Abstract
After coronary drug-eluting stent (DES) implantation, dual antiplatelet therapy (DAPT, usually aspirin in combination with a P2Y12 inhibitor) is necessary. However, older patients are prone to aspirin intolerance or resistance. Indobufen has been reported as an ideal alternative to aspirin. To evaluate the clinical efficacy and safety of indobufen-based DAPT in this population, a total of 251 older patients (aged ≥65 years) undergoing coronary DES implantation in a tertiary healthcare facility in Quzhou, China, were enrolled, of which 110 received indobufen-based DAPT and 141 received aspirin-based DAPT. Baseline data, adverse reactions, adverse events, angina attacks, and repeated revascularization during 1-year follow-up were collected. At 1-year follow-up, creatinine, and left ventricular ejection fraction levels in the indobufen group were higher than those in the aspirin group, whereas estimated glomerular filtration rate levels were lower than those in the aspirin group. No significant difference in the incidence of adverse reactions or adverse events was found between the two groups. At 1-year follow-up, the incidence of angina in the indobufen group was significantly lower than that in the aspirin group, and the repeated revascularization rate was lower than that in the aspirin group. Indobufen-based DAPT was beneficial in avoiding repeated revascularizations (odds ratio 0.433, 95% CI 0.229-0.821, P = 0.010). These findings highlight that, for older patients who underwent coronary DES implantation, indobufen-based DAPT may be a better choice, which can effectively improve patients’ symptoms and prognosis. However, application to older patients with potential renal insufficiency requires caution.
Keywords
Introduction
Coronary artery disease (CAD) is one of the major chronic diseases worldwide, and older people are a high-risk group. Coronary atherosclerosis could cause abnormal coronary blood flow. Patients with severe coronary stenosis or occlusion require coronary revascularization. Compared with coronary artery bypass grafting, percutaneous coronary intervention (PCI) has the advantages of less trauma, lower intraoperative risks, faster postoperative recovery, etc After the introduction of this surgical method, the vascular opening and long-term survival rates of older patients with CAD were significantly improved.1,2 However, in patients with coronary stent implantation, mechanical injury to the endothelium would further activate coagulation cascades and platelets.3,4 Thus, to inhibit platelet activity, these patients should be administered with sufficient doses of preoperative antiplatelet drugs, and postoperative antiplatelet therapy with sufficient dosage and duration is needed to reduce the incidence of thrombotic and cardiovascular events.
Dual antiplatelet therapy (DAPT), which is usually a P2Y12 inhibitor combined with aspirin, is the clinical standard method for antiplatelet therapy after coronary drug-eluting stent (DES) implantation.5,6 The classic antiplatelet aggregation drug aspirin can effectively inhibit platelet aggregation and improve the prognosis of cardiovascular and cerebrovascular diseases, which has been widely recognized clinically. However, many studies have shown that the long-term use of aspirin can lead to serious gastrointestinal reactions, bleeding, hyperuricemia, asthma, etc, which is clinically known as aspirin intolerance. These manifestations may be more severe in the older population, and some patients must stop medications at long-term follow-up. 7 Therefore, safer and more effective antiplatelet drugs to combine with a P2Y12 inhibitor for DAPT are needed. 8
Indobufen is currently the only reversible antiplatelet drug that can reversibly inhibit the activity of platelet cyclooxygenase-1 (COX-1), thereby inhibiting platelet activation, and has anticoagulation, vasodilation, monocyte tissue factor inhibition, antifibrosis, and other effects.9,10 Some clinical trials about indobufen indicate that its biochemical function and clinical efficacy are comparable to those of standard-dose aspirin in conventional applications, with a shorter half-life and less inhibition of prostaglandins, resulting in lower gastrointestinal reactions and lower bleeding risk.11–13 Currently, for populations who cannot tolerate aspirin, indobufen as an alternative drug has already been approved by the Chinese Food and Drug Administration. Older patients have weaker gastrointestinal function and higher risks of bleeding and ischemia and are more prone to aspirin intolerance; however, no sufficient evidence-based medical assessment for indobufen-based DAPT is available in the older population. Therefore, this study aimed to investigate the efficacy and safety of indobufen combined with clopidogrel for DAPT in older patients after coronary DES implantation.
Materials and Methods
This prospective randomized single-center study was conducted among patients in a tertiary healthcare facility in Quzhou, China.
Participants and Randomization
Patients with CAD who were hospitalized in the department of cardiology between June 15, 2021, and March 31, 2023, were selected. The inclusion and exclusion criteria are shown in Table 1. This study was approved by the Ethics Committee of Quzhou People's Hospital and registered with the Chinese Clinical Trial Registration Center (URL: https://www.chictr.org.cn. Registration number: ChiCTR2100046967).
Key Inclusion and Exclusion Criteria.

According to the inclusion and exclusion criteria, 440 participants with CAD were initially screened. Participants were divided with the use of a Web-based randomization system in which participants were assigned, in a 1:1 ratio, to receive the indobufen-based DAPT or to received aspirin-based DAPT. There was no knowledge by the participants or the researchers about in which group the participants would be allocated at the randomization time. Considering the results of coronary angiography and compliance of follow-up, 110 participants in the indobufen group and 141 in the aspirin group were ultimately included (Figure 1).
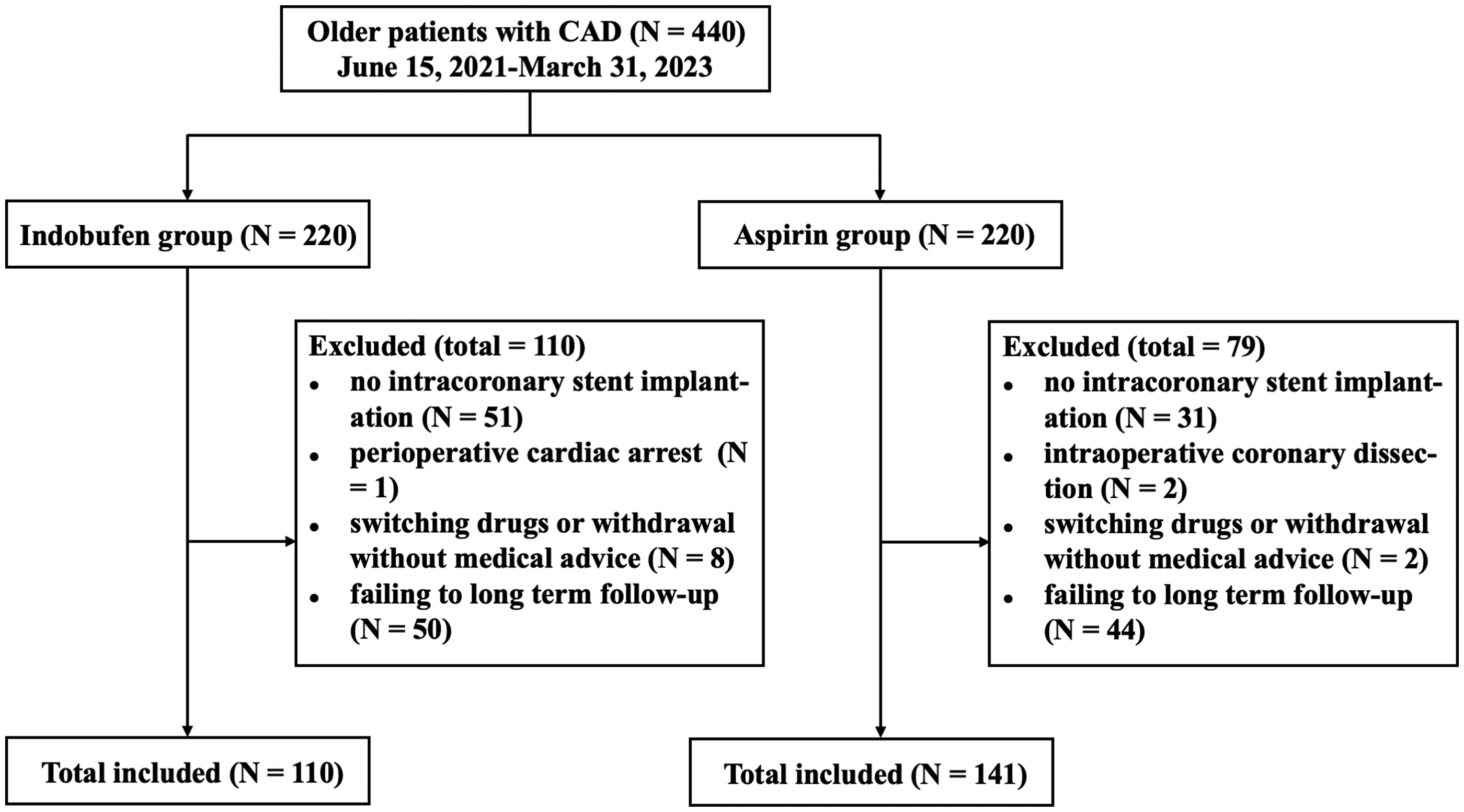
Interventions
Both groups received routine treatment, including dietary control, health education, etc Moreover, statin-based lipid-lowering, antihypertensive, hypoglycemic, anti-ventricular remodeling therapies, etc, should be given. Nitrates should be given if necessary to alleviate symptoms during angina attacks, and proton pump inhibitors (PPIs) should be administered to protect the gastrointestinal mucosa.
Indobufen group: Clopidogrel was administered at a cumulative load of 300 mg at least 6 h before coronary angiography. If the patient took a therapeutic dose of aspirin continuously for ≥3 days preoperatively, indobufen 100 mg twice a day would be continued before coronary angiography. If the patient took a therapeutic dose of aspirin continuously for <3 days preoperatively, 100 mg of indobufen will be given immediately after randomization. If the patient did not take aspirin preoperatively, 200 mg of indobufen was administered immediately after randomization. Within 24 h after coronary DES implantation, indobufen 100 mg twice a day and clopidogrel 75 mg/day were administered for 12 months.
Aspirin group: Clopidogrel was administered with a cumulative load of 300 mg at least 6 h before coronary angiography. If the patient took a therapeutic dose of aspirin continuously for ≥3 days preoperatively, aspirin would be continued by 100 mg/day before coronary angiography. If the patient took a therapeutic dose of aspirin continuously for <3 days preoperatively, 100–300 mg of aspirin would be administered immediately after randomization to achieve a cumulative load of 300 mg before coronary angiography. Within 24 h after coronary DES implantation, aspirin 100 mg/day and clopidogrel 75 mg/day were administered for 12 months.
Coronary angiography and PCI would be performed according to the standard techniques. Patients underwent DES implantation were treated with the everolimus-eluting Xience® stent (Abbott Vascular, Santa Clara, CA, USA), or the zotarolimus-eluting Resolute® stent (Medtronic Incorporation, Santa Rosa, CA, USA), or the everolimus-eluting Promus Premier® stent (Boston Scientific, Natick, Massachusetts).
Observation Indices
Clinical data such as age, sex, body mass index (BMI), systolic blood pressure, smoking history, drinking history, admission diagnosis, and history of comorbidities were collected. Plasma brain natriuretic peptide (BNP) and indices of coagulation function, routine blood, kidney function, inflammation, and color ultrasonography of the heart were measured before and 12 months after medical interventions. At 1-year follow-up, patients returned to the hospital for coronary angiography and would undergo PCI revascularization in the original target vessel or any other coronary vessels if necessary. Follow-up at 3, 6, and 12 months after surgery was performed to record drugs compliance by self-report (measured by the ratio of the number of tablets assumed to have been taken to the total number of tablets that should have been taken, with a maximum possible value of 1), adverse cardiovascular and cerebrovascular events, and adverse reactions, including but not limited to liver and kidney function damage, gastrointestinal response (acid reflux, nausea, gastrointestinal inflammation, gastroduodenal ulcer, etc), bleeding events and classifications according to Bleeding Academic Research Consortium (BARC) 14 (skin and mucous petechia, gingival bleeding, hemoptysis, gastrointestinal hemorrhage, cerebral hemorrhage, urinary hemorrhage, etc), recurrent angina attacks (evaluated by the Rose Angina Questionnaire), all-cause death, cardiovascular death, nonfatal myocardial infarction (MI), ischemic stroke, probable or confirmed stent thrombosis, etc.
Statistical Analysis
In this study, IBM SPSS Statistics for Windows version 24.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism 10.0 (GraphPad Software Inc., La Jolla, CA) were used for statistical analysis and mapping. Quantitative data were analyzed using the t-test or the Wilcoxon rank-sum test, whereas categorical data were analyzed using the chi-square test or the Fisher test. Lists were used to represent adverse reactions and adverse events, and the chi-square test was used to compare the incidence rates between the two groups. Statistical analysis of data before and 12 months after medicine interventions between the two groups was performed using repeated measures of analysis of variance followed by Post-hoc analysis with Bonferroni adjustment for multiple comparisons. Cumulative risk was evaluated by the Kaplan–Meier estimate, and efficacy was evaluated by the log-rank test. Spearman correlation and binary logistic regression analyses were performed to evaluate the risk factors. A two-sided P < 0.05 was considered significant for all tests.
Results
Comparison of Clinical Characteristics of the Indobufen and Aspirin Groups
As shown in Table 2, no significant differences in age, sex, systolic blood pressure, BMI, smoking history, drinking history, CAD classification, and comorbidities were found between the two groups (all P > 0.05).
Clinical Characteristics of the Indobufen and Aspirin Groups.
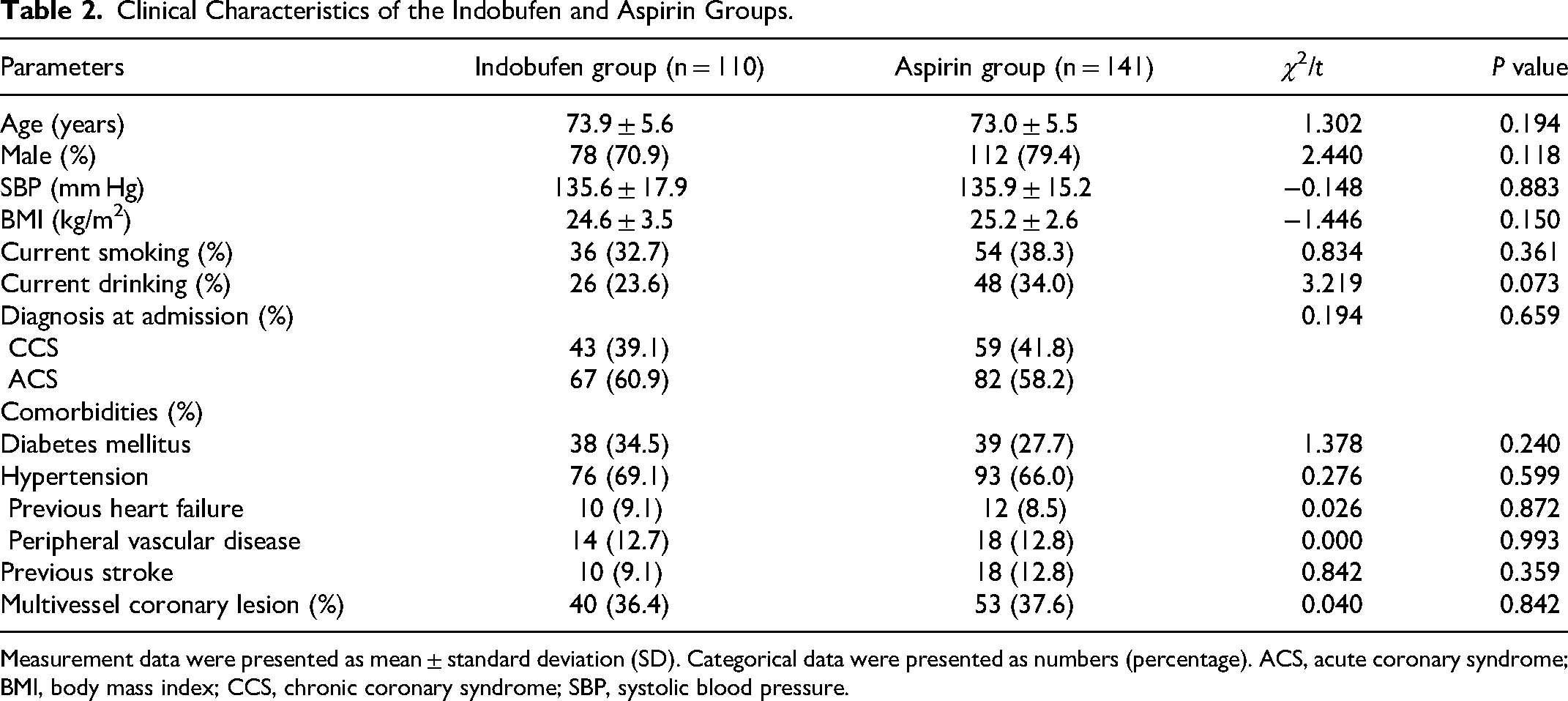
Measurement data were presented as mean ± standard deviation (SD). Categorical data were presented as numbers (percentage). ACS, acute coronary syndrome; BMI, body mass index; CCS, chronic coronary syndrome; SBP, systolic blood pressure.
Comparison of Characteristics at Baseline and 1-Year Follow-up Between the Two Groups
At 1-year follow-up, the creatinine (Cr) level and left ventricular ejection fraction (LVEF) of the indobufen group were both significantly higher than the baseline levels, whereas the estimated glomerular filtration rate (eGFR) showed a significant decrease (all P < 0.05) (Table 3). Compared with baseline, the mean platelet volume and thrombocytocrit of the aspirin group were all significantly lower than the baseline levels (all P < 0.05) (Table 3).
Characteristics at Baseline and 1-Year Follow-up Between the two Groups.
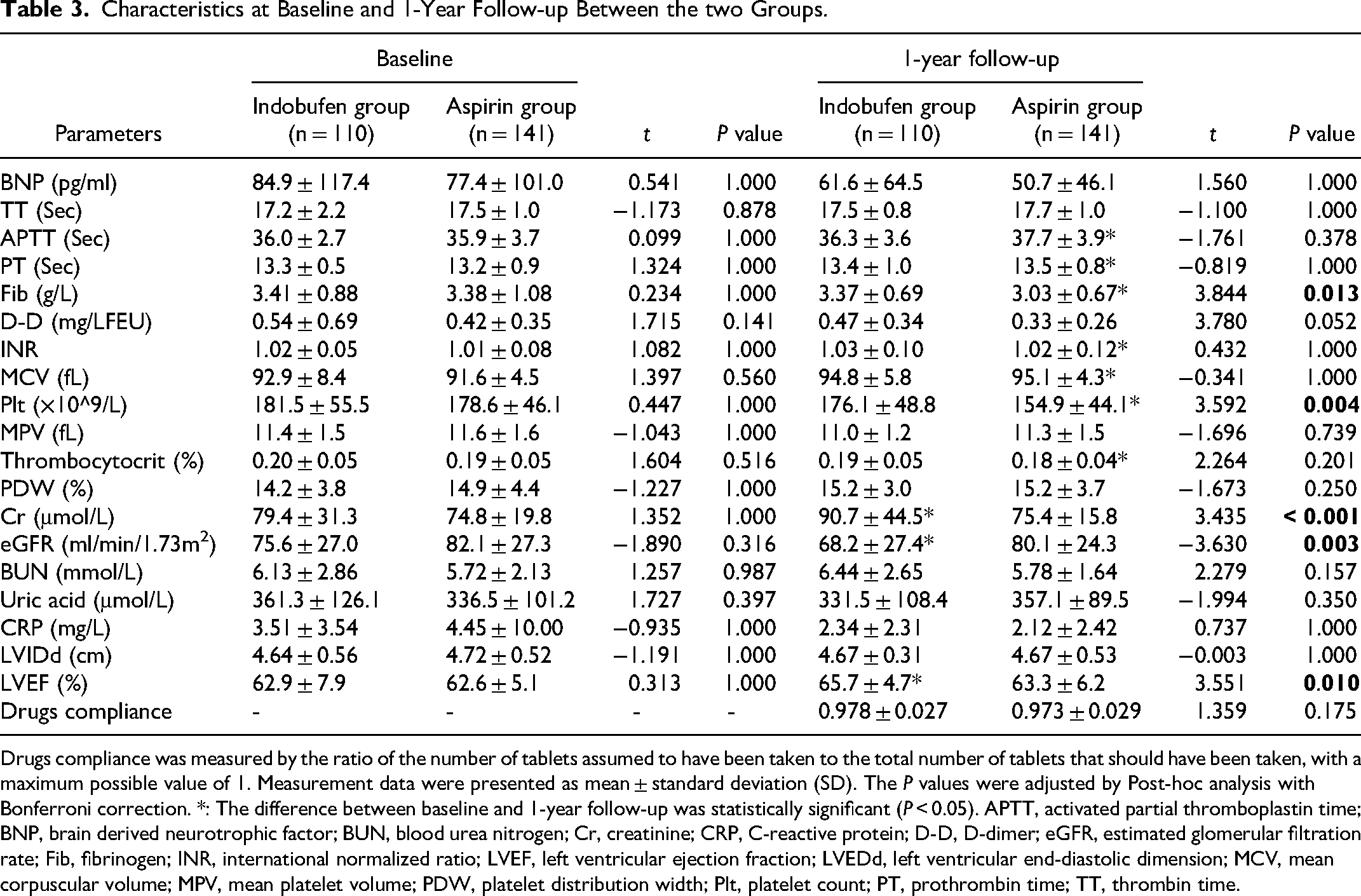
Drugs compliance was measured by the ratio of the number of tablets assumed to have been taken to the total number of tablets that should have been taken, with a maximum possible value of 1. Measurement data were presented as mean ± standard deviation (SD). The P values were adjusted by Post-hoc analysis with Bonferroni correction. *: The difference between baseline and 1-year follow-up was statistically significant (P < 0.05). APTT, activated partial thromboplastin time; BNP, brain derived neurotrophic factor; BUN, blood urea nitrogen; Cr, creatinine; CRP, C-reactive protein; D-D, D-dimer; eGFR, estimated glomerular filtration rate; Fib, fibrinogen; INR, international normalized ratio; LVEF, left ventricular ejection fraction; LVEDd, left ventricular end-diastolic dimension; MCV, mean corpuscular volume; MPV, mean platelet volume; PDW, platelet distribution width; Plt, platelet count; PT, prothrombin time; TT, thrombin time.
At baseline, no significant differences in all indices between the indobufen and aspirin groups (all P > 0.05) (Table 3). At 1-year follow-up, the levels of fibrinogen, platelet count, Cr, and LVEF in the indobufen group were higher than those in the aspirin group, whereas eGFR level was significantly lower, and the differences were all statistically significant (all P < 0.05) (Table 3).
The P values were adjusted by Post-hoc analysis with Bonferroni correction.
Comparison of Adverse Reactions and Events Between the Two Groups
At 1-year follow-up, in the indobufen group, gastrointestinal response occurred in two patients (improved after taking PPI) and skin and mucous petechiae in 1 (no intervention). In the aspirin group, gastrointestinal response occurred in 4 patients (improved after taking PPI), gingival bleeding in 1 (improved after aspirin was reduced to 75 mg/day), gross hematuria in 1 (improved after aspirin was reduced to 50 mg/day), and skin and mucous petechiae in 3 (1 without intervention, and 2 improved after aspirin was reduced to 75 mg/day). No allergies, liver or kidney impairment, or type 3 or 5 bleeding events according to BARC in both groups. The total incidence of adverse reactions in the aspirin group was 6.4% (9/141), which was higher than the 2.7% (3/110) in the indobufen group; however, no significant difference was found between the two groups (P = 0.178) (Table 4).
Adverse Reactions and Adverse Events.
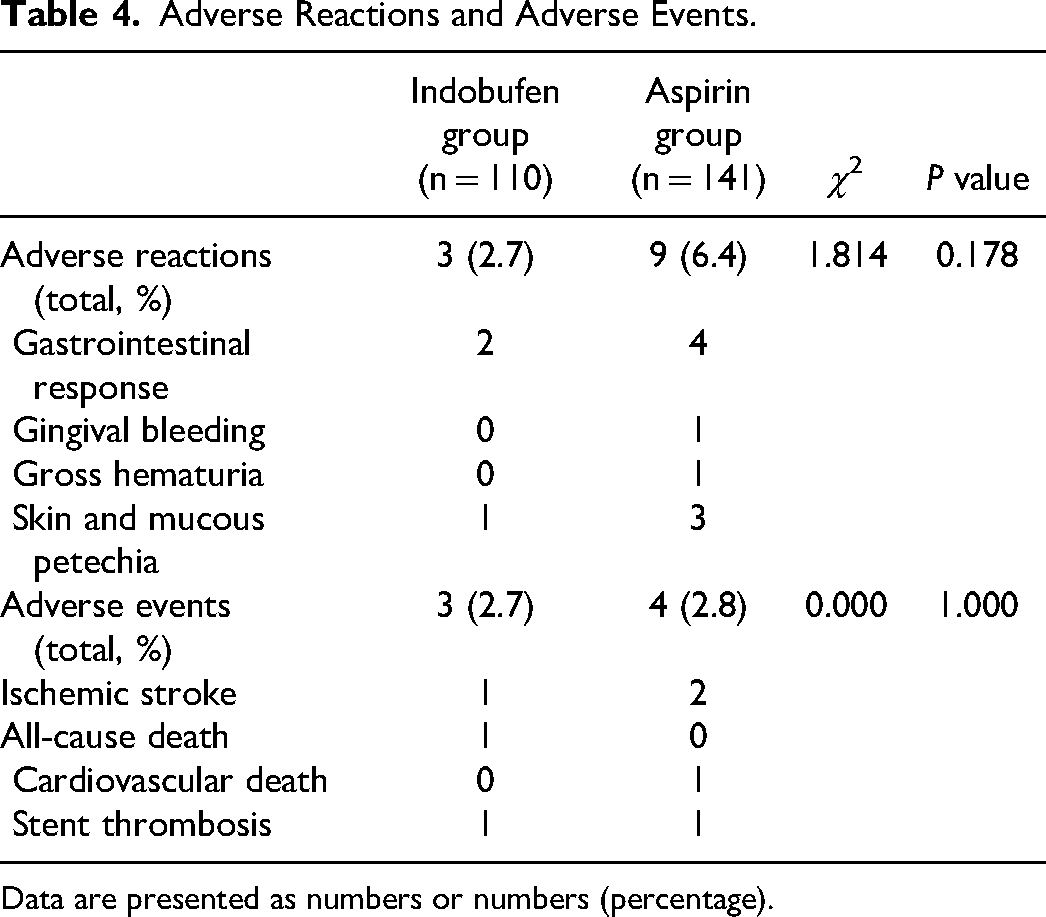
Data are presented as numbers or numbers (percentage).
At 1-year follow-up, no nonfatal MI occurred in both groups. The frequency of adverse events in the indobufen and aspirin groups is shown in Table 4. The rates of adverse events were comparable between the two groups [2.7% (3/110) versus 2.8% (4/141), P > 0.05] (Table 4).
Comparison of Therapeutic Effects Between the Two Groups
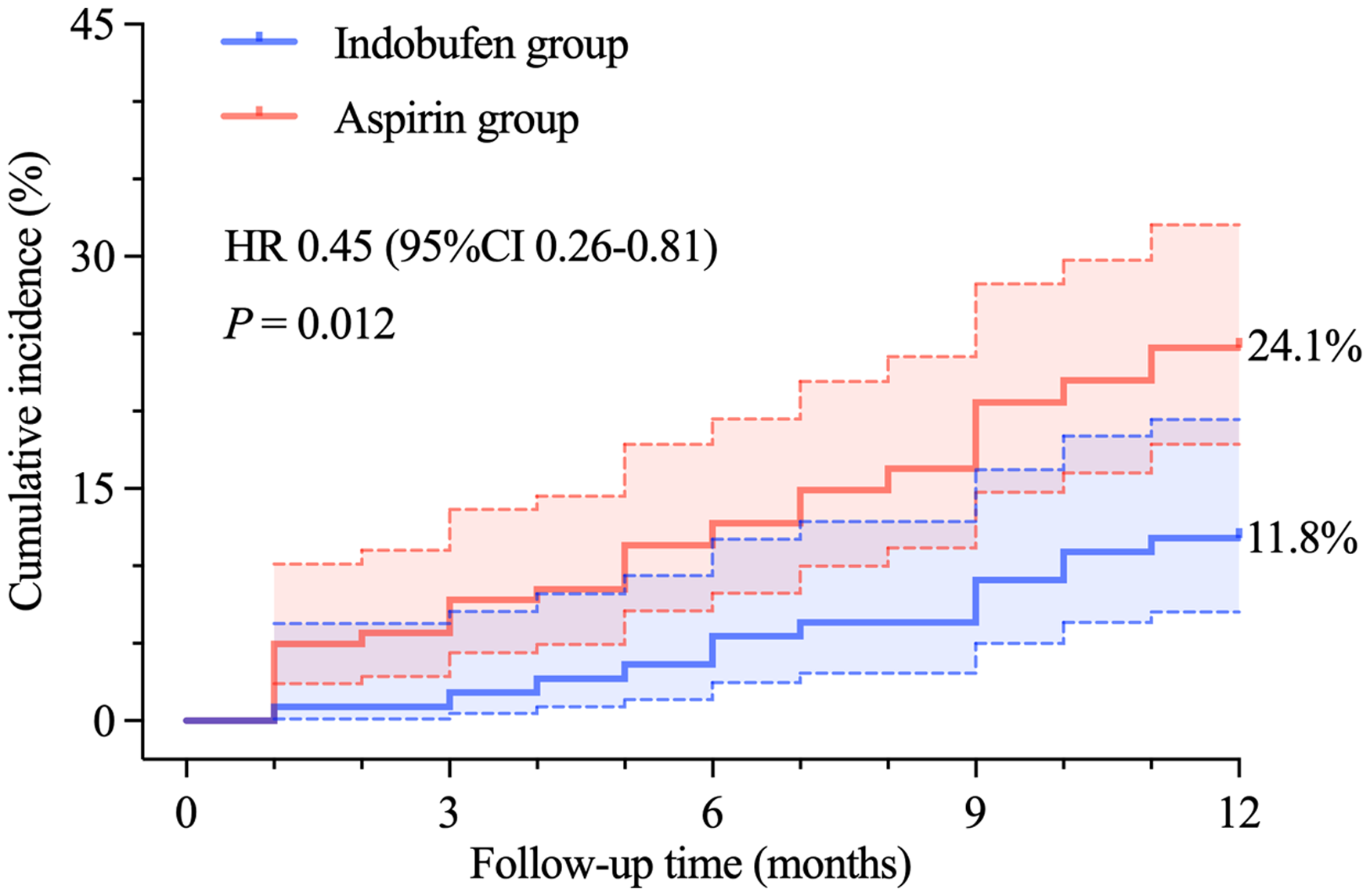
As shown in Figure 2, at 1-year follow-up, angina attacks occurred in 11.8% (13/110) and 24.1% (34/110) of the patients in the indobufen and aspirin groups, respectively. According to the log-rank test, the difference between the two groups was statistically significant (hazard ratio [HR] 0.45, 95% confidence interval [CI] 0.26-0.81, P = 0.012).
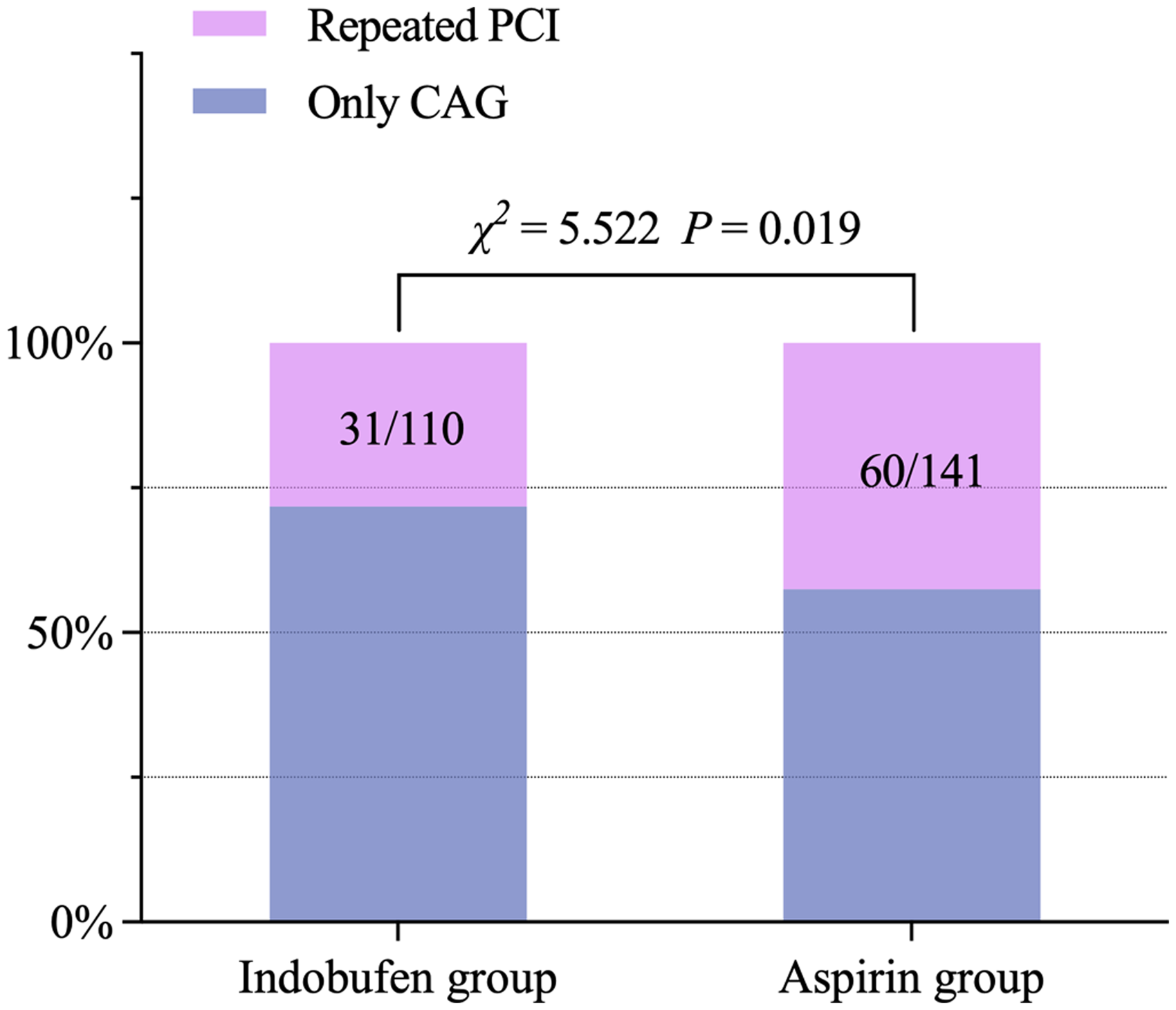
Both groups underwent coronary angiography again at 1-year follow-up and received PCI if necessary. As shown in Figure 3, 28.2% (31/110) of the patients in the indobufen group received repeated revascularization compared with 42.6% (60/141) in the aspirin group. The difference between the two groups was statistically significant (χ2 = 5.522, P = 0.019).
Analysis of Risk Factors of Repeated Revascularizations
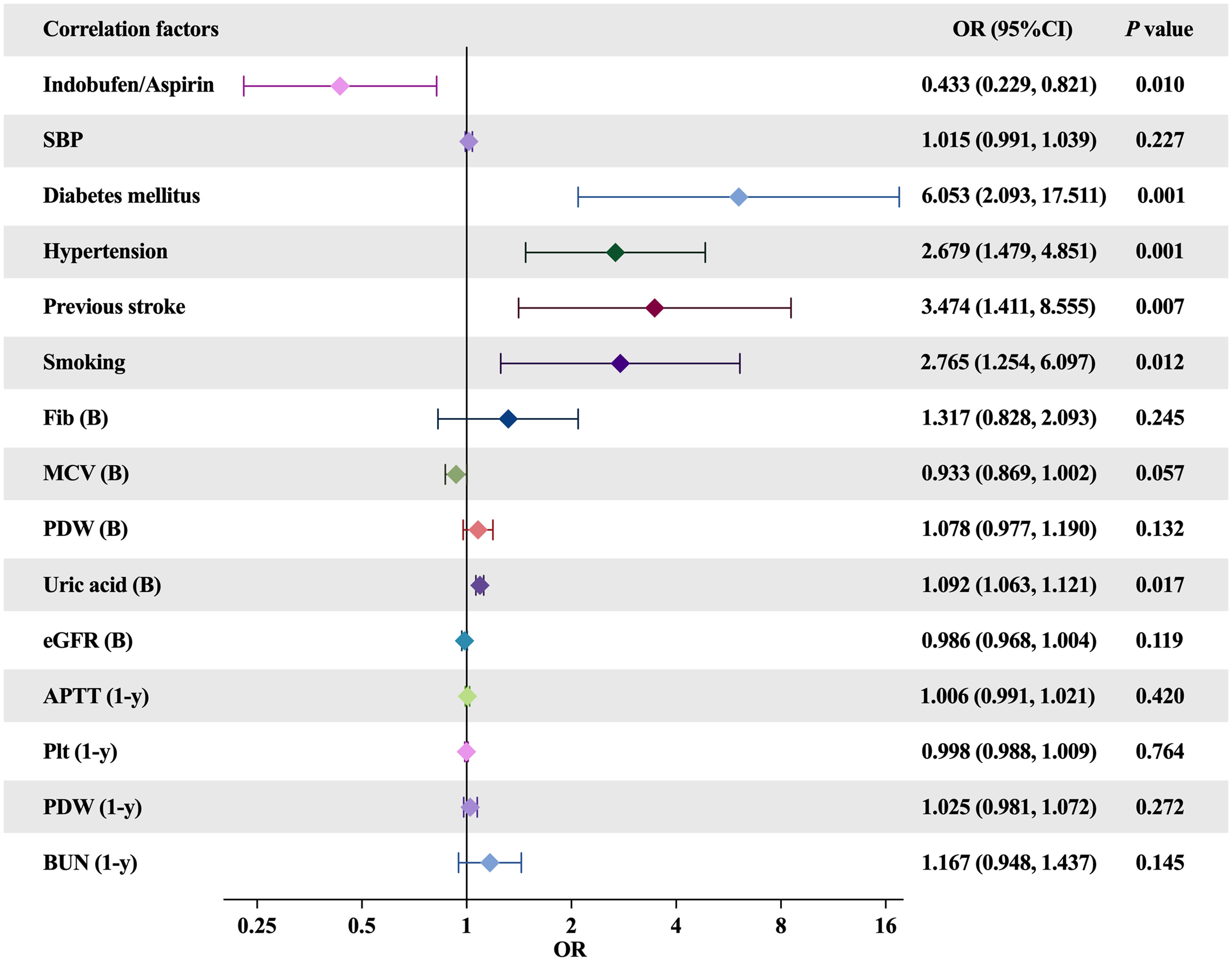
Figure 4 shows the independent risk factors for repeated revascularizations identified through binary logistic regression analysis. After including all possible correlation factors (elected by the Spearman correlation analysis), history of diabetes (odds ratio [OR] 6.053, 95% CI 2.093-17.511, P = 0.001), history of hypertension (OR 2.679, 95% CI 1.479-4.851, P = 0.001), history of stroke(OR 3.474, 95% CI 1.411-8.555, P = 0.007), smoking history (OR 2.765, 95% CI 1.254-6.097, P = 0.012), and high baseline uric acid level (OR 1.092, 95% CI 1.063-1.121, P = 0.017) were found to be independent risk factors for repeated PCI at 1-year follow-up. Indobufen + clopidogrel for DAPT demonstrated a protective effect against repeated PCI at 1-year follow-up (OR 0.433, 95% CI 0.229-0.821, P = 0.010).
Discussion
To the best of our knowledge, this is the first study that evaluated the long-term antiplatelet effects of indobufen and clopidogrel in a selected group of older patients with CAD who underwent coronary DES implantation. The main findings of this study are as follows: (i) After 12 months of DAPT with indobufen and clopidogrel, renal function decreased, whereas the platelet count was less affected compared with DAPT with aspirin and clopidogrel. (ii) The incidence of adverse reactions and adverse clinical events in the indobufen group was similar to that in the aspirin group. (iii) DAPT with indobufen and clopidogrel could effectively improve angina attacks and reduce the risk of repeated revascularizations at 1-year follow-up.
After coronary DES implantation, appropriate dual antiplatelet drugs should be prudently selected, particularly for older patients who have a higher risk of bleeding, ischemia, and other complications. Aspirin is currently the first-line basic antiplatelet drug for the patients who underwent coronary DES implantation, that has been recommended in numerous international clinical guidelines.15,16 However, because of the nonselective and irreversible inhibitory effect of aspirin on COX and prostaglandin synthesis, many patients may have a series of adverse reactions such as gastrointestinal response, bleeding, drug eruption, and even worsened respiratory diseases.7,17 The SYMPHONY study revealed that among patients taking aspirin, 5.4%, 1.7%, 2.6%, and 2.2% experienced nausea, vomiting, gastric indigestion, and abdominal pain, respectively. 18 Even in patients with acute coronary syndrome with high ischemic risk, approximately 9% of them stopped taking aspirin during long-term follow-up because of bleeding, intolerance, etc. 18 Furthermore, a meta-analysis in 2016 indicated that the use of low-dose aspirin (≤100 mg/day) was associated with a 58% increased risk of gastrointestinal bleeding (OR 1.58, 95% CI 1.29-1.95) and a 27% increased risk of hemorrhagic stroke (OR 1.27, 95% CI 0.96-1.68), which may be more severe in the older population. 19 And worse, rapid desensitization with aspirin cannot meet the needs for prompt treatment, and they may be at risk of delayed allergic reactions or desensitization failure.17,20
Indobufen is a nonsteroidal antiplatelet drug, which has a strong inhibitory effect on platelet adhesion, activation, and aggregation. Its characteristics are as follows9,10: (i) Selectivity refers to inhibition of platelet COX-1, reduction of TXA2 generation, lowering the inhibition of prostacyclin production, and fewer gastrointestinal reactions. (ii) Reversibility refers to the reversible inhibition of platelet function and short duration of antiplatelet effect; platelet function is restored within 24 h after discontinuation, which translates to a low risk of bleeding. (iii) Multiple pathways refer to simultaneously inhibiting adenosine diphosphate, platelet-activating factors, collagen, and arachidonic acid to prevent thrombosis rapidly and effectively. Many studies have confirmed that indobufen is more effective than or comparable to standard-dose aspirin in treating ischemic cardiovascular and cerebrovascular diseases caused by arteriosclerosis, with fewer adverse reactions and increased safety.11–13
Latib et al examined 127 patients who after PCI developed aspirin intolerance and argued that indobufen was the most common alternative to aspirin because 64.6% of them received indobufen combined with clopidogrel instead of aspirin. 21 The indobufen-based DAPT reduced the clinical cardiogenic mortality rates and incidence of stent thrombosis, MI, secondary bleeding, and other complications. 21 The strength of evidence of this retrospective analysis is slightly weaker than those of traditional randomized controlled trials; however, it confirmed the efficacy of antiplatelet therapy of indobufen after PCI. In a study of healthy volunteers who received aspirin (200 mg/day for 2 weeks) followed by a 4-week washout period and then indobufen (200 mg twice a day for 2 weeks), 22 the inhibitory rate of blood platelet aggregation 4 h after administration was similar in the two groups (81.07 ± 9.36% vs 96.99 ± 0.29%, P = 0.10). Conversely, at 12 and 24 h, the inhibitory rates of blood platelet aggregation in the indobufen group were significantly decreased (74.04 ± 9.55% vs 97.94 ± 0.28%, P < 0.001; 33.39 ± 11.13% vs 97.48 ± 0.32%, P < 0.001; respectively), which indicated that the initial inhibitory effect of indobufen on platelet aggregation was comparable to aspirin. However, the antiplatelet aggregation effect weakened faster, resulting in a lower bleeding risk. Moreover, the inhibitory effect of indobufen on prostacyclin production was significantly lower than that of aspirin (20.20 ± 7.43% vs 91.80 ± 8.81%, P < 0.001), suggesting that indobufen was generally well tolerated by the gastrointestinal tract.
During a 12-month follow-up, the effect of indobufen on platelet count was less than that of aspirin, and the incidence of adverse reactions and events was basically the same in the two groups. Recently, a large randomized noninferiority trial involving 4551 patients undergoing coronary DEA implantation unveiled that indobufen + clopidogrel for DAPT significantly reduced the 1-year net clinical outcomes (including cardiovascular death, nonfatal MI, ischemic stroke, stent thrombosis, and BARC ≥2 bleeding) compared with aspirin + clopidogrel for DAPT, which was mainly driven by a reduction in bleeding events (2.97% vs 4.71%, HR 0.63, 95%CI 0.46-0.85, P = 0.002) without an increase in ischemic events (1.51% vs 1.40%, HR 1.08, 95%CI 0.67-1.75, P = 0.76). 23 During the follow-up period, MI did not recur, which can be explained by the low probability of bleeding that reduced the discontinuation of DAPT. Further follow-up and a larger sample size are needed for confirmation. At present, indobufen is not yet the preferred option for DAPT because of the lack of sufficient high-quality clinical evidence to support its complete replacement of aspirin.
Coronary atherosclerosis is an ongoing pathological process. Despite the restoration of blood perfusion after PCI, if patients do not comply with the medication therapy and subsequent professional adjustment of diet and exercise, atherosclerotic lesions are easily aggravated and result in myocardial ischemia and hypoxia, which would lead to symptoms such as angina pectoris, heart failure, and recurrent MI. A multicenter study involving 3202 patients who underwent PCI with newer-generation DES showed that 11.84% and 12.66% of patients reported clinically relevant chest pain at 1- and 2-year follow-ups, respectively, and it was more common in women. 24 Furthermore, a posthoc analysis of patient data from two randomized controlled trials in Germany revealed that until 1 year after PCI, the cumulative incidence rates of first target vessel MI or revascularization or first nontarget vessel MI or revascularization were 15.9% and 12.3%, respectively. 25 Studies have suggested that impaired endothelial function and microvascular dysfunction may be the mechanisms underlying persistent chest pain after successful PCI.26,27 In addition to antiplatelet aggregation, indobufen could inhibit the intrinsic coagulation pathway and fibrinogen activation, thereby playing an important role in inhibiting microthrombus formation and improving endothelial function.28,29 Moreover, indobufen could improve microcirculation and hemorheology by increasing erythrocyte deformability and inhibiting potassium channels on arterial smooth muscle cells, causing vasodilation through the nitric oxide pathway,9,30 which would alleviate patient symptoms. In this study, the incidence of angina in the indobufen group was lower than that in the aspirin group (11.8% vs 24.1%, HR 0.45, 95% CI 0.26-0.81, P = 0.012), which indicated that indobufen + clopidogrel for DAPT could provide stable pharmacological support, and improve patient symptoms. In addition, indobufen-based DAPT could significantly reduce the risk of repeated PCIs (OR 0.433, 95% CI 0.229-0.821, P = 0.010). This finding aligns with the previous research of Ren et al In their study, over the 8-to-18-month follow-up period, the incidence of revascularization and thrombotic events were lower in the indobufen group than in the aspirin group, which was generally consistent across all prespecified subgroups. 31 The above results suggest that indobufen's antiplatelet activity, while comparable to that of aspirin, may confer additional benefits in specific clinical situations due to its distinct pharmacological profile by multiple pathways. However, it is important to note that, while repeat revascularization reflects progressive atherosclerosis and may influence health status, quality of life, and angina, higher rates of repeat revascularization in PCI trials should not be assumed as a marker of more frequent all-cause or cardiovascular deaths. 32
The dosage of indobufen varies between young and older patients, as well as in patients with kidney disease; thus, the dosage must be adjusted in these special populations to prevent excessive drug accumulation and subsequent adverse reactions. After taking single or multiple doses of indobufen, older patients had a longer elimination half-life and a slower drug clearance rate than healthy young volunteers, which was mainly due to the decline in renal function. 33 Moreover, in patients with kidney disease, indobufen clearance is related to the degree of renal dysfunction. Early studies have shown that the mean plasma clearance of indobufen in 11 patients with moderate-to-severe renal insufficiency (creatinine clearance <1.2 L/h) was 0.43 L/h, compared with 1.3 L/h in 6 healthy volunteers (creatinine clearance >6 L/h). Patients with moderate-to-severe renal dysfunction also had an extended elimination half-life (15-48 h).33,34 Therefore, the current recommended dose of indobufen is 100–200 mg twice a day, which can be reduced to 100–200 mg/day for older patients or patients with kidney damage. 13 The results of this study revealed that compared with aspirin 100 mg/day, indobufen 200 mg/day could increase the Cr level and decrease the eGFR at 1-year follow-up, suggesting that indobufen may further aggravate the renal burden in the older population. Interestingly, Lou et al showed that indobufen could reduce serum transforming growth factor-β levels and alleviate renal fibrosis in a rat model of chronic kidney disease, and they also demonstrated that the mechanism of indobufen in improving renal fibrosis could be correlated with the upregulation of 6-keto-prostaglandin F1α/thromboxane B2 in renal tissues. 35 However, the true situation might be more complicated in the clinical. As a potential frontline antiplatelet drug, indobufen should not only be observed for its efficacy but also for its complex effect on the kidneys of older patients. Given the above, the dose of indobufen should be reduced for older patients who are more likely to potential renal insufficiency. 13 In addition, the specific mechanism of indobufen on renal function in patients with CAD needs to be elucidated further.
This study still has some limitations. First, the study is based on single-center data, and the small sample size reduces the possibility of discovering significant differences between groups. Thus, to evaluate the correctness and generalizability of our findings, a prospective multicenter study with a large sample is needed. Second, the observation period is relatively short, and the incidence of adverse reactions and events is low, which may influence with statistical results. Third, although multiple regression analysis was performed, the other unmeasured confounding factors that may affect the conclusion could not be adjusted, and lack of blinding at the time of the intervention and the statistical analysis could introduce potential bias. Fourth, during the study, drugs compliance evaluations were generated from patient reports, and recurrent angina attacks were assessed subjectively, which had less credibility. This will be corrected in the future study.
In summary, for older patients with CAD who underwent coronary DES implantation, indobufen + clopidogrel for DAPT may be a better choice, which can effectively improve patient symptoms and prognosis. However, caution is needed when applying it to older patients with potential renal insufficiency. More studies are needed to evaluate indobufen-based DAPT.
Footnotes
Author Contributions
YW and XT performed and designed all the experiments. All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Availability of Data and Material
The data can be obtained by corresponding author upon reasonable request.
Consent for Publication
All authors agreed with publish this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded by Clinical Research Fund Project of Zhejiang Medical Association (2021ZYC-A46) to XT, and Guiding Scientific and Technological Project of Quzhou (2021007) to ZG.
