Abstract
Unfractionated heparin (UFH) is a sulfated glycosaminoglycan that consists of repeating disaccharides, containing iduronic acid (or glucuronic acid) and glucosamine, exhibiting variable degrees of sulfation. UFHs release tissue factor pathway inhibitor (TFPI) which inhibits the extrinsic pathway of coagulation by inactivating factor Xa and the factor VIIa/TF complex. Most heparins used clinically are derived from porcine intestinal mucosa however, heparins can also be derived from tissues of bovine and ovine origin. Currently there are some concerns about the shortage of the porcine heparins as they are widely used in the manufacturing of the low molecular weight heparins (LMWHs). Moreover, due to cultural and religious reasons in some countries, alternative sources of heparins are needed. Bovine mucosal heparins (BMH) are currently being developed for re-introduction to the US market for both medical and surgical indications. Compared to porcine mucosal heparin (PMH), BMH exhibits a somewhat weaker anti-coagulant activity. In this study, we determined the TFPI antigen level following administration of various dosages of UFHs from different origins. These studies demonstrated that IV administration of equigravemetric dosages of PMH and ovine mucosal heparin (OMH) to non-human primates resulted in comparable TFPI antigen release from endothelial cells. In addition, the levels of TFPI were significantly higher than TFPI antigen levels observed after BMH administration. Potency adjusted dosing resulted in comparable TFPI release profiles for all 3 heparins. Therefore, such dosing may provide uniform levels of anticoagulation for the parenteral indications for UFHs. These observations warrant further clinical validation in specific indications.
Keywords
Background
Unfractionated heparin (UFH) is a highly sulfated glycosaminoglycan (GAG) that consists of repeating disaccharide units containing iduronic acid (or glucuronic acid) and glucosamine, exhibiting variable degrees of sulfation. 1 Ever since the discovery of heparins in 1916, mammalian tissues have been used as a source of this anticoagulant. 1 While the first isolation of heparin was from dog liver, 1 lung and intestinal mucosa from cow and pig have been used subsequently to prepare pharmaceutical heparins. 2,3 The initial clinical batches of heparins were obtained from beef lung, however, due to the Bovine Spongiform Encephalopathy (BSE) virus and other cattle borne viral contaminants, its clinical use was disapproved. 4,5 Improved de-loading techniques are now widely used to remove BSE viral contamination in heparin preparations. 6 The FDA requires that all manufactures provides proof of the viral de-loading process for all heparin preparations used clinically. This led to the widespread clinical use of porcine mucosal heparin (PMH) and its depolymerized products. Because of potential supply limitations and quality issues with porcine heparin, the Food and Drug Administration (FDA) has initiated plans to reintroduce bovine mucosal heparin (BMH) and its polymerized products. Besides BMH, ovine mucosal heparins (OMH) are also currently being developed in some countries. 6,4
Tissue factor pathway inhibitor antigen contributes to the anticoagulant effect of these heparins by inhibiting the TF-FVIIa complex. TFPI is a single-chain polypeptide that acts as the primary inhibitor of the initiation of blood coagulation by tissue factor and modulates the severity of a wide variety of bleeding and clotting disorders. 7 –9 The TFPI molecule has an acidic amino-terminal region followed by 3 tandem Kunitz-type proteinase inhibitory domains and a basic carboxy-terminal region. 10 TFPI also directly inhibits factor Xa and, in a factor Xa dependent manner, produces feedback inhibition of the factor VIIa/tissue factor catalytic complex. 11 The plasma concentration of TFPI antigen is low (∼2 nM) and much of the circulating TFPI is bound to lipoproteins, including low-density lipoprotein (LDL), high-density lipoprotein (HDL), and lipoprotein (a). 12 –14 TF is a 45 kDa transmembrane glycoprotein which is located at extravascular sites that are not normally exposed to the blood. It is the main mediator by which thrombin generation is initiated in response to vessel damage (extrinsic pathway). 15 –18 TF availability induces clot formation, activates platelets, and mediates feedback amplification of various earlier steps in clotting, ultimately leading to the deposition of fibrin. 19 UFH and its derivatives release TFPI antigen from the surface of the endothelium. 20 Additionally, UFHs also upregulate TFPI gene expression which causes the elevation of both plasma and cellular TFPI pools. 21
The preferential use of porcine UFH over bovine and ovine counterparts has prompted a number of concerns. 22 First, the limited supply of porcine tissues has led to an increase in heparin cost. Second, over 50% of the world’s population of pigs comes from China, which may prove to be problematic for suppliers in the US health market because of the difficult US-China economic relations. Finally, due to religious and/or cultural practices in various parts of the world, the use of porcine heparins prevents patients from seeking optimal care. In this study, we project that, BMH, when administered at equivalent unit dosages may provide a comparable TFPI antigen release profile as OMH and PMH. while PMH and OMH may be interchangeable, 4 BMH may require potency adjustments, thus exhibiting a comparable profile to ovine and porcine heparins. 23
The primary purpose of these studies was to compare the release of TFPI antigen by bovine, ovine and porcine UFHs following intravenous administration of equigravemetric and equiunit dosages to non-human primates. Equivalent mass/kilogram (equigravemetric) and equivalent USP units (equiunit) dosages to non-human primates, as UFH are usually administered in U/weight to achieve a desirable anticoagulation, it was hypothesized that by increasing the mass of BMH to achieve comparable USP unit potency, BMH will produce similar anticoagulant actions. Most heparin preparations, regardless of their origins are cross referenced against USP reference or European standard (ES) to harmonize their potency. TFPI antigen release is an endogenous process which is not measurable by using invitro assays such as the anti-Xa and anti-IIa methods. Therefore, these studies were designed to compare the levels of TFPI antigen after administration of bovine, ovine and porcine UFHs to non-human primates.
Materials and Methods
Test Agents
Porcine mucosal heparin (PMH) was obtained from Medefil Inc., (Glendale Heights, IL, USA). Bovine mucosal heparin (BMH) was obtained from KinMaster Indústrias Químicas, (Passo Fundo, Brazil). Ovine mucosal heparin (OMH) was obtained from Ronnsi Pharmaceutical, (Jiangsu, China). All samples were obtained as white powders which were stored at room temperature in a desiccator. These samples were weighed on a Mettler balance and 0.9% NaCl was used as a diluent to make stock solutions of 10 mg/ml for the in vivo and in vitro testing.
Commercial Kits
The potency of each heparin was determined by an amidolytic anti-Xa assay in relation to the USP heparin standard (Aniara Diagnostica, OH, USA). Tissue factor pathway inhibitor (TFPI) antigen levels were determined using TFPI kits obtained from Diagnostica Stago, Inc. (Parsippany, NJ, USA).
Primates
Twelve male and female Rhesus monkeys (Macaca mulatta) weighing 4.0-16.0 kg were used for pharmacodynamics studies. All research was conducted in compliance with the guidelines set forth by the Institutional Animal Care and Use Committee (IACUC), Loyola University Chicago Health Sciences Campus and the Principles of Laboratory Animal Care.
Dosing
Each heparin was dissolved in 0.9% physiologic saline prior to administration to primates. Squeeze cages were used to anesthetize the primates with an intramuscular injection of ketamine (10 mg/kg) and xylazine (1-2 mg/kg) based on the most recent charted weight. Before injection of heparins, the primates were weighed to accurately determine the dose of the test agent and skin over the saphenous vein was shaved as necessary and then cleaned using betadine and alcohol. A baseline blood sample was collected by venipuncture of the saphenous vein. Heparins were administered at an equigravemetric dose (0.5 mg/kg) and a potency-adjusted dose (50 or 100 U/kg) intravenously via the saphenous vein. Additional blood samples were collected at 15-, 30-, 60- and 120-minutes post-drug administration. Four primates were dosed with each agent. After administration of the test agent and after collection of each of the subsequent blood samples, primates were returned to their cages and monitored continuously until they awakened from anesthesia and were able to sit up on their own. Furthermore, 10-15 minutes prior to each blood draw, the sedation state of the animal was assessed, and additional doses of ketamine were given intramuscularly as needed.
Sample Collection
All blood samples were drawn using a double syringe technique, employing a 21-gauge butterfly needle. After an initial collection of∼1 ml volume (discarded blood), the syringe was changed, and a 2.7 ml sample was drawn and placed into a tube containing 0.3 ml 3.2% sodium citrate. Citrated blood samples were centrifuged, and aliquots of plasma were stored at 80 °C for analysis of (TFPI) levels.
Sample Analysis
TFPI antigen levels were determined in accordance to the directions provided by the manufacturers. TFPI concentrations were plotted against corresponding sample times using graphing software, GraphPad Prism® Version 8.0 for Windows (GraphPad Software, San Diego, CA, USA). The TFPI plasma concentrations were used to calculate values for the pharmacokinetic parameters such as elimination half-life (t1/2) and area under the plasma concentration time curve (AUC) were calculated by non-compartmental analysis using PKSolver® software. 24
Statistical Analysis
All values were expressed as mean ± standard deviation. Data were analyzed using Student’s unpaired t-test or ANOVA for group wise comparison. Significance was set at p ≤ 0.05. Statistical analysis was performed using GraphPad Prism Version 8.0 (GraphPad Software, San Diego, CA), USA.
Results
In this study, TFPI antigen levels were measured at varying time points after the administration of heparins from various sources. Figure 1 shows the TFPI antigen levels in primates after 0.5 mg/kg IV porcine, bovine and ovine heparin. Following a gravimetric dosage, BMH-treated primates exhibited a significantly lower TFPI level at 15 and 30 mins (p** < 0.01, p* < 0.05) than OMH and PMH, with a maximum value of 200 ± 39 ng/ml at 15 mins post drug administration (Figure 1). OMH and PMH showed comparable TFPI levels at all time points with maximum values of 254 ± 24 ng/ml and 273 ± 42 ng/ml respectively at 15 mins post 0.5 mg/kg drug administration (Figure 1).

The pharmacodynamic effects of 0.5 mg/kg I.V. PMH, BMH and OMH administered to primates as measured by the TFPI release assays. PMH and OMH significantly increased the TFPI antigen release compared to the BMH 15- and 30-mins post drug administration (p** < 0.01, p** < 0.01, p* < 0.05). The data represent the mean ± standard deviation (n = 4).
In a second set of experiments, porcine, bovine and ovine heparins were administered at potency adjusted dosages. Administration of potency adjusted dosages of each UFH resulted in comparable TFPI antigen levels at all time points with a maximum value of 182 ± 13 ng/ml and 252 ± 31 ng/ml at 15 mins post 50 U/kg & 100 U/kg drug administration, respectively (Figure 2A and B). At 60 and 120 mins post IV administration of 100 U/kg, BMH showed slightly higher TFPI levels of 180 ± 26 ng/ml & 132 ± 18 ng/ml, respectively, compared to both OMH & PMH (Figure 2A). The same trend occurred at 60 and 120 mins post IV administration of 50 U/kg of heparins (Figure 2B). BMH showed slightly higher TFPI antigen level of 123 ± 32 ng/ml & 82 ± 15 ng/ml respectively compared to both OMH & PMH.
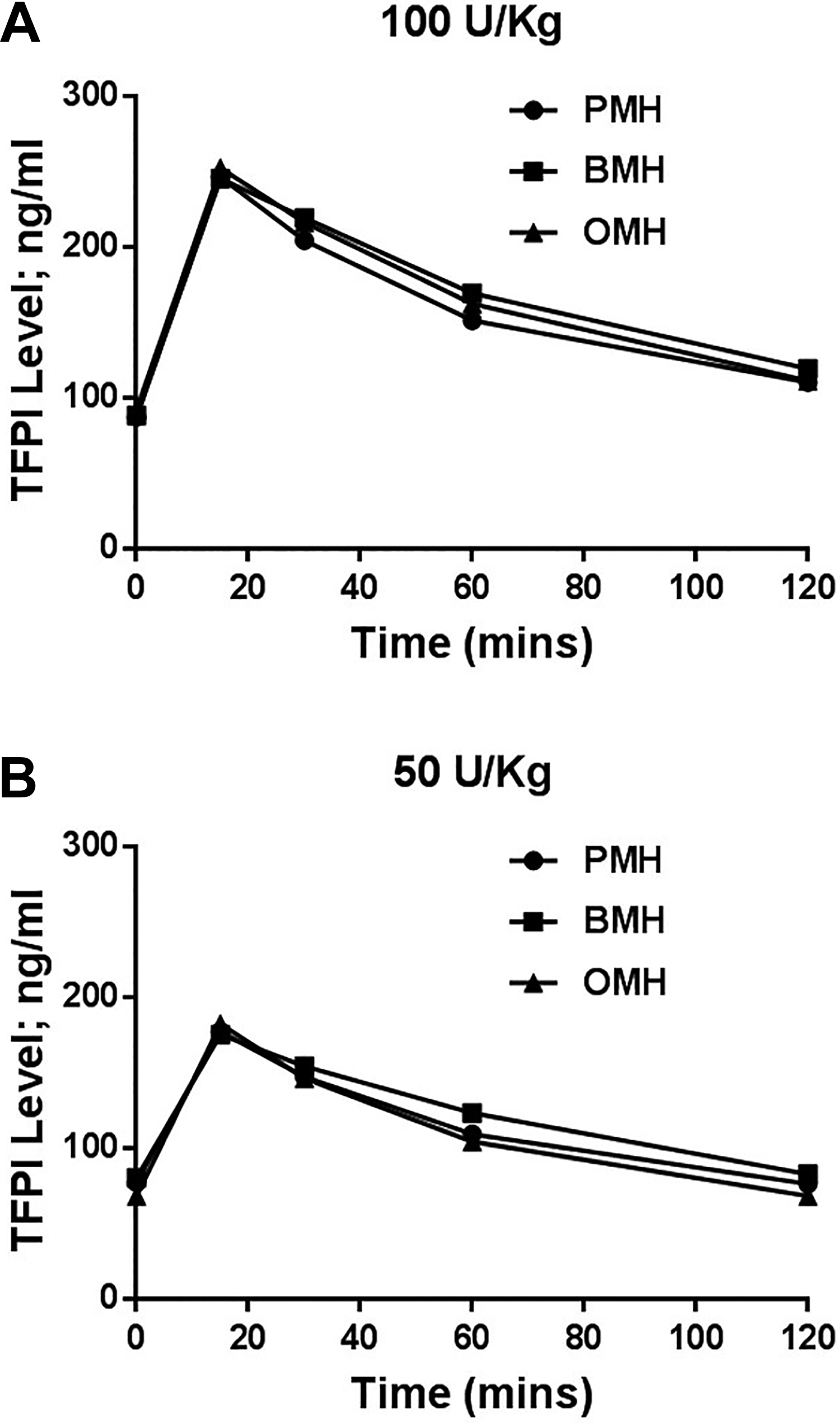
The pharmacodynamic effects of 100 (A) and 50 (B) U/kg IV PMH, BMH & OMH administered to primates as measured by the TFPI release assays. PMH, BMH and OMH showed comparable TFPI release profiles at all time points post I.V. administration of both dosages of PMH, BMH and OMH. The data represent the mean ± standard deviation (n = 4).
The estimation of t½ and AUC calculated using TFPI antigen levels after IV administration of 0.5 mg/kg of various heparins to non-human primates are shown in Figure 3. At the indicated dosage, BMH, OMH and PMH demonstrated a comparable half -life (t½) with a minimum value of 55 ± 6 mins and a maximum value of 61 ± 7 mins (Figure 3A). No significant difference in area under the curve (AUC) was noted between OMH (18639 ± 718 ng/ml*min) and PMH (20135 ± 561 ng/ml*min) after IV administration of 0.5 mg/kg to non-human primates (Figure 3B). However, BMH had a significantly (p* < 0.05) smaller AUC of 16634 ± 698 ng/ml*min compared to PMH as shown in Figure 3B.
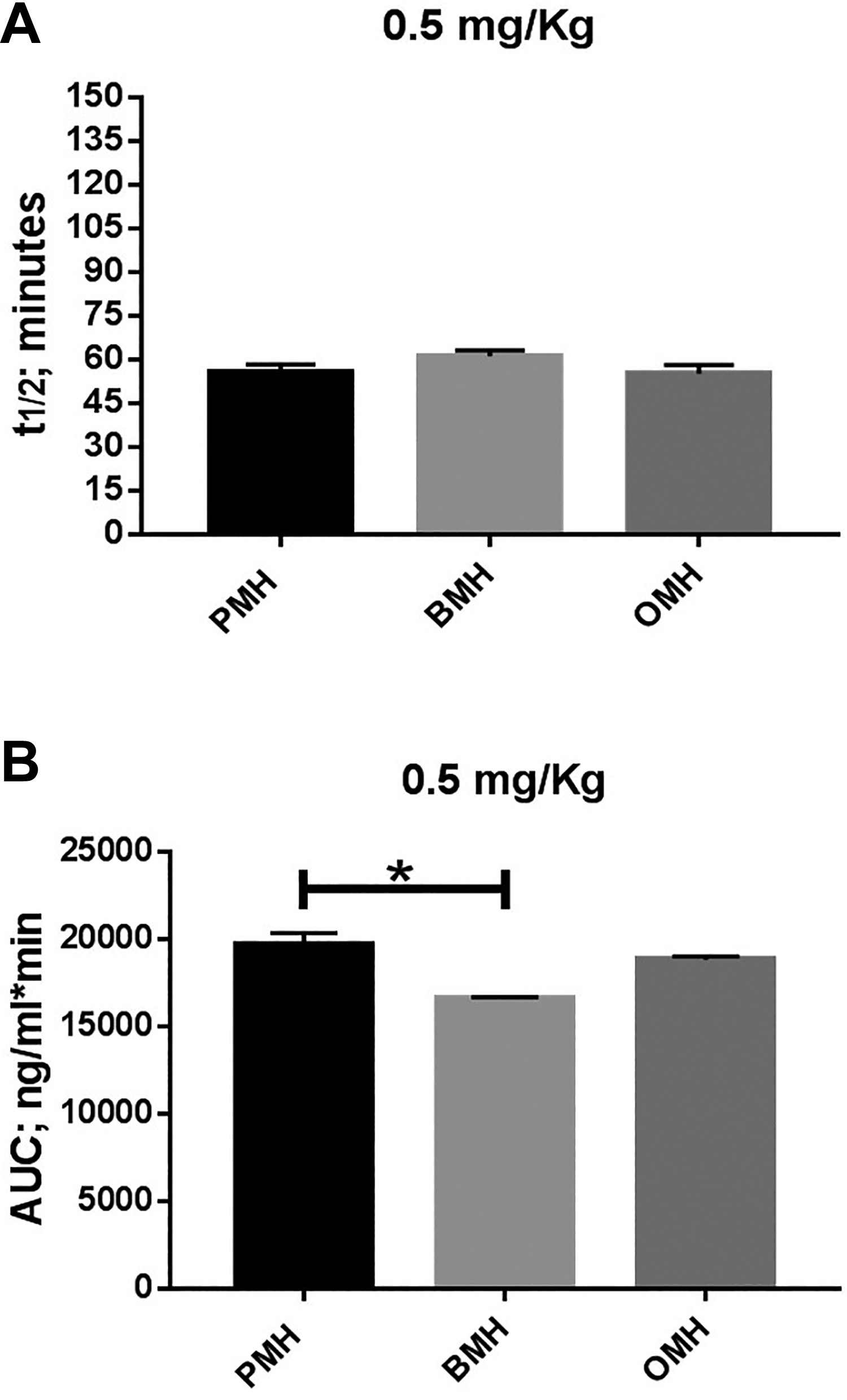
Estimation of pharmacokinetic parameters calculated from TFPI release assay after I.V. administration of 0.5 mg/kg heparins to non-human primates. In panel A, all heparins showed comparable half-life time (t½). BMH showed significant smaller AUC (p* < 0.05) compared to PMH. However, PMH and OMH showed comparable AUC as shown in panel B. The data represent the mean ± standard deviation (n = 4).
The estimation of pharmacokinetic parameters calculated from TFPI antigen release assay after IV administration of 100 and 50 U/kg heparins to non-human primates are shown in Figure 4. At the potency equated dosages (100 and 50 U/kg), all agents demonstrated a comparable half- life with a range of (90-101 mins) and (84-94 mins), respectively (Figure 4A and C). No significant differences in area under the curve were noted among all agents (Figure 4B and D). Post IV administration of 100 U/kg heparins, the AUC of BMH, OMH and PMH were 20435 ± 1698, 19955 ± 1521 and 19030 ± 1398 ng/ml*min, respectively (Figure 4B). Furthermore, the AUC of BMH, OMH and PMH post IV administration of 50 U/kg heparins were 14688 ± 823, 13242 ± 938 and 13721 ± 1034 ng/ml*min, respectively (Figure 4D).

Estimation of pharmacokinetic parameters calculated from TFPI release assay after I.V. administration of 100 and 50 U/kg heparins to non-human primates. In panels A (100 U/kg) and C (50 U/kg), all heparins showed comparable half-life time (t½) ranged from 90 mins to 100 mins. No significant differences in area under the curve (AUC) among all heparins post 100 and 50 U/kg drug administration as shown in panels B and D respectively. The data represent the mean ± standard deviation (n = 4).
Figure 5 shows the plasma concentration-time courses for each agent as determined by their effects on anti-Xa activity post IV administration of various dosages (0.5 mg/kg and 100 U/kg) to non- human primates (n = 4). At 15-, 30- and 60- minutes post-drug (0.5 mg/kg) administration, BMH exhibited significantly lower levels of factor Xa inhibition compared to PMH and OMH (*p < 0.05) as shown in Figure 5A. The peak plasma concentrations achieved following administration of OMH (2.3 ± 0.14 U/ml) and PMH (2.2 ± 0.18 U/ml) were comparable and greater than after the administration of BMH (1.5 ± 0.17 U/ml) (Figure 5A). At the potency equated dosages (100 U/kg), all agents demonstrated comparable levels of factor Xa inhibition at all time pints as shown in Figure 5B.

The pharmacodynamic (PD) effects of various heparins dosages (0.5 mg/kg and 100 U/kg) administered to primates as measured by the anti-Xa assay. Panel A shows the plasma concentration-time courses of each agent (0.5 mg/kg) as determined by effects on anti-Xa activity. At 15-, 30- and 60- minutes, the plasma concentrations of BMH was significantly lower than PMH and OMH (*p < 0.05). However, all agents showed comparable plasma concentrations at all time points after I.V. administration of 100 U/kg, as shown in panel B. The data represent the mean ± standard deviation (n = 4).
Discussion
TFPI is known to be an important physiologic anticoagulant. It plays a major role in the potentiation of the effects of heparins. 25,26 TFPI is released from the endothelial cell surface glycosaminoglycans after the administration of heparin and LMWHs, and accounts for approximately 1/3 of heparin’s anticoagulant activity. 25,26 Several previous studies have reported on the release of TFPI after the administration of heparin and LMWHs prepared from porcine intestinal mucosa. 25 –28
Due to the potential shortage of porcine heparin there has been an interest to use bovine and ovine heparins as an additional source for heparin production. The anticoagulant effects at equigravemetric dosages of these preparations have been previously reported. 29 It was found that bovine heparin showed a lower anticoagulant effect as expected, due to its lower anti-Xa and anti-IIa potency compared to porcine heparin (bovine 140 U/mg vs. porcine 190 U/mg). Besides the anti-Xa and IIa effects of heparin, the release of this inhibitor in circulation, upon parenteral administration of heparin and LMWHs, contributes significantly to the overall pharmacologic effects inhibited by these agents. The studies reported in this manuscript represent the very first investigation where heparins from 3 different sources are directly compared for their ability to release this mediator. As the inhibition of tissue factor is very important in the control of thrombogenesis, the relative release of this inhibitor is an important factor to consider in demonstrating the bioequivalence of unfractionated heparins obtained from different sources.
In the current study, the porcine, bovine and ovine heparins were compared at a fixed dosage of 0.5 mg/kg. The TFPI antigen levels were measured for a period of 2 hours at multiple time points. In these studies, while the porcine and ovine showed somewhat comparable release of TFPI antigen the bovine heparin exhibited relatively lower levels of TFPI release. At 15 minutes, both porcine and ovine heparin exhibited much higher levels of TFPI antigen in comparison to bovine heparin. This increase in TFPI antigen, gradually decreased over a period of time and reverted back to normal levels within 2 hours. Pharmacokinetic parameters such as t ½ were similar between the 3 agents, however, the AUC was significantly lower in the case of BMH. The release of TFPI antigen by heparin is dependent on several factors including degree of sulfation, molecular weight of the chain’s presence and their configuration. It was expected that TFPI would be higher in BMH since it has a higher molecular weight than both porcine and ovine heparin, however, it was lower. The relative differences in the degree of sulfation are minor, therefore, other factors may contribute to the release of TFPI by heparins of different origins.
In addition, studies on the USP potency adjusted heparins were carried out at 2 dosage regimens namely 100 units per kilogram and 50 units per kilogram after intravenous administration. At these dosages all heparins produced comparable release of TFPI which was dose dependent. Elimination kinetics were comparable. The t½ of both the 100 units per kilogram and 50 units per kilogram were comparable. The AUC was also comparable. It was interesting to note that the potency adjusted heparins from different sources in terms of USP units resulted in comparable release of TFPI on heparin. Thus, there may be a competition between binding to AT and the release of TFPI. TFPI release does not require AT binding. Heparin may bind disproportionately to AT and the endothelium to release TFPI.
In a previous report we have demonstrated that both USP potency adjusted heparins have comparable pharmacokinetics in terms of anti-Xa and IIa effects. 23 Therefore, the USP potency adjustment of heparins from different origin results in comparable pharmacodynamic responses for the release of tissue factor pathway inhibitor and inhibition of factor Xa and IIa. Our study further validates that potency adjustment of heparin from different sources will provide products with comparable pharmacodynamic properties. The inclusion of the measurement of TFPI may therefore be useful in the clinical trials designed to compare these agents.
Although, the currently approved invitro anti-Xa and anti-IIa methods are used for the invitro potency evaluation and standardization of heparins. These methods only measure anti-thrombin and heparin cofactor II mediated anticoagulant effects. Since heparin composition is based on only 20-30% components which exhibit this effect, the only approach to determine the additional biological effects is to measure such mediators as the TFPI antigen which is contributory to the biologic actions of this anticoagulant. Therefore, to compare the bioequivalence of potency adjusted heparins of different origin, it is important to demonstrate similar endogenous modulation such as TFPI antigen release. TFPI modulates tissue factor which is an important trigger for endogenous activation of coagulation system.
The heparin preparations used in these studies were all manufactured in accordance to the improved processes which included viral de-loading, and other purification methods to remove contaminants. The regulatory bodies such as FDA require documentation from each manufacturer in regard to the effective removal of contaminants and viruses. Viral de-loading processes are now standard practice included in the manufacturing processes of heparins. In the current studies, the dosages were selected to determine the dose response in terms of the release of TFPI antigen. In addition, they were selected to obtain data for the PK/PD measurements and represent dosages which is between therapeutic and surgical anticoagulation. These studies were designed in a cross over manner and the same animal group was used after applicable wash-out period of 2 weeks.
In this investigation, non-human primates (NHPs) were used because of their similarity to humans in physiology, neuroanatomy, reproduction, development, cognition, and social complexity. The physiological similarity between humans and NHPs means there is greater validity of the data obtained from primate models than other animal models (e.g. PK/PD). A few experimental limitations which could affect the outcome data. First, there is inter-animal variability (i.e. hepatic function, age, sex, and weight). Second, the results could also be influenced by practical limitations on the number of blood samples that were collected and by the number of primates that was included in each treatment group. Additional confirmative studies are required to determine the TFPI functional levels produced by the same agents.
Conclusion
Our studies propose that heparin obtained from bovine, ovine and porcine sources should be considered as biosimilars (a class of drugs) rather than distinct pharmacological entities with reference to their anticoagulant activities. Biologic potency adjustment for heparin substances with the anti-Xa and anti-IIa ratio of 1.0, regardless of the mass per volume considerations will open several options in formulating heparins for wider applications. Beside the Serpins mediated anti-Xa and anti-IIa activities, the release of endogenous TFPI plays an important role in the mediation of its pharmacologic actions. The heparins manufactured from different sources may have compositional differences, invitro cross referencing of these agents against the USP reference standard results in products of comparable anticoagulant and antiprotease potencies. The current studies demonstrate that potency adjusted heparin of bovine, ovine and porcine origin exhibit comparable TFPI antigen release in primates after intravenous administration.
Footnotes
Acknowledgments
The authors are thankful to the staff of the Hemostasis Research Laboratory for their skillful assistance in completing this study. Special thanks to Mr. Jonas Kingo of Aniara (Mason, Ohio) for helpful suggestions for the USP assays and the anti-Xa and anti-IIa kits used in these studies. The authors are also grateful to Ms. Valentina Baigorria of Kin Master, Brazil for providing the bovine mucosal heparin samples and to Mr. Yimimg Yao of Ronnsi Pharmaceutical, China for the supply of ovine heparins. We are also thankful to Ms. Erin Healy Erickson for her expert assistant in preparing this manuscript. We are grateful to Dr. Ken Byron, Interim chair of the Department of Pharmacology and Neurosciences for his support and guidance to facilitate these studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
