Abstract
Introduction:
Currently used unfractionated heparins (UFHs) and low molecular weight heparins (LMWHs) are derived from porcine intestinal mucosa. However, heparins have also been manufactured from tissues of other mammalian species such as cow (Bovine) and sheep (Ovine). Protamine sulphate (PS) is an effective inhibitor of heparin and is used clinically to neutralize both LMWH and UFH. In this study, we determined the PS neutralization profile of these agents in non-human primate model using anti-Xa and anti-IIa methods.
Material and Methods:
UFHs obtained from bovine, ovine and porcine mucosal tissues and their respective depolymerized LMWHs were administered at both, gravimetric (0.5 mg/kg) and potency adjusted (100 U/kg) dosages regimen intravenously to individual groups of primates in cross over studies. PS was administered at a fixed dosage and the relative neutralization of these anticoagulants was measured utilizing amidolytic anti-Xa and anti-IIa methods.
Results:
These studies have demonstrated that, the equi-gravimetric dosages of BMH, PMH and OMH have comparable PS neutralization profiles. At potency adjusted dosages, all UFHs were completely neutralized by PS. Although comparable, the LMWHs were not fully neutralized by PS in both the anti-Xa and anti-IIa assays. PS was more efficient in neutralizing the anti-IIa effects of LMWHs.
Conclusion:
Heparins of diverse origins showed comparable neutralization profiles by PS in the amidolytic anti-Xa and anti-IIa assays.
Background
Unfractionated Heparin (UFH) was first used as an anticoagulant in the early part of the 20th century. 1 The controversy surrounding its discovery stems back to 1916 when a medical student named Jay McLean isolated a fat soluble anticoagulant compound (phosphatides) from the liver of dogs. 1 This took place in the laboratory of William Henry Howell, who later presented findings related to the discovery of a water soluble carbohydrate compound which was distinct from the discovery by McLean. 1 Since then, the pharmaceutical industry has worked to optimize the process of its purification. UFH is a highly sulfated glycosaminoglycan (GAG) that is used extensively as an anticoagulant. It consists of repeating disaccharide units, containing iduronic acid (or glucuronic acid) and glucosamine, exhibiting variable degrees of sulfation. 2 Glycosaminoglycan heparin is processed to pharmaceutical grade heparin, which can be filtered or subjected to controlled depolymerization to produce low molecular weight heparin (LMWH, Mw range: 3500-7500 Da) and ultra low molecular weight heparin (ULMWH, Mw range: 2000-3500 Da). 2,3 The low molecular weight fragment exhibits a molecular weight of approximately one third of its original chain. 3 Each different LMWH has distinct therapeutic and pharmacological properties due to the chemical structure and degree of depolymerization. 4
Although UFH is the cornerstone of treatment of various thrombotic diseases, including venous thromboembolism (VTE), deep vein thrombosis (DVT), and pulmonary embolism (PE), it exhibits some pharmacologic disadvantages such as bleeding and heparin induced thrombocytopenia (HIT). LMWHs are an important alternative to UFH with several advantages, such as higher bioavailability, enhanced safety (less bleeding), greater convenience (fewer injections), and better efficacy (lower rate of thrombotic events). 5,6 Furthermore, LMWHs are used in the management of thromboembolism that is a well-recognized complication of various malignant diseases. 7 UFHs are administered parenterally, often continuously due to its short half-life and over 0.5 billion doses are required per year. On the other hand, LMWHs are subcutaneously administered as they have better bioavailability compared to UFHs. 8 Currently, most of the UFHs used clinically are derived from (porcine intestinal mucosa). However, in various parts of the world heparins are also manufactured from mammalians like cow and sheep. 9
Protamine sulphate (PS) is a reversing agent of heparin and is used clinically to neutralize both LMWH and UFH. In 1868, protamine was identified by Friedrick Miescher as a nitrogenous base bound to nuclear material of Rhine salmon sperm heads. In 1937, Chargraff and Olsen demonstrated that protamine sulfate could neutralize the anticoagulant action of heparin. 10 Today, protamine is usually commercially prepared from salmon milt into a dried powder or solution. It has the ability to reverse the anticoagulant activity of heparin. Protamine is arginine-rich (nearly 67%) and strongly basic, Small nuclear proteins (45 kDa) replace histones late in the haploid phase of spermatogenesis and are believed to be essential for sperm head condensation and DNA stabilization. 11 Protamine has a mild anticoagulant effect when administered alone. However, when it is given in the presence of heparin (which is strongly acidic), ionic forces form a stable precipitate. In this stable precipitate an intermolecular charge neutralization occurs, thereby causing the neutralization of heparin activity. 11 Various factors influence the interaction of protamine and heparin including individual molecular weight of a given protamine or heparin, charge type, and charge density. 12 Heparin’s affinity for protamine is directly influenced by many factors such as molecular weight, degree of sulfation, and position of sulfate residues on the heparin chain. 13 Dawes and Pepper (1982) determined that degree of sulfation is the most important determinant of heparin’s affinity to protamine. The binding of protamine to heparin most likely dissociates heparin from AT, thereby breaking the heparin/AT complex and neutralizing heparin’s anticoagulant activity. 14
Many factors have led to shortages and an increased cost of porcine derived anticoagulants. First, the high demand as open-heart surgeries, hemodialysis and intervention cardiovascular procedures are usually carried out with UFHs. Second, porcine UFHs are widely and extensively used in the manufacturing of LMWHs and ULMWHs. Finally, global pig’s population is at risk as there are about 2 billion pigs worldwide and people kill over one billion pigs annually (an average of 23 million pigs a week). 15 With the advances in technology and manufacturing processes, higher quality and contaminants-free bovine and ovine UFHs are now available for clinical and research purposes. 16 Recently, an outbreak of African swine fever in China has reduced the availability of porcine tissues due to profound reduction in big populations. Hundreds of millions of animals have died or expected to die due to disease or culling resulting in mark reduction of pig population. 17 Currently Chinese authorities and manufactures are exploring the options to solve this problem. As the U.S. heparin supply is dependent on Chinese manufacturers and up to 70% of the U.S. heparin is imported from China, the U.S. government is concerned on the dependence on one source and the potential impact of its shortage on patient usage. 18
In this study, we projected that, BMH, OMH and PMH have comparable PS neutralization profiles when administered at equi-gravimetric dosages. However, potency equated BMH may require higher amount of PS to be completely neutralized. Furthermore, we projected that PS will fail to fully neutralize all LMWHs due to PS neutralization is largely dependent on molecular weight, leading to the implication that LMWHs containing a larger proportion of small oligosaccharides will not be as effectively neutralized. 19 Assay dependent variations in the neutralization profile of both UFHs and LMWHs are well known. In this study, we have focused on the amidolytic anti-Xa and anti-IIa activities which reflect on the molecular weight of the chains and the AT binding sequence.
Materials and Methods
Test Agents
Porcine mucosal heparin (PMH) was obtained from Medefil Inc. (Glendale Heights, IL, USA) and exhibited a USP potency range of 185-200 U/mg. Bovine mucosal heparin (BMH) was obtained from KinMaster Indústrias Químicas (Passo Fundo, Brazil) and exhibited a USP specific activity of 130-140 U/mg. Ovine mucosal heparin (OMH) was obtained from Ronnsi Pharmaceutical (Jiangsu, China) and exhibited a specific activity of 195-208 U/mg. Branded enoxaparin was commercially obtained (Sanofi Aventis, France), bovine and ovine enoxaparins were obtained from Ronnsi Pharmaceutical (Jiangsu, China). The USP anti-Xa and Anti-IIa potencies of all 3 LMWHs were comparable and ranged of 95-105 U/mg and 30-40 U/mg respectively. For the potency adjustment studies of BMH, mass per volume adjustments were made to equate the potency to USP referenced porcine UFH. No adjustments were needed for the LMWHs as the unit potency for these agents are comparable at 100 U/mg for anti-Xa and 30-40 U/mg for the anti-IIa activities. All samples were obtained as white powders which were stored at room temperature in a desiccator. These samples were weighed on a Mettler balance and 0.9% NaCl was used as a diluent to make stock solutions of 10 mg/ml for the in vivo and in vitro testing.
Reagents and Commercial Kits
Protamine sulfate single dose vials were obtained from Fresenius Kabi pharmaceutical company (Melrose Park, IL, USA). Heparin anti-Xa and anti-IIa USP kits were obtained from HYPHEN Biomed (France). The anti-Xa and anti-IIa USP kits reference numbers were 221010 and 221025 respectively.
Primates
Twelve male and female Rhesus monkeys (Macaca mulatta) weighing 4.0-16.0 kg were used for PS neutralization studies. All research was conducted in compliance with the guidelines set forth by the Institutional Animal Care and Use Committee (IACUC), Loyola University Chicago Health Sciences Campus and the Principles of Laboratory Animal Care.
Dosing
Each heparin was dissolved in 0.9% physiologic saline prior to administration to primates. Squeeze cages were used to anesthetize the primates with an intramuscular injection of ketamine (10 mg/kg) and xylazine (1-2 mg/kg) based on the most recent charted weight.
Before injection of heparins, the primates were weighed to accurately determine the dose of the test agent, skin over the saphenous vein was shaved as necessary and then cleaned using betadine and alcohol. A baseline blood sample was collected by venipuncture of the saphenous vein. Heparins were administered at an equi-gravimetric dose (0.5 mg/kg) or potency-adjusted dose (100 U/kg) intravenously via the saphenous vein. However, LMWHs were administered at a dose of 1 (mg/kg) intravenously via the saphenous vein. 15 minutes post drug administration, a second blood sample was collected, and primates were immediately treated with a 1 mg/kg IV dose of protamine sulfate or an equivalent volume of saline. Additional blood samples were collected at 15-, 45- & 120-minutes post PS administration. These experiments detailed here are non-terminal. As such, following a minimal 1-week drug washout period, a primate was used in another treatment group. Four primates were dosed with each agent. After administration of the test agent and after collection of each of the subsequent blood samples, primates were returned to their cages and monitored continuously until they had awakened from anesthesia and were able to sit up on their own. 10-15 minutes prior to each blood draw, the sedation state of the animal was assessed. Additional doses of ketamine were given intramuscularly as needed.
Sample Collection
All blood samples were drawn using a double syringe technique, employing a 21-gauge butterfly needle. After an initial ∼1 ml volume (discard blood) was collected, the syringe was changed, and a 2.7 ml sample was drawn and placed into a tube containing 0.3 ml 3.2% sodium citrate. Citrated blood samples were centrifuged, and aliquots of plasma were stored for ex-vivo analysis.
Sample Analysis
Plasma concentrations of the various agents was plotted against corresponding optical densities in the factor IIa or factor Xa assays using graphing software, SigmaPlot for Windows version 12.3 (Systat Software, San Jose, CA) and best-fit curves was made. 20,21 The drug concentration in each primate blood sample in terms of anti-IIa and anti-Xa activities was determined by extrapolation. The area under the plasma concentration time curve (AUC) was calculated from the extrapolated plasma concentrations using the PKSolver® software for Microsoft Excel. 22,23
Statistical Analysis
All values were expressed as mean ± standard deviation. Data were analyzed using Student’s unpaired t-test or ANOVA for group wise comparison. Based on previous studies, it was determined that, individual groups of animals comprised of 4 primates was sufficient to achieve statistical significance in each experiment. In all cases a P value < .05 was considered statistically significant. Computed P values were presented for each analysis. Graphs with data and error bars were drawn using Microsoft Excel (Microsoft ® Corporation, Redmond, WA) or GraphPad Prism version 8.0. for Windows (GraphPad Software, San Diego, CA).
Results
The time courses of the amidolytic anti-Xa and anti-IIa activities of PMH, BMH and OMH (100 U/kg & 0.5 mg/kg) post I.V. administration of saline (control) and PS (0.5 mg/kg) are shown in Tables 1 & 2 respectively. Administration of potency adjusted dosages (100 U/kg) of each UFH followed by saline I.V. injection resulted in comparable drug levels through all time points as determined by anti-Xa and anti-IIa assays with a maximum value of 1.25 ± 0.2 U/ml and 1.26 ± 0.25 U/ml respectively (Table 1 & Figure 1). However, the gravimetric dosage (0.5 mg/kg) of BMH exhibited a significant lower drug levels at 15 and 45 mins (P* <.05) post drug administration as determined by anti-Xa assay; 0.81 ± 0.09 U/ml & 0.56 ± 0.02 U/ml respectively (Table 1 & Figure 1A). Figure 1B shows the plasma concentration-time courses for each agent as determined by their effects on anti-IIa activity. BMH-treated primates exhibited a significant lower drug levels at 15 and 45 mins (P* < .05) post drug administration; 0.79 ± 0.12 U/ml & 0.58 ± 0.09 U/ml respectively compared to the potency adjusted dosages (Table 1 & Figure 1B). PMH, BMH and OMH at various dosages showed comparable PS neutralization profiles at all time points as determined by both assays. However, slightly higher residual levels observed in the BMH (100 U/kg) treated primates as determined by both assays (Figure 1A&B). These observed differences were not significant when compared to OMH and PMH.
Comparative Plasma Concentration-Time Courses of Various Unfractionated Heparins Post Saline I.V. Injection as Determined by Various Antiprotease Assays in Non-Human Primates (n = 4).

Abbreviations: BMH, bovine mucosal heparin; OMH, ovine mucosal heparin; PMH, porcine mucosal heparin; Xa, factor Xa; IIa, thrombin. All results represent the mean (±) standard deviation (n = 4).
Comparative Plasma Concentration-Time Courses of Various Unfractionated Heparins Post Protamine Sulfate (PS) I.V. Injection at a Dose of 0.5 (mg/Kg) as Determined by Various Antiprotease Assays in Non-Human Primates (n = 4).

Abbreviations: BMH, bovine mucosal heparin; OMH, ovine mucosal heparin; PMH, porcine mucosal heparin; Xa, factor Xa; IIa, thrombin. All results represent the mean (±) standard deviation (n = 4).

Comparative neutralization profile of various dosages of UFHs post protamine sulfate (PS) I.V. injection at a dose of 0.5 (mg/kg) as determined by various antiprotease assays in non-human primates (n = 4). Anti-Xa assay (A) and Anti-IIa assay (B). PMH, BMH and OMH at various dosages showed comparable PS neutralization profiles at all time points as determined by both assays. However, BMH at 100 U/kg wasn’t completely neutralized at 15- and 45-mins time points post PS injection as determined by both assays. The data represent the mean ± standard deviation (n = 4).
The time courses of the amidolytic anti-Xa and anti-IIa activities of various LMWHs (1 mg/kg) post I.V. administration of saline (control) and PS (0.5 mg/kg) are shown in Tables 3 & 4 respectively. Administration of various LMWHs followed by saline I.V. injection (Control) resulted in comparable drug levels through all time points as determined by anti-Xa and anti-IIa assays with a maximum value of 1.29 ± 0.19 U/ml and 0.32 ± 0.01 U/ml respectively (Table 3 & Figure 2). All LMWHs were partially neutralized by PS by the same degree as determined by both assays (Figure 2). 15 minutes post PS administration, plasma concentration of all LMWHs were comparable and ranged from 0.94 ± 0.14 U/ml to 0.97 ± 0.13 U/ml and from 0.14 ± 0.03 U/ml to 0.16 ± 0.02 U/ml as determined by anti-Xa (Figure 2A) and Anti-IIa (Figure 2B) assays respectively. The same trend was observed at all time points (Figure 2 & Table 4).
Comparative Plasma Concentration-Time Courses of Various Low Molecular Weight Heparins Post Saline I.V. Injection as Determined by Various Antiprotease Assays in Non-Human Primates (n = 4).

Abbreviations: B, bovine; O, ovine; P, porcine; LMWH low molecular weight heparin; Xa, factor Xa; IIa, thrombin. All results represent the mean (±) standard deviation (n = 4).
Comparative Plasma Concentration-Time Courses of Various Low Molecular Weight Heparins Post Protamine Sulfate (PS) I.V. Injection at a Dose of 0.5 (mg/Kg) as Determined by Various Antiprotease Assays in Non-Human Primates (n = 4).

Abbreviations: B, bovine; O, ovine; P, porcine; LMWH low molecular weight heparin; Xa, factor Xa; IIa, thrombin. All results represent the mean (±) standard deviation (n = 4).

Comparative neutralization profile of various LMWHs post protamine sulfate (PS) I.V. injection at a dose of 0.5 (mg/kg) as determined by various antiprotease assays in non-human primates (n = 4). Anti-Xa assay (A) and Anti-IIa assay (B). All LMWHs were partially neutralized by PS by the same degree as determined by both assays. The data represent the mean ± standard deviation (n = 4).
Figure 3 shows AUC for protamine sulfate (PS) neutralization time curves of various UFHs post I.V. injection at a dose of 0.5 (mg/kg) as determined by various antiprotease assays. BMH at 0.5 mg/kg showed significant smaller AUC (P* < .05) of 105 ± 7.9 post saline I.V. injection compared to others as measured by both assays. All UFHs at 100 U/kg dosages showed comparable AUC post saline injection at a range of (150.8 ± 14.5) to (171 ± 11.8) U/kg*min as determined by anti-Xa assay (Figure 3A) and a range of (150.5 ± 9.9) to (165.6 ± 16.4) U/kg*min as determined by anti-IIa assay (Figure 3B). AUC % reductions for PMH, OMH and BMH as determined by anti-Xa were comparable and which were 75.5% ± 2.3%, 77.9% ± 4.6% and 73.8% ± 2.8% respectively. Similar AUC % reductions were observed when heparin levels were determined using an anti-IIa assay, which were 75.8% ± 3.1%, 79.3% ± 2.6% and 74.9% ± 1.8% for PMH, OMH and BMH respectively (Figure 3).
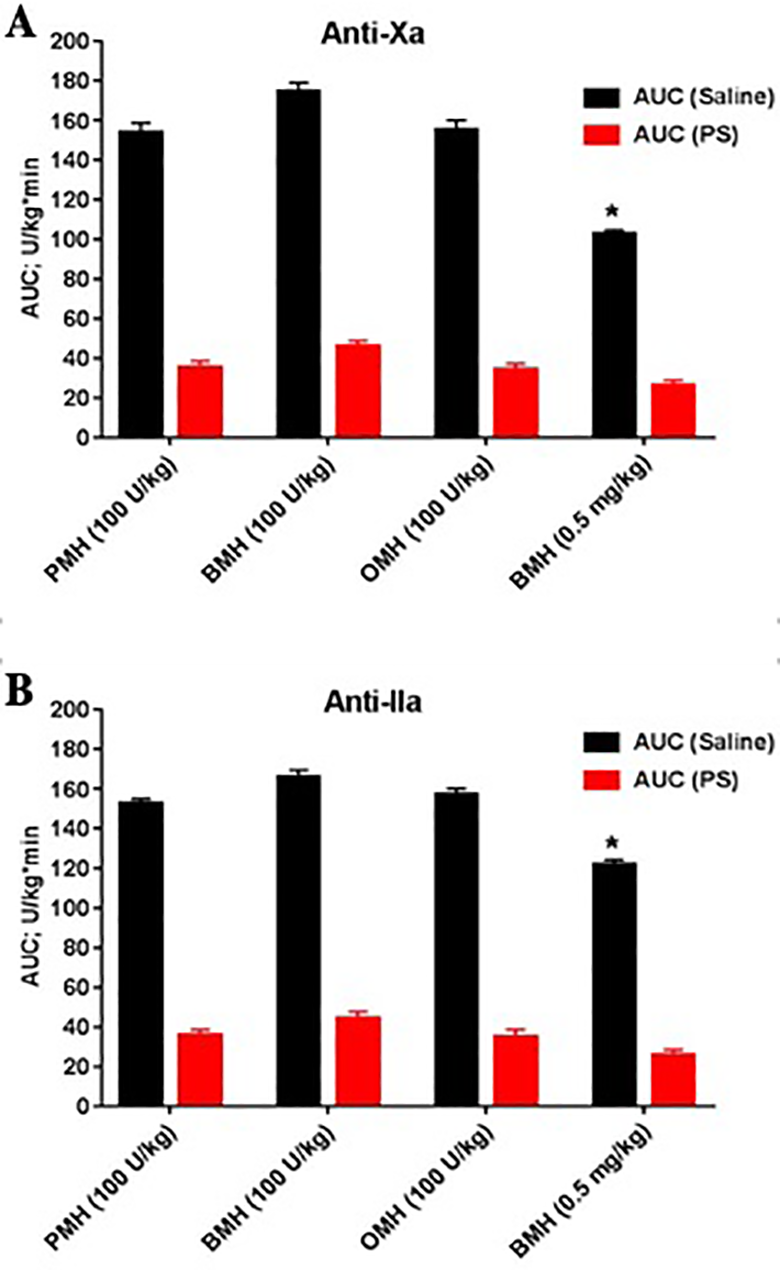
Comparative AUC for protamine sulfate (PS) neutralization time curves of various UFHs post I.V. injection at a dose of 0.5 (mg/kg) as determined by various antiprotease assays in non-human primates (n = 4). Anti-Xa assay (A) and Anti-IIa assay (B). BMH at 0.5 mg/kg showed significant smaller AUC (P* < .05) post saline I.V. injection compared to others as measured by both assays. All UFHs at various dosages showed comparable AUC-% reduction as determined by both assays. The data represent the mean ± standard deviation (n = 4).
AUC for protamine sulfate (PS) neutralization time curves of various LMWHs post I.V. injection at a dosage of 0.5 (mg/kg) as determined by various antiprotease assays (Figure 4). All LMWHs at 1 mg/kg dosages showed comparable AUC post saline injection in a range of (149.5 ± 5.8) to (153.5 ± 7.1) U/kg*min as determined by anti-Xa assay and in a range of (34.3 ± 2.3) to (35.6 ± 4.9) U/kg*min as determined by anti-IIa assay. All LMWHs at 1 mg/kg dosage were partially neutralized post PS I.V. injection as determined by both assays. Each showed comparable AUC-% reductions of a range of (19.2% ± 2.3% to 22% ± 3.8%) and (50.5% ± 3.8% to 53.4% ± 6.8%) as determined by anti-Xa and anti-IIa assays respectively (Figure 4).
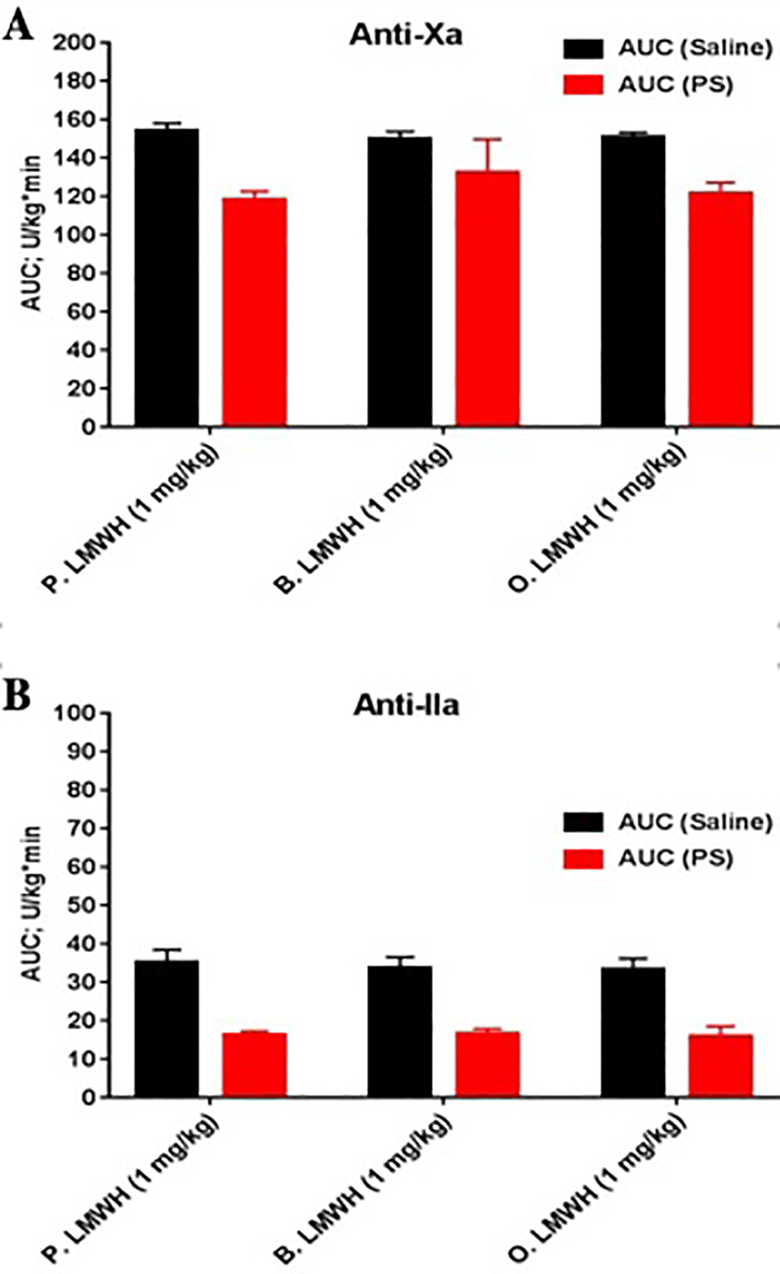
Comparative AUC for protamine sulfate (PS) neutralization time curves of various LMWHs post I.V. injection at a dose of 0.5 (mg/kg) as determined by various antiprotease assays in non-human primates (n = 4). Anti-Xa assay (A) and Anti-IIa assay (B). All LMWHs showed comparable AUC-% reduction as determined by both assays. The data represent the mean ± standard deviation (n = 4).
Discussion
The ability to neutralize UFHs is an important factor in their clinical use. PS is a polycationic polymer rich in basic amino acids such as proline, histidine and arginine. 11,24 PS is used for the neutralization of UFHs and LMWHs such as enoxaparin, 25 -27 however its neutralization of LMWHs is not as effective as with UFHs. Other polycationic substances including PF4 have also been tested for their ability to neutralize UFHs. 28,29 However, the results have been inconclusive and the use of PF4 may lead to heparin induced thrombocytopenia (HIT). Additionally, a catatonically modified polysaccharide antidote to neutralize UFHs has been reported in non-clinical settings. 29 There have been several other antidotes with diverse chemical structures which have been proposed for the neutralization of UFHs. 29 Heparinases are members of a class of enzymes which can degrade heparin with varying specificities. 30 Heparinase-I is a flavobacterial enzyme which has been tested in both preclinical and clinical settings for the neutralization of UFHs. 31 Despite these developments, PS remains the sole antagonist for the control of bleeding and neutralization of circulating heparin following surgical procedures.
The studies reported in this manuscript describe the in vivo neutralization of UFHs and LMWHs obtained from bovine, ovine and porcine sources following their administration at equigravemetric and potency-adjusted dosages. In this investigation, LMWHs produced by benzylation followed by alkaline hydrolysis were used for the neutralization studies. A previous publication has reported on the comparison of UFHs and LMWHs produced by using mucosal tissue of bovine, ovine and porcine origins. 18 Additional publication provide evidence of the equivalence of bovine and porcine UFHs at potency-adjusted levels. 32 It is noteworthy that, the bovine heparin preparations exhibit a lower potency in comparison to porcine heparin at equivalent mass levels. However, supplementation or administration of these heparins on a unit basis results in comparable anticoagulant activities. A study comparing the neuralization profiles of heparins from different origins has not been reported previously. This study represents the first integrated investigation on the in vivo neutralization profile of UFHs and LMWHs in a primate model. This model has been extensively used in the study of the PK of heparins following both IV and SC administration. 32
The neutralization profile of UFHs from various origins was investigated at a dosage of 100 U/kg after IV administration and at a gravimetric dosage of 0.5 mg/kg IV. In this study PS administered at a dose of 0.5 mg/kg effectively neutralized all 3 UFHs in an identical manner as measured by anti-Xa and anti-IIa assays.
Since all LMWHs exhibited a comparable in vitro potency of 100 U/mg, these agents were administered at 1 mg/kg intravenously and their neutralization was studied after administration of PS at a dose of 0.5 mg/kg. The LMWHs exhibited major differences in the anti-Xa and anti-IIa profiles in contrast to heparins which were comparable in both assays. The relative neutralization of the anti-Xa activity was much weaker in contrast to the anti-IIa activities of LMWHs. At the same time the elimination of the anti-IIa actions after PS neutralization was faster than that observed with the anti-Xa assay. This observation suggests that, the higher MW chains responsible for the anti-IIa activity were more effectively complexed with PS in contrast to the lower molecular weight chains which only express anti-Xa activity. This data is consistent to previous findings where differential neutralization of LMWHs was reported. 25
BMH at the 0.5 mg/kg dosage showed lower anti-Xa and anti-IIa activities in comparison to PMH and OMH at comparable dosages of 100 U/kg (equivalent to 0.5 mg/kg). Therefore, the anti-Xa and anti-IIa activities of BMH were lower in comparison to PMH and OMH. However unlike with LMWHs, PS completely neutralized the anti-Xa and anti-IIa effects of UFHs, effects which were sustained during the study period of 2 hours. There were no differences observed between the different UFHs studied in terms of the extent of neutralization. This observation is in discordance with the previous findings where BMH required higher amounts of PS for the neutralization of bleeding in CABG (Cardiopulmonary Bypass Surgery). 33 However, in this clinical trial relatively higher dosages of PMH and BMH were used. In this clinical study, higher amounts of PS were required to neutralize UFHs which were dosed at comparable level.
In the current study, PS was used at a fixed dosage of 0.5 mg/kg with both the UFHs and LMWHs. The average molar concentration of UFH components was much lower than the average molar concentration of LMWHs. This may be one reason for the marked differences in the neutralization profile of the 2 groups of heparins. Regardless of these differences, the relative neutralization of UFHs and LMWHs from various origins was comparable. It would be of interest to determine the impact of higher doses of protamine on LMWH neutralization. Although LMWHs are usually administered subcutaneously, these agents have also been administered intravenously for surgical and interventional procedures. Limited available data suggests that intermittent bleeding complications with LMWHs may be manageable by repeated administration of PS. 34
The PK profile of the UFHs of different origins showed comparable trends and the AUC for both the anti-Xa and anti-IIa activities were similar for the potency adjusted agents. BMH at 0.5 mg/kg showed a proportionately lower AUC with both the anti-Xa and anti-IIa assays. This is due to the lower potency of this anticoagulant in comparison to PMH and OMH. PS administration resulted in comparable reductions of the anti-Xa and anti-IIa activities of all heparins.
The PK profile of the LMWHs was comparable in the anti-Xa and anti-IIa assays, however unlike heparins, the LMWHs exhibited markedly reduced anti-IIa activities. The AUC values for the anti-Xa activities after PS administration showed comparable trends however unlike UFHs, only showed partial decrease in the range of 20%-30% for the anti-Xa activity. While minor differences were noted however there were not statically significant. The anti-IIa effects as calculated on the basis of the AUC were comparable among the LMWHs tested but were much lower when compared to the anti-Xa activity. The anti-IIa activity of each LMWH was equally neutralized (∼40%-50%) by PS.
The current studies have several limitations. A fixed dose of PS was used with both the UFHs and LMWHs. A higher PS dosage of 1-2 mg/kg may have provided additional insight of the neutralization of the LMWHs. The duration of the study was also limited to a 2-hour period, which precluded determining whether the extent of heparin rebound differs with the various heparins. Such a study may relevant data in a time period of 12 hours. Additionally, in this study only anti-Xa and anti-IIa activities were monitored. Global anticoagulant assays such as the activated partial thromboplastin time (aPTT) and thrombin generation inhibition (TGA) assays may provide additional information of the relative neutralization of UFHs and LMWHs. Due to the limited availability of the number of primates used and other regulatory restrictions, a predetermined statistical power analysis and sample size calculations were not carried out.
Despite these limitations, these studies clearly showed that, potency adjusted UFHs are effectively neutralized by PS. However, the LMWHs are only partially neutralized by PS at the dosages studied. Nonetheless, the neutralization profile of all LMWHs were comparable at the dosages studied. An increased dosage of PS may provide more effective neutralization of these agents for the IV settings.
The studies reported in this manuscript were carried out with IV dosing of both the UFHs and LMWHs. Thus, this data is relevant to surgical and interventional indications for these agents. LMWHs are mostly used SC and the absorption profile follows a different PK profile which will require periodic administration of PS which is only administered IV and follows much faster elimination kinetics. The preliminary results presented in this manuscript suggest that additional studies are needed to develop effective protocols for the neutralization of the SC administered LMWHs by PS. Furthermore, the data generated in these studies is based on the use of non-human primate model and warrant validation studies in clinical trials in human.
Conclusions
In conclusion, the data presented in this manuscript validate the hypothesis that administration of potency adjusted UFH doses produce comparable anti-Xa and anti-IIa levels, which are effectively neutralized. In contrast, while the LMWHs exhibit differential anti-Xa and anti-IIa activities when compared to UFHs, all agents exhibited comparable profile and no species dependent differences were noted. PS differentially neutralized the anti-Xa and anti-IIa activities of LMWHs which was much weaker when compared to UFHs. Nevertheless, the LMWH of various origins exhibited similar in vivo neutralization profile. Moreover, these findings require clinical validation in adequately powered human trails.
Footnotes
Acknowledgments
The authors are thankful to the staff of the Hemostasis Research Laboratory and the comparative medicine facility of Loyola University, Health science center for their skillful assistance in completing this study. Special thanks to Mr. Jonas Kingo of Aniara (Mason, Ohio) for helpful suggestions for the USP assays and the anti-Xa and anti-IIa kits used in these studies. The authors are also grateful to Ms. Valentina Baigorria of Kin Master, Brazil for providing the bovine mucosal heparin samples and to Mr. Yiming Yao of Ronnsi Pharmaceutical, China for the supply of ovine heparins. We are also thankful to Dr Meharvan Singh, Vise provost for research at Loyola University and Dr Eva Wojcik, Chair of the Department of Laboratory Medicine and Surgery for their encouragement and support for these studies. The authors also acknowledge the partial funding of the studies through Cardiovascular Institute and the Department of Pharmacology and Neuroscience.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Walter Jeske received research funding from KinMaster, Brazil, whereas Jawed Fareed received research funding from Ronnsi, China.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Received partial funding from Cardiovascular Institute and the Department of Pharmacology and Neuroscience.
