Abstract
The two major classes of drugs that target the RAS are the angiotensin-converting enzyme (ACE) inhibitors and the selective AT1 receptor blockers (ARBs). Although both of these drug classes target angiotensin II, the differences in their mechanisms of action have implications for their effects on other pathways and receptors that may have therapeutic implications. Both ACEIs and ARBs are effective antihypertensive agents that have been shown to reduce the risk of cardiovascular and renal events. Direct inhibition of renin –the most proximal aspect of the RAS –became clinically feasible from 2007 with the introduction of aliskiren. This latter drug has been shown to be efficacious for the management of hypertension. Combined therapy of direct renin-inhibitors with ACEIs or ARBs has been tested in some clinical situations as congestive HF and proteinuria with diverse results. This article tries to offer an updated review of current knowledge on the use of RAS blocking drugs in clinical settings.
The Renin–Angiotensin System
The renin–angiotensin system (RAS) preserves fluid volume during periods of restricted dietary salt and also prevents ischemia during acute volume loss. The main effector peptide of the RAS is angiotensin II. It induces vasoconstriction and sympathetic activation, raises aldosterone levels, and promotes renal salt and water retention via the angiotensin II type 1 (AT1) receptor. 1 Over the last few decades, the RAS has been a drug target of particular interest because of its involvement in cardiovascular disease (CVD) and renovascular disease. The CVD and renovascular disease can be understood as a continuum of risk factors, target organ damage, events, and mortality. Risk factors (such as hypertension, dyslipidemia, diabetes, and smoking) lead to the development of target organ damage including atherosclerosis, left ventricular hypertrophy (LVH), and renal impairment. Target organ damage progressively worsens, leading ultimately to myocardial infarction (MI), heart failure (HF), end-stage renal disease (ESRD), stroke, or death. 2 Angiotensin II, the main effector peptide of the RAS, plays an active role during all stages of this continuum. The first step in the RAS cascade is the formation of angiotensin I from the precursor angiotensinogen under the action of renin; indeed, early evidence for the importance of RAS in CVD came from the consistent finding that renin activity is predictive of the risk of cardiovascular (CV) events. Angiotensin I is then converted to angiotensin II, the principal effector peptide of the RAS, by angiotensin-converting enzyme (ACE). In addition, angiotensin II can be produced in tissues by enzymes such as chymase. This locally produced angiotensin II is believed to mediate paracrine and autocrine functions. Angiotensin II acts via 2 receptor subtypes, AT1 and AT2. Activation of AT1 receptors results in vasoconstriction, aldosterone and vasopressin secretion, sodium retention, and decreased renal perfusion. Hence, these receptors mediate the deleterious effects of angiotensin II, including elevated blood pressure (BP) and cardiac and vascular remodeling. 3 The effects of the AT2 receptors have been less clearly defined because of the limited expression of these receptors in adults, because of their unconventional signaling pathways, and because many AT2-mediated actions are masked by opposing AT1-mediated effects. However, it is now recognized that AT2 receptors generally oppose the actions of AT1 receptors, mediating various antiproliferative and anti-inflammatory effects and promoting tissue differentiation and regeneration and apoptosis. 4
Additional components of the RAS have been identified in the last decade, including bioactive angiotensin peptides, such as angiotensin III, angiotensin IV, and angiotensin-(1-7), the effects of which have not yet been fully elucidated for the CV and renal system. 5
The discovery of the renin receptor has shed further light on the biology of the RAS. Renin, simply considered until recently as the rate-limiting enzyme of RAS activation, has also turned out to be the ligand for a protein known as the renin/prorenin receptor that binds renin and prorenin about equally, regardless of their biologic activities. Prorenin, which represents 70% to 90% of total circulating renin, when bound to the receptor induces an increase in the catalytic efficiency of angiotensinogen conversion to angiotensin I, which contributes to local production of angiotensin II and its systemic levels, as well as binding of renin/prorenin to the renin/prorenin receptor, exerting physiologic effects that are independent of angiorensin II, including activation of intracellular signal pathways, enhanced synthesis of DNA, and stimulation of the release of plasminogen activator inhibitor 1, collagen 1, fibronectin, and transforming growth factor β-1. 6
Renin–Angiotensin Axis Blocking Drugs
The 2 major classes of drugs that target the RAS are the ACE inhibitors (ACEIs) and the selective AT1 receptor blockers (ARBs). Although both of these drug classes target angiotensin II, the differences in their mechanisms of action have implications for their effects on other pathways and receptors that may have therapeutic implications. 7 Both ACEIs and ARBs are effective antihypertensive agents that have been shown to reduce the risk of CV and renal events (see below).
Direct inhibition of renin—the most proximal aspect of the RAS—became clinically feasible from 2007 with the introduction of aliskiren (Rasilez; Novartis Pharmaceuticals, Switzerland). Aliskiren has been shown to be efficacious for the management of hypertension, congestive HF, and proteinuria either as monotherapy or in combination with ACEIs or ARBs. 8 Figure 1 shows the effect site for every drug class.

The RAS system: effect site for each kind of RAS blocking drugs. AT indicates angiotensin; RAS, renin–angiotensin system.
Angiotensin-Converting Enzyme Inhibitors
The ACEIs reduce RAS activation by blocking the conversion of angiotensin I to angiotensin II, leading to decreased activation of both AT1 and AT2 receptors. Angiotensin II type 1 receptors predominantly mediate the pathological effects of angiotensin II, including vasoconstriction and other mechanisms that raise BP as well as vascular hypertrophy, endothelial dysfunction, atherosclerosis, inflammation, and apoptosis. Angiotensin II type 2 receptors, in contrast, mediate mostly opposing and beneficial effects, promoting antiproliferation, differentiation, regeneration, anti-inflammation, and apoptosis. In addition to blocking the conversion of angiotensin I to angiotensin II, ACEIs block the enzymatic degradation of bradykinin. Increased levels of bradykinin contributes to the positive effects of ACEIs, since activation of β-2 receptor leads to release of nitric oxide with vasodilatory and tissue protective results. In addition, evidence is accumulating that ACE itself can act as a cell surface receptor and that binding of an ACEI to the ACE triggers a signaling cascade that leads ultimately to prostaglandin I2 (PGI2) generation and additional vasodilatory effects. 9
Although acute treatment with an ACEI reduces circulating angiotensin II to negligible levels, chronic treatment has been associated with reemergence of angiotensin II, a phenomenon referred to as “reactivation.” In a study of patients with congestive HF on long-term ACEIs therapy, reactivation of angiotensin II occurred in approximately 1 in every 6 patients and reactivation of aldosterone in about 1 in every 3 patients. 10 These late changes in patients receiving ACEIs have been linked to poorer outcomes. 11
The ACE inhibition has been associated with angioedema (very uncommon, 0.1%) and cough. 12,13 A theoretical disadvantage of ACEIs is that it reduces the activity of AT2 receptor along with the AT1 receptor. Despite their drawbacks, ACEIs are an important drug class because of their proven effectiveness for reducing CV events and mortality in patients with advanced CVD evidenced by HF or a history of MI. However, ACEIs may also reduce CV risk in patients with less advanced coronary heart disease (CHD). This was demonstrated in the Heart Outcomes Prevention Evaluation (HOPE) study, which compared ramipril with placebo. 14
Table 1 shows currently approved ACEIs. The most recently approved ACEI is moexipril hydrochloride; it is a nonsulfhydryl-containing precursor of the ACEI moexiprilat. Its only indication is hypertension treatment with the same contraindications and warnings as others ACEIs. 15
Angiotensin-Converting Enzyme Inhibitors.
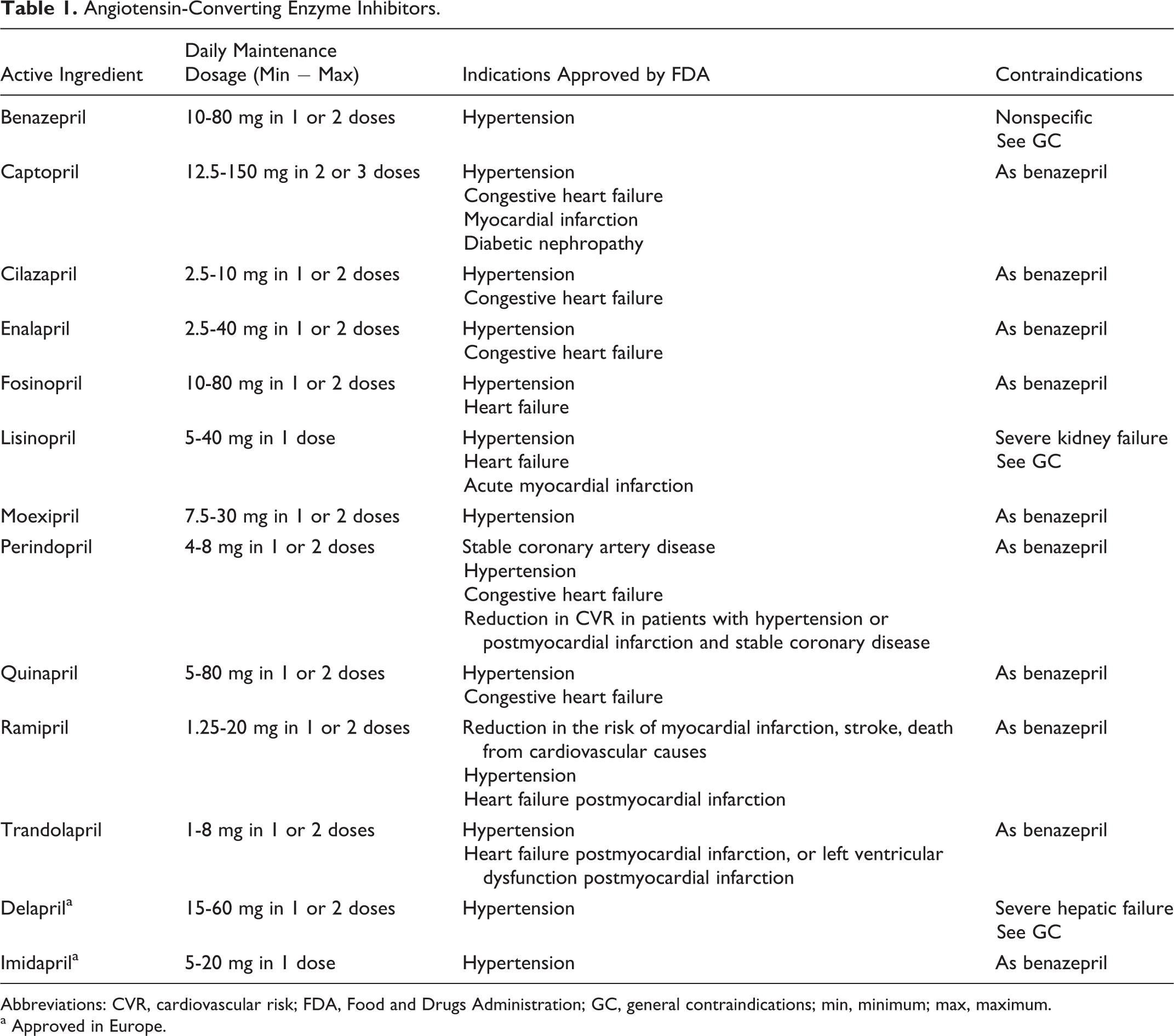
Abbreviations: CVR, cardiovascular risk; FDA, Food and Drugs Administration; GC, general contraindications; min, minimum; max, maximum.
a Approved in Europe.
Angiotensin Receptor Blockers
The ARBs block the RAS by antagonizing the binding of angiotensin II to the AT1 receptor. Because the ARBs are selective for the AT1 receptor, these agents have a greater potential for providing a complete inhibition of the RAS than ACEIs. In addition, ARBs have minimal affinity for the AT2 and thus permit activation of the AT2 receptor by angiotensin II, possibly providing beneficial (but unproven) antiproliferative and anti-inflammatory effects. Unlike ACEIs, ARBs have no effects on bradykinin accumulation and do not induce nitric oxide and PGI2 via the β-2 receptor. The clinical implications of this difference between the ARBs and the ACEIs, aside from the absence of kinin-related cough with ARBs, are not known. The ARBs have been associated with a highly favorable tolerability profile, better than that of any of the other classes of antihypertensives. 16 The clinical evidence for the efficacy of ARBs in reducing CV events has become quite strong as a result of recent major outcome trials. 17
Table 2 shows currently approved ARBs. The most recently approved ARB is azilsartan. The drug substance used in the drug product formulation is the potassium salt of azilsartan medoxomil. Dose adjustment is not required in patients with mild-to-severe renal impairment or ESRD nor it is necessary for patients with mild or moderate hepatic impairment. The only indication is treatment of high BP with the same contraindications and warnings as others ARBs. 18
Angiotensin II Receptor Blockers.

a Taken from KDOQI Clinical Practice Guidelines on Hypertension and Antihypertensive Agents in Chronic Kidney Disease. Am J Kidney Dis. 2004 May;43(5 Suppl 1): S1-290.
b Including renal artery stenosis in the kidney transplant or in a solitary kidney.
Direct Renin Inhibitors
Renin is not only the rate-limiting step in angiotensin II formation but also shows remarkable substrate specificity for angiotensinogen, making it an attractive target for therapeutic inhibition. Furthermore, the recent identification of a renin receptor in the glomerular mesangium and in arterial subendothelium suggests the possibility of additional advantages of renin inhibition beyond those of ACEIs and ARBs. 19 For instance, binding of renin to this receptor not only increases its catalytic efficiency in angiotensin I formation but also converts otherwise inert prorenin into an active moiety. Moreover, the interaction of either renin or prorenin with its receptor, in the absence of angiotensin peptide formation, leads to activation of the potentially pathogenetic mitogen-activated protein kinase pathway. 20
Aliskiren is an orally active renin inhibitor that became the first drug in its class to receive regulatory approval for the treatment of hypertension in 2007. Aliskiren inhibits the first rate-limiting step in the RAS cascade, the conversion of angiotensinogen to angiotensin I, thereby reducing synthesis of all subsequent components of the cascade. In contrast, use of an ACEI or ARB leads to a compensatory rise in the upstream components of the RAS cascade, including plasma renin activity. 21 Clinical studies in patients with hypertension having aliskiren have demonstrated impressive reductions in BP, with similar safety and tolerability to ARBs. This study also demonstrated that side effects in the aliskiren-treated group were comparable to those seen in placebo-treated patients. In clinical trials thus far, the most common adverse events reported have been headache, diarrhea, dizziness, fatigue, and back pain; the frequencies of these are similar to both ARBs and placebo. 22
New Evidences
Angiotensin-Converting Enzyme Inhibitors
In addition to their original indication for hypertension, ACEIs are currently indicated for the treatment of patients at high risk of coronary artery disease (CAD), after MI, with dilated cardiomypathy or chronic kidney disease. Despite the absence of formal studies or indications, they also have widespread off-label use in vascular conditions such as peripheral arterial disease and scleroderma. No new trials have been published in recent years, but several reports about the Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation (ADVANCE) trial (published in 2007) have been released. This trial assessed the CV effects of the administration of a perindopril–indapamide fixed combination, irrespective of initial BP levels. The risk of death from CVD and death from any cause was significantly reduced. A separate report on renal outcomes has been published. Active treatment reduced the risk of renal events, which was driven by reduced risks of developing microalbuminuria and macroalbuminuria. Lower systolic BP levels during follow-up, even to <110 mm Hg, was associated with progressively lower rates of renal events. 23 The relative treatment effects on major adverse cardiovascular events (MACEs) were similar across all stages of chronic kidney disease (CKD). In contrast, the absolute treatment effects approximately doubled in those with CKD stage ≥3 when compared to those with no CKD. 24
Several meta-analyses about the therapeutic effects of ACEIs have been published. All of them were directed to the effects of RAS inhibitors (including ARBs) on CVD. They are described in Table 3. Van Vark et al analyzed the effects of RAS inhibitors as a class of drugs on all-cause mortality. Overall, RAS inhibition was associated with a reduction in all-cause mortality and CV mortality. The observed treatment effect resulted entirely from the class of ACEIs, which were associated with a significant 10% reduction in all-cause mortality, whereas no mortality reduction could be demonstrated with ARBs treatment. 25 The meta-analysis of Baker et al also found that the ACEIs reduce the risk of total mortality and nonfatal MI but increase the risk of syncope and cough compared with placebo. 26 In a third meta-analysis including all antihypertensive drugs, ACEIs came out inferior to calcium channel blockers (CCBs) regarding stroke risk but superior regarding HF risk. 27 A recent review made with only 6 recent outcome trials of BP-lowering drugs in patients with hypertension or high-risk patients concluded that ARBs might offer less protection against MI than ACEIs; therefore, the latter should remain the preferred RAS inhibitor for CV prevention in ACEI-tolerant patients. 35 In a cohort study of hospitalized older patients with HF and preserved ejection fraction not receiving ARBs, discharge initiation of ACEIs was associated with a modest improvement in the composite end point of total mortality or HF hospitalization. 36
Effects of ACEIs/ARBs on CV Mortality and Morbidity.

Abbreviations: ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin II type 1 receptor blocker; BB, β-blocker; CAD, coronary artery disease; CV, cardiovascular disease; CI, confidence interval; HF, heart failure; HR, heart rate; OR, odds ratio; RR, relative risk; MI, myocardial infarction.
a No differences with other drug classes, including ACEIs, were found.
b Versus placebo, CV mortality did not differ between ARB-treated and non-ARB-treated patients, OR 0.98 (0.94-1.02).
c No differences versus other kind of drugs were found.
d CV mortality. % ARBs/ACEIs were evaluated together.
A Cochrane review evaluated the benefits and harms of ACEIs and ARBs or both in people with stage 1 to 3 CKD who do not have diabetes mellitus. The ACEIs had no impact on all-cause mortality or MACE in people with stage 3 CKD. Only a study compared ACEIs with ARB and reported little difference in effect between the treatments when urinary protein, BP, or creatinine. 37
Safety Issues
Geng et al evaluated in a meta-analysis the effect of ACEIs on the development of new-onset type 2 diabetes. The ACEIs treatment was associated with significant reduction in the risk of new-onset diabetes (NOD) in patients with hypertension, CHD or CVD, or HF. 38 Sipahi et al conducted a meta-analysis aimed to determine the effect of ACEIs on cancer occurrence and cancer death; ACEIs therapy did not have any effect on occurrence of cancer, cancer death, or gastrointestinal cancer. 39 Angioedema is a potentially life-threatening adverse event of RAS inhibitors. In a meta-analysis, the risk of angioedema with ACEIs was 2.2 times higher than that with ARBs. Incidence of angioedema was higher in HF trials compared to hypertension or CHD trials without HF. Weighted incidence of angioedema with ACEIs was 0.30% compared to 0.11% with ARBs, 0.13% with direct renin inhibitors (DRIs), and 0.07% with placebo (ARBs and DRIs not significantly different from placebo). 40 A significant association between ACE insertion/deletion polymorphism and ACEI-related cough in studies with mean age <60 years, but not in studies with mean age >60 years, has been reported. 41
A retrospective cohort study concluded that maternal use of ACEIs in the first trimester has a risk profile similar to the use of other antihypertensives regarding malformations in live born offspring. The apparent increased risk of malformations associated with use of ACEIs (and other antihypertensives) in the first trimester may be likely due to the underlying hypertension rather than the medications. 42
Pharmacogenetics
Information on the reasons of heterogeneities in treatment effect between subgroups of patients could be used to develop an evidence-based guidance for the institution of ACEIs therapy. No heterogeneity in risk reduction by ACEIs has been observed in relation to relevant clinical characteristics. A new approach to such “guided therapy” could be to integrate the patients’ genetic information. The angiotensin receptor type 1 (AGTR1) gene has been proposed by many as an important genetic determinant of the efficacy of RAS system modulators, in particular of ACEIs and ARBs. 43,44 Nevertheless, results regarding the clinical impact of genetic polymorphisms of this gene have been inconsistent. 45 The PERGENE study is a large pharmacogenetic substudy of the EUROPA trial, aimed to assess the achievability of pharmacogenetic profiling. In the BK1 receptor gene, rs12050217 was a strong modifier of the treatment benefit of perindopril; additionally, in the AT1 receptor gene, rs275651 and rs5182 significantly modified the treatment benefit of perindopril. 46 Another interesting aspect of this study is that the 3 single-nucleotide polymorphisms associated with the CV benefits of perindopril were not associated with its BP reductions. In the same way, the angiotensinogen (M235T) gene polymorphism is significantly associated with essential hypertension. Patients carrying TT genotype had a greater BP-lowering response when treated with enalapril those carrying MM and MT genotypes. 47 In a nested case–control study, the risk of MI was significantly lower among users of ACEIs compared with that in users of other antihypertensives. In patients using ACEIs, the largest risk reduction was found in patients carrying the ACE-4656-G allele. The risk of MI was reduced in those having the AGTR1-1166AC or AA genotype compared with that in users of ACEIs with the AGTR1-1166CC genotype. None of the polymorphisms modified the effectiveness of ARBs regarding the risk of MI. 48
Angiotensin Receptor Blockers
A large amount of information about the clinical effects of ARBs has been cumulated in recent years. The effectiveness of ARBs to reduce CV has been studied in several RCTs. The Ongoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial (ONTARGET) compared ramipril, telmisartan, and the combination of the 2 drugs in patients with vascular disease or high-risk diabetes. The primary composite outcome was CV mortality, MI, stroke, or hospitalization for HF. Mean BP was lower in both the telmisartan group and the combination therapy group than in the ramipril group. The primary outcome frequency was similar in all the groups, and the telmisartan group had lower rates of side effects. 49 The Telmisartan Randomised AssessmeNt Study in ACE iNtolerant subjects with cardiovascular Disease (TRANSCEND) assessed whether telmisartan would be effective in high-risk patients intolerant to ACEIs. The BP was lower in the telmisartan group compared with placebo, but no differences in the primary outcome or the secondary outcomes were found. Fewer patients receiving telmisartan were hospitalized for a CV reason, and fewer patients permanently discontinued study medication in the telmisartan group. 50 Effects on LVH have also been reported. Prevalence of LVH at entry in TRANSCEND was reduced by telmisartan compared with placebo, and new-onset LVH occurred less frequently with telmisartan compared with placebo. The LVH regression was similar in 2 groups. In ONTARGET, differences between the groups in the primary outcomes were not significant. 51 In 2009, the Food and Drugs Administration (FDA) approved telmisartan as the first treatment in its class to reduce the risk of heart attack, stroke, or death from CV causes in patients at high CV risk who are unable to take ACEIs. 52 In Europe, the European Medicines Agency (EMA) granted further indication of telmisartan, for the reduction in CV morbidity in patients with history of CHD or stroke or peripheral arterial disease; or type 2 diabetes mellitus with documented target organ damage. 53
The Heart Institute of Japan Candesartan Randomized Trial for Evaluation in Coronary Artery Disease (HIJ-CREATE) studied patients with CHD having hypertension who were randomly assigned to receive either candesartan-based or non-ARB-based pharmacotherapy (including ACEIs). No significant differences existed between the groups in terms of MACE or HF. The NOD was diagnosed significantly less frequently with candesartan than with non-ARBs. Incidence of study drug discontinuation due to adverse events was lower with candesartan. 54 In the Candesartan Antihypertensive Survival Evaluation in Japan (CASE-J) trial, comparable efficacy was noted between candesartan and amlodipine in the incidence of CV morbidity and mortality. Candesartan suppressed NOD more effectively than amlodipine. Afterward, an observational study investigated this effect for another 3 years beyond the experimental period of the CASE-J trial. The incidence of CV events was not different between both the groups. 55
Overall, no superior benefits in terms of CV mortality or CV morbidity have been found, but ARBs seem to have a lesser incidence of NOD and better tolerance than other antihypertensive drugs. This benefit has been confirmed by a meta-analysis that pooled the results of 17 studies in order to evaluate drug adherence by class. The pooled mean adherence by drug class ranged from 28% for β-blockers (BBs) to 65% for ARBs. There was better adherence to ARBs compared with ACEI, CCBs, diuretics, and BBs. 56
Several meta-analyses have also addressed the effects of ARBs on CVD, they are shown in Table 3. 25 –34 Taken all together, it seems that ARBs are not inferior to other kinds of antihypertensive drugs in order to prevent MACE. A Cochrane review quantified the dose-related, BP-lowering efficacy of ARBs versus placebo in the treatment of primary hypertension. The evidence from this review suggests that there are no clinically meaningful BP-lowering differences between the available ARBs. The BP-lowering effect of ARBs is similar to ACEIs as a class. 57 Bramlage and Harsford made a systematic review about the BP reduction and compliance with treatment of antihypertensive drugs. They found that ARBs have a competitive BP-lowering efficacy compared with ACEI, CCBs, β-blockers, and diuretics. Patients were more compliant on ARBs than on ACEi and CCBs, BBs, and diuretics. Although the price per tablet of ACEi and ARBs is higher than that of older ones (diuretics and BBs), the newer drugs result in a more favorable cost-to-effect ratio when direct drug costs, and indirect costs were also considered. 58
The FDA has approved an expanded indication for olmesartan medoxomil for the treatment of hypertension in pediatric patients aged 6 to16 years. The approval was based on data from a randomized, double-blind study of pediatric patients. Results of the initial dose–response phase show that olmesartan significantly reduced BP in both a weight-adjusted and a dose-dependent manner. As observed in adult populations, BP reductions were smaller in black patients as compared with the nonblack pediatric patient cohort. Pediatric use of olmesartan was well tolerated; adverse events were similar to those reported in adult studies, with dizziness most commonly reported (incidence, 3%). The recommended starting and maintenance doses for patients weighing from 20 to <35 kg are 10 mg and 20 mg once daily, respectively; patients weighing ≥35 kg should receive an adult starting dose of 20 mg once daily and be maintained at a daily dose of 20 to 40 mg. An extemporaneous suspension may be prepared for children unable to swallow tablets. 59
Diabetic Retinopathy
Three separate RCTSs have tried to assess whether candesartan could reduce the incidence of retinopathy in type 1 diabetes (DIabetic REtinopathy Candesartan Trials [DIRECT]-Prevent 1); progression of retinopathy in type 1 diabetes (DIRECT-Protect 1); and progression of retinopathy in type 2 diabetes (DIRECT-Protect 2). Candesartan had no beneficial effect on retinopathy progression and only a trend to reduced incidence of retinopathy could be shown. 60 Contrariwise, a small multicenter controlled trial involving patients with normotension having type 1 diabetes and normoalbuminuria show that enalapril and losartan did not slow nephropathy progression but slowed the progression of retinopathy. 61 In the DIRECT-Protect 2, the risk of progression of retinopathy was nonsignificantly reduced in patients on candesartan compared with those on placebo, but regression was increased. 62
Heart Failure
Although the prevention of HF remains a major therapeutic challenge, the established association between BP reduction and the prevention of stroke, CAD, and CV death in patients with hypertensive or high-risk patients has not been confirmed for HF. In this regard, Verdecchia et al conducted a meta-analysis of trials comparing ACEIs, ARBs, or CCBs, with diuretics, BBs, or placebo in patients with hypertensive or high-risk patients without HF. In trials versus placebo, the risk of HF was reduced with ACEIs, whereas the effect of ARBs and CCBs was not significant. In trials versus diuretics/BBs, no differences were found between ACEIs and comparators. The risk of HF was significantly lesser with ACEIs/ARBs than CCBs. The ARBs significantly reduced the risk of the composite outcome, stroke, and NOD. 63
Inhibition of the RAS by ACEI or ARBs forms the foundation of the evidence-based therapy for patients with HF and reduced ejection fractions. National HF guidelines vary in their recommendations regarding whether ACEIs or ARBs should be used as the drugs of first choice. 64 A systematic review has evaluated the effect of ARBs in patients with HF on total mortality and HF hospitalizations. The ARBs did not show any beneficial effect on mortality when used in combination with ACEI or when compared with ACEI alone, although a reduction in hospital admissions was observed. 65 A Cochrane review evaluated the benefit and harm of ARBs compared with ACEIs or placebo in patients with symptomatic HF. The ARBs did not reduce total mortality or total hospitalizations compared with placebo. Total mortality or morbidity did not differ between ARBs and ACEIs, but withdrawals due to adverse effects were lower with ARBs. Combinations of ARBs plus ACEIs increased the risk of withdrawals due to adverse effects but did not reduce total mortality or total hospital admissions versus ACEI alone. 66 In a meta-analysis, ARBs did not show any beneficial effect on mortality when used in combination with ACEIs or when compared with ACEIs alone, but a reduction in hospital admissions was observed. 56 So, ARBs do not seem to be inferior to ACEIs in HF treatment.
However, RCTs have often excluded patients with HF with preserved ejection fraction (HFpEF). Meune et al have performed a meta-analysis of the randomized trials of ACEIs or ARBs in patients with HF and HFPEF. Compared with placebo, ACEIs, or ARB, significantly lowered risks of hospitalization for HF and CV mortality for HF at 1 year. However, they have no significant effect on mortality during more prolonged follow-up. 67 Contrariwise, another meta-analysis found no significant difference between pharmacotherapy (including ARBS and ACEIs) and placebo in terms of CV mortality, hospitalization, and worsening HF. 68 This was also the conclusion of another pooled analysis of the 3 major randomized trials of RAS inhibition in HFPEF (CHARM-Preserved, I-PRESERVE, and Perindopril in Elderly People with Chronic HF). 69 Nevertheless, more recently a prospective study from the Swedish HF Registry found that among patients with HF and preserved ejection fraction, the use of RAS antagonists was associated with lower all-cause mortality. 70 Therefore, the only conclusion is that evidence is inconclusive.
New-Onset Diabetes
Numerous analyses have demonstrated that antihypertensive therapies promote the development of type 2 diabetes mellitus. Studies indicate that the application of ACEIs and ARBs lead to lesser NOD incidence compared to BBs, diuretics, and placebo. Given that BBs and diuretics impair glucose metabolism, the metabolic effects of different antihypertensive drugs should be considered. In this regard, although the cost of ACEIs and ARBS are higher, the use in patients with metabolic disorders could be cost effective in the long term if NOD is avoided. 71 Three meta-analyses have tried to determine whether the administration of ACEI or ARBs may reduce the incidence of NOD in placebo-controlled clinical trials. All 3 concluded that both ACEIs and ARBs reduced NOD as compared to placebo. 72 –74 A systematic literature review has also concluded that ARBs as well as ACEIs have a preventive effect regarding the development of NOD. They found that treatment with candesartan could lead to savings in total costs of 549 US dollar per patient and in incremental costs of 30 000 US dollars per diabetes mellitus avoided. 75
Cancer
The fear that drugs chronically administered for the treatment of hypertension and other CVDs might increase the risk of cancer has surfaced several times in the past, particularly in relation to the use of diuretics. 76 It has been reported that the use of ARBs could be associated with an increased risk of cancer in a meta-analysis of some trials comparing this kind of drug with other antihypertensive drug. 77 The methodology of this trial has been criticized, since some RCTs with ARBs were not included. For instance, if the number of patients with cancer from VALUE was added to the equation, any cancer signal with ARB disappeared. 78 Afterward, several other meta-analyses 79 –82 and cohort studies 83 –87 addressed this issue (they are shown in Table 4). The final conclusion is that only the first trial found the association of treatment and ARBs and cancer. Moreover, Mc Menammin et al conducted a systematic review for observational/interventional studies that used clinically relevant outcomes for cancer progression and survival. They concluded that the evidence suggested that ACEIs or ARBs use may be associated with improved outcomes in patients with cancer. 88
The ARBs and Cancer.

Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin II type 1 receptor blocker; CI, confidence interval; HF, heart failure; HR, heart rate; OR, odds ratio; RR, relative risk.
a For ACEIs: 1.12 (0.87-1.47).
b There was some evidence of an increased risk of breast and prostate cancer but there was a decreased risk of lung cancer.
c For ARBs, the use of angiotensin-converting enzyme inhibitors and calcium channel blockers was associated with an increased rate of lung cancer (RR: 1.13; 95% CI: 1.06-1.20 and RR: 1.19; 95 CI: 1.12-1.27, respectively).
d For ACEIS: RR 1.17 (95% CI 1.14-1.20).
e For ACEIs, 0.99 (0.80-1.24).
Chronic Kidney Disease
Prevention of renal dysfunction is one of the most important properties of an antihypertensive treatment. A meta-analysis studied whether RAS inhibitors reduce the incidence of renal dysfunction when compared to other antihypertensive treatments in patients with essential hypertension and no preexistent renal disease. Patients randomized to RAS inhibitors did not show a significant reduction in the risk of developing renal dysfunction as compared to other antihypertensive strategies. 89 This is not surprising when compared with the cumulated evidence. 90
Microalbuminuria is a surrogate marker usually used in RCTs, although its signification in the prognosis of nondiabetic CKD remains to be confirmed. 91,92 In the ONTARGET trial, the renal effects were assessed through changes in renal function and proteinuria. The primary renal outcome was a composite of dialysis, doubling of serum creatinine, and death. The number of events for the composite primary outcome was similar for telmisartan and ramipril but was increased with combination therapy. The GFR declined least with ramipril compared with telmisartan or combination therapy. Conversely, the increase in urinary albumin excretion was less with telmisartan or with combination therapy than with ramipril. 93 In the TRANSCEND, no important difference was found in the composite renal outcome with telmisartan versus placebo but a larger sample sizes serum creatinine in the telmisartan group. Albuminuria increased less with telmisartan than with placebo. Decreases in estimated GFR were greater with telmisartan. 94 A post hoc analysis of ONTARGET trial and the TRANSCEND one shows that in ONTARGET there was no CV or renal benefit from dual over monotherapy in any subgroup. Moreover, in TRANSCEND, in the comparison of ARB with placebo, there was a significant interaction between the main renal outcome in the direction of harm for patients with normoalbuminuria (hazard ratio, 2.35) and no benefit in patients with microalbuminuria. 95 In this regard, a meta-analysis assessing ARBs treatment in patients with normotension and proteinuria found a significant reduction in urinary protein excretion. Subgroups analysis shows that ARB therapy resulted in a significant decrease in urinary protein excretion in diabetic patients with microalbuminuria or nondiabetic nephropathy with overt proteinuria. 96
Microalbuminuria is an early predictor of diabetic nephropathy and premature CVD. The Randomized Olmesartan and Diabetes Microalbuminuria Prevention (ROADMAP) study show that olmesartan delays the onset of microalbuminuria in patients with type 2 diabetes. Surprisingly, an unexpected higher rate of fatal CV events was found with olmesartan among patients with preexisting CHD. 97,98
A meta-analysis has evaluated the effect of ACEIs and ARBs on CV mortality in patients with diabetic nephropathy. Use of ARBs, but not ACEIs, was associated with a significant reduction in ESRD risk. Both the drug classes were associated with reduction in the risk of doubling serum creatinine, but neither affected all-cause mortality. 99 Another meta-analysis aimed to compare the renal outcomes between ACEIs/ARBs and other antihypertensive drugs or placebo in type 2 diabetics. The ACEIs/ARBs had significantly lower risk of serum creatinine doubling, macroalbuminuria, and albuminuria regression than other antihypertensive drugs, mainly CCBs. The risks of ESRD and microalbuminuria were not lower in the ACEIs/ARBs group. The ACEIs/ARBs benefit over placebo was significant for all outcomes except microalbuminuria. The lack of any differences in BP decrease between ACEI/ARB and active comparators suggest this benefit is not simply due to the antihypertensive effect. 100
Renal dysfunction and microalbuminuria are major independent risk factors for CVD in diabetic and nondiabetic patients. 101 A substudy of the CASE-J trial examined the effects of candesartan and amlodipine on CV events in patients with hypertension having CKD. The CV event rate did not differ between the 2 groups. In the subgroup analysis based on the CKD stage, there were no significant differences in the incidence rates of CV events between the 2 groups in stages 1 + 2 and 3 CKD. In stage 4 CKD, however, candesartan reduced the incidence of CV events, particularly of renal events (doubling serum creatinine and ESRD), compared with amlodipine. 102 Two meta-analyses have tried to examine the CV benefits and risks of ARBs in people with albuminuria or CKD. One of them found a significant reduction in the risk of nonfatal CV events with ACEIs versus placebo but not with ARB versus placebo, ACEIs versus ARBs or with combined therapy versus monotherapy. Development of ESRD and progression of microalbuminuria to macroalbuminuria were reduced significantly with ACEIs and ARBs but not with combined therapy versus monotherapy. 103 A second meta-analysis reported that the risk of CV outcomes was decreased with RAS blockade in nondiabetic nephropathy, diabetic nephropathy, and proteinuric patients with CKD when compared with control therapy or placebo. 104
Atrial Fibrillation
There is compelling evidence that the RAS is involved in the pathogenesis of atrial fibrillation (AF). Currently, it is recognized that there is a sustained reduction in new-onset AF in patients with significant underlying heart disease (eg, left ventricular dysfunction and hypertrophy) treated with ACEIs or ARBs, but evidence is less convincing in patients with moderate structural heart disease and recurrent AF. 105 –107 No benefits have been recently reported in 2 large prospective studies with ARBs in patients with mild structural disease, and this clearly raises the issue that these therapies may not offer the same benefit in all patients. 108,109 For instance, a meta-analysis found that in primary prevention, RAS inhibition was effective in patients with HF and those with hypertension and LVH but not in post-MI patients overall. 110 Table 5 shows the result of reported meta-analyses. 111 –113,117,118 The biggest meta-analysis suggests a protective effect, but it was not found in the smaller ones. This could suggest a publication bias that may result in an overestimation of the treatment effect, since bigger samples make easier to find significant results. Therefore, the question remains open. It has also been reported a nested case–control analysis using data from the United Kingdom-based General Practice Research Database; the authors found that long-term therapy with ACEIs, ARBs, or BBs was associated with a lower risk of atrial fibrillation as compared with CCBs therapy. 114
The ARBs/ACEIs for Atrial Fibrillation Prevention.
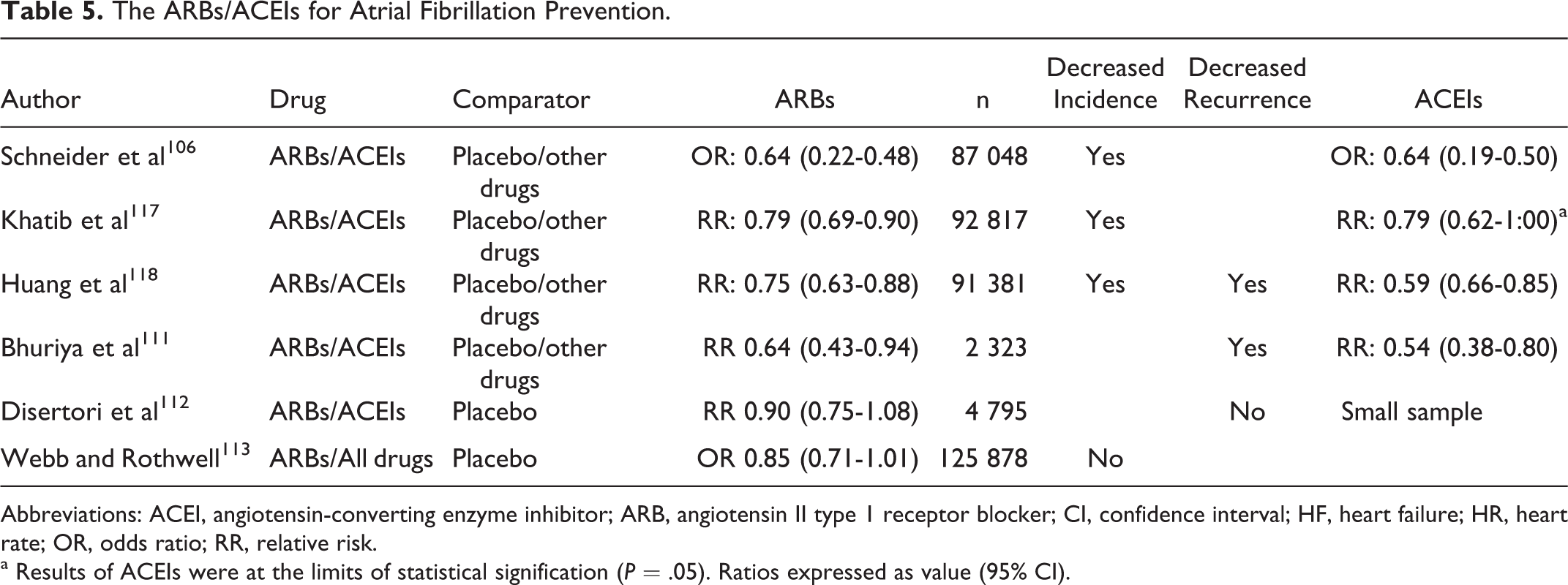
Abbreviations: ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin II type 1 receptor blocker; CI, confidence interval; HF, heart failure; HR, heart rate; OR, odds ratio; RR, relative risk.
a Results of ACEIs were at the limits of statistical signification (
Pharmacogenetics
As with ACEIs, genetics may influence the effect of ARBs. For instance, a lower BP reduction response was observed to conventional doses of valsartan in African American patients than non-African American patients, but at 640 mg, a paradoxical higher response was observed in African American patients than in non-African American patients. 115 In this way, patients with the ATR1 A1166C AC genotype had no significant reduction in proteinuria when treated with valsartan, and no effect was found in GFR changes. 116 In the RENAAL study, proteinuric type 2 diabetic patients with the D allele of the ACE gene treated with placebo group were more likely to reach the composite end point, but within the losartan group, the genotype did not correlate with reaching the composite end point. 119 After treatment with irbesartan in Chinese patients with hypertension, patients with genotype (−344 TC) in the CYP11B2 and those ones with the 174MM genotype of the angiotensinogen gene had a significantly greater systolic (but not diastolic) BP reduction. 120 Polymorphisms in the chromosome 11q21 region have also been associated with response to candesartan. 121
Stroke
Some, but not all, other studies with ARBs suggested that these agents especially reduce stroke incidence. For instance, in the Losartan Intervention For End point reduction (LIFE) study, losartan-based therapy reduced stroke better than BB-based therapy. 122 In contrast, in the VALUE study, valsartan was not better than amlodipine in stroke prevention, although this may have been attributable to less effective BP control. 123 The MOrbidity and mortality after Stroke, Eprosartan compared with nitrendipine for Secondary prevention (MOSES ) study compared eprosartan or nitrendipine in patients with a history of proven stroke or transient ischemic attack. There was a significant reduction in the all-cause mortality and the number of CV and cerebrovascular events, including all recurrent events and in the recurrence of stroke, as well as in first-time CV events. 124 Although there has been some concern on the reliability and relevance of the study, 125 it was the first trial directly aimed to assess the effects of ARBs on stroke compared with a CCB. In this way, a meta-analysis on the effects of ARBs on the risk of stroke found that ARBs were associated with a significant reduction in the risk of stroke versus placebo, but they were differences with ACEIs and calcium antagonists. 126 In the Acute Candesartan Cilexetil Therapy in Stroke Survivors (ACCESS) trial, candesartan (or placebo) was given earlier 7 days after onset of stroke. Neurological outcome after 1 year in the ARB-treated group was better than in the placebo group. 127 The mechanism of this possible beneficial effect may lie in the selective blocking by ARBs of the deleterious effects mediated through the AT1 receptor, whereas the effects on the AT2 receptor are unaffected or even enhanced. 128 The Scandinavian Candesartan Acute Stroke Trial (SCAST) was designed to confirm the findings of ACCESS. During the 7-day treatment period, BPs were significantly lower in patients allocated candesartan than in those on placebo, but the risk of the composite vascular end point did not differ between the treatment groups. Moreover, no differences in stroke incidence were found. 129 A substudy of the factorial Prevention Regimen for Effectively Avoiding Second Strokes (PRoFESS) trial addressed this issue. Combined death or dependency and stroke recurrence did not differ between the treatment groups although in comparison with placebo telmisartan lowered BP. 130 Another predefined substudy using MRI examinations failed to demonstrate that telmisartan could prevent the progression of white-matter lesions. 131 This evidence may be summarized as contradictory. Therefore, it can be concluded that ARBs reduce the risk of stroke when compared with placebo, but they are not better than other antihypertensive drugs. No special effect has been demonstrated using ARBs in acute stroke in spite of the results of the ACCESS trial.
Dementia
An important issue of antihypertensive treatment is the theoretical ability to prevent or delay cognitive impairment. This was investigated in the ONTARGET and the TRANSCEND trials as secondary outcomes, but the pooled data from these studies did not show that the different approaches to blocking of the RAS had different effects on cognitive outcomes. 132 In the same way, in the PRoFESS trial, disability due to recurrent stroke and cognitive decline is not affected by the preventive use of telmisartan. 133 The Observational Study on Cognitive function And Systolic Blood Pressure Reduction (OSCAR) evaluated the impact of eprosartan-based therapy on cognitive function. Use of eprosartan, either as monotherapy or in combination regimens, was associated with a substantial reduction in BP. Treatment with eprosartan gets a small, although significant, beneficial effect on cognitive function. Since this was not a randomized comparative trial, it cannot be concluded a special effect of ARBs on cognitive impairment, since the benefit can be explained by the BP reduction. 134,135 In this regard, 3 large cohort studies have investigated whether ARBs can protect against Alzheimer disease and dementia or reduce the progression of both the diseases. The first is a prospective cohort analysis using the Setting Administrative database of the US Veteran Affairs. The ARBs were associated with a significant reduction in the incidence and progression of Alzheimer disease and dementia compared with ACEIs or other CV drugs. 136 The second is a nationwide, population-based cohort database in Taiwan. No significant difference in the dementia occurrence rate between patients exposed to ARBs and nonexposed controls was found. 137 The third is a retrospective study using a large national cohort of beneficiaries of the Department of Veterans Affairs who have diabetes. It examined incidence of dementia over a 2-year follow-up period. Antihypertensive medications decreased risk, ranging from 24% for ARBs to 4% for BB. In a stratified analysis of patients without hypertension, ACEIs (HR = 0.81) and ARBs (HR = 0.55) continued to show protective effects. 138 As conclusion, there is no enough evidence coming from RCTs to support the protective effect of ARBs against dementia. Larger cohort studies suggested this protective ability, but this issue is still controversial.
Untoward Effects
Between 5% and 20% of patients treated with ACEIs develop intolerance; in this regard, angioedema is a rare, potentially life-threatening adverse event of ACEIs, but it had been occasionally described with ARBs. A meta-analysis found that with ACEIs and ARBs, incidence of angioedema was higher in HF trials compared to hypertension or CHD trials without HF. Weighted incidence of angioedema with ACEIs was 0.30% compared to 0.11% with ARBs, 0.13% with DRIs, and 0.07% with placebo. Therefore, incidence of angioedema with ARBs and DRIs was significantly different from placebo. 139 In this regard, it has been reported a retrospective cohort study when compared with BBs, ACEIs or aliskiren was associated with an approximately 3-fold higher risk of angioedema, although the number of exposed events for aliskiren was small. The risk of angioedema was lower with ARBs than with ACEIs or aliskiren. 140
The ARBs are routinely used as an alternative treatment for patients who develop intolerance with ACEIs. A recent meta-analysis has evaluated the tolerability of ARBs in patients with intolerance to ACEIs; ARBs had fewer cough events versus ACEIs and drug discontinuation, and cough risk rates were similar to placebo. Angioedema risk with ARBs was also similar to placebo. Although compared with placebo, hypotension, renal dysfunction, and hyperkalemia were more frequent with ARBs, this kind of drug seems to be a safe alternative in ACEI-intolerant patients. 141 A second meta-analysis has addressed this issue, the risk of angioedema after switching treatment was 9.4% for possible cases and 3.5% for confirmed cases. Most important, no fatal events were reported. 142 Thus, the risk of recurrent angioedema after switching the treatment from ACEIs to ARBs seems to be low.
A systematic review of published case reports and case series dealing with intrauterine exposure to ACEIs or to ARBs has been reported. Overall, 52% of the newborns exposed to ACEIs and 13% of the newborns exposed to ARBs did not exhibit any complication. Neonatal complications were more frequent following exposure to ARBs and included renal failure, oligohydramnios, death, arterial hypotension, intrauterine growth retardation, respiratory distress syndrome, pulmonary hypoplasia, hypocalvaria, limb defects, persistent patent ductus arteriosus, or cerebral complications. The long-term outcome without complications occurred in only 50% of the exposed children. 143 A prospective, observational, controlled cohort study in the first trimester of pregnancy found no differences in rates of major malformations, although both the ACE-ARBs and disease-matched groups exhibited significantly lower birth weight and gestational ages than the healthy controls. There was a significantly higher rate of miscarriage noted in the ACE/ARB group. 144
High Doses
Treatment for hypertension has traditionally been commenced with low doses, with subsequent up-titration as needed. This pattern was primarily due to fear of side effects associated with older therapies, especially adrenergic blockers that were associated with dose-related side effects such as postural hypotension, drowsiness, and dry mouth. In comparison with these other classes of drugs, side effects associated with ARBs can hardly be differentiated from placebo. Thus, the presumption that increasing doses of drug automatically means more side effects, which have limited starting doses with the older classes of antihypertensives, does not appear to be a clinically relevant factor with ARBs. 145 So, increasing the doses of ARBs above the maximal dosage for its antihypertensive effect could be a way to get intensive blockade of RAS. In fact, when alternate doses of ARBs or ACEIs in patients with type 2 diabetes and microalbuminuria were compared, the percentage decline in albumin excretion rate from baseline was greater with higher doses, and the regression to normoalbuminuria was greater, with less progression to macroalbuminuria. Adverse events were not significantly different. 146 The Heart failure Endpoint evaluation of Angiotensin II Antagonist Losartan (HEAAL) study compared clinical outcomes in patients with HF, reduced LVEF, and intolerance to ACEIs who were randomly assigned to a high (150 mg) or low (50 mg) daily dose of losartan. Losartan 150 mg daily reduced the rate of death or admission for HF in patients with HF-reduced left-ventricular ejection fraction and intolerance to ACEIs compared with losartan 50 mg daily. These findings show the value of up-titrating ARB doses to confer clinical benefit. 147 Finally, a meta-analysis found that in type 2 diabetes with microalbuminuria, higher doses of ARBs were associated with significantly reduced albumin excretion rate and increased regression to normoalbuminuria as compared with lower dose treatment. Adverse events were more frequent but not significantly so. 148
What About Dual Blockade?
Dual blockade of RAS has been tested for CV morbidity prevention, after MI, in the treatment of renal disease associated with micro- or macroalbuminuria and in congestive HF. Dual blockade does not seem to more intensively reduce BP. 149 The results of ONTARGET have been previously commented. The superiority of dual blockade of RAS for the treatment of hypertension in patients with high CV risk could not be proved. Moreover, these results prompted an urgent reappraisal of the European Guidelines on Hypertension Management due to the finding of a clear J curve effect on CV morbidity and mortality. 150 The ONTARGET results cast some shadows on the use of dual blockade RAS in CHD, since near one-half of the sample had MI and one-quarter more had other manifestations of CAD. These results resemble closely those of the VALIANT study, in which the combination of a full dose of captopril plus valsartan (the latter at a lower dose than that used in the valsartan-only group) did not significantly reduce the occurrence of the primary outcome but did increase hypotension. 151 Taken together, the final results of these 2 studies show no additive effect of ARBs in conjunction with a full dose of proven ACEIs to improve the prognosis of CAD. Contrariwise, a substantial increase in the undesirable secondary effects was detected.
Several authors have reported a superior effect of the combination of ACE inhibition and ARB on microalbuminuria/macroalbuminuria in patients with primary nephropathies 152 –156 and in diabetic patients. 157,158 Two meta-analyses have addressed this issue suggesting that concomitant therapy with an ARB and an ACEI leads to greater reductions in proteinuria than monotherapy without excessive side effects. 159,160 An RCT has examined the effects of olmesartan on primary composite outcome of doubling of serum creatinine, ESRD, and death in type 2 diabetic patients with overt nephropathy. Olmesartan significantly decreased BP, proteinuria, and rate of change in reciprocal serum creatinine. Unfortunately, CV was higher in the olmesartan group than the placebo group, whereas MACE and all-cause death were similar between the 2 groups. Hyperkalemia was more frequent in the olmesartan group. 161 Most recently, Luño et al reported the results of a multicenter, open-label randomized controlled trial to compare the efficacy of combining the lisinopril and the irbesartan with that of each drug in monotherapy in slowing the progression of type 2 diabetic nephropathy. There were no significant differences in proteinuria reduction or BP control between the groups. The number of adverse events, including hyperkalemia, was similar in all the 3 groups. They were unable to show a benefit of the combination of lisinopril and irbesartan compared to either agent alone on the risk of progression of type 2 diabetic nephropathy. 162 Therefore, in this moment, we can only assure that dual blockade of RAS using an ACEI and an ARB may get a further decrease in proteinuria, but there is no evidence in the long run that this combination will better retard CKD progression.
The main concern of dual blockade is the risk of adverse events. A recent meta-analysis found that patients receiving combination therapy had an increased risk of developing any adverse effect, hypotension, worsening renal function, and hyperkalemia; but there was no difference in angioedema or cough. 163 Other reports suggested that in proteinuric renal disease, dual blockade of the RAS could be safely used in most patients even in the stage III or KDOQI Guidelines, although it should be closely monitored. 164 In the Valsartan in HF trial (Val-HeFT), valsartan (added on ACEI treatment) reduced the estimated glomerular filtration rate by the same amount in patients with and without CKD and reduced the risk of the first morbid event in patients with CKD. 165 A part of the problem is that combination therapy is frequently prescribed for patients without established indications, and this is associated with an increased risk of adverse renal outcomes. 166
Dual blockade of RAS has also been tested in the treatment of HF. The combination of ACEIs and ARBs may produce more reduction in left ventricle size than either agent alone. 167 In the CHARM-Added trial, the use of candesartan resulted in a reduction in CV death or HF hospitalization. 168 In a meta-analysis, dual blockade of RAS was not associated with any significant benefit for all-cause mortality and CV mortality compared with monotherapy, but it was associated with a reduction in admissions to hospital for HF. However, dual therapy was associated with a sharp increase in the risk of adverse events. 169 A second meta-analysis found no difference between patients treated with combination therapy and ACEI therapy alone but fewer hospital admissions for HF. Patients treated with combination therapy had a higher risk of untoward effects. 170 Thus, based on current evidence, combination therapy with ARBs and ACEIs may be reserved for patients with HF who remain symptomatic on therapy with ACEIs under strict monitoring for any signs of worsening renal function and/or symptomatic hypotension.
Direct Renin Inhibitors
Harel et al have performed a meta-analysis of RCTs using DRI; combination therapy with aliskiren and ACEIs or ARBs significantly increased the risk of hyperkalemia compared with monotherapy. The risk of acute kidney injury did not differ significantly between the combined therapy and the monotherapy groups. 171 Contrariwise, another meta-analysis concluded that the safety and tolerability profile of aliskiren in combination with the ARBs valsartan or losartan, or diuretic, is similar to aliskiren, ARBs, or diuretics alone. 172,173 They agreed with a study where treatment with aliskiren plus ARB in adult patients with hypertension was associated with significantly better compliance/persistence and fewer hospitalizations and medical visits compared with ACEI plus ARB therapy. 174
In humans, the target organ-protective effects of aliskiren are being investigated in ASPIRE HIGHER, the largest clinical trials program underway in the cardiorenal disease area, involving 14 randomized, double-blind studies including more than 35 000 patients with hypertension, diabetes, HF, renal dysfunction, and previous MI. 175 Due to ethical issues, most of the clinical trials included in this program were developed using dual blockade of the RAS instead of using aliskiren alone. The Aliskiren Observation of Heart Failure Treatment (ALOFT) trial recruited patients with hypertension who had stable HF and were randomized to treatment with aliskiren or placebo in addition to prior treatment with an ACEI/ARB and BB. Aliskiren reduced plasma BNP, plasma N-terminal proBNP, and urinary aldosterone. An improvement in echocardiographic measurement of left ventricular filling pressure was also observed. There was no significant difference between aliskiren and placebo for the safety assessment of renal dysfunction, symptomatic hypotension, and hyperkalemia. 176 The aliskiren and valsartan to reduce NT-proBNP via renin–angiotensin–aldosterone system blockade (AVANT-GARDE) study investigated whether aliskiren, alone or in combination with valsartan, could improve ventricular hemodynamics as assessed by reductions in NT-proBNP levels. There were no differences in clinical outcomes, but there were more adverse events in patients treated with active therapy. 177
Aliskiren in Left Ventricular Hypertrophy (ALLAY) compared the effects of aliskiren and losartan alone and in combination with LVH assessed by magnetic resonance imaging. Aliskiren monotherapy was statistically noninferior to losartan in reducing left ventricular mass index. The aliskiren–losartan did not achieve a significant combination reduction in left ventricular mass index compared with losartan monotherapy. 178 The ASPIRE evaluated the effects of adding aliskiren to an ACEI or an ARB, on left ventricular volume and function after MI. No differences were observed. In addition, the combined rates of CV deaths, hospitalization for HF, recurrent heart attack, resuscitated sudden death, and stroke were similar. In contrast, patients receiving aliskiren had a higher rate of adverse events. 179 The results of ASPIRE are consistent with those of the ONTARGET and VALIANT trials, dual RAS blockade did not produce any significant clinical advantage in this high-risk population but significantly increased the risk of adverse effects.
The first study to assess the renoprotective effects of aliskiren was Aliskiren in the Evaluation of Proteinuria in Diabetes (AVOID). Adding aliskiren to losartan provided a further significant 20% reduction in urinary albumin–creatinine ratio compared with the addition of placebo. This effect appeared to be independent of BP reduction. 180 The primary objective of the Aliskiren Trial in Type 2 Diabetes Using Cardio-Renal Endpoints (ALTITUDE) trial was to determine whether aliskiren reduces CV and renal morbidity and mortality compared with placebo when added to conventional treatment (including ACEI or ARBs). 181 Unfortunately, on December 20, 2011, Novartis announced that following the interim review of data from the ALTITUDE study, a decision to terminate the trial has been taken on the recommendation of the independent Data Monitoring Committee overseeing the trial. The committee concluded that patients were unlikely to benefit from treatment and identified higher adverse events in patients receiving aliskiren in addition to standard of care in the trial. Specifically, in the trial arm in which aliskiren was added to the standard of care, there was an increased incidence of nonfatal stroke, renal complications, hyperkalemia, and hypotension. 182 After the interruption of ALTITUDE, the EMA banned the use of dual blockade with aliskiren in diabetic patients and patients with moderate-to-severe renal failure. 183 Afterward, the FDA contraindicated this use in diabetic patients and warning to avoid use of aliskiren with ARBs or ACEIs in patients with moderate-to-severe renal impairment (ie, GFR < 60 mL/min). 184 These results contrast with a recent report about the combination therapy in diabetics in the ONTARGET trial; dual therapy did not increase stroke or altered other major CV or renal events in patients with diabetes, irrespective of the presence of nephropathy. 185
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
