Abstract
LCZ696, a first-in-class angiotensin receptor neprilysin inhibitor, has been demonstrated to have greater advantages in the treatment of heart failure compared with angiotensin-converting enzyme inhibitors, enalapril, or angiotensin receptor blockers (ARBs). However, studies that compared the efficacy and safety of LCZ696 against valsartan in patients with hypertension are limited. To provide further evidence for the benefits of LCZ696 and to make this assessment, a meta-analysis of randomized controlled trials (RCTs) was performed. The Cochrane Central Register of Controlled Trials (CENTRAL), EMBASE, MEDLINE, PubMed, and ClinicalTrials.gov were searched for RCTs. Twelve studies involving 3816 patients were eligible for inclusion. Seven studies compared LCZ696 with valsartan, and 5 studies compared LCZ696 with olmesartan. LCZ696 showed a significantly greater reduction in systolic blood pressure (BP; mean difference [MD] = −5.43 mm Hg; 95% confidence interval [CI]: −6.36 to −4.49 mm Hg; P < .001), diastolic BP (MD = −2.34 mm Hg; 95% CI: −2.67 to −2.01 mm Hg; P < .001), 24-hour ambulatory systolic BP (MD = −3.57 mm Hg, 95% CI: −4.29 to −2.85 mm Hg; P < .001), and 24-hour ambulatory diastolic BP (MD = −1.32 mm Hg, 95% CI: −1.77 to −0.78 mm Hg; P < .001) from the baseline than ARBs. LCZ696 was more effective in reducing BP (odds ratio [OR] = 5.34; 95% CI: 4.49-6.36; P < .01) and had a higher rate of BP control compared with ARBs (OR = 1.52; 95% CI: 1.37-1.69; P < .01). LCZ696 had no difference in the incidence of adverse events (OR = 1.05; 95% CI: 0.94-1.18; P = .38) or serious adverse events (OR = 0.80; 95% CI: 0.51-1.24; P = .31) compared to ARBs. This meta-analysis revealed that LCZ696 has a greater antihypertensive efficacy and an equal tolerability profile.
Summary table
Introduction
Hypertension is an independent risk factor for cardiovascular (CV) events accounting for 9.4 million CV deaths worldwide every year. By 2025, the number of patients with hypertension is predicted to reach 1.56 billion people. 2 Despite the strong positive relationship between blood pressure (BP) levels and CV disease risks, two-thirds of the patients have remained untreated or have been controlled ineffectively. Activation of the sympathetic nervous system and renin–angiotensin–aldosterone system (RAAS) are the major pathophysiologic mechanisms of hypertension. Therefore, the RAAS has become one of the most important targets for CV disease therapy. Among the 5 main classes of antihypertensive drugs, angiotensin receptor blockers (ARBs) have been proved to play an important role in regulating BP, with the absolute benefits for patients with heart failure (HF). 3,4 Recently, studies have reported a new pharmacological class of angiotensin receptor neprilysin inhibitor. As detailed above, LCZ696 comprises molecular moieties of valsartan and AHU377, which are present in a 1:1 molar ratio. AHU377 is a prodrug that is metabolized to the active neprilysin (NEP) inhibitor LBQ657 by cleavage of its ethylester. The Prospective comparison of ARNI with ARB on Management Of heart failUre with preserved ejectioN fracTion: a phase 2 double-blind randomised controlled trial (PARAMOUNT-HF) trial was a phase II trial that assesses the efficacy and safety of LCZ696 as compared to valsartan in 300 patients with signs and symptoms of HF and heart failure with preserved ejection fraction (HFpEF). The primary outcome N-terminal prohormone of brain natriuretic peptide (NT-proBNP) level, a marker of disease severity in patients with HFpEF, was significantly reduced from the baseline by LCZ696 compared with valsartan. 5 The study also reported that LCZ696 had a significantly greater BP reduction than valsartan and no significant difference in adverse events (AEs) between the 2 groups. The results of the PARAMOUNT trial have now contributed to the recently initiated Prospective comparison of ARni with ARB Global Outcomes in heart failure with preserved ejectioN fraction (PARAGON-HF) trial (ClinicalTrials.gov identifier NCT01920711) which aims to evaluate the effect of LCZ696 compared with valsartan in reducing the rate of the composite CV death and HF hospitalization in approximately 4000 patients with HFpEF and New York Heart Association (NYHA) class II to IV. This ongoing study is expected to be completed in 2019.
The superior effects of LCZ696 might be explained by synergistic benefits based on the concurrent effect of NEP inhibition and AT1 receptor blockade in patients with HF. Hypertension is a reversible factor for HF in the early stages. If BP can be decreased effectively, the left ventricle relaxing and filling can be improved by reducing the left atrial and end-diastolic pressure. 6 LCZ696 reduced the BP better than valsartan; however, regression results of the BP changes showed that the benefits for NT-proBNP and left atrial size reduction were independent of the lowered BP. 5
All participants in the current clinical trials included patients with HF and excluded hypertension. It is difficult to estimate the role of LCZ696 in those patients with hypertension without HF. LCZ696 might be a better choice for patients with hypertension with high-risk CV events due to its greater hypotension property and additive CV protective effect.
It is hypothesized that LCZ696 can delay the progression of hypertension to HF and have a reversible effect on CV remolding for patients with hypertension, such as hypertrophy induced by high BP. The objective of this meta-analysis was to provide useful data on the efficacy and safety of LCZ696 compared with valsartan in patients with hypertension.
Materials and Methods
Search Strategy for Identification of Studies
For this study, a search was made in The Cochrane Central Register of Controlled Trails (CENTRAL), EMBASE, MEDLINE, PubMed, and ClinicalTrials.gov until December 31, 2016, with the medical subject heading. The keywords included “hypertension,” “high blood pressure,” “angiotensin receptor blockers,” “angiotensin receptor antagonists,” “angiotensin II receptor antagonists,” “angiotensin II receptor blockers,” “azilsartan,” “candesartan,” “irbesartan,” “losartan,” “olmesartan,” “telmisartan,” “valsartan,” “eprosartan,” “LCZ696,” “ARNI,” and “angiotensin receptor neprilysin inhibitor.” A further search of the conference proceeding from the European Society of Cardiology, the American College of Cardiology, and American Heart Association was conducted. The search was restricted to randomized controlled trials (RCTs) of LCZ696 versus ARBs in patients with hypertension published in peer-viewed journal. All included studies were needed to report the systolic BP (SBP), diastolic BP (DBP), 24-hour ambulatory systolic BP (AMSBP), 24-hour ambulatory diastolic BP (AMDBP), BP control rate, and incidence of AEs as outcomes. Reports completed recently were also included.
Study Selection
Two reviewers (Y.Z. and X.Z.) abstracted data from eligible studies separately. Studies were selected if they fulfilled the following inclusion criteria—(1) The studies were prospective, randomized, controlled trials; (2) The patients in the studies had essential hypertension; (3) Patients were randomly assigned to LCZ696 and ARBs; (4) The studies reported clinical outcomes including SBP, DBP, 24-hour AMSBP and AMDBP, BP control rate, and incidence of AEs; (5) Patients with “hypertension” enrolled in this study were restricted to participants with BP >140/90 mm Hg or on antihypertensive therapy; and (6) No limitations of sex or age. After full-text assessment, studies that complied with the above-mentioned criteria and that possessed sufficient data were included in the analysis.
Data Extraction and Quality Assessment
According to the prespecified selection criteria and Cochrane reviewer’s handbook, the 2 reviewers (Y.Z. and X.Z.) extracted data independently. Inconsistencies were resolved by discussion. The following data were extracted from each trial: publication year, study design, random sequence generation, allocation sequence concealment, blinding method, total number, age, gender distribution, duration of therapy, and BP of patients at baseline. The quality of this study was assessed by the Cochrane risk of bias. 7 Y.Z. and X.Z. assessed the risk of bias of all included trials and prepared a risk of bias table as described in chapter 8 of the Cochrane handbook (Figures 1 and 2).

Methodological quality graph: reviewer author's judgments about each methodological quality item presented as percentage across all included studies.

Methodological quality summary: review authors' judgments about each methodological quality.
Outcome Measures
The following end points were compared in the LCZ696 versus ARBs groups: (1) change from the baseline in SBP and DBP; (b) change from the baseline in 24-hour AMSBP and 24-hour AMDBP; (c) number of participants who achieved BP control, defined as SBP/DBP <140/90 mm Hg; (d) AEs, defined as hyperuricemia, nasopharyngitis, dizziness, and headache; and (e) serious AEs, defined as serious cardiac disorders, that is, arrhythmia, edema, malignancy, pruritus, diarrhea, and dyspnea.
Statistical Analyses
Eligible clinical results were BP reduction, 24-hour ambulatory BP (AMBP) reduction, BP control rate, and AEs (serious and nonserious). Changes in BP were presented as the mean difference (MD) with a 95% confidence interval (CI). Each dichotomous outcome was summarized by a percentage. A summary of an odds ratio (OR) and their corresponding 95% CI for BP control rates and AEs was also obtained. All analyses were compiled using review manager software (Review Manager; RevMan version 5.3.3 Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). Although all studies were included independently and followed the exclusion and inclusion criteria strictly, heterogeneity was still inevitable. The heterogeneity of studies was assessed with the Q statistic and I 2 statistic and I 2 value of 0%, 25%, 50%, and 75% showing no, low, moderate, and high heterogeneity, respectively. I 2 > 50% indicated statistically significant heterogeneity between studies.
Funnel plots of symmetry were used to graphically assess publication bias. All tests in the analysis were considered statistically significant and were 2 sided with P < .05. All results were reported in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement. 8
Results
Study Characteristics
The flow diagram of study selection is shown in Figure 3. A total of 441 articles were identified and screened for retrieval. After removing all duplicates (232 articles), the 209 full-text articles were assessed and then 164 studies were excluded from the analyses by Y.Z. and X.Z. after reading titles and abstracts. Because they did not report outcomes of interest for this research, 17 studies were excluded. In 16 studies, the outcomes have not been published, so they were also excluded. Finally, 12 studies were included in the quantitative synthesis. The 12 trials were conducted in a number of different countries. Almost 50% of the studies were conducted in the United States (5 studies). 9 –13 This review also included other countries: China (6), 9,11,12,14 –16 Japan (4), 12,17 –19 Argentina, Canada, Philippines, Hungary, India, Romania, Greece, Italy, Russian, Spain, Korea, Singapore, and Thailand. All trials used a parallel group design.

Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram of study selection.
A total of 3816 individuals with hypertension from 12 clinical trials were included in this meta-analysis. Table 1 lists the design and baseline characteristics of the included studies. Patients enrolled in these studies were all older than 18. Four studies included patients over 60 years of age. 12,18 LCZ696 was compared with olmesartan in 5 trials 10,12,14,16,17 and with valsartan in 7 trials. 9,11,13,19 –22 Only 1 study compared LCZ696 at doses of 100, 200, and 400 mg/d with ARBs; 1 study compared 200 mg/d LCZ696 with ARBs. Two studies compared 200 and 400 mg/d LCZ696 with ARBs, and 8 studies compared LCZ696 400 mg/d with ARBs. 11 Study durations ranged from 8 to 52 weeks.
Baseline Characteristics of Trials included in the Meta-Analysis.

Abbreviations: DB, double-blind; LCZ, LCZ696/angiotensin receptor neprilysin inhibitor; msDBP, mean seated diastolic blood pressure; msSBP, mean seated systolic blood pressure; OL, open-label; Olm, olmesartan; PG, parallel groups; Val, valsartan.
Blood Pressure Reduction Effects
In Figures 4 and 5, results showed a significant reduction in SBP (−5.43 mm Hg; 95% CI: −6.36 to −4.49 mm Hg; P < .001) and DBP (MD = −2.34 mm Hg; 95% CI: −2.67 to −2.01 mm Hg; P < .001) in patients with hypertension receiving LCZ696 therapy when compared with ARBs. As compared with ARBs, LCZ696 significantly decreased the 24-hour AMSBP (MD = −3.57 mm Hg; 95% CI: −4.29 to −2.85 mm Hg; P < .001) and DBP (MD = −1.32 mm Hg; 95% CI: −1.77 to −0.78 mm Hg; P < .001; Figures 6 and 7). However, substantial heterogeneity was observed in BP and 24-hour AMBP, hence random-effects model was used to incorporate heterogeneity among studies.

Comparison of LCZ696 with angiotensin receptor blockers (ARBs) on the outcome of systolic blood pressure.

Comparison of LCZ696 with angiotensin receptor blockers (ARBs) on the outcome of diastolic blood pressure.

Comparison of LCZ696 with angiotensin receptor blockers (ARBs) on the outcome of 24-hour ambulatory systolic blood pressure.

Comparison of LCZ696 with angiotensin receptor blockers (ARBs) on the outcome of 24-hour ambulatory diastolic blood pressure.
Blood Pressure Control Rates
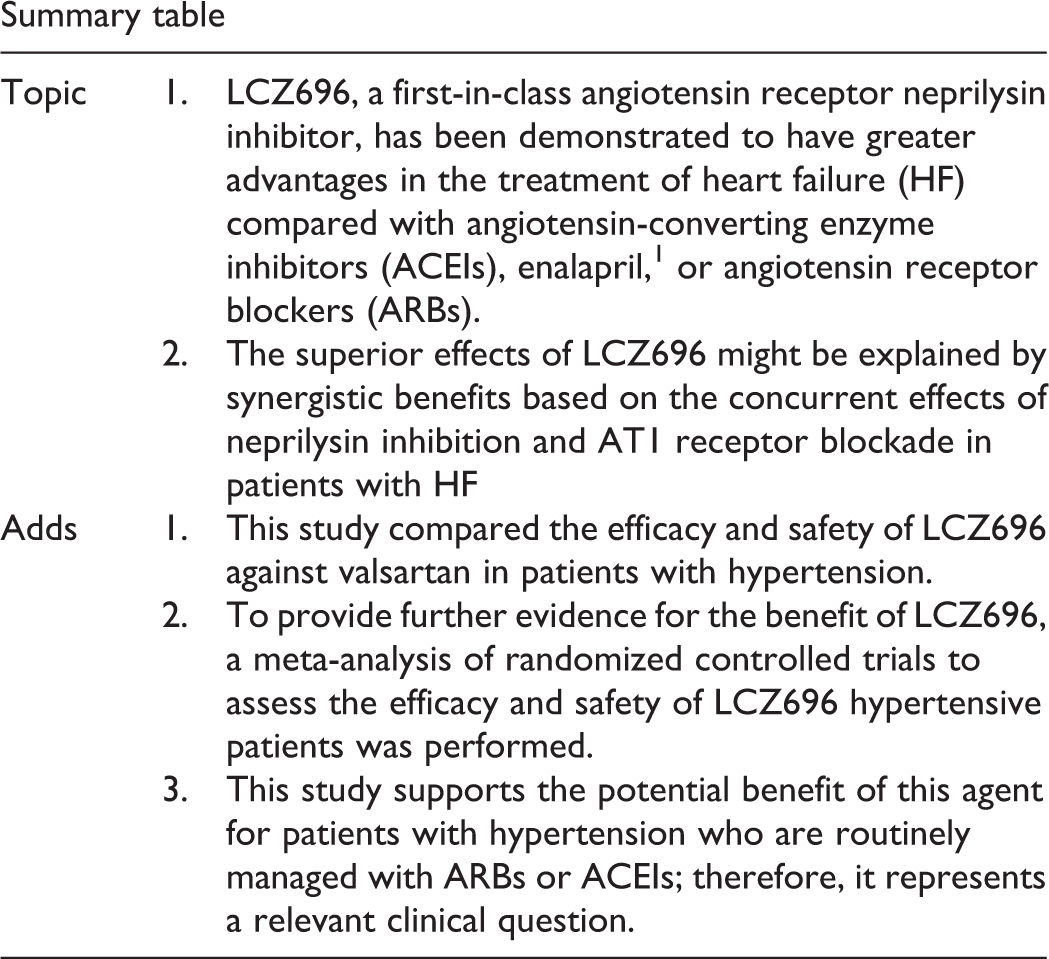
The response of BP to drug treatment was defined as SBP ≤140 mm Hg and DBP ≤90 mm Hg. In this meta-analysis, LCZ696 was significantly more effective in reducing BP than ARBs, especially in patients who were administered a high dose of LCZ696 (400 mg; Figure 8). A fixed-effects model was used due to the slight heterogeneity among these studies.
Comparison of LCZ696 with angiotensin receptor blockers (ARBs) on the blood pressure control rate.
Adverse Events (Serious and Nonserious)
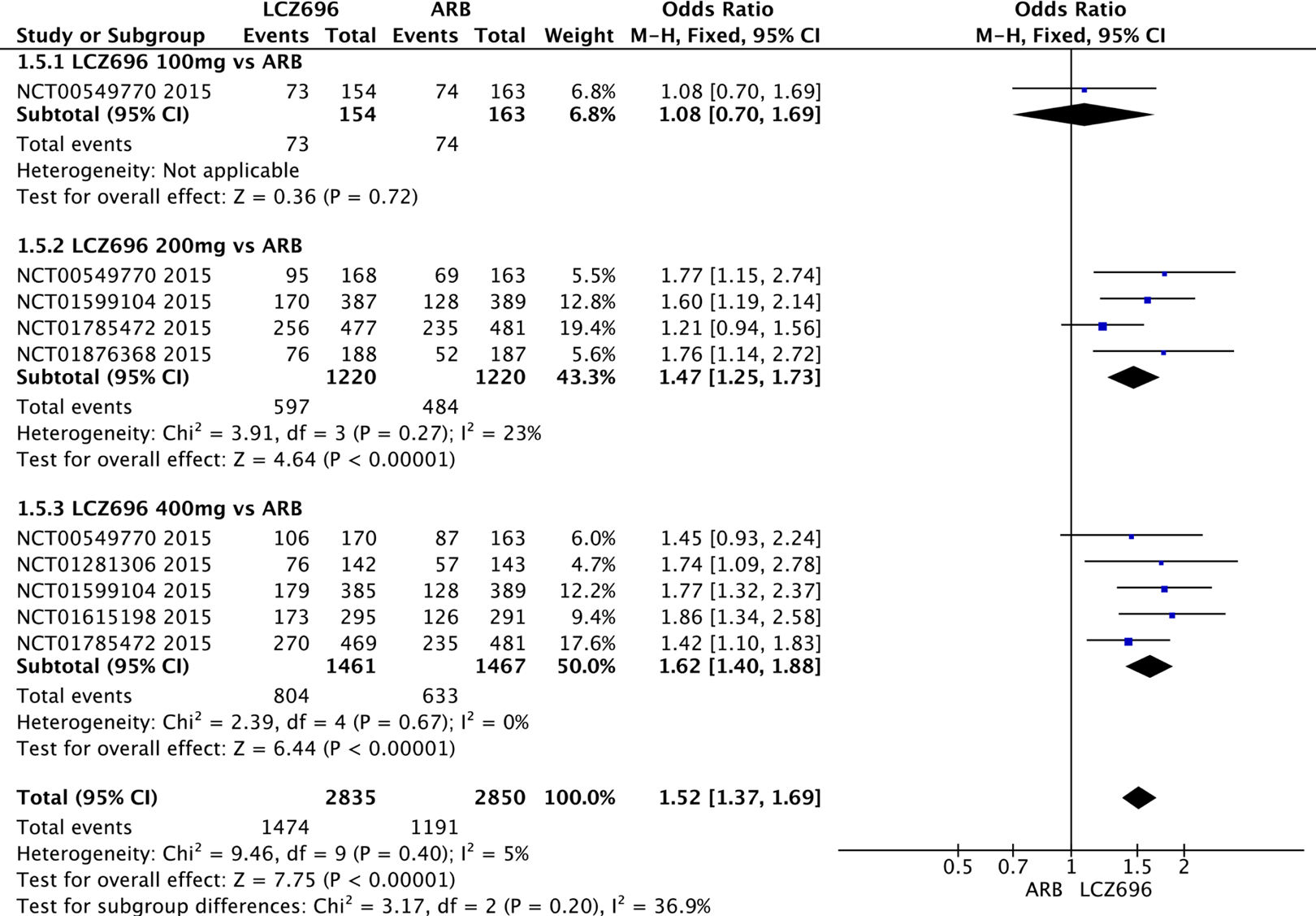
There was no significant difference between LCZ696 and ARBs in the rates of AEs (OR = 1.05; 95% CI: 0.94-1.18; P = .38; Figure 9A) or serious AEs (OR = 0.80; 95% CI: 0.51-1.24; P = .31) in both groups (Figure 9B).
A, Relative risks of adverse events (AEs) for LCZ696 and angiotensin receptor blockers (ARBs). B, Relative risks of serious AEs for LCZ696 and ARBs.
Effects of Different Doses of LCZ696 Versus ARBs
Evaluation of the efficacy and safety of different LCZ696 doses was performed in this meta-analysis. After treatment with 400 mg/d LCZ696, SBP (MD = −5.21 mm Hg; 95% CI: −5.26 to −5.16 mm Hg; P < .001), DBP (MD = −2.29 mm Hg; 95% CI: −2.32 to −2.26 mm Hg; P < .001), 24-hour AMSBP (MD = −3.74 mm Hg; 95% CI: −3.80 to −3.68 mm Hg; P < .001), and 24-hour AMDBP (MD = −1.70 mm Hg; 95% CI: −1.74 to −1.67 mm Hg; P < .001) were significantly reduced when compared with ARB. LCZ696 at 200 mg/d also showed a reduction in SBP (MD = −3.50 mm Hg; 95% CI: −3.56 to −3.44 mm Hg; P < .001), DBP (MD = −1.65 mm Hg; 95% CI: −1.69 to −1.62 mm Hg; P < .001), 24-hour AMSBP (MD = −3.37 mm Hg; 95% CI: −3.43 to −3.31 mm Hg; P < .001), and 24-hour AMDBP (MD = −1.55 mm Hg; 95% CI: −1.58 to −1.51 mm Hg; P < .001) when compared with ARBs. LCZ696 at 100 mg/d was associated with a slight reduction in SBP (MD = −1.31 mm Hg; 95% CI: −1.56 to −1.06 mm Hg; P < .001), DBP (MD = −0.83 mm Hg; 95% CI: −0.99 to −0.67 mm Hg; P < .001), and 24-hour AMSBP (MD = −0.51 mm Hg; 95% CI: −0.90 to −0.12). Notably, compared with 100 mg/d LCZ696, 24-hour AMDBP (MD = 0.76 mm Hg; 95% CI: 0.49-1.03 mm Hg; P < .001) was slightly reduced after treatment with ARBs. 11 In the subgroup analysis, no beneficial effect was found on the achievement of target BP by 100 mg/d LCZ696 (OR = 1.08; 95% CI: 0.70-1.69; P = .72). LCZ696 200 and 400 mg provided significantly greater power versus ARBs on the target BP achievement (OR = 1.47, 95% CI: 1.23-1.73, P < .001; OR = 1.62, 95% CI: 1.40-1.88, P < .001; Figure 8). However, there was no significant difference between the 100 mg/d LCZ696 and ARBs on the BP control rate. Compared with ARBs, there was no difference in the rate of AEs or serious AEs in LCZ696, regardless of 100 mg (AEs: OR = 0.83, 95% CI: 0.22-3.16, P = .79; serious AEs: OR = 1.05, 95% CI: 0.06-16.86, P = .98), 200 mg (AEs: OR = 0.94, 95% CI: 0.78-1.13, P = .52; serious AEs: OR = 0.42, 95% CI: 0.17-1.05, P = .06), or 400 mg (AEs: OR = 1.13, 95% CI: 0.98-1.13, P = 1.10; serious AEs: OR = 0.99, 95% CI: 0.59-1.67, P = .98; Figure 9A and B).
Heterogeneity
Systolic BP (MD = −5.21 mm Hg; 95% CI: −5.26 to −5.16 mm Hg), DBP (MD = −2.34 mm Hg; 95% CI: −2.67 to −2.01 mm Hg), 24-hour AMSBP (MD = −3.57 mm Hg; 95% CI: −4.29 to −2.85 mm Hg), and 24-hour AMDBP (MD = −3.57 mm Hg; 95% CI: −4.29 to −2.85 mm Hg) were all significantly reduced after being treated with LCZ696 compared to ARBs. There was evidence of high statistical heterogeneity for all of these outcomes (I 2 = 97.7% for SBP, I 2 = 97.9% for DBP, I 2 = 97.1% for 24-hour AMSBP, and I 2 = 97% for 24-hour AMDBP). There was low to moderate statistical heterogeneity for the outcomes of BP control rate (I 2 = 5%), AEs (I 2 = 22%), and serious AEs (I 2 = 32%).
Bias of Included Studies
All of the included trials did not describe the method used to conceal the allocation sequence in sufficient detail, so the bias of allocation concealment was unclear. All trials were randomized and double-blinded and reported all prespecified outcomes. Six trials were parallel group, multicenter trials. Overall, the included studies were of high quality.
Discussion
This meta-analysis evaluated the efficacy and safety of LCZ696 in patients with hypertension compared with ARBs. It was found that (a) LCZ696 significantly reduced BP versus ARBs, without an increase in AEs; (b) the BP-lowering capacity of LCZ696 seemed to be higher than ARBs observed in both office BP and 24-hour AMBP; and (c) the 400 mg/d LCZ696 dose in reducing BP was stronger than 100 and 200 mg/d doses without increasing the risk of AEs.
This meta-analysis showed that LCZ696 has a greater antihypertensive efficacy and an equal tolerability profile. LCZ696 represents a new alternative treatment for patients with hypertension, particularly in terms of its superiority over standard ARBs in patients with HF. Hypertension is a worldwide but controllable disease, 23 probably influencing 30% to 45% of the general population. 24 High BP is associated with a high risk of CV events and increased CV mortality exponentially when levels of BP ≥115/75 mm Hg. Although antihypertensive drugs have successfully been developed, many people with high BP don’t even know they have it, due to no symptoms or warning signs of hypertension, so there is still room for improvement.
Thus, there is an emergent medical demand for novel agents to treat hypertension. Recently, the attention of the medical research teams in the use of LCZ696 in hypertension has grown, as clinical trials and animal experiments have shown its cardioprotective and nephroprotective capacities. 25 –28 The superior clinical benefits of LCZ696 in CV disease were expected. However, it is worth noting that 1 moiety of LCZ696, AHU377, an inhibitor of NEP, can break down the Aβ peptide that forms amyloid plaques in the brain. For patients with hypertension treated for decades, LCZ696 might potentially increase Aβ levels and increase the risk of Alzheimer disease. 29 The Prospective comparison of ARNi with ACEi to Determine Impact on Global Mortality and morbidity in Heart Failure trial (PARADIGM-HF) trial does not indicate any adverse effects related to cognitive impairment with LCZ696. Further study on this issue is required.
Mechanisms
LCZ696 inhibits both AT1 receptor and natriuretic peptide (NP) degradation. The NP system plays a key role in the pathophysiology of the CV system, including atrial natriuretic peptide (ANP), brain natriuretic peptide, C-type natriuretic peptide, and D-type natriuretic peptide. 30 Neprilysin inhibitors accelerate vasodilation and natriuretic by increasing the plasma concentration of the vasoactive peptide substance P 31 and preventing the breakdown of bradykinin and endogenous NPs. However, NEP inhibition leads not only to an increased concentration of vasodilator peptides but also to an increased levels of vasoconstrictor peptides. 32 This may explain the variable effects of NEP inhibitor on BP, which can be unchanged, increased, or decreased in normotensive and hypertensive people. It has been demonstrated that RAAS also has a “protective arm.” The protective effect is conducted by stimulating the AT2 receptor and the Mas receptor through Ang-(1-7), respectively. 33 The vasodilation effects induced by the stimulated AT2 receptor can produce only if the AT 1 receptor is blocked as well. 34 LCZ696 combines NEP inhibition with an ARB moiety, increasing the levels and effects of ANP while blocking the effects of Ang II. It is speculated that the presence of higher levels of Ang II tends to bind AT2 R and produce the positive biology response. All of these mechanisms appear to be responsible for the greater hypotensive effect by LCZ696 compared to ARBs. It is tentatively put forward that the greater capacity of BP reduction in LCZ696 can provide a better cardioprotective effect, especially in patients with hypertensive heart disease.
Strength and Limitations of This Meta-Analysis
This is the first meta-analysis that reports on the efficacy and safety of LCZ696 on BP in patients with hypertension. It includes only RCTs. All trials included in this analysis are double-blind trials and half of these trials are multicentered; therefore, the quality is high. LCZ696 presents a new alternative treatment for patients with hypertension, particularly in light of its superiority over a standard angiotensin receptor blockade in patients with HF. It is crucial to investigate whether superior efficacy results associated with favorable safety can also be expected when LCZ696 is used as an antihypertensive drug. This study provides useful data on which to base future research.
The evidence presented here is most convincing for patients older than 18 years. Only 2 trials recruited participants older than 60 years. One was the Prospective Comparison of Angiotensin Receptor Neprilysin Inhibitor with Angiotensin Receptor Blocker Measuring Arterial Stiffness in the Elderly trial and a study from Izzo et al which evaluated the efficacy of LCZ696 on these patients. This study indicates that LCZ696 is suitable to diminish pulse pressure in elderly patients. 35 Izzo et al concluded that LCZ696 causes greater SBP reduction than valsartan in those aged 65 years or older but only reduced DBP slightly in those under 65 years. 20
Some limitations in this article should be acknowledged. First, despite the substantial data on the effectiveness of antihypertensive LCZ696 and ARBs in patients with hypertension, there were only 2 types of ARBs (valsartan and olmesartan), which might lead to low statistical power. Second, it is difficult to control the confounding factors such as age, sex, and other potential factors. Also, a subgroup analysis in different populations, such as patients with resistant hypertension, elderly patients with hypertension, and patients from different races could not be conducted, which may cause a significant heterogeneity in BP and ABPM analysis. Third, the effects of antihypertensive agents may be different from the variation in how and when the BP was measured. Fourth, unpublished data were not included and only selected articles published in English were used. Fifth, different duration, different study groups, and different antihypertensive drugs used at the baseline may lead to confounding bias and affect the antihypertensive effect. Finally, this meta-analysis may be underpowered for a long-term AE comparison between LCZ696 and ARBs due to the relatively short duration of the included RCTs.
Conclusions
In conclusion, compared with ARBs, LCZ696 significantly decreased office BP and AMBP without increasing AEs. The meta-analysis indicates that the superiority BP-lowering capacity of LCZ696 may be due to the concomitant effect of the NEP inhibitor and valsartan. As a result, LCZ696 might provide greater CV protection. Although the precise mechanism remains to be elucidated, the analysis supports the potential benefit of this agent for patients with hypertension who are routinely managed with ARBs or angiotensin-converting enzyme inhibitors; therefore, this situation represents a relevant clinical question. Further RCTs of this agent within the hypertensive population are required to establish the benefits within the patient subpopulation and the impact on morbidity and mortality for long-term treatment. More research is required to evaluate which hypertensive populations without HF are most likely to benefit. 25 Thus, which hypertensive patients populations would most benefit from LCZ696 needed to be identified.
Footnotes
Author Contributions
Yang Zhao contributed to conception and design of the work and drafting the article. Xu Zhao contributed to data collection. Ruixin Ma contributed to data analysis and modifying the manuscript. Ningyin Li and Heng Yu contributed to revision of the article. Jing Yu contributed to final approval of the version to be finished.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (NSFC-81270332), Gansu Province Natural Science Foundation (1104FKCA150, 1205TCYA042), and the Gansu Administration of Traditional Medicine Foundation (GZK- 2010-Z-1).
