Abstract
Among patients with pulmonary embolism (PE), various permutations of normal or abnormal cardiac troponin results and normal or abnormal echocardiographic right ventricular function are encountered in clinical practice. We aimed to explore whether there is a true gradient of risk based on troponin and echocardiographic results. This study included normotensive patients with PE from the PROgnosTic valuE of CT scan in hemodynamically stable patients with acute symptomatic pulmonary embolism (PROTECT) study. Patients were categorized as having -Troponin/-Echo, -Troponin/+Echo, +Troponin/-Echo, and +Troponin/+Echo. The primary outcome was 30-day “complicated course,” including death from any cause, hemodynamic collapse, or recurrent PE. Secondary outcomes included individual adverse event rates. Of the 834 patients who had echocardiographic and troponin results, 569 patients (68%) had -Troponin/-Echo, 126 patients (15%) had -Troponin/+Echo, 74 patients (8.9%) had +Troponin/-Echo, and 65 patients (7.8%) had +Troponin/+Echo. The incidence of 30-day complicated course was 4.6% in patients with -Troponin/-Echo, 11.9% in patients with -Troponin/+Echo, 13.5% in patients with +Troponin/-Echo, and 16.9% in patients with +Troponin/+Echo (P for trend <0.001). In the subgroup of patients with a high-risk sPESI (i.e., intermediate-risk according to the ESC guidelines) (n = 527), the incidence of 30-day complicated course was 14.9% in patients with -Troponin/+Echo, 18.5% in patients with +Troponin/-Echo, and 17.5% in patients with +Troponin/+Echo (P for trend <0.01). In patiens with PE, there seems to be a risk gradient based on troponin and echocardiographic results. This study did not detect a significant risk difference in those with +Troponin/-Echo compared with -Troponin/+Echo.
Introduction
The key to an effective treatment of symptomatic pulmonary embolism (
For identification of low-risk patients, studies have validated the use of clinical prognostic models such as the Pulmonary Embolism Severity Index (PESI), the simplified PESI (sPESI), and the Hestia criteria. 6 –8 Alternatively, a positive troponin testing is integrated with the results of an echocardiogram showing right ventricle (RV) dysfunction to identify patients with intermediate-high risk PE. 1 In the pathophysiological cascade after an episode of acute PE, RV dilation usually precedes myocardial injury. 1 However, it is not uncommon to encounter patients who have a positive cardiac troponin result, but normal RV function and size on echocardiography, or viceversa. The prognostic significance of this discordant finding of positive troponin and negative echocardiography (+Troponin/-Echo) is unclear. Prior studies evaluating patients with +Troponin/-Echo have shown conflicting data regarding the prognosis of these patients, compared to that of patients with negative troponin testing and echocardiographic RV dysfunction (−Troponin/+Echo) 9,10 In a smaller study of 124 patients who had confirmed symptomatic acute PE, Binder and colleagues found that patients with +Troponin/-Echo did not have a higher risk of death compared to patients with -Troponin/+Echo. 9 In contrast, another study of 141 patients who had acute PE and underwent baseline echocardiographic evaluation and concomitant troponin measurement found that those with +Troponin/-Echo had a 30-day all-cause mortality of 23%, compared with 9% for those with -Troponin/+Echo. 10
In light of this uncertainty, we aimed to explore whether there is a true gradient of risk based on troponin and echocardiographic result. We were also interested in profiling the +Troponin/-Echo population and assessed whether these discordant results have a prognostic implication. To evaluate this hypothesis, we studied a large cohort of patients who enrolled the prospective multicenter PROgnosTic valuE of CT scan in hemodynamically stable patients with acute symptomatic pulmonary embolism (
Methods
PROTECT is a prospective, multicenter observational cohort study designed by the authors and sponsored by the Institute of Health Carlos III, Spain (NCT00880737). 12,13 This PROTECT substudy was a posteriori designed to test the prognostic significance of isolated myocardial injury (i.e., without concomitant echocardiographic RV dysfunction) in normotensive patients with acute symptomatic PE. Local ethics committees approved the study. All patients provided written informed consent.
Patients
Only patients diagnosed with acute PE (first symptoms occurring within 14 days) by multidetector CT were eligible. 14 Exclusion criteria consisted of treatment with thrombolytics at the time of PE diagnosis, life expectancy less than 3 months, pregnancy, geographic inaccessibility precluding follow-up, age younger than 18 years, severe renal insufficiency (creatinine clearance <30 mL/ min), inability to complete CT testing (e.g., allergy to intravenous contrast agents, unavailability of CT), or hemodynamic instability at presentation (defined as cardiogenic shock, systolic blood pressure < 90 mmHg, or use of inotropic support). We also excluded patients who did not successfully complete the protocol-required transthoracic echocardiography.
Baseline Examinations
The study required that patients undergo echocardiography within 24 hours after diagnosis of PE. The study defined echocardiographic dysfunction as the presence of at least 2 of the following: dilatation of the right ventricle (end-diastolic diameter >30 mm from the parasternal view or the right ventricle appearing larger than the left ventricle from the subcostal or apical view), hypokinesis of the right ventricle free wall (any view), or tricuspid regurgitant jet velocity >2.6 m/s. 15 Echocardiograms were reviewed by trained cardiologists according to the requested criteria.
Cardiac Biomarker Determinations
PROTECT used a core laboratory for blood biomarker measurement. The core laboratory personnel, blinded to the patients’ baseline characteristics and clinical outcome, measured cardiac troponin I (cTnI) levels quantitatively by using a microparticle enzyme immunoassay (Abbot, USA). PROTECT defined cTnI concentrations of >0.05 ng/mL as the 99th percentile of a healthy reference population with a coefficient of variation <10%, and indicative of myocardial injury (cTnI positive). 16
Study Outcome Measures
The study used a complicated course within 30-days from the diagnosis of PE as the primary outcome. A complicated course was defined as death from any cause, hemodynamic collapse (defined as need for cardiopulmonary resuscitation, systolic blood pressure < 90 mm Hg for at least 15 minutes, need for cathecolamine administration, or need for thrombolysis), or adjudicated recurrent PE within the 30-days of follow-up. Secondary outcomes included individual adverse event rates (i.e., all-cause mortality, PE-related death, hemodynamic collapse, and recurrent PE). An independent Adjudication Committee, whose members were blinded to initial prognostic test results, adjudicated all serious adverse events.
Statistical Analyses
Comparisons of baseline characteristics and outcome rates were made between the -Troponin/-Echo, -Troponin/+Echo, +Troponin/-Echo, and +Troponin/+Echo groups. Differences between +Troponin/-Echo and -Troponin/+Echo were assessed using χ2 test and a 2-tailed, t test for comparisons for categorical and continuous variables, respectively, and analysis of variance or Kruskal-Wallis were used, when appropriate, to compare all groups. Categorical variables are reported as counts with percentages and continuous variables are reported as mean (SD). Multivariable logistic regression was used to identify variables associated with +Troponin/-Echo; -Troponin/-Echo served as the reference group. We adjusted models for age, sex, coexisting conditions (i.e., cancer, immobilization, chronic lung disease, chronic heart disease), and severity of PE (i.e., heart rate, systolic blood pressure, arterial oxyhemoglobin saturation, sPESI) at hospital admission. Event curves for the outcomes were estimated by the Kaplan-Meier method and compared by the log-rank test. Analyses were based on time to first event, and if no event occurred, event time was censored at the end of follow-up. Since identification of intermediate-risk PE patients requires a positive prognostic rule, 1 we repeated analyses in this subgroup of patients with a high-risk sPESI. In addition, multiple logistic regression analysis compared the complication risk of 4 patient groups defined by the combination of troponin testing plus echocardiography. No adjustments for other baseline parameters were made in this latter model.
Statistical significance was defined a s a 2-tailed P-value of <0.05 for all analyses. Analyses were performed using SPSS, version 26 for the PC (SPSS, Inc. Chicago, IL, USA).
Results
Study Population
We screened 848 consecutive normotensive patients diagnosed with acute PE by multidetector CT in the emergency department for eligibility (Figure 1). The study excluded 14 patients because they did not have complete baseline troponin data required for analyses (1.7%; 95% confidence interval [CI], 0.9% to 2.8%). No statistically significant difference was observed between included and excluded patients regarding demographics, medical history, and clinical presentation. A total of 834 patients (410 men and 424 women; 98% of the screened population) were included in the present study. Of these, 569 patients (68.2%) had -Troponin/-Echo, 126 patients (15.1%) had -Troponin/+Echo, 74 patients (8.9%) had +Troponin/-Echo, and 65 patients (7.8%) had +Troponin/+Echo.

Study flow diagram.
The characteristics of the patient population (mean [SD] age, 67 17 years) are presented in Table 1. Compared with patients with -Troponin/+Echo, patients with +Troponin/-Echo were older (mean [SD] age, 72 16 years vs 69 16 years), had a greater burden of comorbidities (e.g., immobilization, recent surgery), and more signs of clinical severity (tachycardia, hypoxemia and hypotension). Compared with patients with -Troponin/+Echo, patients with +Troponin/-Echo were less likely to have concomitant deep vein thrombosis (DVT), but more likely to have higher BNP levels (mean [SD] pg/mL, 420540 pg/mL vs 342607 pg/mL). Patients with +Troponin/+Echo were younger and had a higher prevalence of female gender than patients with +Troponin/-Echo. Patients with +Troponin/+Echo more frequently manifested the signs of clinical severity (syncope, high-risk according to the sPESI, tachycardia, hypoxemia and hypotension), and had higher BNP levels (mean [SD] pg/mL, 569593 pg/mL vs 420540 pg/mL), compared with those with +Troponin/-Echo.
Baseline Clinical Characteristics.

† Active or under treatment in the last year.
‡In the previous month.
y Immobilized patients are defined in this analysis as non-surgical patients who had been immobilized (i.e., total bed rest with bathroom privileges) for ≥4 days in the month prior to PE diagnosis.
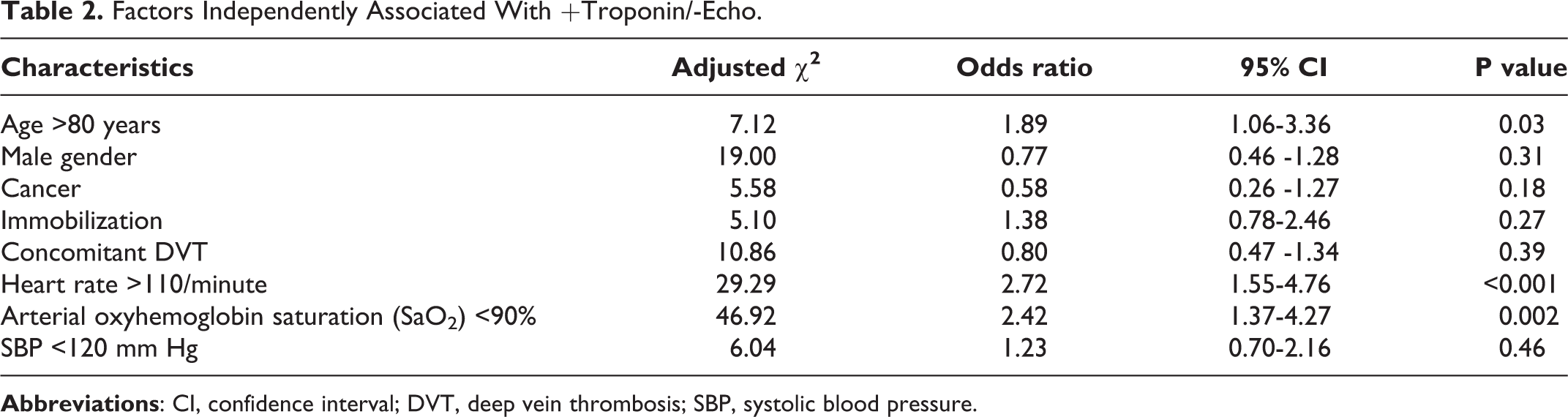
In multivariable models, factors significantly associated with +Troponin/-Echo were age >80 years (odds ratio [OR], 1.89; 95% CI, 1.06-3.36), heart rate >110/min (OR, 2.72; 95% CI, 1.55-4.76), and arterial oxyhemoglobin saturation <90% (OR, 2.42; 95% CI, 1.37-4.27) (Table 2).
Factors Independently Associated With +Troponin/-Echo.
Clinical Outcomes
A complicated course during the 30-day follow-up period occurred in 62 patients (7.4%; 95% CI, 5.8% to 9.4%). Thirty-seven of the 834 patients (4.4%; 95% CI 3.1% to 6.1%) died, and 3 patients (0.4%; 95% CI 0.1% to 1.1%) had recurrent symptomatic PE (1 non-fatal and 2 fatal). In addition to the 37 deaths, complicated course was due to hemodynamic collapse in 28 patients (3 were fatal and 1 was associated with non-fatal recurrent PE) (Table 3).
30-Day Adverse Events by Troponin/Echo Result.
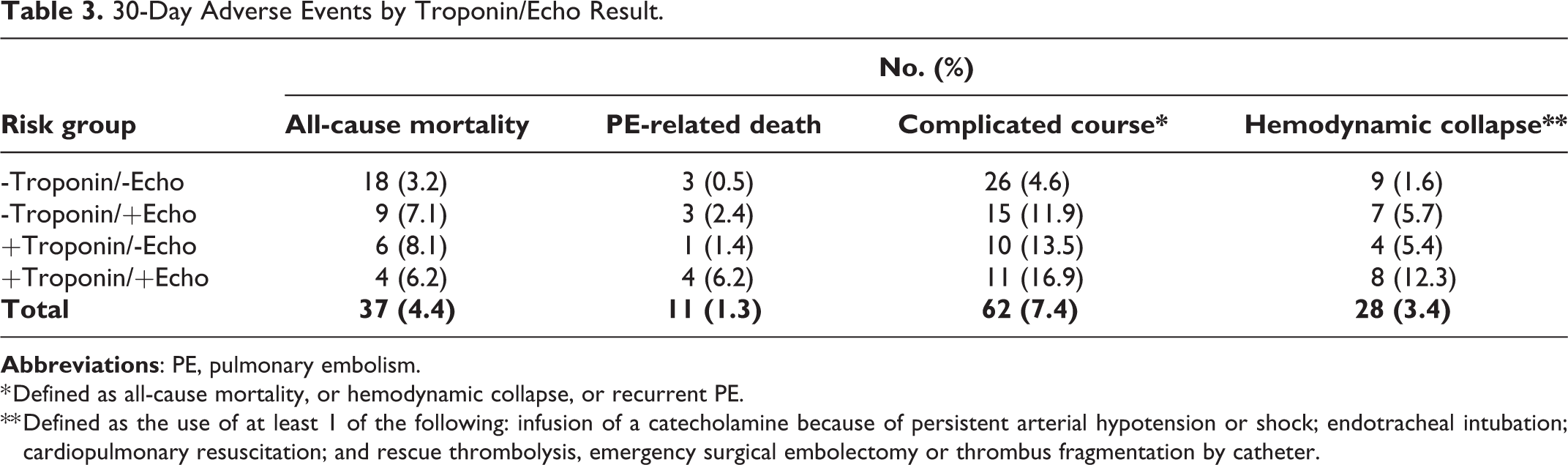
* Defined as all-cause mortality, or hemodynamic collapse, or recurrent PE.
** Defined as the use of at least 1 of the following: infusion of a catecholamine because of persistent arterial hypotension or shock; endotracheal intubation; cardiopulmonary resuscitation; and rescue thrombolysis, emergency surgical embolectomy or thrombus fragmentation by catheter.
The incidence of 30-day complicated course was 4.6% (95% CI, 3.0% to 6.6%) in patients with -Troponin/-Echo, 11.9% (95% CI, 6.8% to 18.9%) in patients with -Troponin/+Echo, 13.5% (95% CI, 6.7% to 23.5%) in patients with +Troponin/-Echo, and 16.9% (95% CI, 8.8% to 28.3%) in patients with +Troponin/+Echo (P <0.001) (Figure 2). Thirty-day all-cause mortality occurred in 18 patients with -Troponin/-Echo (3.2%), 9 patients with -Troponin/+Echo (7.1%), 6 patients with +Troponin/-Echo (8.1%), and 4 patients with +Troponin/+Echo (6.2%) (P = 0.07). Thirty-day PE-related death occurred in 3 patients with -Troponin/-Echo (0.5%), 3 patients with -Troponin/+Echo (2.4%), 1 patient with +Troponin/-Echo (1.4%), and 4 patients with +Troponin/+Echo (6.2%) (P = 0.001). The incidence of hemodynamic collapse was 1.6% with -Troponin/-Echo, 5.7% with -Troponin/+Echo, 5.4% with +Troponin/-Echo, and 12.3% with +Troponin/+Echo (P < 0.001).

Cumulative incidence of the primary endpoint: 30-day complicated course.
In the subgroup of patients with a high-risk sPESI (i.e., intermediate-risk according to the European Society of Cardiology [
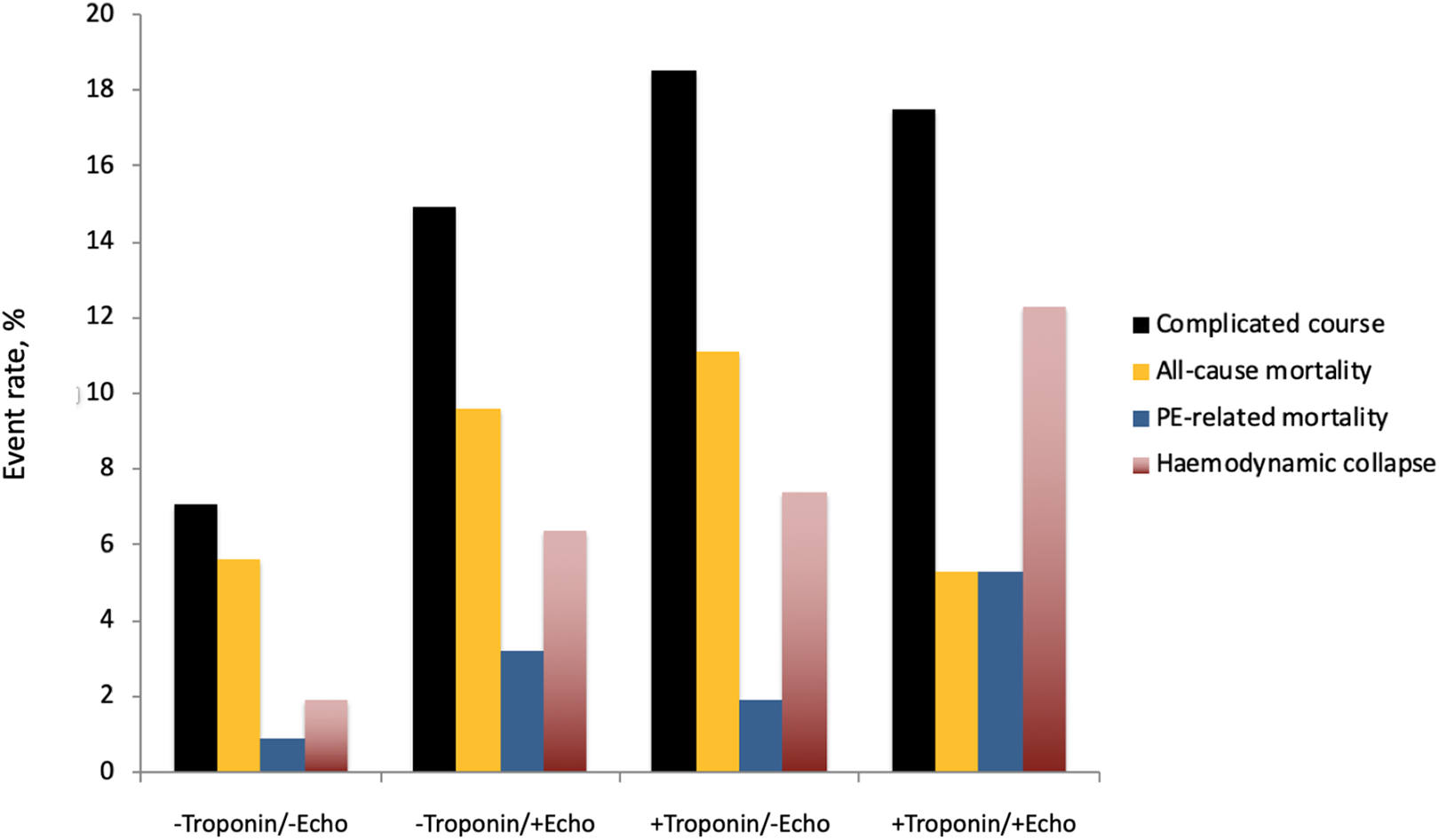
30-day adverse event rates in the subgroup of patients with high-risk sPESI.
Compared with the reference group (-Troponin/-Echo), -Troponin/+Echo was associated with a 2.8-fold increase in the odds of 30-day complicated course, while +Troponin/-Echo was associated with a 3.3 fold increase (Table 4).
Combination of Cardiac Troponin I With Echocardiography for Definition of Risk Groups in Patients With Acute Pulmonary Embolism.
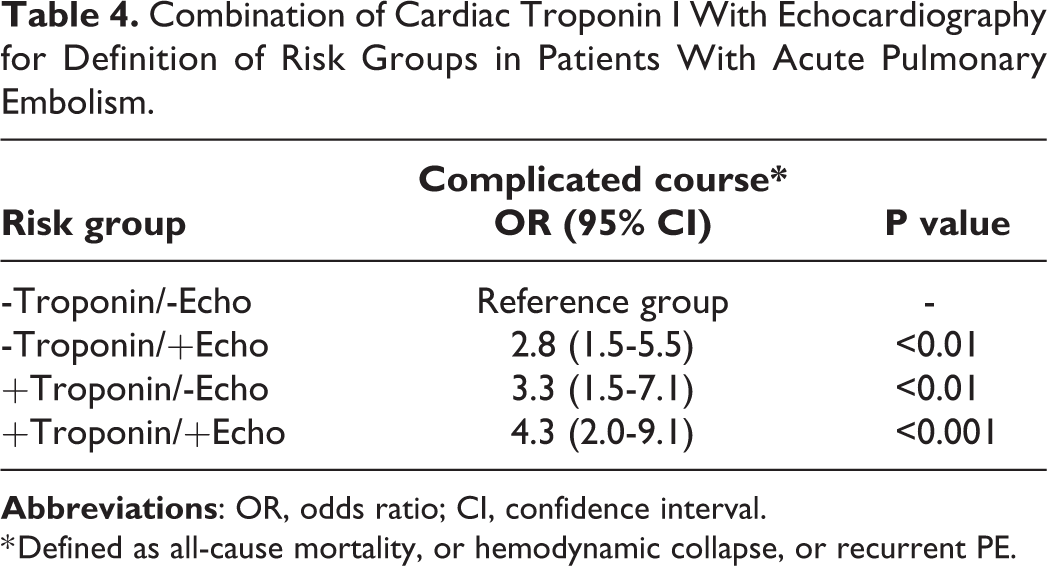
* Defined as all-cause mortality, or hemodynamic collapse, or recurrent PE.
Discussion
This study showed that in a large cohort of normotensive patients with acute symptomatic PE, 8.9% of patients had the discordant findings of +Troponin/-Echo. Though there was a risk gradient based on troponin and echocardiographic results, patients with +Troponin/-Echo, compared to -Troponin/+Echo, were not at a significantly increased risk for a complicated course.
Patients diagnosed with acute symptomatic PE should undergo risk stratification. 17 According to the ESC prognostic scheme, intermediate-low risk patients are those with a positive clinical prognostic score and either echocardiographic RV dysfunction (-Troponin/+Echo), elevated troponin levels (+Troponin/-Echo), or none (-Troponin/-Echo). 1 Previous studies have shown conflicting data regarding the short-term prognosis of this intermediate-low risk subgroup. 18,19 While one study found that the proportion of 30-day PE-related complications markedly increased with each higher ESC risk group (10.5% in intermediate-low risk group vs 17.5% in the intermediate-high risk), 18 another study that enrolled 906 stable and unstable patients with acute PE showed that the difference in the risk of death between intermediate-low and intermediate-high risk patients was relatively modest (6.0% in intermediate-low risk group vs 7.7% in the intermediate-high risk). 19 Within the intermediate-low risk category of patients, a different proportion of +Troponin/-Echo patients in each study might explain the discrepant findings.
Pathophysiologic consequences of occlusion of a pulmonary artery include RV dilation and dysfunction. 20 The increase in RV pressure and volume leads to an increase in wall tension and myocyte stretch that may cause RV ischemia, further reducing contractile forces. Compared with patients with -Troponin/+Echo, we found that patients with +Troponin/-Echo were less likely to have concomitant residual DVT, but more likely to have tachycardia. It is possible that patients with +Troponin/-Echo might have a higher clot burden (reflected by complete embolization of the thrombus from the leg veins), causing tachycardia as a compensatory sign, indicative of impending hemodynamic collapse. Tachycardia has been associated with elevated troponin levels, even before RV dilation and dysfunction occur. 21
Observational studies have suggested that the combination of abnormal clinical variables (alone or in combination as clinical prediction rules), laboratory evidence of myocardial injury, and imaging evidence of RV dysfunction identifies a subgroup of normotensive patients who have about a 20% risk of short-term major PE-related complications. 22,23 This study further supports the applicability of the sPESI, in combination with imaging tesing and cardiac biomarkers, to identify the sickest normotensive patients with acute symptomatic PE. In patients with +Troponin/+Echo, a 30-day complicated course occurred in 17.5% of those with a positive sPESI, compared with 12.5% of those with a negative sPESI.
What are the clinical implications of our findings? First, this study has evaluated a possible at-risk population that is commonly encountered in clinical practice but has been underrecognized and not well characterized. Second, previous studies have been too small to definitively differentiate patients with +Troponin/-Echo from those with -Troponin/+Echo, and thus clinical practice guidelines consider these populations to have an equivalent prognosis (intermediate-low risk PE). 1,16 This study suggests that, compared with patients with -Troponin/-Echo, patients with +Troponin/-Echo are not at significantly higher risk for PE-related adverse outcomes, even in the subgroup of patients with a positive sPESI. However, the event rates were numerically different and we cannot exclude the possibility that our study, despite being larger than most prior studies, was still underpowered. Clinical practice guidelines suggest that normotensive patients with acute PE and +Troponin/+Echo (i.e., intermediate-high risk PE) should be monitored over the first hours or days due to the risk of early hemodynamic decompensation and circulatory collapse. Our results indicate that monitoring might not be extended to patients with +Troponin/-Echo, since their short-term prognosis is similar to that of patients with -Troponin/+Echo.
This study has several limitations. Despite the large number of patients assessed for this study, the low number of primary endpoints did not allow for more precision in our estimates. One weakness of the study included the restriction of recruitment to patients who could safely undergo the extra tests within 24 hours after diagnosis of PE. However, it is not clear whether this enrollment restriction had a substantive effect on the results. Since this study did not directly assess the impact of our results on the management of these patients, further studies are needed to address this important question.
In conclusion, there seems to be a risk gradient based on troponin and echocardiographic results in stable patients with acute PE. Compared to -Troponin/+Echo, patients with +Troponin/-Echo were not at an increased risk for a complicated course, and might not benefit from a differentiated management. Future studies may further validate these findings, and if they can change clinician behavior and improve patient outcomes.
Footnotes
Author Contributions
Study concept and design: Mirambeaux, Bikdeli, Jimenez. Acquisition of data; analysis and interpretation of data; statistical analysis: Mirambeaux, Le Mao, Muriel, Pintado, Pérez, Rodríguez, Velasco, Morillo, Bikdeli, Jimenez. Drafting of the manuscript: Le Mao, Bikdeli, Jimenez. Critical revision of the manuscript for important intellectual content: Mirambeaux, Le Mao, Muriel, Pintado, Pérez, Rodríguez, Velasco, Morillo, Bikdeli, Jimenez. Study supervision: Bikdeli, Jimenez. The corresponding author, David Jiménez, had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr. Bikdeli reports that he is a consulting expert, on behalf of the plaintiff, for a litigation related to a specific type of IVC filters.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
