Abstract
Although pharmaceutical grade heparin is obtained almost exclusively from porcine intestinal mucosa, there is interest in diversifying heparin sourcing to address potential supply shortages and economically motivated adulteration. Since ovine-derived heparin is structurally similar to porcine heparin, it is expected that ovine-derived low-molecular-weight heparin (LMWH) will be comparable to porcine-derived LMWH. This study compared the pharmacokinetic (PK) behavior of 3 batches of ovine LMWH with that of enoxaparin in nonhuman primates. Blood samples were collected prior to and at 2, 4, and 6 hours post-administration of a 1 mg/kg subcutaneous dose of LMWH. Circulating drug concentrations determined using anti-Xa and anti-thrombin assays were used to calculate values for PK parameters. Tissue factor pathway inhibitor (TFPI) levels were measured by enzyme-linked immunosorbent assay. The ovine LMWHs tested met pharmacopoeial potency and molecular weight distribution requirements for enoxaparin. In the post-administration samples, comparable levels of branded enoxaparin and ovine enoxaparin were observed using anti-Xa and anti-thrombin assays, with the concentration versus time curves being nearly superimposable. Consistent with this similarity, no significant differences were observed between PK parameters calculated for branded enoxaparin and ovine LMWH. The TFPI levels returned to baseline levels by 6 hours in ovine LMWH-treated animals but remained slightly elevated in animals treated with branded enoxaparin. It is concluded that the pharmacokinetics of ovine enoxaparin were not only comparable between different batches but also similar to the branded product. Thus, LMWH prepared from ovine mucosal heparin is comparable to its porcine-derived counterpart.
Background
Heparin products have been used for more than 75 years for the prevention and treatment of venous and arterial thrombosis. 1 Low-molecular-weight forms of heparin offer several pharmacologic advantages over unfractionated heparin including a reduced level of polydispersity, increased subcutaneous bioavailability, and more predictable drug levels. 2 Several low-molecular-weight heparins (LMWHs) are marketed in the United States, including Lovenox (enoxaparin sodium), Fragmin (dalteparin sodium), and Innohep (tinzaparin sodium). Each of these products is derived from porcine mucosal heparin.
Direct chemical measurement of heparin levels in plasma is difficult because of the large number of heparin saccharide chains and a lack of sensitive assays. The time course of heparin or LMWH action in circulation is typically described in terms of its anticoagulant activity. For this purpose, anti-Xa and anti-IIa levels are most commonly used, although studies have been carried out using activated partial thromboplastin time (aPTT), activated clotting time (ACT), and other point-of-care assays. 3
Approximately 80% of the world supply of porcine heparin originates from China. Following the heparin contamination crisis in 2007 to 2008, 4 there is renewed interest in diversifying the source of heparin. 5 Heparins derived from different tissues and/or species are known to have subtle differences in chemical structure as related to their degree of sulfation and acetylation. Additionally, isolation conditions can impact the structural and functional characteristics of the final product. The extent to which such structural differences translate to functional differences is not completely clear. Heparin isolated from sheep intestine (ovine) has been shown to have a high degree of structural and functional similarity to porcine-derived heparins. 6,7
It was, therefore, hypothesized that low-molecular-weight forms of ovine heparin would also be similar to their porcine-derived counterparts. This study compares the pharmacokinetic (PK) behavior of porcine- and ovine-derived LMWHs following subcutaneous administration to nonhuman primates.
Materials and Methods
Test Agents
Enoxaparin sodium (Lovenox) was from Sanofi-Aventis, Bridgewater, New Jersey (Lot #7L010A). Three lots of ovine LMWH (OES L170401D, OES L170402D, OES L170403D) were obtained from Suzhou Ronnsi Pharma, Suzhou, China. The ovine LMWHs were obtained as white powder that were stored at room temperature in a desiccator. These samples were weighed on a Mettler (Columbus, Ohio) balance, and 0.9% NaCl2 was used as a diluent to make stock solutions of 10 mg/mL for the in vivo and in vitro testing.
Primates
Rhesus monkeys (
Dosing
Primates were anesthetized by the intramuscular administration of ketamine (10 mg/kg) and xylazine (1-2 mg/kg) based on their most recent charted weight. Following attainment of the appropriate depth of anesthesia (assessed by a lack of response to foot pinch), primates were freshly weighed to accurately determine the dose of the test agent. The procedure room was maintained at an ambient temperature of 78°F to minimize the chance that primates would become hypothermic while under anesthesia. A baseline blood sample was collected by venipuncture of the saphenous vein. An area in the abdominal region was shaved and cleansed with a combination of betadine and alcohol. The test agent was administered at a dose of 1 mg/kg subcutaneously. Additional blood samples were collected at 2, 4, and 6 hours post-drug administration. Four primates were dosed with each LMWH.
Sample Collection
All blood samples were collected using a double syringe technique, employing a 21-gauge butterfly needle. After an initial ∼1 mL volume (discard blood) was collected, the syringe was changed and a 2.7 mL sample was drawn and placed into a tube containing 0.3 mL 3.2% sodium citrate. Citrated blood samples were centrifuged at 1100 ×
Sample Analysis
Anti-Xa and anti-IIa activities were determined using in-house amidolytic assays 9 employing human thrombin or bovine factor Xa (Enzyme Research Laboratories, South Bend, Indiana) and Spectrozyme TH or Spectrozyme Xa (Biomedica Diagnostics, Windsor, Nova Scotia, Canada). Assays were performed on an ACL Elite (Werfen, Bedford, Massachusetts). The aPTT was determined using TriniCLOT reagents (Tcoag, Wicklow, Ireland). The TFPI levels were determined using the Asserachrom Total TFPI Enzyme-Linked Immunosorbent Assay (ELISA; Stago, Parsippany, New Jersey).
In vitro concentration–response curves were made by supplementing the various LMWHs into pooled plasma. Plasma concentrations of various LMWHs were plotted against corresponding optical densities in the anti-Xa or anti-IIa assays using graphing software, SigmaPlot Version 12.0 for Windows (Systat Software, San Jose, California). Best-fit curves were made, and the drug concentration in each primate blood sample was determined by extrapolation.
Molecular weight determinations were made using gel permeation chromatography in a high-performance liquid chromatography (HPLC) system as reported previously. 10 Briefly, the system was equilibrated using freshly degassed mobile phase (0.3 M sodium sulfate) until a stable baseline was obtained. Analysis was carried out by injecting 20 μL of sample (10 mg/mL in 0.3 M sodium sulfate) into the HPLC system. The flow rate for the mobile phase was 0.5 mL/min, and the run time for each sample was 65 minutes. The internal temperature for the refractive index (RI) detector was set at 35°C, and UV determination was made at 205 nm. All analyses were made at room temperature. The elution profile of each sample was analyzed in relation to a calibration curve prepared using 13 heparin fractions ranging in molecular weight from 3.0 to 40 kDa. The molecular weight profile consists of parameters such as weight average molecular weight, number average molecular weight, peak molecular weight, and polydispersity.
Pharmacokinetic/Pharmacodynamic Calculations
The extrapolated plasma concentrations were used to calculate values for PK parameters such as elimination half-life (
Statistical Analysis
Statistical analysis was performed using SigmaPlot Version 12.0 for Windows. Circulating drug levels were compared using 2-way repeated measures analysis of variance. Differences with
Results
The elution profiles of the 3 lots of ovine LMWH were observed to be nearly superimposable; however, the elution profile of Lovenox appeared to have a slightly larger tail at the early elution times (higher molecular weight; Figure 1A). The values for number average molecular weight and weight average molecular weight were slightly higher for Lovenox (3.82 and 4.49 kDa, respectively) compared to ovine LMWH (3.72 and 4.24 kDa, respectively; Figure 1B).
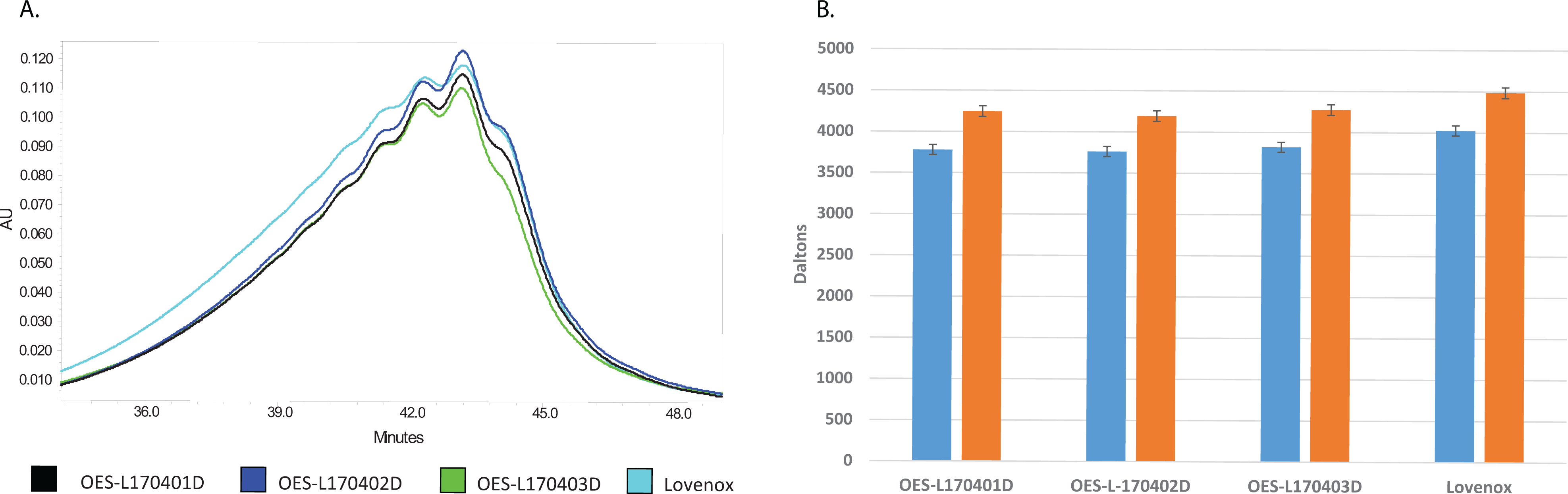
Molecular weight profiling of Lovenox and ovine low-molecular-weight heparin (LMWH). Molecular weights were determined by size exclusion chromatography (SEC)-high-performance liquid chromatography (HPLC) in relation to 13 narrow range heparin calibrators. A, Elution profile determined by UV detection. B, Number average molecular weight (blue) and weight average molecular weight (orange).
The anticoagulant and antiprotease activities of Lovenox and ovine LMWH were compared following in vitro supplementation to plasma. Comparable activities were observed in terms of the inhibition of factor Xa and thrombin as well as for aPTT prolongation (Figure 2).
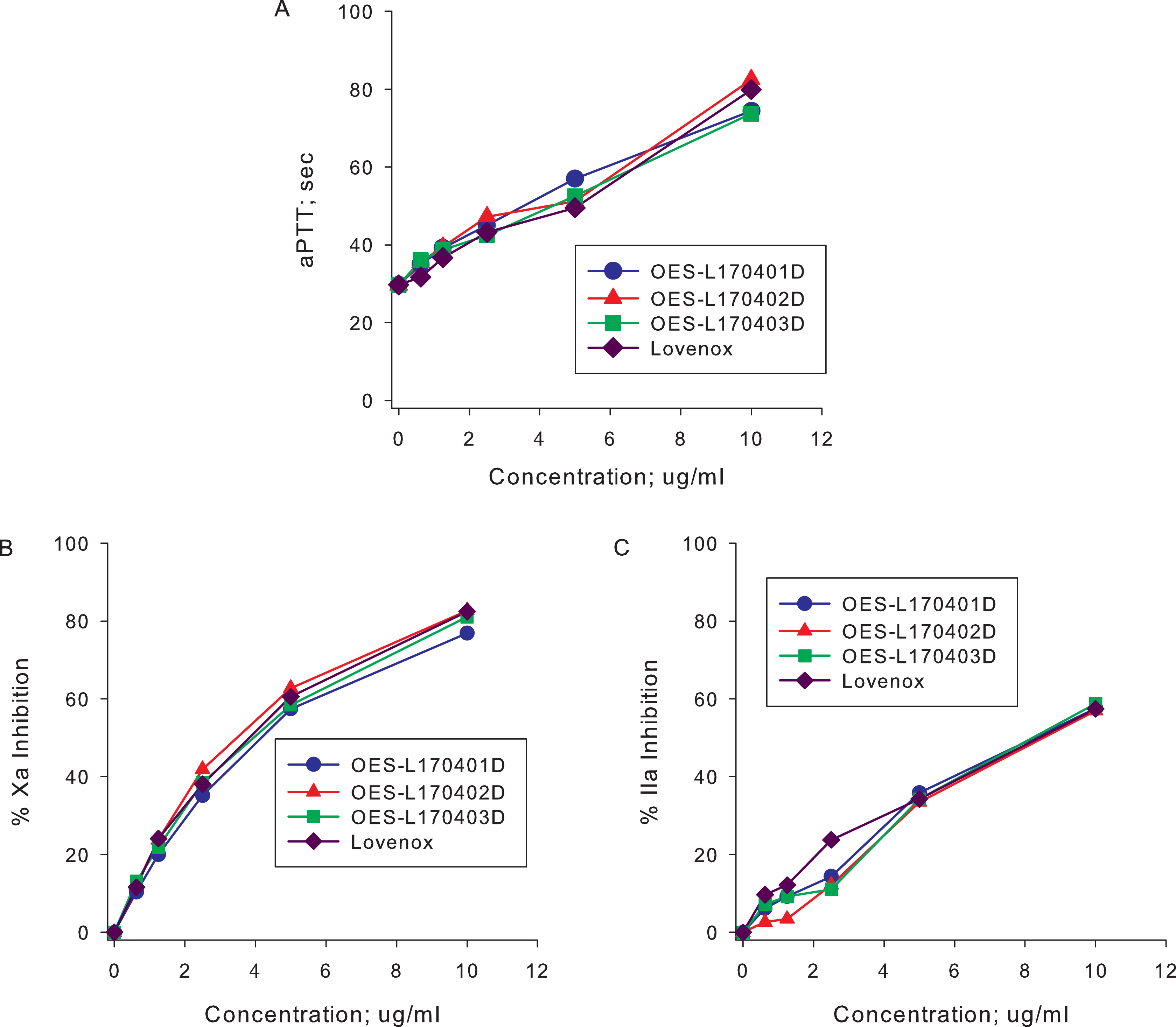
In vitro activity of Lovenox and ovine low-molecular-weight heparin (LMWH). (A) Anticoagulant activity was determined by measuring the activated partial thromboplastin time (aPTT). (B) Anti-Xa and (C) anti-IIa activities were measured using amidolytic assays. Lovenox and ovine LMWH produced comparable anticoagulant and antiprotease activities in all of these assays.
Primates were administered Lovenox or ovine LMWH at a 1 mg/kg subcutaneous dose. Blood samples were subsequently drawn at 2, 4, and 6 hours post-administration. Circulating drug levels were determined using amidolytic anti-Xa and anti-IIa assays (Figure 3). By anti-Xa assay, peak drug levels of approximately 10 µg/mL were observed following administration of each LMWH. Only minor differences were observed between batches of ovine LMWH. By anti-IIa assay, peak drug levels of approximately 4 µg/mL were observed following administration of each LMWH, with only minor differences observed between batches of ovine LMWH.
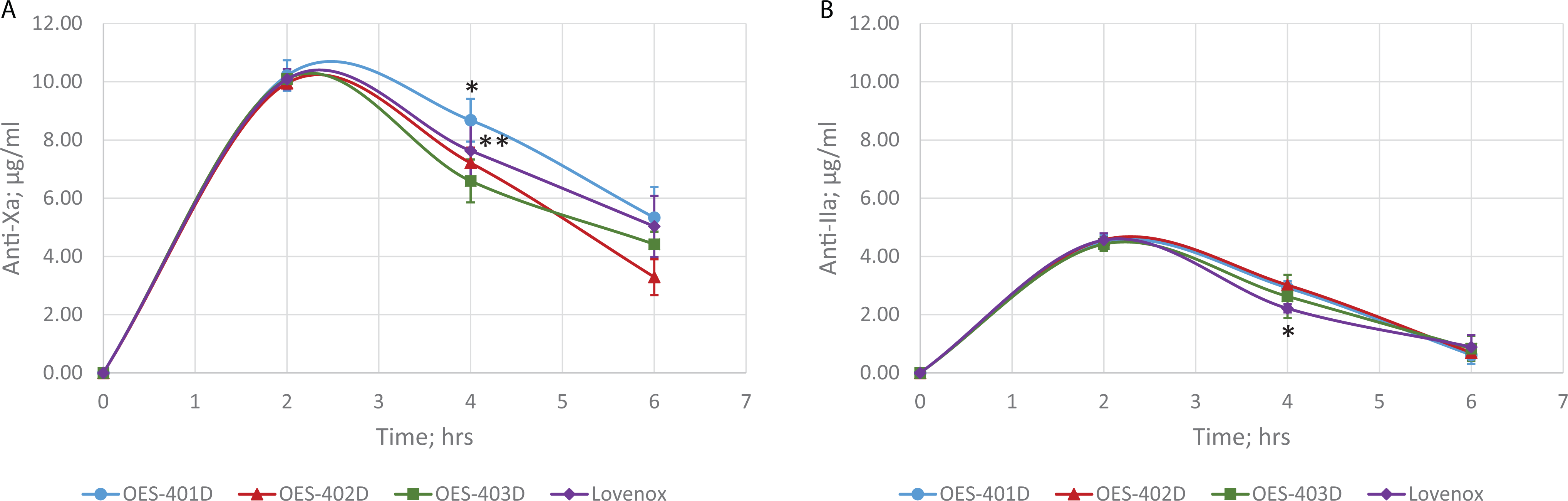
Circulating drug levels in primates treated with Lovenox or ovine low-molecular-weight heparin (LMWH). Circulating drug levels were determined using amidolytic anti-Xa (A) and anti-IIa (B) assays relative to batch-specific calibration curves. Good batch-to-batch consistency was observed between drug levels obtained with different batches of ovine enoxaparin. Drug levels in ovine LMWH-treated animals were comparable to those observed following Lovenox administration.
The TFPI levels in all plasma samples were measured by ELISA (Figure 4). Some variability in TFPI release was observed between lots of ovine LMWH. As a whole, peak TFPI levels at 2 hours were the same following administration of ovine LMWH and Lovenox. Differences were observed, however, at later time points, where TFPI levels were observed to remain elevated for a longer time following Lovenox treatment [AUC0-6 hours Lovenox = 104.75 (ng × h)/mL vs AUC0-6 hours ovine LMWH = 87.50 (ng × h)/mL].
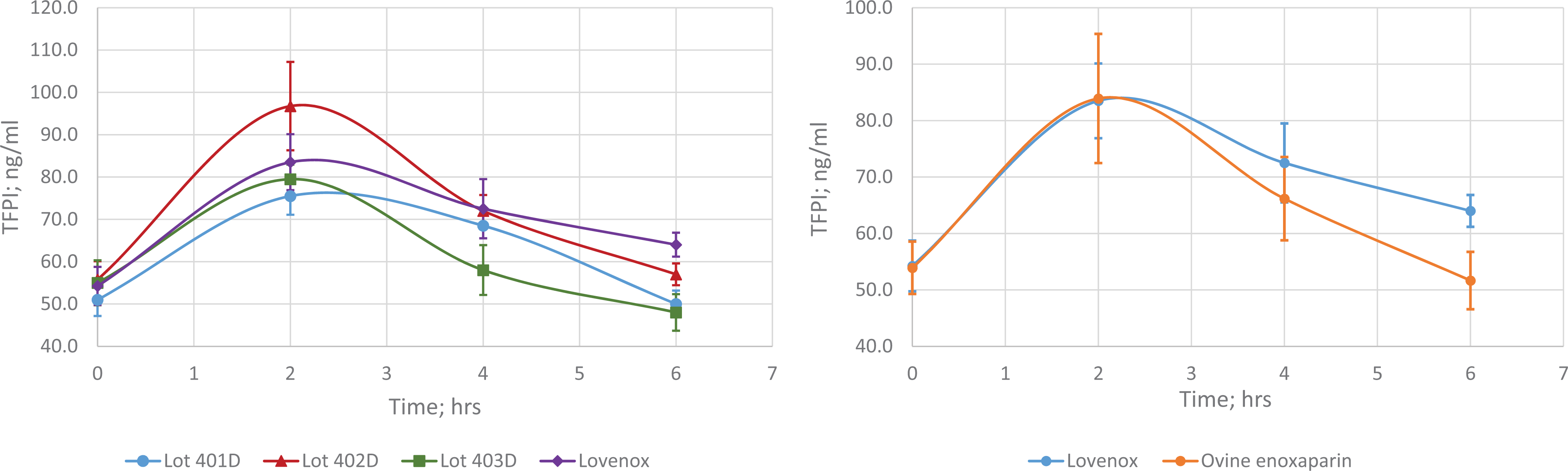
Plasma tissue factor pathway inhibitor (TFPI) levels in primates treated with Lovenox or ovine low-molecular-weight heparin (LMWH). Circulating TFPI levels were determined by enzyme-linked immunosorbent assay (ELISA). *
Values for PK parameters were derived by applying a noncompartmental model to circulating drug levels determined by anti-Xa and anti-IIa assays (Figure 5). The PK profiles of ovine LMWH and Lovenox were observed to be comparable, with no statistically significant differences observed.
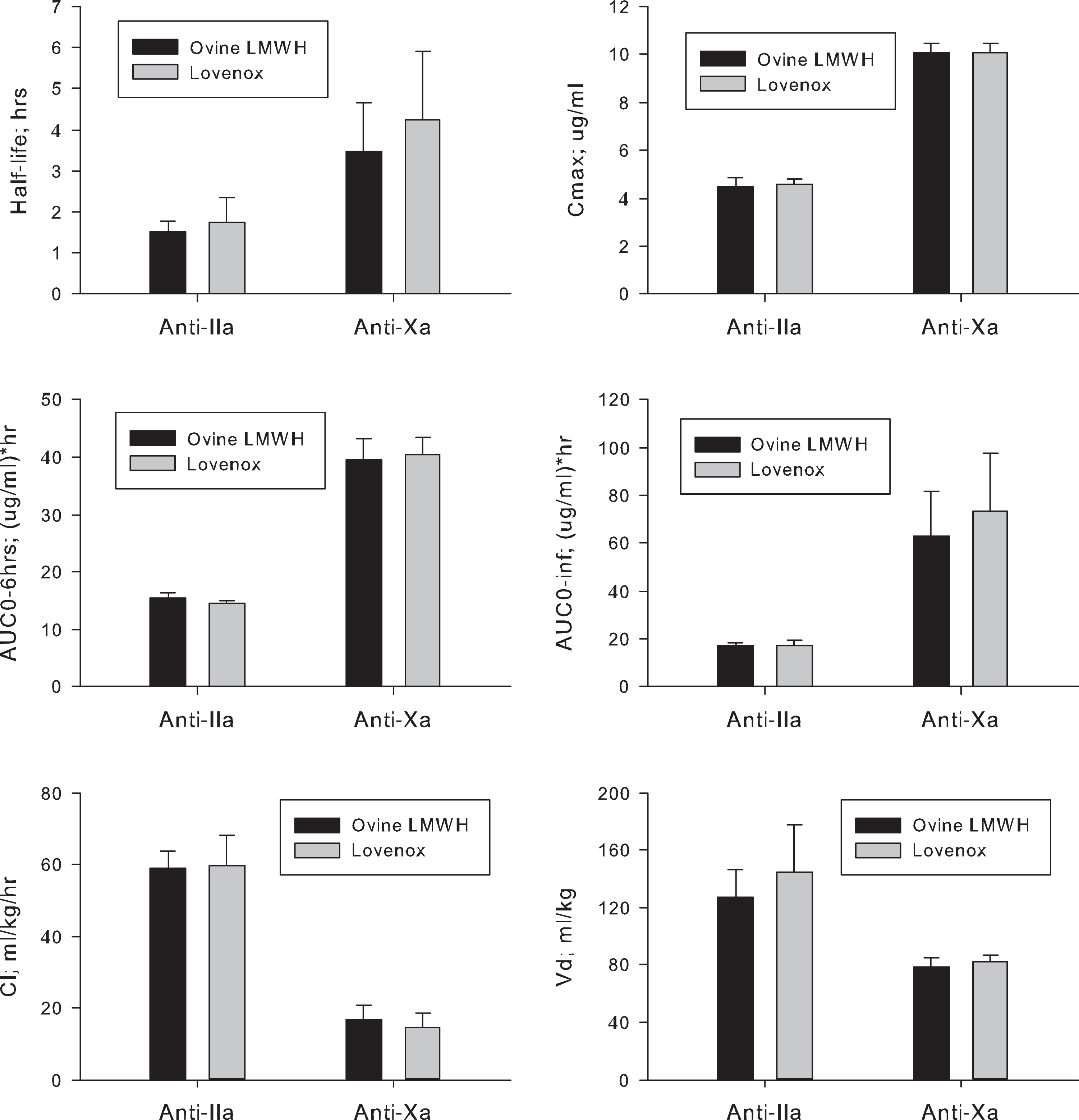
Pharmacokinetic profile of subcutaneously administered ovine low-molecular-weight heparin (LMWH) and Lovenox. Values for pharmacokinetic parameters were calculated using circulating drug levels determined by anti-IIa and anti-Xa assays.
Discussion
Despite the introduction of small-molecule direct oral anticoagulants, the usage of LMWHs continues to increase. Currently, commercially available LMWHs are made exclusively from porcine mucosal heparin. Owing to a possible shortage of porcine heparin starting material, it is of interest to evaluate the feasibility of using other common heparin sources to produce LMWHs. Heparins can be obtained in commercial quantities from bovine lung, bovine intestine, and ovine intestine. Bovine unfractionated heparins have been shown to differ from porcine heparin in terms of molecular weight distribution, the degree of sulfation, and the biologic potency. 12 Unfractionated ovine heparin more closely matches porcine heparin in terms of both structure and activity. 6,7
There have been several previous studies to evaluate LMWHs derived from non-porcine heparins using either β-elimination/alkaline hydrolysis or nitrous acid-based depolymerization processes. In one study, LMWHs derived from bovine lung and bovine intestinal heparin were produced by β-elimination reaction and subjected to compositional and potency analysis. 13 Despite the fact that the starting bovine heparins were different than porcine heparin, depolymerized versions of bovine lung and intestinal heparins met the specifications for Lovenox in terms of molecular weight and potency. In terms of their disaccharide composition, the parent heparins and daughter LMWHs were highly correlated. Another comparison of LMWHs produced by benzylation/alkaline hydrolysis of porcine mucosal heparin and bovine lung heparin demonstrated that while such products are comparable in terms of molecular weight and 1,6-anhydo content, bovine-derived LMWHs have lower degrees of N-acetylation and a lower content of antithrombin (AT)-binding sites leading to an anti-Xa potency that was approximately 70% that of porcine-derived LMWHs. 14 In another study, LMWHs were produced from ovine, bovine, and porcine heparins by nitrous acid and hydrogen peroxide–induced depolymerization. 15 Here, 13C nuclear magnetic resosnance (NMR) spectra and disaccharide analyses indicated that LMW ovine heparin shows a higher degree of similarity to porcine- and bovine-derived LMWHs than the parent heparins. The LMWHs were produced from porcine and ovine intestinal heparin and bovine lung heparin by nitrous acid depolymerization and compared in terms of their composition and functional activity. 16 Overall, current literature suggests that bioequivalent LMWHs can be generated when appropriate modifications to the production process are employed.
In addition to a demonstration of structural comparability and bioequivalent potencies, Food and Drug Administration guidance for the development of biosimilar LMWHs also requires demonstration of bioequivalence in terms of pharmacodynamic profile. 17 The current study compares the pharmacodynamic profile of LMWH derived from ovine heparin with that of Lovenox following subcutaneous administration to primates. Primates have previously been shown to respond similarly to humans to the administration of heparin-like anticoagulants.
The current study assessed plasma drug levels in terms of anti-Xa and anti-IIa activities. By both assays, the time course of circulating LMWH levels was comparable for Lovenox and the ovine LMWHs, and the ratio of anti-Xa to anti-IIa activities as assessed by area under the curve were as expected for LMWHs. Heparin administration results in an increase in the circulating level of TFPI. In the current study, comparable peak levels of TFPI were observed following administration of Lovenox and ovine LMWH; however, the increase in TFPI levels was more prolonged at later time points in Lovenox-treated primates. This may result from subtle differences in chemical composition of the tested LMWHs as properties such as charge density and chain length have previously been shown to impact circulating TFPI levels. 18
The studies described here demonstrate that LMWH derived from ovine heparin and Lovenox exhibit comparable anticoagulant, antiprotease, and PK profiles and suggest that these LMWHs will exhibit similar PK profiles when administered to humans.
Footnotes
Acknowledgments
The authors are grateful to Dr Atul Laddu, CEO of the Georgia Thrombosis Forum, for facilitating research fellowships for Mr Rangnekar and Mr Niverthi.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yiming Yao is an employee of Ronssi Pharma.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Yiming Yao is an employee at Ronssi Pharma, the company that produced the ovine LMWHs that were used in this study.
