Abstract
Purpose:
The role of low-molecular-weight heparin (LMWH) in managing nonmassive pulmonary embolism (PE) is well known. In unstable cases, especially after thrombolytic therapy for massive PE, unfractionated heparin (UFH) is preferred for PE management. This study aimed to investigate the effectiveness and safety of LMWH after thrombolytic therapy.
Methods:
A prospective, observational multicenter trial was performed in 249 patients with acute PE who required thrombolysis. Massive and submassive PEs were categorized into 2 groups depending on whether they were treated with LMWH or UFH after thrombolytic treatment. The primary end point was all-cause mortality during the first 30 days; the secondary end point included all-cause mortality, nonfatal symptomatic recurrent PEs, or nonfatal major bleeding.
Results:
The mean age at diagnosis was 60.7 ± 15.5 years. The PE severity was massive in 186 (74.7%) patients and submassive in 63 (25.3%). The incidence of all-cause 30-day death was 8.2% and 17.3% in patients with LMWH and UFH, respectively (P = .031). Major hemorrhage occurred in 4% (n = 5) and 7.9% (n = 10) of patients and minor hemorrhage occurred in 9% (n = 11) and 13.4% (n = 17) of the cases treated with LMWH and UFH, respectively.
Conclusion:
These results suggest that LMWH treatment can be used safely in patients with PE after thrombolytic therapy.
Introduction
Pulmonary embolism (PE) remains poorly understood, despite advances in diagnosis and therapy. 1 Early anticoagulation has been the cornerstone of PE treatment for many years and is lifesaving. The European and American guidelines recommend unfractionated heparin (UFH) as the only compound for concomitant use with thrombolysis. 2,3 However, patients with low-risk PE may be candidates for anticoagulation with low-molecular-weight heparin (LMWH). The LMWH and UFH have been shown to have equivalent efficacy and safety for the management of nonmassive PE. 4 On the other hand, LMWH has been shown to be superior to UFH in patients with deep venous thrombosis (DVT) and in those with DVT having PE because LMWH reduces mortality, recurrent thrombosis, and serious bleeding events. 5 The effect of using LMWH after thrombolytic treatment when managing acute massive PE is not clear; many clinicians continue to treat acute PE, which is a high-risk situation, with UFH. We aimed to compare UFH and LMWH management after thrombolytic treatment in acute massive and submassive PEs.
Methods
Study Design
This study was conducted between January and November 2013 as part of a large prospective, multicenter, and observational study in 25 centers in Turkey. The study population included 249 patients with the diagnosis of PE confirmed by multidetector computed tomography (CT). The study protocol was approved by the local ethics committee at the coordinating center, and written informed consent was obtained from all patients.
Patients and Setting
Consecutive patients older than 18 years of age with clinically suspected acute PE were considered for the study. The study consisted of 249 patients who were admitted with massive and submassive PEs. Before the patients could be included, PE had to be objectively documented by multidetector CT. All patients’ demographic characteristics and clinical and laboratory parameters were obtained from the hospital’s electronic database. A Wells score for PE was calculated for each patient. The results of imaging studies and laboratory tests (
The PE severity was classified into 3 groups according to European Society of Cardiology guidelines: high risk (patients with shock or hypotension), intermediate risk (presence of a positive marker for myocardial injury or right ventricular dysfunction), or low risk (absence of right ventricular dysfunction or negative marker of myocardial injury). 6
Inclusion criteria were hemodynamic instability and cardiogenic shocks accompanied by vital signs and echo findings. Exclusion criteria were absolute contraindications for thrombolytic therapy or nonmassive PE. A total of 186 (74.7%) patients with massive PE and 63 (25.3%) patients with submassive PE were classified. High and moderate risks of mortality were taken into consideration, after which the decision to use thrombolytic therapy was made. Thrombolytic therapy was administered to each patient in the first 14 days after onset of the symptoms. The patients received either LMWH or UFH after thrombolytic therapy. Both groups continued with warfarin, with the target-activated partial thromboplastin time being defined as 60 to 80 seconds; oral anticoagulants were used for at least 3 months.
Outcomes
Thirty-day clinical follow-up data were obtained for all patients. The primary end point was all-cause mortality during the first 30 days; the secondary end point included all-cause mortality, nonfatal symptomatic recurrent PE, or nonfatal major bleeding.
Nonfatal bleeding events were classified as major if they were overt and (1) occurred in a critical organ (eg, intracranial, intraocular, and retroperitoneal hemorrhage), (2) were associated with a drop in the hemoglobin level of 2.0 g/dL or more, and (3) required a transfusion of 2 units of blood or more. 7 Recurrent PE was diagnosed either by a new perfusion scan or by the presence of a new intraluminal filling defect, or an extension of a previous filling defect on helical thoracic CT. 8
Patients who were discharged from the hospital were followed by the outpatient polyclinic or by telephone interviews.
Statistical Analyses
Statistical analyses were performed with SPSS for Windows, version 17.0 (SPSS Inc, Chicago, Illinois). We used Pearson chi-square test to compare the probability of occurrence of the primary outcome between the LMWH and UFH groups and binary logistic regression to calculate unadjusted odds ratios and 95% confidence intervals. A P value <.05 was regarded as statistically significant in all analyses.
Results
Overall, 1355 patients with acute PE were evaluated during the study period. Of these, 249 (18.4%) were enrolled in the study who were treated with thrombolysis for PE. The mean age at diagnosis was 60.7 ± 15.5 years, and 109 (46.5%) patients were male. Of these, 186 (74.7%) had massive PE and 63 (25.3%) had submassive PEs and were treated with either LMWH (n = 122) or UFH (n = 127). Patient characteristics are shown in Table 1. In both treatment groups, there were no significant differences in mean age and clinical features. The most frequent symptoms at presentation were dyspnea (94%) followed by chest pain (63%), hemoptysis (12%), and syncope (25.7%). The following comorbidities were present in 127 (51%) patients: COPD, heart failure, cancer, renal failure, cerebrovascular disease, or hypertension. The most common concomitant conditions included arterial hypertension (10.8%) and coronary artery disease (12%). Active or previous malignancy was found in 10% of patients and COPD in 8.4% of patients, whereas heart failure and cerebrovascular occlusion were noted less frequently (Table 1).
Demographics and General Patient Characteristics.
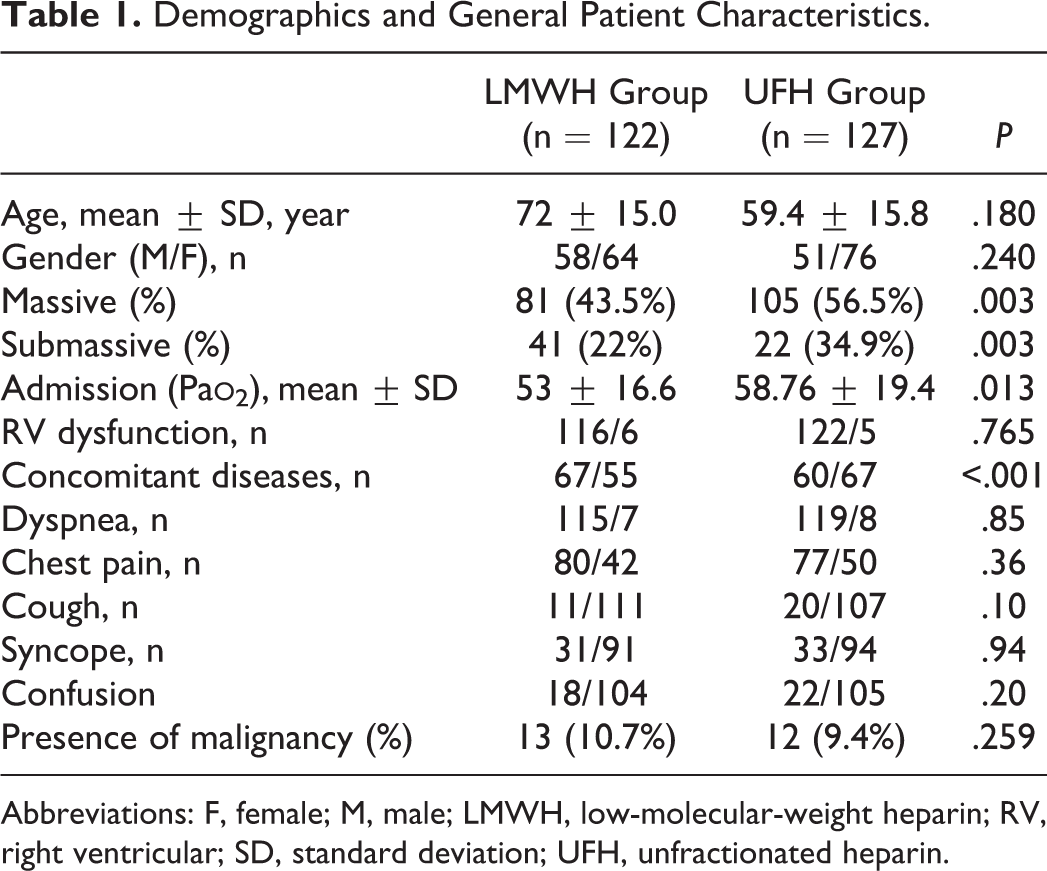
Abbreviations: F, female; M, male; LMWH, low-molecular-weight heparin; RV, right ventricular; SD, standard deviation; UFH, unfractionated heparin.
There were 15 (6%) patients with major bleeding events. In all, 5 (4%) patients had major bleeding events in the LMWH group, and 10 (7.9%) patients had major bleeding events in the UFH group. Minor hemorrhage occurred in 17 (13.4%) patients in the UFH group compared with 11 (9%) patients in the LMWH group. The frequency of bleeding complications was not significantly different between the 2 groups, but the UFH group had a tendency toward more frequent major bleedings but, conversely, less frequent bleedings compared with the LMWH group (P = .211; Table 2). There were 5 (7.9%) patients with major bleeding events in the submassive PE group and 10 (5.4%) patients with major bleeding events in the massive PE group (P = .22); the incidence of minor bleeding was 8 (12.7%) and 20 (10.7%) in the submassive and massive PE, respectively (P = .37; Table 3).
Adverse Events, Death, and Hemorrhage According to Treatment Groups for All Patients.a
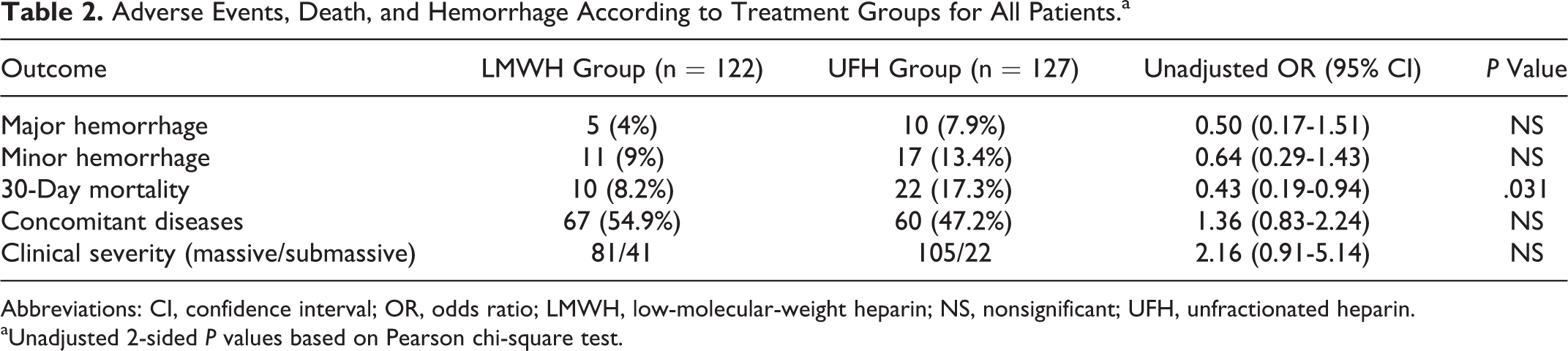
Abbreviations: CI, confidence interval; OR, odds ratio; LMWH, low-molecular-weight heparin; NS, nonsignificant; UFH, unfractionated heparin.
aUnadjusted 2-sided P values based on Pearson chi-square test.
Mortality Rate, Recurrence of Pulmonary Embolism (PE), and Hemorrhage According to Clinical Severity.a
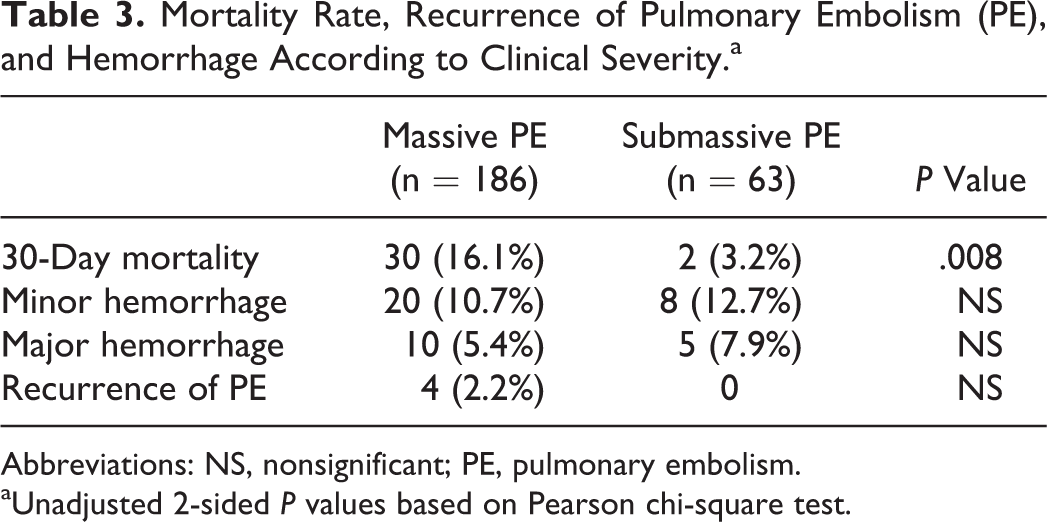
Abbreviations: NS, nonsignificant; PE, pulmonary embolism.
aUnadjusted 2-sided P values based on Pearson chi-square test.
Thirty-two patients (17.3%) died during the 30-day follow-up period, with causes including recurrent emboli (n = 4), respiratory failure (n = 9), major hemorrhage (n = 3), acute renal failure (n = 1), acute cardiac failure (n = 3), and multiple organ failure. 2 Most of the recurrence (n = 4) and all major hemorrhage (n = 3) events were observed in the UFH group. Mortality rates in the present study were 30 (16.1%) and 2 (3.2%) in the massive and submassive PE groups, respectively (P = .008). The incidence of all-cause death was 10 (8.2%) and 22 (17.3%) in patients with LMWH and UFH, respectively (P = .031).
Ignoring confounding factors when Pearson chi-square test was performed showed that treatment with UFH was associated with an increased risk of 30-day mortality. Additionally, conditional logistic regression analysis, which eliminated the effect of comorbid conditions, bleeding complications, and severity of embolism, was used to estimate the odds ratios and the 95% confidence intervals for associations between the 30-day mortality and treatment option. When we performed a logistic regression analysis for confounders including comorbid conditions, major and minor bleeding complications, and severity of embolism, we found that treatment option was not associated with an increased risk of 30-day mortality (Table 4).
Logistic Regression Analysis of Risk Factors and 30-Day Mortality.
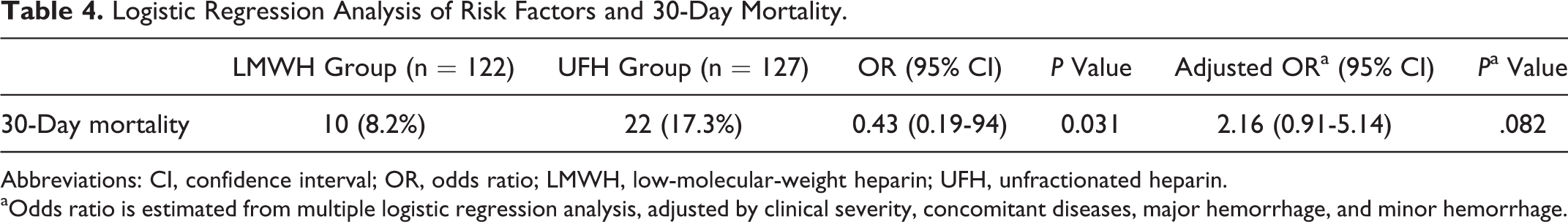
Abbreviations: CI, confidence interval; OR, odds ratio; LMWH, low-molecular-weight heparin; UFH, unfractionated heparin.
aOdds ratio is estimated from multiple logistic regression analysis, adjusted by clinical severity, concomitant diseases, major hemorrhage, and minor hemorrhage.
Because clinical severity is an important risk factor for 30-day mortality, we performed a Pearson chi-square test and conditional logistic regression analysis for patients with massive PE. Our data showed that disregarding confounding factors and eliminating the effect of comorbid conditions revealed that bleeding complication treatment options were not associated with 30-day mortality (Table 5).
Logistic Regression Analysis of Risk Factors and 30-Day Mortality for Massive Pulmonary Embolism (PE).
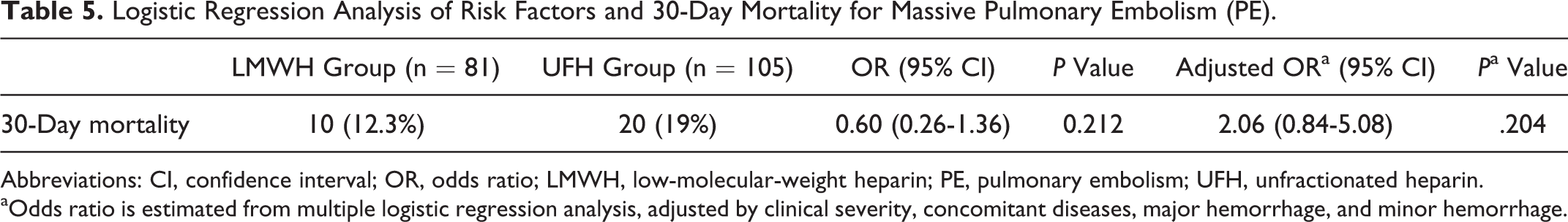
Abbreviations: CI, confidence interval; OR, odds ratio; LMWH, low-molecular-weight heparin; PE, pulmonary embolism; UFH, unfractionated heparin.
aOdds ratio is estimated from multiple logistic regression analysis, adjusted by clinical severity, concomitant diseases, major hemorrhage, and minor hemorrhage.
In the submassive PE group, there was no mortality in patients treated with LMWH, but there were 2 (9.1%) cases of mortality among patients treated with UFH after exclusion of comorbidity and bleeding complications. Using binary logistic regression analysis, there was no significant differences in treatment options in the submassive PE group (P = .997; data not shown).
Discussion
The physicians continue to treat PE with UFH because of doubts that patients with PE are at a higher risk of fatal outcomes and because evidence that LMWH is effective and does no harm during PE treatment is believed to be insufficient. Our study indicates that LMWH is as effective and safe as UFH for the treating massive and submassive PEs.
The 2008 American College of Chest Physicians Evidence-Based Clinical Practice Guidelines recommend fibrinolysis for patients who show evidence of hemodynamic compromise unless there are major contraindications from bleeding risk. 9 Hemodynamically unstable patients with massive PE are at high risk of adverse events and mortality. Fibrinolytic therapy is the accepted treatment in these patients. However, normotensive patients with acute PE evidence of right ventricular dysfunction are categorized as having submassive PE and compose a population at intermediate risk of adverse events and early mortality. The largest clinical trial, thus far, on fibrinolysis for submassive PE recommends the administration of fibrinolytic therapy. 10 In the present study, 186 (74.7%) patients treated with thrombolytics had massive PE, and 63 (25.3%) patients treated with thrombolytics had submassive PE.
The LMWH, which was first studied and approved for DVT prevention, 13 has many advantages over UFH, including more predictable and reliable anticoagulation, increased patient satisfaction, and the ability to be self-administered, making home-based treatment a possibility. 11 The medication cost of LMWH was higher; however, injection and laboratory monitoring costs were lower, making the total cost lower. 12 Studies with LMWH have demonstrated that it has similar morbidity and mortality outcomes to UFH in treating DVT and nonmassive PE. 4,14,15
The LMWH and UFH are the most common anticoagulants recommended for treating confirmed nonmassive PE in ACCP guideline in 2012. 3 Many studies showed that LMWH is an effective drug for nonmassive PE; however, the role of LMWH in therapy for patients with massive PE is unclear. Many clinicians continue to treat PE with UFH because of doubt that patients with PE are at higher risk of fatal outcomes and because evidence that LMWH is effective and safe for the treatment of PE is believed to be insufficient. However, DVT and PE are regarded as different manifestations of the same disease process, and physicians are often expected to respond similarly by treating with LMWH. 4,16 Only 1 retrospective study in the literature has shown the effectiveness and safety of LMWH in cases with acute massive PE. However, there is no head-to-head comparison of 2 drugs as a means to investigate the efficacy and safety of LMWH in cases with massive PE. 17 This is the first multicenter, observational study trial comparing the thrombolytic use with LMWH and with UFH.
Treatment with fibrinolysis, especially in massive PE, is suggested to decrease all-cause mortality according to available data. Treatment with heparin anticoagulation in submassive PE shows a short-term mortality rate of 3.0%. Although adjunctive fibrinolytic therapy may have a 30% relative reduction in mortality, the effect size on mortality as a result of submassive PE is probably <1%. Thus, secondary adverse outcomes such as persistent right ventricular dysfunction, chronic thromboembolic pulmonary hypertension, and impaired quality of life represent appropriate substitute goals of treatment. 18,19 Because of this, we applied thrombolytic treatment to 63 (25.3%) patients with submassive PE (manifested as right ventricular pressure overload and dysfunction). The 2014 European Society of Cardiology guidelines were presented after we completed our study. The guidelines categorized massive as the high-risk group, and the submassive (manifested as right ventricular pressure overload and dysfunction) PEs treated with a thrombolytic were categorized as the intermediate high-risk group. 20
Both UFH and LMWH are associated with complications such as hemorrhage and thrombocytopenia. 21,22 Many studies have revealed that LMWH treatment of PE resulted in less hemorrhage and thrombocytopenia than treatment with UFH. 23,24 In our study, treatment with UFH and LMWH resulted in major hemorrhage in 10 (7.9%) and 5 (4%) patients, respectively. No thrombocytopenia was observed. Our results suggest that there was no increased risk of major hemorrhage or significant difference in patients treated with LMWH versus UFH for acute massive and submassive therapies. In our study, 30-day mortality occurred in 17.3% of the cases who received UFH after thrombolytic therapy and in 8.2% of the cases who received LMWH.
Clinical severity may be the most important factor affecting mortality rates; when we repeated the same analysis in patients with massive PE, we reported no effect of treatment option on 30-day mortality day in the LMWH and UFH groups. There was a difference between groups in clinical severity and presence of concomitant diseases (Table 5).
This study has 2 limitations. First, data were collected prospectively, but the study design was retrospective. Second, some centers used different LMWH active substances.
These studies are conducted with small patient groups in single centers. To our knowledge, this is the first prospective, multicenter, and observational study comparing LMWH and UFH after thrombolytic use in patients with submassive and massive PEs.
Conclusion
This study showed that LMWH regimens exhibited similar efficacy in 2 groups of patients with PE and massive PEs. Although the death rates and PE recurrence rates were similar between the 2 treatments, the LMWH regimen had a lower bleeding tendency. Thus, compared with the LMWH regimen, the UFH regimen is as effective and perhaps safer in treating patients with PE. These findings support the belief that optimizing LMWH is valuable when treating patients following thrombolytic use with acute massive PE.
Footnotes
Authors’ Note
The TUPEG Study Investigators includes Aygul Guzel, MD, Department of Pulmonary Medicine, 19 Mayıs University, School of Medicine, Samsun, Turkey. Dursun Tatar, Suat Seren Training and Research Hospital, Department of Chest Diseases, İzmir, Turkey. Aydanur Ekici, Department of Pulmonary Medicine, Kırıkkale University, School of Medicine, Kırıkkale, Turkey. Ersin Günay, Department of Pulmonary Medicine, Afyon Kocatepe University, School of Medicine, Afyon, Turkey. Ezgi Demirdogen, Department of Pulmonary Medicine, Uludağ University, School of Medicine, Bursa, Turkey. Servet Kayhan, MD, Department of Pulmonary Medicine, Recep Tayyip Erdoğan University, School of Medicine, Rize, Turkey. Serhat Çelikel, Department of Pulmonary Medicine, Medipol University, School of Medicine, İstanbul, Turkey. Nuri Tutar, MD, Department of Pulmonary Medicine, Erciyes University School of Medicine, Kayseri, Turkey.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
