Abstract
This study aimed to investigate the long-term safety and benefits of antiplatelet therapy in patients with cerebral infarction with thrombocytopenia, as evidence regarding this was limited. This cohort trial assessed patients with acute cerebral infarction with thrombocytopenia treated in the Neurology Department of Shanghai Tenth People’s Hospital from January 2016 to December 2018, and enrolled patients were followed up for 9 months. The patients were divided into non-antiplatelet and antiplatelet groups based on the actual intake of antiplatelet drugs. Primary endpoints included hemorrhagic events, recurrence of cerebral infarction, and activity of daily living (ADL) score changes. To balance baseline clinical data, propensity score matching was applied, and there were finally 65 matched patients, including 30 and 35 in the antiplatelet and non-antiplatelet groups, respectively. There were no differences in hemorrhagic and cerebral infarction recurrence rates between the 2 groups. ADL score change was higher in the antiplatelet group than in the non-antiplatelet group (10 vs 5, p = 0.039). In multivariate regression analysis, antiplatelet therapy significantly predicted a positive change in ADL scores [B = 8.381, 95% confidence interval (0.56-16.19)]. In patients with acute cerebral infarction with thrombocytopenia, antiplatelet therapy could the improve the quality of life in the chronic stage.
Introduction
It is estimated that a great deal of deaths occurring globally are attributable to stroke, which is considered the second leading cause of death; 70-80% of stroke patient cases are ischemic infarction cases. 1 This imposes a heavy economic burden on society and families due to its association with high morbidity, mortality, and disability. 2 With population aging, poor diet, irregular exercise, and other factors, the incidence of cerebral infarction in patients has sharply increased, with this pathology becoming one of the major neurological diseases worldwide. Antiplatelet therapy is the cornerstone of cerebral infarction therapy. Oral aspirin or P2Y12 receptor inhibitor (clopidogrel) is currently the most widely used antiplatelet therapy and is highly recommended by multiple guidelines. 3 However, weighing the pros and cons of this therapy and reducing the potential cerebrovascular risks while avoiding adverse hemorrhagic events is a common problem in clinical practice, 4 especially in individuals with thrombocytopenia. Antiplatelet therapy increases the risk of hemorrhagic events while preventing the recurrence of stroke. 5 There is little evidence on the safety and benefits of this treatment in patients with cerebral infarction with thrombocytopenia. 6 According to large stroke clinical trials, aspirin significantly reduces mortality and disability in patients with cerebral infarction as well as disease recurrence, but slightly increases the risk of symptomatic intracranial hemorrhage. 7 –9 A recent study retrospectively analyzed 6 patients with cerebral infarction with thrombocytopenia, and proposed that antiplatelet therapy should be administered to patients with platelet counts above 50 × 109/L, but its sample size was too small and solid evidence was not provided. 6 We collected data for almost 3 years in our hospital and retrospectively analyzed the long-term safety and benefits of antiplatelet therapy in patients with cerebral infarction with thrombocytopenia.
Materials and Methods
Patients and Ethnics
Patients with acute cerebral infarction with thrombocytopenia hospitalized in the Department of Neurology, Shanghai Tenth People’s Hospital, from January 2016 to December 2018 were enrolled. The diagnosis of cerebral infarction was confirmed by magnetic resonance imaging (MRI)/head computed tomography (CT). The diagnosis of thrombocytopenia was reflected by a platelet count < 100 × 109/L, 10 thrombocytopenia is a common clinical problem defined as a platelet count < 150 × 109/L, although many feel that a cutoff value of 100 × 109/L is more appropriate to identify clinically significant thrombocytopenia. Individuals with a history of traumatic bleeding, active ulcers, and other contraindications to antiplatelet therapy were excluded. This study was approved by the local ethics committee of Shanghai Tenth People’s Hospital (SHSY-IEC-4.1/19-199/01).
Study Design
Patients with acute cerebral infarction and thrombocytopenia were divided into non-antiplatelet and antiplatelet groups according to whether they were treated with antiplatelet drugs, and the patients were followed up for 9 months. The obtained baseline data included gender, age, risk factors of cerebrovascular disease (smoking history, drinking history, hypertension, diabetes, atrial fibrillation, and coronary heart disease), admission National Institutes of Health Stroke Scale (NIHSS) score, cerebral arterial stenosis degree, and laboratory test results (blood glucose, blood lipids, glycated hemoglobin, homocysteine, etc.). The primary end points included hemorrhagic events, recurrence of cerebral infarction, and changes in activity of daily living (ADL) scores.
Antiplatelet drug dosage
According to the guidelines for the early management of patients with stroke, in this study, patients in the antiplatelet groups took aspirin 100 mg QID or clopidogrel 75 mg QID. 3
Activity of daily living
The functional capacity of patients was measured using the Barthel Index, which was applied to assess the participants’ current level of ability to perform the following 10 items: feeding, bathing, grooming, dressing, bowel control, bladder control, toileting, transferring in and out of bed to chair and back, mobility on level surface, and mobility on stairs. 11
Bleeding Academic Research Consortium (BARC) definition for bleeding
The BARC definition contains the 6 types of bleeding that were hierarchically defined from type 0 in which there was no bleeding to type 5 in which there was fatal bleeding. 12
Data Analysis
SPSS 20.0 (IBM, Armonk, NY, USA) was used for statistical analysis. The research subjects were not randomly assigned, which could render the distribution of confounding factors in both groups uneven. Due to the potential bias and confounding variables between the 2 groups, propensity score matching (PSM) was performed to retrieve relatively balanced baseline data. PSM was applied to study the associations of antiplatelet therapy with outcomes to be interfered with by all other confounding factors. Normally distributed data were expressed as mean ± standard deviation and assessed by the t-test. Non-normally distributed data were expressed by median and quartile intervals and compared using a nonparametric test. Enumeration data were compared by the x 2 test and expressed as number and percentage. Multivariate regression analysis was performed to determine whether treatment had an impact on the end event. All statistical assessments were 2-tailed, and P < 0.05 was considered statistically significant.
Results
Baseline Data of Patients Administered Antiplatelet and Non-Antiplatelet Treatments
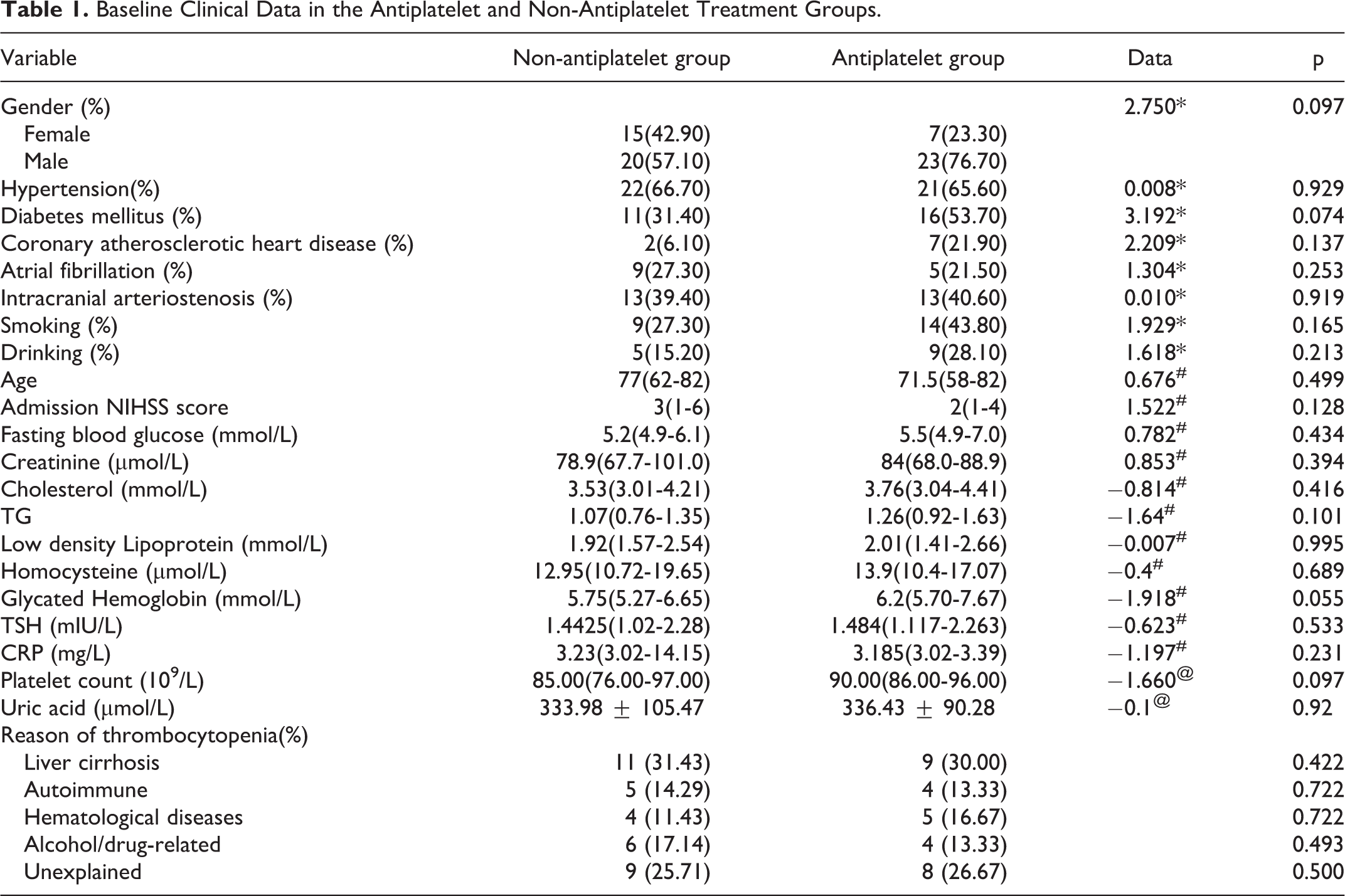
There were 2,198 patients with cerebral infarction in the Department of Neurology, Shanghai Tenth People’s Hospital, from January 2016 to December 2018, including 93 with thrombocytopenia. To match baseline data, PSM was performed. Finally, 65 patients were matched, based on the actual intake of antiplatelet drugs, including 35 in the non-antiplatelet group and 30 in the antiplatelet group, of which 15 were on aspirin, 9 were on clopidogrel, and 6 were on aspirin plus clopidogrel. Thus, there were no significant differences in baseline platelet count, discharge NIHSS score, smoking history, drinking history, hypertension history, diabetes history, atrial fibrillation history, intracranial artery stenosis or occlusion, uric acid, total cholesterol, triglycerides, low-density lipoprotein, thyrotropin, creatinine, fasting blood glucose, and homocysteine (Table 1).
Baseline Clinical Data in the Antiplatelet and Non-Antiplatelet Treatment Groups.
Outcome Indicators in the Antiplatelet and Non-Antiplatelet Groups
The whole hemorrhage rate, cerebral infarction recurrence rate, and quality of life improvement at 9 months of follow-up were compared between the non-antiplatelet and antiplatelet groups. In the non-antiplatelet group, there was one case of intracranial hemorrhage and 2 cases of skin/mucosal hemorrhage. In the antiplatelet group, there was one case of intracranial hemorrhage, 2 cases of skin/mucosal hemorrhage, and one case of gastrointestinal hemorrhage. According to the BARC criteria of bleeding non-antiplatelet group: 2 cases of type 1, 2 cases of type 2, 1 case of type 3c, and antiplatelet group: 1 case of type 1, 2 cases of type 2, and 1 case of type 3c. No differences were found in hemorrhage or infarction rates. Meanwhile, a slight difference in ADL score change was detected (Z = −2.06, p = 0.039; Table 2).
Outcome Indicators in the Antiplatelet and Non-Antiplatelet Groups.

Antiplatelet Therapy Predicts ADL Score Improvement
In multivariate regression analysis, antiplatelet therapy significantly predicted positive ADL score change after 9 months (p = 0.036, B = 8.381, 95% CI [0.56. 16.19]).
Discussion
In this study, we did not find a significant difference in the recurrence and hemorrhage events, which was inconsistent with other studies, while the ADL showed improvement. The ADL score is an assessment of the functional status of activities of daily living, and studies have shown that 1.85 point is the minimal clinically important difference. 13 The improvement in the ADL value may imply that antiplatelet drugs have a positive effect on the recovery of neuroplasticity. 14 A previous case series of idiopathic thrombocytopenia combined with acute cerebral infarction has yielded controversial results for hemorrhagic and infarction events 15 –18 due to small sample sizes and incomplete follow-up. 19 Other studies have also pointed out that decreased platelet count stimulates the increase of platelet particles, leading to stenosis of intracranial arteries; meanwhile, antiplatelet drugs may inhibit platelet microparticles. 20 –23 This may also be because we only used the consultation and physical examination during the 9-month follow-up, and did not examine the MRI/CT to clarify certain silent cerebral infarctions.
Aspirin mainly prevents the synthesis of TXA2 by inhibiting cyclooxygenase. Clopidogrel selectively and irreversibly prevents the binding of ADP to the platelet P2Y12 receptor, thereby suppressing platelet aggregation, 24 which also increases the risk of hemorrhage. 25,26 Anti-platelet therapy after cerebral infarction significantly reduces mortality and disability rates and decreases disease recurrence, but slightly increases the risk of symptomatic hemorrhage. 7,8,27 This study further proves that active antiplatelet therapy in stroke patients with mild to moderate thrombocytopenia could help patients improve their daily activities. Studies have shown that abnormal platelet counts are related to hospital mortality, but with no significant difference after adjusting for confounding factors. 28
There were limitations in this study. For instance, the patients in the cohort had mild to moderate thrombocytopenia only, and it is unknown whether this result can be generalized to cases of severe thrombocytopenia. The sample size was not large enough, and the PSM process automatically excluded some severe cases so that the baseline NIHSS scores of the enrolled patients were not very high. We hope that this retrospective cohort study can help eliminate an unfounded fear of hemorrhage associated with antiplatelet therapy use with thrombocytopenia 29
Conclusion
In our propensity score-matched analysis, we did not find a significant difference in hemorrhage or secondary cerebral infarction among patients with mild to moderate thrombocytopenia on antiplatelet therapy. However, higher ADL scores were seen in the antiplatelet therapy group, which supports the use of antiplatelet therapy in the antiplatelet group.
Footnotes
Authors’ Note
SG, XL, and GM conceived and designed the experiments; SG and XM performed the experiments; XM, YZ, YL, and AJ analyzed the data; SG and GM wrote the manuscript. This study was approved by the local ethics committee of Shanghai Tenth People’s Hospital (SHSY-IEC-4.1/19-199/01).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Shanghai Sailing Program (No. 19YF1438400), and the Shanghai Science and Technology Commission Project (No. 18411961700 and 19441908500).
