Abstract
Background
Defibrotide is a DNA-derived polyelectrolyte with anti-ischemic and antithrombotic effects, primarily used in post-bone marrow transplantation (BMT) veno-occlusive disease (VOD). Currently, it is produced from porcine mucosal tissue, which presents cultural, economic, and supply limitations.
Aim
This study compared coagulation activity, molecular profiles, and DNA content of porcine- versus ovine-derived defibrotide.
Methods
Active pharmaceutical ingredient (API) samples of 19 porcine and 9 ovine defibrotide batches were dissolved in saline (1 mg/mL) and added to pooled plasma and whole blood (100 µg/mL). Coagulation was assessed using activated partial thromboplastin time (aPTT) and thrombin time (TT). Anti-Xa and anti-IIa activities were measured by amidolytic assays. DNA content was quantified via the heparin red method, and molecular weight was determined by size exclusion chromatography.
Results
Neither porcine nor ovine preparations showed anticoagulant effects in aPTT. Both produced mild, comparable anticoagulant activity in TT. Anti-Xa and anti-IIa values were similar across sources. Molecular weight profiles were also comparable (ovine: 16.8 ± 0.24 kDa vs porcine: 18.02 ± 0.32 kDa). DNA content showed no significant difference (ovine: 75.78 μg vs porcine: 79 μg/10 μg API).
Conclusion
Ovine- and porcine-derived defibrotide demonstrated equivalent coagulation, molecular, and DNA content profiles. These findings suggest ovine mucosa is a viable alternative source for defibrotide, potentially improving accessibility, reducing cost, and addressing dietary restrictions associated with porcine-derived products.
Introduction
Defibrotide is a DNA-derived polyelectrolyte drug that is currently used in the management of post-bone marrow transplantation-associated veno-occlusive disease (VOD). The pathogenesis of VOD, also known as sinusoidal obstruction syndrome (SOS), begins with injury of the sinusoidal endothelial cell due to various factors (ie microbial damage, cytokine production, and conditioning regimen used during transplantation). 1 The occurrence of VOD has been reported in up to 60% of patients following stem cell transplantation. 2 Defibrotide is currently the only approved drug for the treatment of moderate and severe life-threatening VOD. 3 It is a natural product obtained through controlled depolymerization of porcine intestinal mucosal DNA. It consists of a mixture of single-stranded (90%) and double-stranded (10%) phosphodiester oligonucleotides, which may provide some nuclease resistance, allowing the strands to remain stable long enough to reach the drug's target, the liver. 4
It has been shown that defibrotide has anti-ischemic, antithrombotic, profibrinolytic, and anti-inflammatory effects that result in endothelial protection. 5 Defibrotide has a complex mechanism of action, but it appears to act as an adenosine receptor agonist and as a stimulus to the production of prostaglandins. 6 It has also been found to modulate endothelial cell function, causing an increase in tissue plasminogen activator (t-PA) levels, a decrease in plasminogen activator inhibitor levels, and an increase in prostaglandin I2 (PGI2) formation in humans. 7 Additionally, defibrotide has not been shown to demonstrate any significant systemic anticoagulant effects. 8
Recently, pharmaceutical manufacturers have shown interest in developing ovine and bovine heparins. Since defibrotide is a byproduct of the heparin manufacturing process, it is possible to obtain sizeable amounts of defibrotide from bovine and ovine intestinal mucosa. Moreover, the supply and demand chain limitations necessitate the resourcing of defibrotide from non-porcine tissues. The purpose of this study is to compare a generic version of defibrotide derived from ovine sources with porcine-derived defibrotide in various in vitro settings. Specifically, we analyzed nineteen porcine and nine ovine active-pharmaceutical-ingredient (API) batches obtained from commercial manufacturers. Each preparation was evaluated under identical laboratory conditions across multiple in vitro assays, including activated partial thromboplastin time (aPTT), thrombin time (TT), anti-Xa, and anti-IIa amidolytic activity, DNA content quantification, and molecular-weight determination by size-exclusion chromatography. The goal was to determine whether ovine-derived defibrotide demonstrates comparable coagulation, molecular, and biochemical profiles to the currently marketed porcine formulation. Each batch represented an independent production lot of pharmaceutical-grade defibrotide produced under standardized manufacturing conditions.
Beyond laboratory comparison, expanding defibrotide sourcing also carries cultural and accessibility implications. In the United States, there are diverse religious and cultural beliefs. Recent studies estimate that Islam is the third-largest religion in the United States and the second-largest religion worldwide. 9 In Islam, pork and anything derived from it is forbidden. Currently, available defibrotide is manufactured using porcine mucosal tissue. It is important to explore other agents to accommodate the beliefs of Muslims, as well as other individual or groups with similar preferences. Other mammalian tissues, such as bovine and ovine intestines, may also be suitable for defibrotide production. The current price of defibrotide is prohibitively expensive, which limits its use in wider patients, especially in underdeveloped countries. The development of lower-cost generic defibrotide would make it more accessible for a wider range of patients.
Materials and Methods
Porcine-derived defibrotide was obtained from Gentium Pharmaceuticals (Villa Guardia, Italy) and represented the branded formulation. Ovine-derived defibrotide was obtained from Ronnsi (Suzhou, China) as research-grade API. At the time of this study, no FDA-approved or commercially available generic ovine defibrotide formulation available. Accordingly, this was designed to compare source-dependent biochemical and coagulation characteristics of ovine- and porcine-derived defibrotide rather than to assess branded versus generic drug equivalent. A total of 19 porcine batches and 9 ovine batches were analyzed. Each batch represented an independent API production lot, with its own manufacturer's reference number.
Sample Preparation
Each batch of defibrotide was dissolved in saline to prepare a 1 mg/mL solution. The individual batch of reconstituted solutions were then supplemented into citrated plasma preparations and citrated whole blood at a final concentration of 100 µg/mL. Blood bank plasma was purchased from the Loyola University Medical Center Blood Bank, and control blood samples were collected from multiple healthy adult volunteers aged 18–40 years. Blood bank plasma consisted of citrated frozen plasma prepared according to standard blood bank protocols, stored under controlled frozen conditions, and thawed immediately prior to coagulation analysis. Plasma obtained from healthy volunteer donors was processed and analyzed individually and was not pooled across donors. All blood and plasma samples were exposed to defibrotide under identical experimental conditions; untreated control samples were not included.
Coagulation Assays
Activated Partial Thromboplastin Time (aPTT) was measured using the ACL TOP 300 (Instrumentation Laboratory, Lexington, MA).
Thrombin Time (TT) was assessed using the ST4 clot-based analyzer (Diagnostica Stago, Paris, France).
Citrated whole blood samples were also centrifuged to obtain plasma, which was assayed for aPTT and TT as well.
Reference ranges for coagulation parameters were based on manufacturer specifications for the respective instruments used. Normal activated partial thromboplastin time (aPTT) values on the ACL TOP 300 system range from 26–38 s, while normal thrombin time (TT) values on the ST4 clot analyzer range from 14–21 s. Anti-Xa and anti-IIa activities in plasma without anticoagulant exposure are expected to be near zero (below 5%), reflecting the absence of heparin or direct thrombin inhibitors.
Anti-Xa and Anti-IIa Activity
Anti-Xa and anti-IIa activities were determined using amidolytic assays performed in microtiter plates with purified enzymes.
DNA Quantification
The DNA content in defibrotide was quantified using the Heparin Red method, a fluorescence dye-based technique. The heparin red probe was provided by Dr Kramer (University of Heidelberg, Germany).
Molecular Weight Determination
Molecular weight (MW) was assessed via size exclusion chromatography (SEC) using unfractionated heparin as a standard. SEC was performed on a Waters high-performance liquid chromatography (HPLC) system using a tendam column method using tandam column PSK 2000 and 3000. Calibrators included were the molecular fractions of heparin in the range of 2000–50,000.
Statistical Analysis
All results were compiled and expressed as mean ± standard deviation using Microsoft Excel. GraphPad Prism was used to generate bar graphs and calculate p-values.
Results
The mean ± standard deviation (SD) values for ovine and porcine preparations are summarized in tables, while figures display the mean and standard errors for each preparation with p-values included where calculated. Unless otherwise stated, all mean ± SD values represent measurements from 19 independent porcine batches and 9 independent ovine defibrotide batches. For interpretive purposes, values within established reference ranges were considered unchanged, whereas prolongations beyond reference ranges were considered indicative of anticoagulant effect. Table 1 and Figure 1 present data for whole blood (WB) in aPTT and TT. Ovine values were 38.75 ± 4.10 s in aPTT and 25.42 ± 4.53 s in TT. Porcine values were 39.74 ± 2.60 s in aPTT and 20.42 ± 4.77 s in TT. Figure 1 illustrates aPTT values in group A and TT values in group B. Both groups displayed comparable values, indicating no anticoagulation effect in the aPTT assay and a mild anticoagulant effect in TT. The comparison between ovine and porcine defibrotide in aPTT showed no statistically significant difference (p = 0.385), while the TT comparison demonstrated a statistically significant difference (p = 0.0024).

Comparison of ovine and porcine defibrotide mean clotting times in whole blood in aPTT (A) and TT (B). Data are presented as mean ± SD. P-values calculated via unpaired two-tailed t-test.
A Comparison of Ovine and Porcine Defibrotide in Whole Blood.

Table 2 and Figure 2 represent the data for retrieved plasma (RP) preparations. The ovine values were 34.28 ± 0.92 s in aPTT and 37.31 ± 6.41 s in TT. For porcine, aPTT was 33.04 ± 1.07 s, and TT was 31.51 ± 1.59 s As shown in Figure 2, group A represents aPTT values, while group B depicts TT values. The values were comparable in both preparations, showing no anticoagulation in aPTT and mild anticoagulation in TT. In retrieved plasma preparations, both aPTT (p = 0.0006) and TT (p = 0.0005) values between ovine and porcine defibrotide were significantly different, despite their mean values being relatively close.
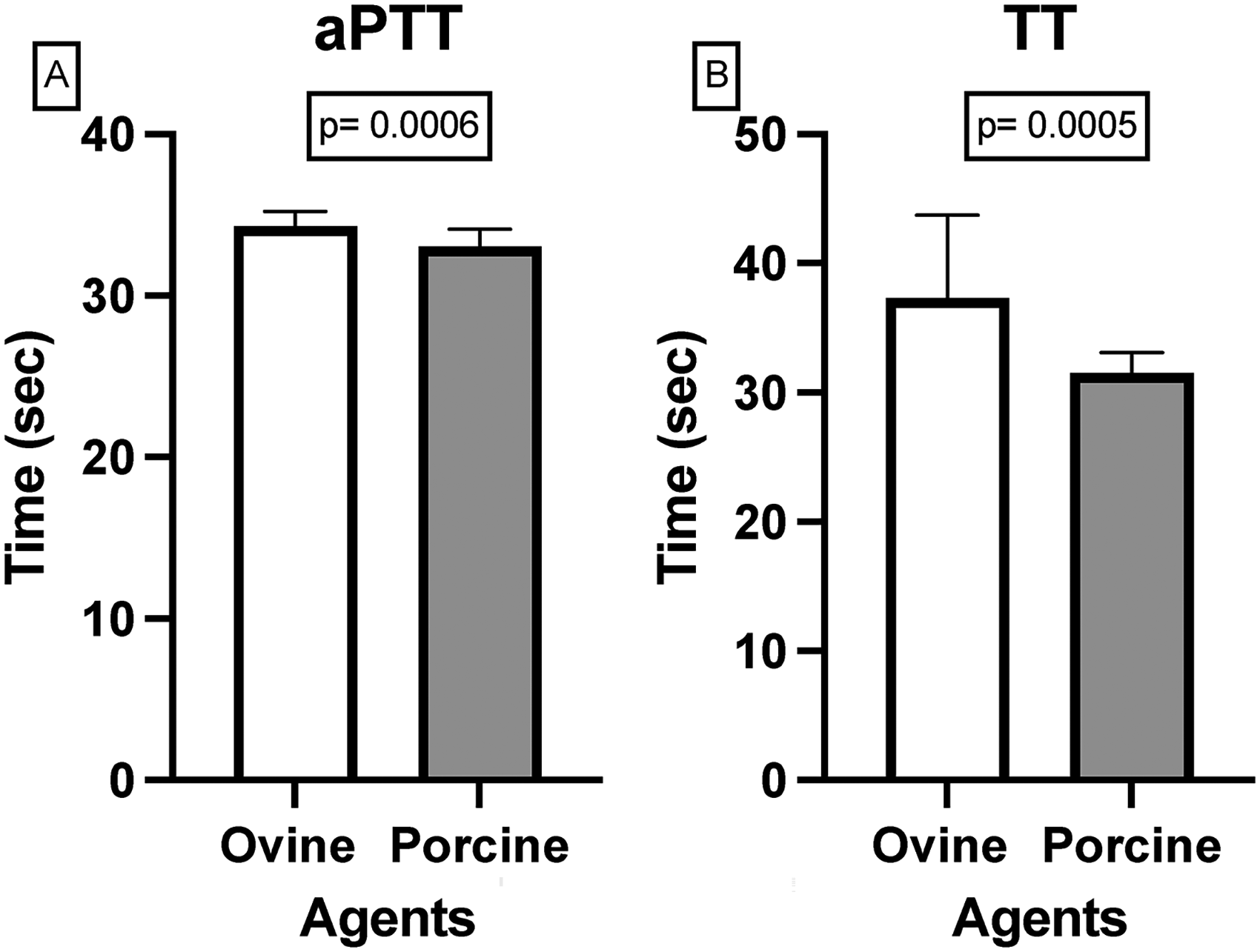
Comparison of ovine and porcine defibrotide mean clotting times in retrieved plasma measured by aPTT (A) and TT (B). Data are presented as mean ± SD. P-values calculated via unpaired two-tailed t-test. Retrieved plasma refers to plasma obtained by centrifugation of citrated whole blood immediately after collection.
A Comparison of Ovine and Porcine Defibrotide in Retrieved Plasma.

Additionally, Table 3 and Figure 3 represent ovine and porcine preparations in blood bank plasma (BBP). The ovine values were 31.26 ± 0.67 s in aPTT and 33.39 ± 1.85 s in TT. The porcine values were 39.74 ± 2.57 s in aPTT and 25.57 ± 5.12 s in TT. Figure 3 represents aPTT values in Group A and TT values in Group B. Similarly, to WB and RP, BBP preparations showed no anticoagulation in aPTT and mild anticoagulant effects in TT. The difference in aPTT values was highly significant (p < 0.0001), as was the difference in TT values (p < 0.0001), indicating stronger divergence in blood bank plasma samples.
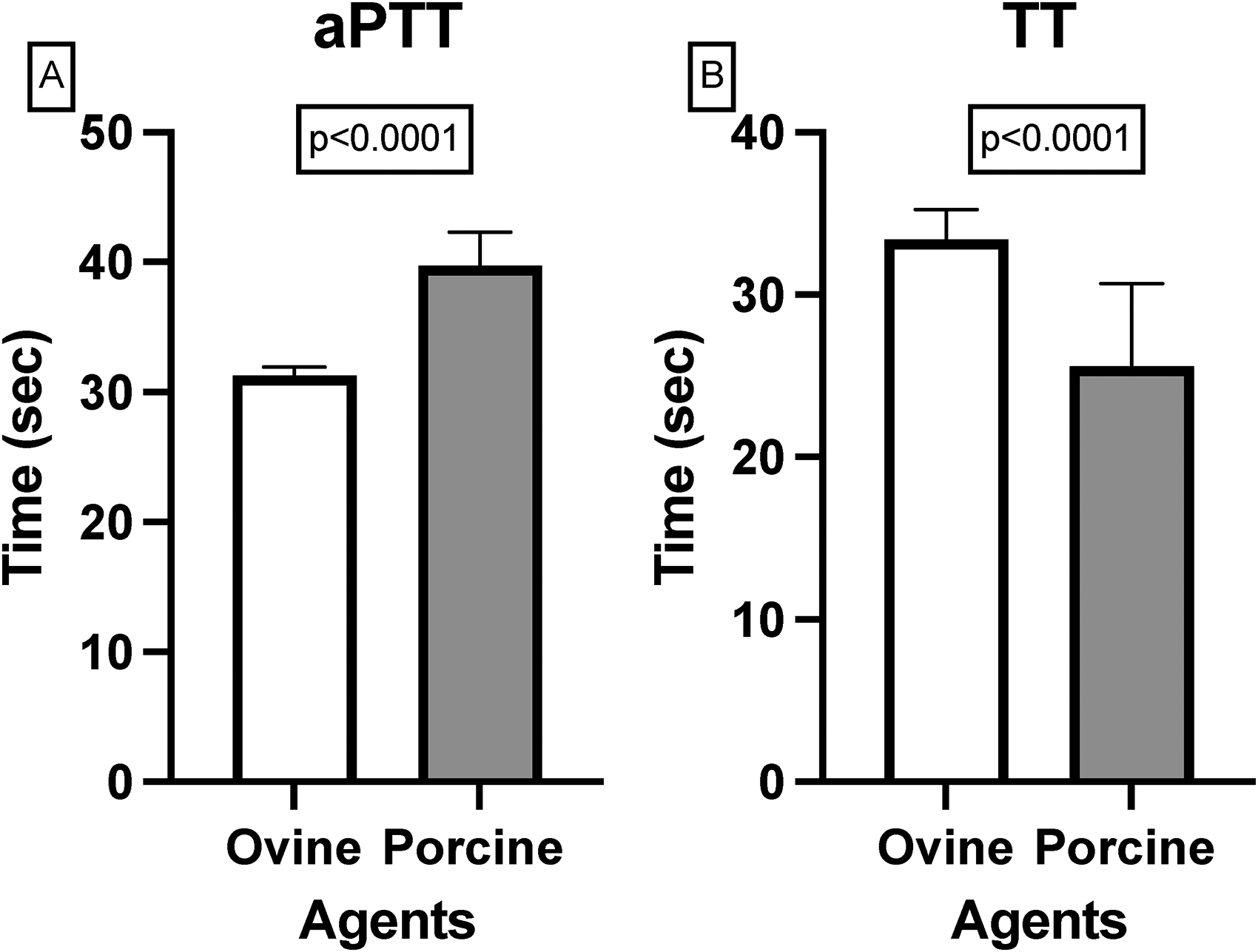
Comparison of ovine and porcine defibrotide mean clotting times in blood bank plasma measured by aPTT (A) and TT (B). Data are presented as mean ± SD. P-values calculated via unpaired two-tailed t-test.
A Comparison of Ovine and Porcine Defibrotide in Blood Bank Plasma.

Ovine and porcine defibrotide activities in anti-Xa and anti-IIa are depicted in Tables 4 and 5, with Figure 4 displaying group A for anti-Xa and group B for anti-IIa. For anti-Xa (Table 4) ovine was 7.44 ± 2.70 s while porcine was 7.19 ± 1.44 s For anti-IIa (table 5) ovine was 7.44 ± 2.70 s while porcine was 7.24 ± 1.25 s Both preparations showed comparable activities in anti-Xa and anti-IIa assays. Anti-IIa comparison showed no statistically significant difference (p = 0.667). A p-value was not calculated for Anti-Xa, as only summary values were analyzed.

A comparison of ovine and porcine defibrotide mean clotting times in Anti-Xa (A) and Anti-IIa (B). Data are presented as mean ± SD. P-values calculated via unpaired two-tailed t-test.
Ovine and Porcine Defibrotide in Anti-Xa.
Ovine and Porcine Defibrotide in Anti-IIa.

Table 6 shows the DNA content of ovine and porcine defibrotide. Ovine was 75.78 ± 4.74 μg while porcine was 79 ± 6.39 μg, relative to heparin. Figure 5 shows that the mean values for

Comparison of DNA content in ovine and porcine defibrotide. Data are presented as mean ± SD. P-values calculated via unpaired two-tailed t-test.
DNA Content of Ovine and Porcine Defibrotide.

DNA content in both preparations were similar. The comparison of DNA content between ovine and porcine defibrotide showed no statistically significant difference (p = 0.191).
Finally, Table 7 shows the molecular weight of ovine and porcine defibrotide. Ovine was 16.8 ± 0.24 kDA, while porcine was 18.02 ± 0.32 kDA. Figure 6 further illustrates the comparable molecular weights between the two preparations. A p-value was not calculated for molecular weight, as only the mean and standard deviation were available.

Comparison of the molecular weights of ovine and porcine defibrotide. Data are presented as mean ± SD.
Molecular Weight of Ovine and Porcine Defibrotide.

Discussion
In this study, we conducted various in vitro experiments that showed generic defibrotide from ovine has similar effects to porcine defibrotide. Of the various batches of defibrotide obtained, no changes were observed. In the activated partial thromboplastin time (aPTT) assay, as shown in Figures 1A–3A, both defibrotide's demonstrated similar trends across all plasma types. No anticoagulant effects were observed in whole blood, and only slight variations were seen in retrieved and the blood bank plasma. In the thrombin time (TT) assay (Figures 1B–3B), both preparations showed mild anticoagulant effects, with differences that remained consistent within biologically acceptable ranges. The effect of blood cellular components binding on defibrotide was studied by supplementing various defibrotide samples in whole blood and retrieved plasma. The objective of these studies was to demonstrate any significant binding of defibrotide and its fractions with blood cells or its components. We did not find any significant differences between the whole blood and retrieved plasma studies. Therefore, defibrotide and its fractions did not show any significant indifferent binding affinity to the blood cells.
Figure 4 shows that anti-Xa and anti-IIa activity were also comparable between the two preparations. While anti-IIa was statistically evaluated, anti-Xa was reviewed based on summary values. Figure 5 and Figure 6 demonstrate that DNA content and molecular weight profiles were highly similar across both preparations, further supporting their biochemical equivalence; a p-value was calculated for DNA content, while molecular weight was reviewed based on summary values.
While p-values provide helpful insight into statistical significance, it is important to consider the broader biological relevance of these findings. The overall trends across all parameters, even where p-values reached significance, consistently point toward functional comparability between ovine and porcine defibrotide. Slight statistical differences in clotting times may arise due to inherent variability in biological sources or batch processing but do not undermine the clinical potential of ovine defibrotide as a viable alternative.
To place these in vitro findings into clinical and translational context, the following discussion highlights the therapeutic relevance and sourcing considerations of defibrotide.
Currently, porcine defibrotide is important for the management of post-bone marrow transplantation-associated veno-occlusive disease (VOD), also known as sinusoidal obstruction syndrome (SOS). VOD is an unpredictable, potentially life-threatening complication of hematopoietic stem cell transplantation (HSCT). It is caused by conditioning regimens that generate toxic metabolites, which damage hepatocytes and lead to sinusoidal endothelial cell activation. 10 These endothelial cells swell up, leading to progressive narrowing of the venous lumen, a reduced sinusoidal venous outflow, and ultimately post-sinusoidal portal hypertension. 11 It is characterized by hyperbilirubinemia, painful hepatomegaly, and fluid retention. 12 The mean incidence of VOD is estimated to be 13.7% following myeloablative conditioning for HSCT. 13 The mortality risk of patients who develop severe VOD, characterized as VOD with multiorgan failure, was 84.3%. 13
Defibrotide is a mixture that consists of 90% single-stranded phosphodiester oligonucleotides and 10% double stranded phosphodiester oligonucleotides. 14 Defibrotide has shown anti-ischemic, antithrombotic, profibrinolytic, and anti-inflammatory effects that result in endothelial protection. 5 The polyanionic nature of defibrotide is important because it interferes with the cationic proteins that cause vascular instability. 15 Currently, defibrotide is produced from porcine intestinal DNA and is indicated for treatment of adult or pediatric patients with VOD following HSCT in the United States. 16
Recently, pharmaceutical manufacturers have been interested in the development of ovine and bovine heparins. Obtaining sizeable amounts of ovine and bovine defibrotide, a byproduct of heparin manufacturing, along with the supply and demand limitations of porcine defibrotide, necessitates the resourcing of defibrotide from non-porcine sources. Furthermore, porcine defibrotide is prohibitively expensive, which limits its use in underdeveloped countries. Therefore, we compared a generic defibrotide from ovine origin to porcine through various in vitro settings. It is crucial to expand the source of defibrotide because of its importance in the treatment of VOD, making defibrotide more accessible and reducing its productions costs.
Additionally, religions such as Islam, have prohibitions on pork and anything derived from it. Muslims are not allowed to consume these products, except under extremely exceptional conditions. 17 Addressing these dietary restrictions will help to overcome limitations on the usage of porcine defibrotide in certain populations. This shift could make treatment both more inclusive and economically viable.
Within the scope of the present in vitro analyses, these findings support the feasibility of ovine-derived defibrotide as a functionally comparable alternative to porcine-derived material.
Conclusion
In conclusion, this study demonstrates that ovine-derived defibrotide exhibits comparable characteristics to porcine-derived defibrotide across various in vitro assays, including activated partial thromboplastin time (aPTT), thrombin time (TT), anti-Xa, anti-IIa assays, molecular weight, and DNA content. This successful comparison suggests that ovine defibrotide could serve as a viable alternative to porcine defibrotide, addressing supply limitations, high costs, and dietary restrictions associated with porcine-derived products. Exploring defibrotide sources beyond non-porcine options could improve accessibility and affordability, making this essential treatment more widely available, particularly in underdeveloped regions and among populations with religious dietary restrictions. Further research and clinical trials are warranted to validate these findings and confirm the therapeutic equivalence of ovine defibrotide in a clinical setting.
Limitations of the Study
This study was conducted entirely using the in vitro system, and therefore the findings may not fully reflect the pharmacodynamic or clinical effects of ovine- versus porcine-derived defibrotide in patients. While multiple batches from each source were analyzed, batch-to-batch variability and manufacturing differences between suppliers could influence results. Additionally, the sample size for ovine defibrotide batches was smaller than that for porcine, which may limit statistical power for certain comparisons. Future studies, including in vivo and clinical trials, are necessary to confirm the therapeutic equivalence and safety profile of ovine-derived defibrotide in clinical settings.
Footnotes
Abbreviations
Acknowledgments
Special thanks are extended to the Hemostasis and Thrombosis Research Laboratory staff for facilitating the characterization of defibrotide. The authors are thankful to Dr Eva Wojcik, the Chairman of the Department of Pathology and Laboratory Medicine for supporting this study.
Ethical Approval
The blood bank plasma was purchased from Loyola University Medical Center Blood Bank, and the control blood samples were collected from volunteers under the IRB LU#9191.
Authors’ Contributions
Mr Yiming Yao and Dr Jawed Fareed designed this study. Hussein Abourahma collected samples, performed experiments, collected data, and drafted the manuscript. Dr Prakasha Kempaiah revised the manuscript for important intellectual content. Mr Yiming Yao and Dr Jawed Fareed supervised the project and interpreted the data. All authors have approved the final article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
