expression, induced a progressive increase in the surface expression of TF in monocytes that reached a maximum of approximately 3-fold that of unstimulated monocytes. Pretreatment with lidocaine significantly inhibited LPS-induced surface expression of TF (Figure 1A). In time course experiments, this inhibitory effect of lidocaine (100 μg/mL) was first evident after a 3-hour incubation and continued for 18 hours (Figure 1B). Ropivacaine and bupivacaine also substantially inhibited TF expression on LPS-stimulated monocytes and, like lidocaine, did so in a dose-dependent manner (Figure 1C). None of the 3 local anesthetics had any effect in basal TF expression in unstimulated monocytes. We also evaluated the inhibitory effect of local anesthetics on TF expression in the THP-1 monocytic cell line. In contrast to the results with primary monocytes, lidocaine did not significantly inhibit TF expression in LPS-stimulated THP-1 cells. However, at a high concentration (300 μg/mL) pretreatment with ropivacaine or bupivacaine led to a significant inhibition of TF expression (data not shown). In endothelial cells, the 3 local anesthetics showed a tendency to attenuated TNF-α-stimulated TF expression although this difference did not reach statistical significance (Figure 2).
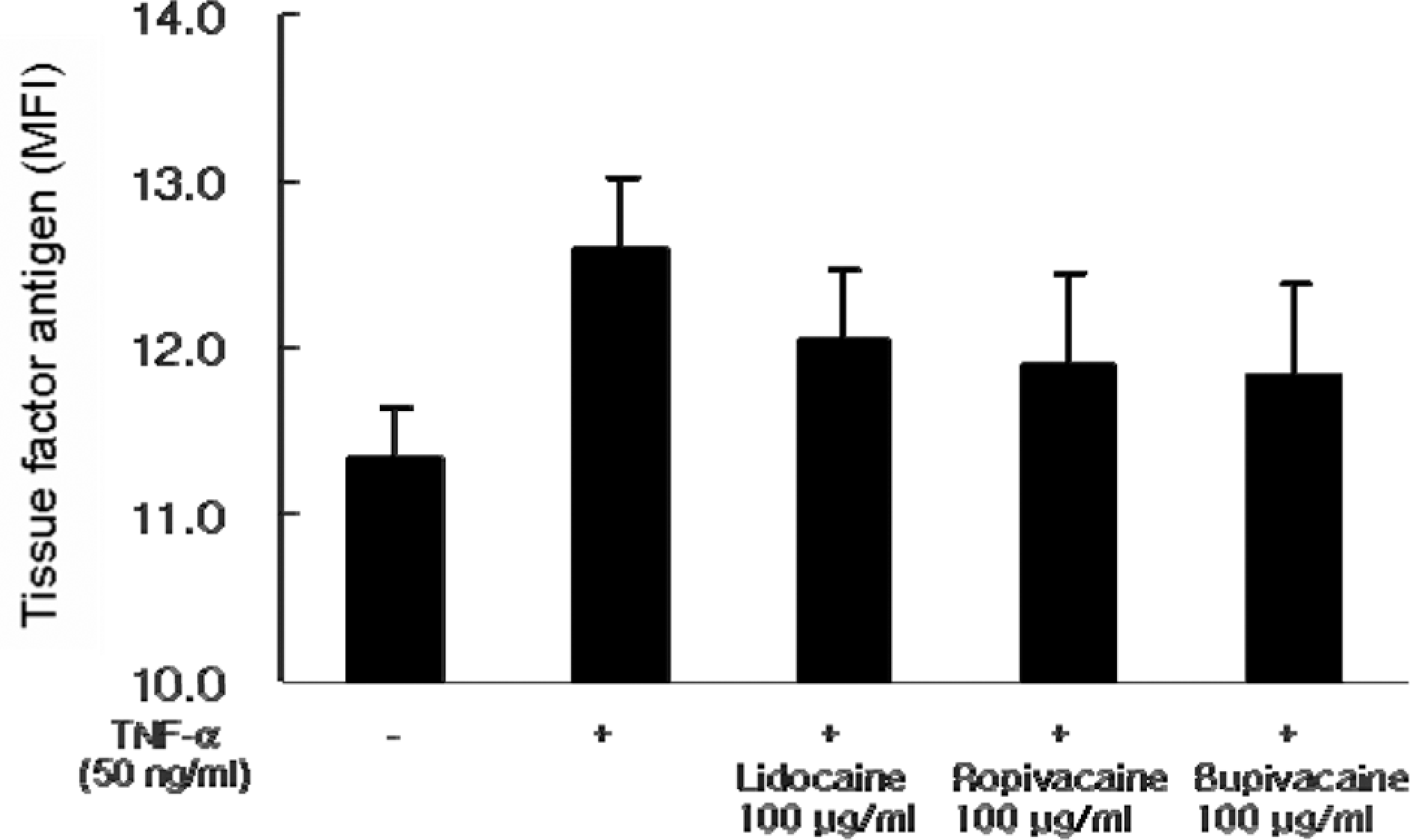
Effect of local anesthetics on surface expression of tissue factor in tumor necrosis factor α (TNF-)-stimulated endothelial cells human umbilical vein endothelial cells (HUVECs) were pretreated for 1 hour with 100 µg/mL lidocaine, ropivacaine, or bupivacaine. After a 5-hour incubation with 50 ng/mL TNF-α, the surface expression of tissue factor on HUVEC was determined by flow cytometry. The results, depicting the inhibition of tissue factor antigen expression on TNF-a-stimulated HUVECs by lidocaine, ropivacaine, or bupivacaine, are expressed as the mean fluorescence intensity (MFI) of tissue factor (mean ± SEM of 3 separate experiments).
Effect of Local Anesthetics on the TF Activity in Monocytes
Using a chromogenic assay to measure TF activity, we analyzed the effect of local anesthetics on TF function. Lipopolysaccharide induced a marked increase in monocyte TF activity that was attenuated by pretreatment with each of the 3 local anesthetics (Figure 3).
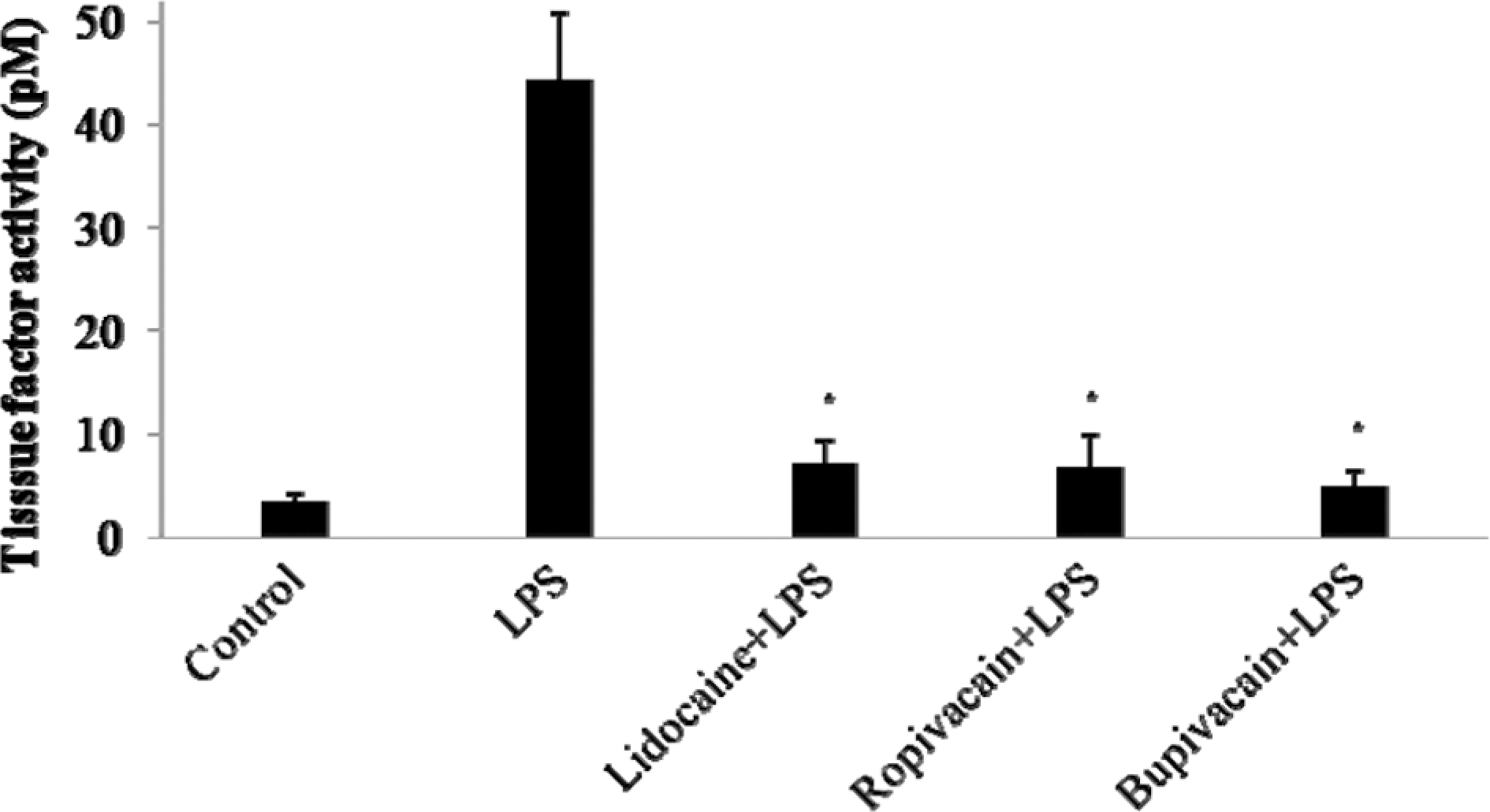
Effect of local anesthetics on tissue factor activity in peripheral monocytes stimulated by lipopolysaccharide (LPS). Peripheral blood mononuclear cells (PBMC) were pretreated for 1 hour with 300 mg/mL lidocaine, ropovacaine and bupvacaine. After 5 hours incubation with LPS (20 ng/mL), the tissue factor activity was measured with chromogenic assay. The tissue factor activity was calculated by comparing with standard curve. The results are expressed as the mean activity of tissue factor ± SEM of 3 separate experiments. *P < .05 vs LPS-stimulated control.
Effect of Local Anesthetics on TF mRNA Levels of Monocytes
To determine the effect of local anesthetics on the steady state level of TF mRNA, we measured TF mRNA levels using real-time RT-PCR. As shown in Figure 4, LPS stimulation induced approximately a 10-fold increase in TF mRNA in monocytes. Pretreatment with lidocaine, ropivacaine, or bupivacaine reduced LPS-induced increases in TF mRNA by approximately 56%, 40.4%, and 70.2%, respectively.
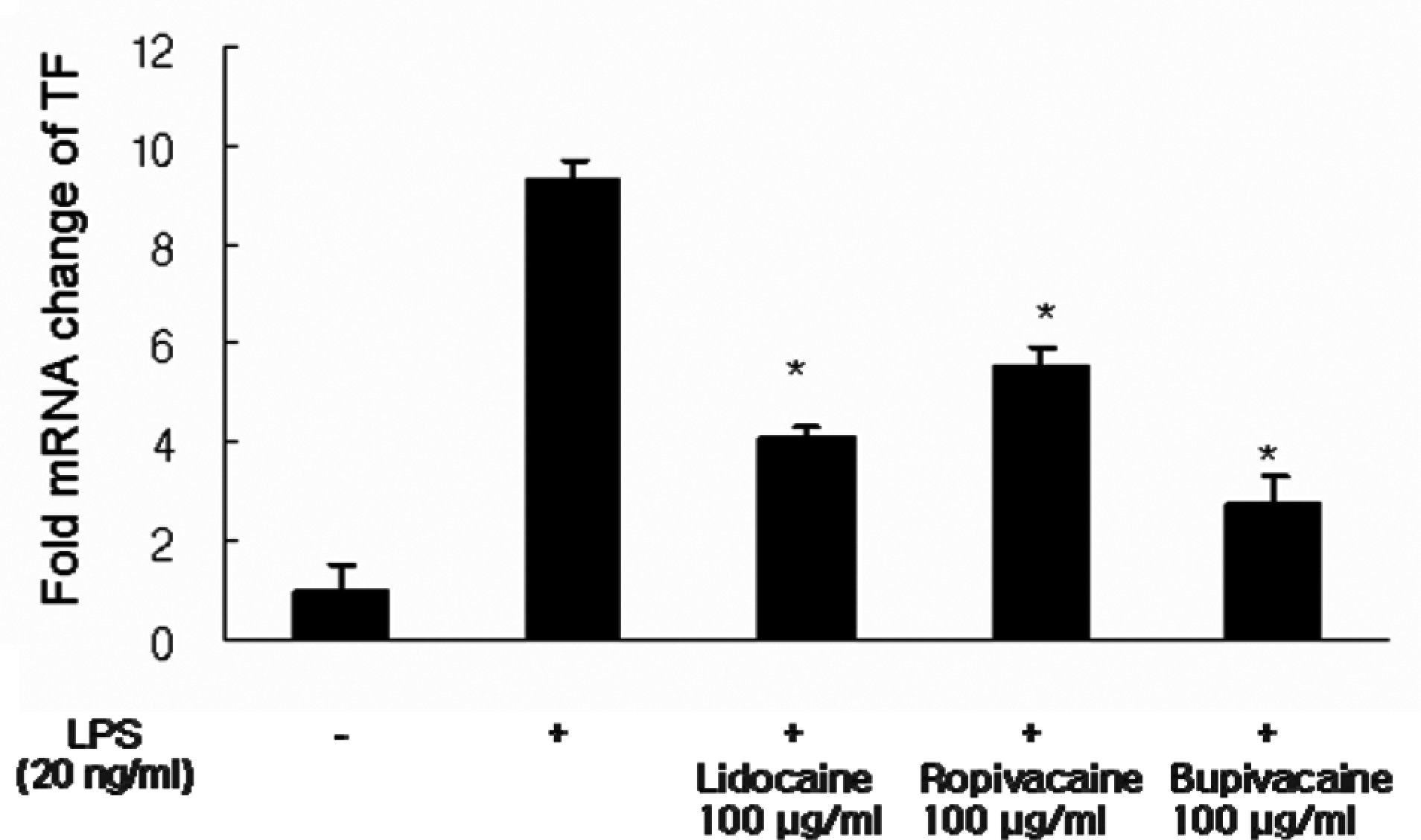
Effect of lidocaine on tissue factor (TF) messenger RNA (mRNA) in LPS-stimulated peripheral monocytes. Peripheral blood mononuclear cells (PBMC) were preincubated with and without 100 mg/mL lidocaine, ropivacaine, or bupivacaine and then treated with 20 ng/mL lipopolysaccharide (LPS) for 2 hours. Tissue factor (TF) mRNA levels were measured by real-time RT-PCR analysis and the results are expressed as the mean fold (± SD) mRNA change relative to the glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA (control) levels. *P < .05 vs. LPS-stimulated controls.
Effect of Local Anesthetics on Apoptosis and Viability in Peripheral Monocytes
Because the inhibitory effect of these local anesthetics on TF expression could result from drug-mediated cytotoxicity, we measured apoptosis and viability using annexin V and 7-AAD, respectively. None of the 3 drugs had a significant effect on apoptosis specifically (Figure 5A) or cellular viability generally (Figure 5B). Even the treatment of both lidocaine (100 μg/mL) and LPS (20 ng/mL) on monocytes did not significantly induce apoptosis. These results indicate that lidocaine, ropivacaine, and bupivacaine reduce TF production through a mechanism that is independent of cytotoxic effect.
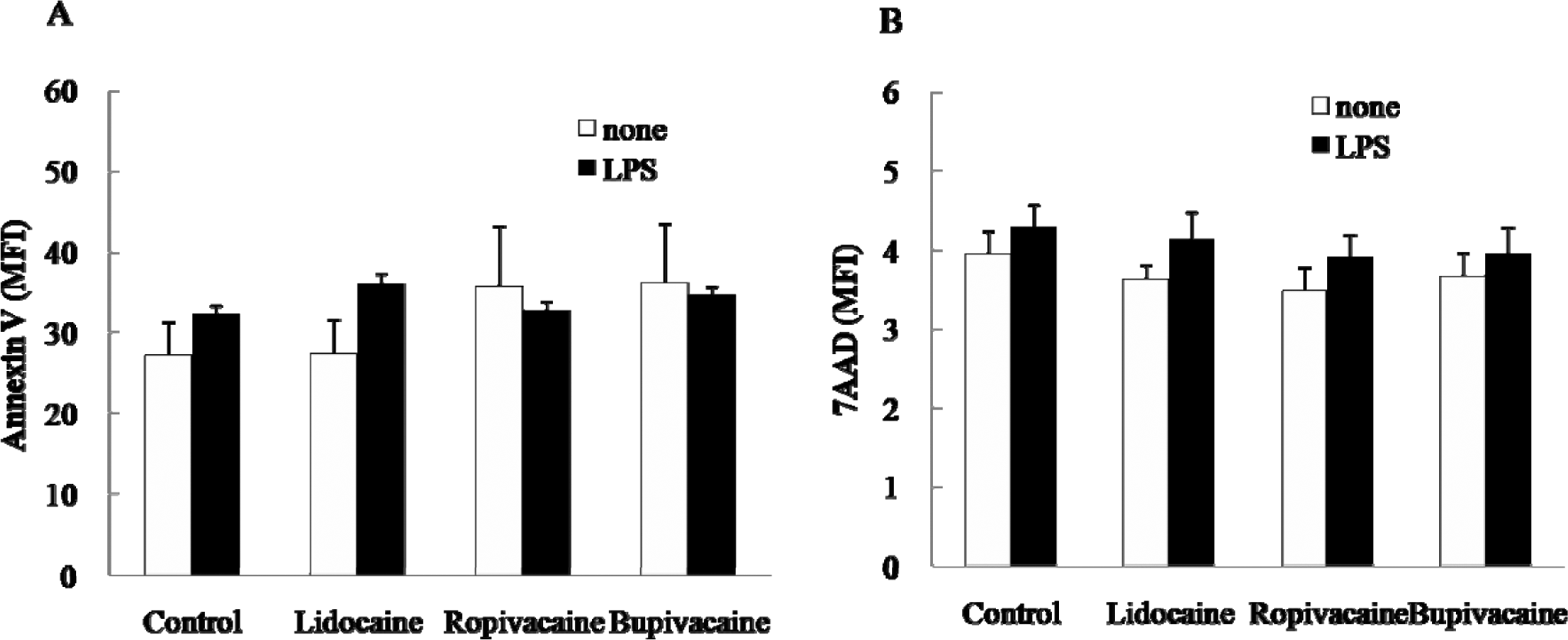
Effect of local anesthetics on apoptosis in peripheral monocytes. Peripheral blood mononuclear cells (PBMC) were preincubated at 37°C with lidocaine, ropivacaine, and bupivacaine at concentration of 100 mg/mL and stimulated with 20 ng/mL lipopolysaccharide (LPS) or vehicle. Apoptotic and dead cells were determined quantitatively by (A) annexin V-PE and (B) 7-amino-actinomycin D (7AAD) viability dye, respectively. The results are expressed as the mean fluorescence intensity (MFI) of the annexin V and 7AAD (±SEM) of 3 separate experiments.
Discussion
Local anesthetics are widely used in various clinical anesthesia and analgesia applications. In addition to their anesthetic actions, these agents have been hypothesized to possess anticoagulant properties Nielsen et al. 13 suggested that epidural anesthesia and analgesia decrease platelet aggregation through a reduction in the stress response. Moreover, Cooke et al 14 documented direct effects of local anesthetics on thrombosis reporting that intravenous (IV) lidocaine prevented deep venous thrombosis after elective hip surgery. However, it was not clear which molecular component was involved in the anticoagulant activity of local anesthetics.
We hypothesized that TF was the target molecule through which local anesthetics exerted their anticoagulant activity. As predicted, local anesthetics effectively downregulated TF expression in monocytes and endothelial cells. This downregulation of TF was also manifested at the mRNA level. Moreover, concentrations of local anesthetics that showed an anticoagulant effect were not associated with significant cell death. To our knowledge, this is the first report of an effect of local anesthetics on TF expression.
Surface expression of TF is an important effector function of activated monocytes in inflammation. Monocytes isolated from the peripheral blood of healthy individuals have undetectable levels of TF, but express TF strongly after stimulation with LPS or other inflammatory mediators. 15 Vascular endothelial cells can be induced to express TF robustly in response to inflammatory stimuli and TF is expressed in vivo by monocytes and endothelial cells during sepsis.16,17 Tissue factor has also been found in a number of thrombotic locations, such as atheromas and tumor cells. 18 Finally, this induced expression of TF promotes a more generalized activation of the clotting system. Therefore, attention has turned toward reducing TF expression as a means to attenuate sepsis and prevent thrombosis. Many studies have attempted to inhibit TF expression in monocytes, focusing in the possible use of anti-inflammatory cytokines (IL-4, IL-10, and IL-13), certain prostanoids, pentoxifyllin, retinoic acid, and antisense olidonucleotides. 19
Recent studies have reported an anti-inflammatory effect of lidocaine through inhibition of Nuclear factor of kappa B (NF-κB) activation.20,21 Lidocaine has been shown to inhibit the expression of the proinflammatory mediators, IL-2 and TNF-α in T cells, 22 supporting the idea that lidocaine is a potential anti-inflammatory agent. Inflammatory stimuli can induce a hypercoagulable state by inducing TF in monocytes and endothelial cells. Thus, because they exert both anti-inflammatory and anti-coagulant activity, local anesthetics may be good therapeutic candidates against inflammatory hypercoagulability.
We demonstrated that local anesthetics, lidocaine, ropivacaine, and bupivacaine, have the ability to reduced TF expression by activated monocytes. The clinical application of local anesthetics as agents for anesthesia and analgesia could be beneficial in a range of inflammatory and hypercoagulable conditions such as pregnancy and gynecology, sepsis, and trauma. In addition to its role in inflammation and initiation of coagulation, TF has recently been shown to participate in tumor growth and angiogenesis. 22 Thus, the ability of local anesthetics to reduce TF expression might be applicable in patients with cancer. In future, the design of appropriate formulations using these local anesthetics could make efficient therapeutic approaches possible.
The concentration of local anesthetics used to inhibit TF expression was quite high (˜100 μg/mL). Because lidocaine concentrations are reportedly much lower than these levels after epidural infusion (2.0 ± 0.4 μg/mL) and IV administration (1.9 ± 0.8 μg/mL) in humans, the usual clinical dosage used for anesthesia may not be sufficient to achieve an anticoagulant effect. The high concentrations used in our study can be used as an anticoagulant, because they were not associated with significant cell death. The clinical application of local anesthetics in this context requires further exploration.
In conclusion, the local anesthetics, lidocaine, ropivacaine, and bupivacaine, reduced the expression of TF antigen and activity in activated monocytes by inhibiting TF mRNA synthesis. A more refined understanding of the anticoagulant properties of local anesthetics may ultimately prove the feasibility of using local anesthetics in hypercoagulable and proinflammatory states.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Grant of the Korea Healthcare Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A080720), and a grant from the SNUH Research Fund (04-2007-040).
