Abstract
The interplay between vitamin D, the renin–angiotensin system (RAS), and collagen remodeling has been implicated in the pathogenesis of various cardiovascular diseases. This study sought to explore this relationship in atrial fibrillation (AF) by profiling plasma levels of 25-hydroxyvitamin D, RAS biomarkers, and collagen remodeling biomarkers using the enzyme-linked immunosorbent assay method. We hypothesized that 25-hydroxyvitamin D levels would inversely correlate with RAS biomarkers and that levels of RAS and collagen remodeling biomarkers would positively correlate with each other. Although our AF cohort (n = 37) did not exhibit decreased 25-hydroxyvitamin D levels compared to normal controls (n = 26), these levels inversely correlated with renin (Spearman r = −0.57, P = 0.005). Renin levels were elevated in patients with AF compared to normal controls (1233 ± 238 ng/mL vs 401 ± 27 ng/mL, P = 0.0002) and positively correlated with levels of matrix metalloproteinase 1 (MMP-1; Spearman r = 0.89, P = 0.01) and MMP-2 (Spearman r = 0.82, P = 0.03). These data suggest that 25-hydroxyvitamin D may influence RAS activation, and renin may help mediate the collagen remodeling process in AF. Understanding mediators of RAS dysregulation in AF may elucidate targets for therapeutic intervention to prevent collagen remodeling.
Introduction
Atrial fibrillation (AF) is the most common cardiac arrhythmia seen in clinical practice and is associated with significant morbidity and mortality. The 2010 Global Burden of Diseases, Injuries, and Risk Factors Study estimated that 33.5 million patients have AF globally, while another study estimated that the number of Americans affected by AF in that year (3-6 million) is projected to triple by 2050. 1,2 According to Rahman et al, the clinical management of AF costs the United States approximately US$26 billion annually, a projection that does not include the costs associated with treating complications such as stroke, heart failure, myocardial infarction, chronic kidney disease, and dementia. 3 Although the exact pathophysiology of AF remains unknown, it is thought to be due to multifactorial structural and electrical changes in the atria. Because of its increasing prevalence, incidence, and burden, the pathophysiology of AF must be further clarified in order to better treat symptoms, reduce hospitalizations, and prevent complications.
25-Hydroxyvitamin D and Renin–Angiotensin System
Vitamin D is a fat-soluble vitamin that is best known for regulating calcium, parathyroid hormone, and bone mineral homeostasis. New research suggests that vitamin D may also have pleotropic effects. In a study done by Cantorna et al, vitamin D was found to be an inflammatory modulator and antioxidant, inhibiting the proliferation of T lymphocytes that produce interferon-γ and interleukin 17. 4 Researchers have since investigated the role of vitamin D deficiency in various cardiovascular diseases. A meta-analysis by Burgaz et al demonstrated an inverse relationship between 25-hydroxyvitamin D and hypertension, while a randomized controlled trial by Nasri et al demonstrated the positive impact of vitamin D supplementation on blood pressure lowering in patients with type 2 diabetes mellitus. 5,6 A double-blinded, placebo-controlled intervention study by Shedeed et al noted an improvement of ejection fraction in pediatric patients with congestive heart failure supplemented with vitamin D. 7,8
The relationship between vitamin D deficiency and AF has so far been inconclusive. 9,10 Chan et al demonstrated that there was an increased risk of developing AF in patients genetically deprived of, but not deficient in, vitamin D due to single nucleotide polymorphisms in its metabolic pathways. 11 Foreman et al revealed vitamin D’s role in negatively regulating renin–angiotensin system (RAS) in healthy individuals. 12 Vitamin D deficiency has been speculated to attenuate the benefit of RAS blockade in patients with AF. 12 –14
Renin–Angiotensin System and Collagen Remodeling
Prior studies have investigated the roles of RAS and collagen remodeling in cardiovascular diseases such as hypertension and heart failure. 15,16 The role of renin in collagen remodeling has not been investigated in the context of AF. Rather, angiotensin II is thought to be the main player that binds to its AT1 receptor and upregulates the JAK/STAT3 signaling pathway which then increases atrial extracellular matrix deposition via matrix metalloproteinase (MMP) 1 and MMP-2. 17,18 These MMPs break down type I and type III collagens which are most abundant in the heart, releasing breakdown products of collagen-like C-telopeptide of type I collagen (CITP). Subsequent alterations in cardiac structure and function may increase the risk of cardiovascular diseases like hypertension and congestive heart failure. 15,16
Renin–angiotensin system and collagen remodeling have been separately studied as potential mechanisms involved in the pathophysiology of AF. The role of RAS in the development of AF has been postulated in patients with comorbid hypertension and heart failure but not in patients with AF as a primary concern. 19,20 Other studies have also noted the importance of collagen turnover factors like transforming growth factor β in the pathogenesis of AF. 21,22 Little is known about the interaction between RAS and collagen remodeling in patients with AF. Our study sought to further investigate the interplay between vitamin D, RAS biomarkers, and collagen remodeling biomarkers in patients having AF with the goal of better understanding the pathophysiology of this disease process and exploring potential targets for intervention.
Materials and Methods
This study is a nonrandomized case–control study in which patients with nonvalvular AF undergoing catheter ablation at Loyola University Medical Center were consented and enrolled. Whole blood was collected from these patients in two, 5-mL citrated tubes (n = 37) prior to ablation. Blood samples were centrifuged and the separated plasma was aliquoted and stored at −80°C. Control plasma samples purchased from a commercial vendor (George King Biomedical, Orland Park, Kansas) represented a cohort of healthy, nonsmoking volunteers (n = 26) who were not taking any medications. No other demographic information was provided for these control samples. Each sample was labeled with the study number of the patient under the approved IRB#205530. These samples were analyzed without access to clinical information until after biomarker levels were obtained. Sandwich enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems, Minneapolis, Minnesota) were used to evaluate plasma levels of renin, CITP, and 25-hydroxyvitamin D in patients with AF and normal controls. Competitive ELISA kits (LSBio, Seattle, Washington) were used to evaluate plasma levels of angiotensin II in patients with AF and normal controls. We included previously obtained data on plasma levels of MMP-1 and MMP-2 on the first 7 patients in this cohort as additional biomarkers of collagen remodeling.
Eligibility Criteria
Patients who were 18 years or older receiving care for AF at Loyola University Medical Center were recruited to be a part of this study. Exclusion criteria included patients with acute stroke, acute myocardial infarction, severe heart failure, sepsis, mechanical ventilation, cognitive impairment, and pregnancy.
Electronic Medical Record Review
After plasma levels of these biomarkers were recorded, demographic information including age, sex, and race/ethnicity was obtained for this cohort from the EPIC electronic medical records (Table 1). Medications that were used at the time of blood draw were also noted. Atrial fibrillation complications like stroke, heart failure, myocardial infarction, and chronic kidney disease were noted if they occurred any time after the blood draw until August 2019, a date that was approximately 2 years after the last patient was recruited into the study.
Demographics of Patients With Atrial Fibrillation.

a Age range.
We also included explored information on postcatheter ablation recurrence. Of the 37 patients enrolled in this study, 5 decided not to undergo catheter ablation. Of the 32 who did undergo catheter ablation, 9 experienced AF recurrence sometime between the procedure date and August 2019. Recurrence was defined as having an episode of AF that was caught on an electrocardiogram at any point after catheter ablation. We analyzed how biomarker levels compared between those who experienced post catheter ablation recurrence and those who did not.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism software. Plasma levels of biomarkers were recorded as means ± standard errors. Two-tailed Mann-Whitney U tests were carried out to compare biomarker levels between patients with AF and normal controls. Sensitivity analysis was performed by excluding patients taking spironolactone for renin measurements, excluding patients taking angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) for angiotensin II measurements, and excluding patients taking vitamin D supplementation for 25-hydroxyvitamin D measurements. In the AF cohort, Spearman coefficients were used to measure correlations between RAS biomarkers (renin and angiotensin II) and collagen remodeling biomarkers (CITP, MMP-1, MMP-2) or 25-hydroxyvitamin D. Statistical significance was defined as P < 0.05.
Results
The mean age of patients in this study was 61.8 years, ranging from 29.6 to 78.2 years. Males comprised 73% of the cohort while females comprised 27%. Most patients identified as Caucasian/white (97.2%) and the remaining 2.7% identified as Asian/Pacific Islanders. No other race/ethnicity was represented in this study group. Many of these patients were on ACE inhibitors (18.9%), ARBs (16.2%), spironolactone (5.4%), and vitamin D3 supplementation (32.4%). Some of these patients had acute complications of AF including stroke (2.7%), heart failure (18.9%), myocardial infarction (16.2%), and chronic kidney disease (13.5%).
Renin was significantly elevated in patients with AF compared to normal controls (1233 ± 238 ng/mL vs 401 ± 27 ng/mL, P = 0.0002), even after performing sensitivity analysis for spironolactone use (Figure 1A and Table 2). Angiotensin II was significantly decreased in patients with AF compared to normal controls (837.6 ± 34.8 ng/mL vs 976.5 ± 26.3 ng/mL, P = 0.005), even after performing sensitivity analysis for ACE inhibitor and ARB use (Figure 1B and Table 2). C-telopeptide of type I collagen was significantly elevated in patients with AF (16.02 ± 1.03 ng/mL vs 12.48 ± 0.64 ng/mL, P = 0.02; Figure 1C and Table 2). 25-Hydroxyvitamin D levels did not differ in patients with AF compared to controls (58.95 ± 19.99 ng/mL vs 56.33 ± 16.42 ng/mL, P > 0.05) after performing sensitivity analysis for vitamin D supplementation (Table 2).

A-C, Two-tailed Mann-Whitney U tests comparing plasma renin levels between patients with atrial fibrillation (AF) and George King (GK) controls (1233 ± 238 ng/mL vs 401 ± 27 ng/ mL), comparing plasma angiotensin II levels between patients with AF and GK controls (837.6 ± 34.8 ng/mL vs 976.5 ± 26.3 ng/mL), and comparing plasma C-telopeptide of type I collagen (CITP) levels between patients with AF and GK controls (16.02 ± 1.03 ng/mL vs 12.48 ± 0.64 ng/mL).
Results of 2-Tailed Mann-Whitney U Tests Comparing Plasma Levels of 25-Hydroxyvitamin D, Renin, Angiotensin II, and CITP Between Patients With AF and Normal Controls.
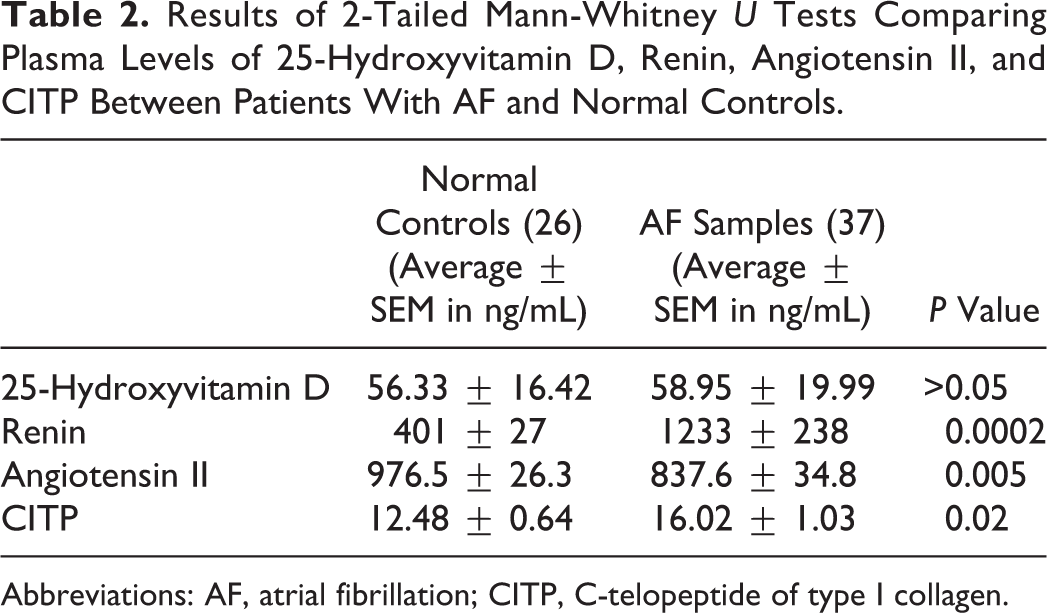
Abbreviations: AF, atrial fibrillation; CITP, C-telopeptide of type I collagen.
Renin did not differ significantly in patients who experienced recurrence after catheter ablation and those who did not (2069 ± 821 ng/mL vs 945 ± 184 ng/mL, P > 0.05), even after performing sensitivity analysis for spironolactone use. Angiotensin II did not differ significantly in patients who experienced recurrence after catheter ablation and those who did not (701.6 ± 80.5 ng/mL vs 840.6 ± 59.3 ng/mL, P > 0.05) after performing sensitivity analysis for ACE inhibitor and ARB use. C-telopeptide of type I collagen did not differ significantly in patients who experienced recurrence after catheter ablation and those who did not (14.32 ± 0.86 ng/mL vs 16.95 ± 1.49 ng/mL, P > 0.05). 25-Hydroxyvitamin D did not differ significantly in patients who experienced recurrence after catheter ablation and in those who did not (61.38 ± 9.29 ng/mL vs 69.83 ± 4.27 ng/mL, P > 0.05), even after performing sensitivity analysis for vitamin D supplementation.
Renin negatively correlated with 25-hydroxyvitamin D (Spearman r = −0.57, P = 0.005) and positively correlated with MMP-1 (Spearman r = 0.8929, P = 0.0123) and MMP-2 (Spearman r = 0.82, P = 0.03; Figures 2A-C). No significant correlations were found between renin and CITP or between angiotensin II and CITP, MMP-1, MMP-2, and vitamin D (Table 3).

A-C, Spearmen correlational analysis between 25-hydroxyvitamin D and renin (Spearman r = −0.57, P = 0.005), between renin and matrix metalloproteinase 1 (MMP-1; r = 0.89, P = 0.01), and between renin and matrix metalloproteinase 2 (MMP-2; r = 0.82, P = 0.03) in patients with atrial fibrillation.
Results of Spearmen Correlation Analysis Relating Renin–Angiotensin System Biomarkers (renin and angiotensin II) with 25-Hydroxyvitamin D or Collagen Remodeling Biomarkers (CITP, MMP-1, and MMP-2) in Patients With Atrial Fibrillation.
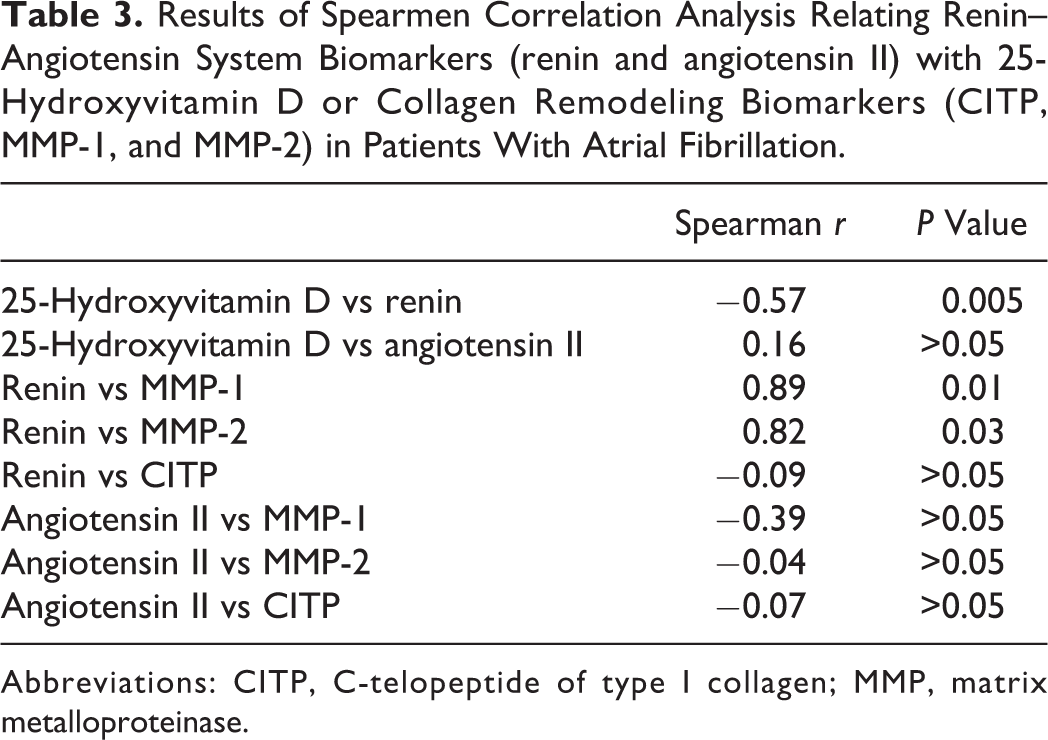
Abbreviations: CITP, C-telopeptide of type I collagen; MMP, matrix metalloproteinase.
Discussion
In this study, we investigated the interplay between vitamin D, RAS biomarkers, and collagen remodeling biomarkers in patients with AF compared to normal controls. Our findings are as follows: (1) renin levels are significantly elevated in AF; (2) angiotensin II levels are significantly decreased in patients with AF; (3) CITP levels are significantly elevated in patients with AF; and (4) renin levels negatively correlate with 25-hydroxyvitamin D levels and positively correlates with MMP-1and MMP-2 levels.
Vitamin D and the Renin–Angiotensin System
Our data are consistent with prior studies that have demonstrated an inverse association between 25-hydroxyvitamin D and activation of RAS. 12 However, measured plasma levels of 25-hydroxyvitamin D did not significantly differ between our cohort of patients with AF and normal controls. Therefore, this study does not necessarily suggest that deficiency of 25-hydroxyvitamin D is implicated in the elevation of renin and angiotensin II levels in AF. We considered the idea that the ELISA kits may not have had the required specificity to distinguish between 25-hydroxyvitamin D and the various vitamin D analogues existing in the human body. This may have caused measured vitamin D levels to appear higher than anticipated. However, if this were the case, both the AF group and the control group would have been similarly affected and would have displayed higher levels of vitamin D. Had any significant differences existed between these 2 groups, the statistical analysis would have reflected this regardless of lack of ELISA specificity. Ultimately, both groups reveal levels of 25-hydroxyvitamin D above 20 ng/mL, indicating sufficiency. Further investigation of what actually causes elevation of RAS in AF may allow for a means of intervention by which to reduce the risk of developing AF.
Utility of Renin and CITP as Prognostic Biomarkers
In the literature, renin and CITP have both been described as potential biomarkers in risk stratification of various cardiovascular diseases. The Health Outcomes Prevention Evaluation study by Verma et al noted that high plasma renin activity was an independent risk factor for major vascular events and mortality in patients with diabetes and atherosclerosis. 23 Gonzalez et al in another study demonstrated that increased plasma renin activity correlated with mortality in patients with hypertension. 24 Otaki et al found that CITP levels were predictive of clinical outcomes in patients with peripheral artery disease after endovascular procedures. 25 Because renin and CITP were significantly elevated in our cohort of patients with AF, we investigated the role of these biomarkers in risk stratification of patients with AF. It would have been interesting to study how types of AF affects these biomarker levels. Unfortunately, only a few patients in this cohort had established information on specific type of AF which limited our ability to do this analysis. The incidence of complications such as stroke, heart failure, myocardial infarction, and chronic kidney disease after the blood sample collection date was noted. In our cohort, we did not find any significant correlations between the biomarkers and these life-threatening complications, each of which was tested individually. However, this subgroup analysis lacks significant power since it reduces the already small sample size into even smaller groups. Therefore, it would be beneficial to investigate the predictive value of renin and CITP with larger samples sizes to determine the clinical utility of these biomarkers as prognostic factors in risk stratification of patients with AF.
Renin and Collagen Remodeling
In terms of the general relationship between RAS and collagen remodeling, previous studies have described angiotensin II as the main mediator of cardiac structural and electrical remodeling in various cardiovascular diseases. 26 –28 Some studies have even demonstrated the reduction in new onset AF in patients with heart failure or end-stage renal disease by using ACE inhibitors and ARBs. 29,30 Our data suggest that renin may also be involved in this collagen remodeling process, thus providing another potential target for therapeutic intervention. In a canine model of AF, Satoh et al demonstrated the utility of aliskiren, a direct renin inhibitor, in suppressing structural and electrical remodeling. 31 Investigating the effect of using direct renin inhibitors in humans with AF may elucidate their potential utility in clinically targeting renin to modulate collagen remodeling in this disease process.
Paradoxically, patients with AF in this cohort had decreased plasma levels of angiotensin II compared to normal controls even after doing the sensitivity analysis excluding patients on ACE inhibitors and ARBs. This was an unexpected finding considering prior studies that suggest the utility of ACE inhibitors and ARBs in reducing the incidence of AF in patients with other diseases, implying that angiotensin II would be elevated in these patients. 18,19 A study done by Walters et al supports our hypothesis that this be may be due to upregulation of angiotensin-converting enzyme 2 (ACE2) which degrades angiotensin II into smaller fragments in the AF disease process. 32
Limitations
This study has several limitations. As mentioned previously, the small sample size limits the power of the study. Unfortunately, samples were collected from a cohort of patients prior to undergoing catheter ablation, and recruitment was much slower at our institution than anticipated. Additionally, plasma levels of MMP-1 and MMP-2 were only determined for the first 7 patients who were recruited in this cohort, limiting the correlational analysis between these 2 biomarkers and renin and angiotensin II. Due to the small sample size and lack of sex and racial/ethnic diversity in this study, generalizability is reduced.
As mentioned in the Materials and Methods section, control samples were obtained from a biomedical company that did not provide any demographic information beyond ensuring that samples were from healthy, nonsmokers who do not take any medications. In retrospect, we could have recruited a cohort of healthy age, gender, and race-matched individuals without AF. This may have prevented any biomarker variations that occur with demographic differences.
Lastly, the fact that a number of patients in the AF cohort also had other comorbid cardiovascular illnesses and were using medications to treat these diseases could have interfered with baseline functioning of RAS and vitamin D and thus impacted biomarker profiling despite sensitivity analysis. We considered making vitamin D supplementation an exclusion criteria; however, many patients under consideration for recruitment for this study were unaware of their supplementation status and/or what type of vitamin D they were receiving. We also considered making presence of comorbid cardiovascular diseases and use of their medications exclusion criteria. However, most patients qualifying for catheter ablation inevitably had other cardiovascular complications. In other words, lone AF was difficult to find in this cohort of patients. Thus, we included these patients regardless of comorbid conditions and medication use and did our best to counter for their influence in retrospect.
Future Directions
Future studies using larger sample sizes could be helpful in determining the utility of renin and CITP as markers of risk stratification in patients with AF. Studies focusing on the use of direct renin inhibitors in humans with AF could also provide more clarity on their utility as agents that prevent structural and electrical remodeling of the heart. Correlating biomarker levels with echocardiogram findings of left atrial diameter or volume could help in determining the role of biomarkers in approximating structural change in AF. Finally, more studies are needed to better understand why angiotensin II is reduced in this cohort.
Conclusion
Renin and CITP elevations suggest that RAS and collagen remodeling may be involved in the pathogenesis of AF. Although our data do not suggest the idea that 25-hydroxyvitamin D deficiency is associated with AF, it does demonstrate that 25-hydroxyvitamin D and renin have an inverse relationship in patients with AF. Additionally, renin may be an important mediator in the collagen dysregulation based on its positive correlations with collagen remodeling biomarkers. Further investigation is necessary to determine whether therapeutically targeting renin could affect collagen remodeling in AF. Further investigation is also needed to elucidate why angiotensin II is decreased in these patients. Understanding mediators of RAS dysregulation may provide targets for therapeutic intervention in patients with AF.
Footnotes
Authors’ Note
Ethical approval to report this case was obtained from Loyola University Chicago Health Sciences Institutional Review Board (IRB#205530). Written informed consent was obtained from patients for their anonymized information to be published in this article.
Acknowledgments
The authors are grateful to the patients and staff involved with the collection of samples at the Loyola University Medical Center Department of Cardiology. We would like to thank the Hemostasis and Thrombosis laboratories at Loyola University Chicago Health Sciences Campus for their mentorship and guidance. Research reported in this manuscript was supported by T35 training grant from the National Heart Lung and Blood Institute (NHLBI) of the National Institutes of Health (NIH) under the award number T35 HL120835.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this manuscript was supported by the T35 training grant from the National Heart Lung and Blood Institute (NHLBI) of the National Institutes of Health (NIH) under the award number T35 HL120835.
