Abstract
We aimed to detect alterations and deficits in hemostasis during cardiac surgery with cardiopulmonary bypass (CPB) using point-of-care-supported coagulation analysis (rotational thromboelastometry, impedance aggregometry), in addition to single factor assays for the measurement of fibrinogen (FI) and factor XIII (FXIII) levels. Forty-one patients scheduled for elective cardiac surgery with CPB were enrolled in this observational study. Perioperative measurement (pre-, postheparin, 30-minutes before the end of bypass, 1-hourpostoperatively) of standard laboratory variables, additional rotational thromboelastometry (ROTEM; International GmbH, Munich, Germany), Multiplate analysis (Roche, Switzerland), and an assay of FXIII activity were performed as well as the collection of epidemiological data and blood loss. The FI and FXIII levels as well as the measured ROTEM and Multiplate parameters correlated weakly with the blood loss. Clotting time and maximum clot firmness (MCF) of the intrinsically activated ROTEM showed a good correlation (rCT-INTEM = 0.378; P < .05, rMCF-INTEM = 0.305; P < .05) with postoperative drainage loss, suggesting a dependence of blood loss on the initial intrinsic activity. Additionally, perioperative FI or FIBTEM levels and the FXIII levels correlated with each other. Intrinsically activated ROTEM showed a good correlation with postoperative drainage loss, thus suggesting a dependence of blood loss on the initial intrinsic activity and therefore facilitating clinicians to assess postoperative bleeding complications. Based on the FI level or the MCFFIBTEM measured by ROTEM, it may also be possible to assess the FXIII concentration. Especially in chronically ill and massive bleeding cardiac surgery patients with significantly decreased FXIII levels, the knowledge of FXIII deficiency may help clinicians to treat coagulation disorders more adequately.
Introduction
Bleeding disorders after extracorporal circulation are still a significant problem. 1 High blood loss and consecutive administration of blood products represent an independent risk factor for perioperative mortality in cardiac surgery patients. 2
The body’s confrontation with an exogenous surface, mechanical destruction, hemodilution, and inflammatory processes lead to an activation of coagulation, fibrinolysis, platelet dysfunction, and hemolysis. 3 Randomized trials have proven that point-of-care (POC)-based hemotherapy results in less blood loss and a lower transfusion rate than treatments based on the results of conventional laboratory analyses. 4 –10
Especially coagulation factors with long half-lives, which are needed for clot formation and stabilization, such as fibrinogen (FI, 3-4 days) and factor XIII (FXIII, 12 days), reach critically low plasma concentrations at an early stage, thus having an important influence on bleeding. 11 Therefore, particularly, the quick measurement of FI by FIBTEM has proven beneficial. When it comes to evaluating bleeding, apart from FI, we will also have to pay attention to FXIII, as it is the factor, which completes the cross-linking of fibrin and protects the clot from fibrinolysis. The focus on FXIII levels during cardiac surgery is due to the fact that extracorporeal circulation reduces the FXIII plasma level which, in turn, adversely affects the postoperative blood loss. 12,13 Global testing methods, however, do not measure FXIII. Laboratory detection of FXIII is expensive and takes up to several hours to be measured, so it is not suitable for dynamic therapy control. Additionally, attempts to adequately estimate FXIII levels by means of thromboelastometry remained so far unsuccessful. 11,14 As FI and FXIII are activated on the same activation pathway and are similarly compromised by cardiopulmonary bypass (CPB), the possibility of a diagnostic connection will have to be investigated.
With respect to the potential methods and apart from analyzing the plasmatic coagulation, there is the option of measuring the proportion of corpuscular constituents with impedance aggregometry (Multiplate; Roche). By using this method, we can, firstly, evaluate the effect of cyclooxygenase inhibitors, ADP-receptor antagonists and glycoprotein IIa inhibitors, which are often encountered in the medication history of cardiac patients, and, secondly, we can assess CPB-induced effects on platelet function. As of now, the intake of antiplatelet drugs seems to negatively affect bleeding after cardiac surgery; it would be furthermore of interest for the differential diagnosis of bleedings to first check whether the patient’s platelet function is sufficient. 15
The scope of the present observational study was to analyze the perioperative hemostasis of patients undergoing elective cardiac surgery with CPB. We gathered details of the intrinsic or extrinsic clotting system in addition to clot strength and platelet function. Furthermore, we therefore also consider hyperfibrinolysis and both FI and FXIII as individual parameters. A further objective of the present study was to identify patients at risk of postoperative blood loss. Moreover, we aimed at examining the correlation between FI and FXIII, especially with respect to postoperative blood loss. To complement our set of diagnostic methods, rotational thromboelastometry (ROTEM; International GmbH) and impedance aggregometry were used as POC methods in addition to the traditional laboratory methods.
Methods
Patients and Methods
This prospective observational study included 41 adult patients undergoing elective heart surgery with the use of CPB/extracorporeal circulation. The research protocol was approved by the local ethics committee of the University of Halle (approval number: HAL 213). All patients gave written informed consent to participate.
Procedure
Perioperative care in the study group did not differ from the standard procedure in cardiothoracic surgery at the University Hospital of Halle, including the application of CPB with extracorporeal circulation, mild hypothermia (33°C-35°C) during cardioplegia, and intraoperative administration of tranexamic acid.
Blood samples from all patients were obtained through an indwelling artery catheter immediately before the induction of anaesthesia (measuring point/MP 1), 5 minutes after the administration of heparin according to activated clotting time (ACT; measuring point/MP 2), 30 minutes before the end of bypass (measuring point/MP 3), and 1 hour after the end of surgery (measuring point/MP 4). In addition to standard laboratory analyses International Normalized Ratio (INR), partial thromboplastin time, and FI measurement as well as EDTA blood samples for platelet analysis and hemoglobin/hematocrit (HCT) level determination), we furthermore conducted additional measurements for all patients using ROTEM (citrate blood for APTEM, EXTEM, INTEM, and FIBTEM), Multiplate (hirudin blood for ASP, ADP, and TRAP assay), and FXIII determination (citrate blood; Berichrom factor XIII test kit; Siemens Healthcare Diagnostics GmbH, Eschborn, Germany). Thromboelastometry is a viscoelastometric method for hemostasis testing in whole blood. A whole blood sample is placed into a cuvette, and a cylindrical pin is immersed. Between the pin and cuvette remains a gap, bridged by the blood. The pin is rotated by a spring to the right and the left. As long as the blood is liquid, the movement is unrestricted. When blood starts clotting, the clot increasingly restricts the rotation of the pin with rising clot firmness. This kinetic is detected mechanically and calculated by an integrated computer to the typical curves and numerical parameters. With a set of dedicated assays, hypo- and hyperfunctional stages of the clotting process, hemostasis disorders, hyperfibrinolysis, the extent of dilutional coagulopathy, requirement for FI, or platelet substitution can be examined.
At the second and third time of measurement (ie, MP2 and MP3) taking place during the heparinization of the patient, we refrained from measuring INTEM and PTT. The perioperative hemotherapy followed the guidelines of the German Medical Association, 16 and the coagulation therapy was conducted by the anaesthesia team in compliance with conventional treatment guidelines.
After the end of surgery, the mechanically ventilated patients were moved to the intensive care unit (ICU). Finally, after the operation, the thoracic drainage loss was measured every 2 hours.
Fibrinogen and FXIII
Fibrinogen was determined by 2 different measuring methods (laboratory or MCF-FIBTEM) at corresponding measuring points to standard laboratory, that is, at MP 1-4. The measured levels of FI and FXIII showed a significant and moderate to strong positive correlation (β = .497-.766; P < .00; MP: 1-4; Table 4). Similar to the conventional FI testing, FXIII also showed dependence to the measurement of FI by FIBTEM (Table 4). Regression analysis revealed an estimation of the FXIII concentration based on the FI or the MCF-FIBTEM (regression equation, scatter dot plots [Figures 1 and 2]). In every case, the correlation coefficient resulting from the MCF-FIBTEM measuring method was slightly lower compared to the function of laboratory FI activity measurement.
Demographic and Clinical Variables of Patients.
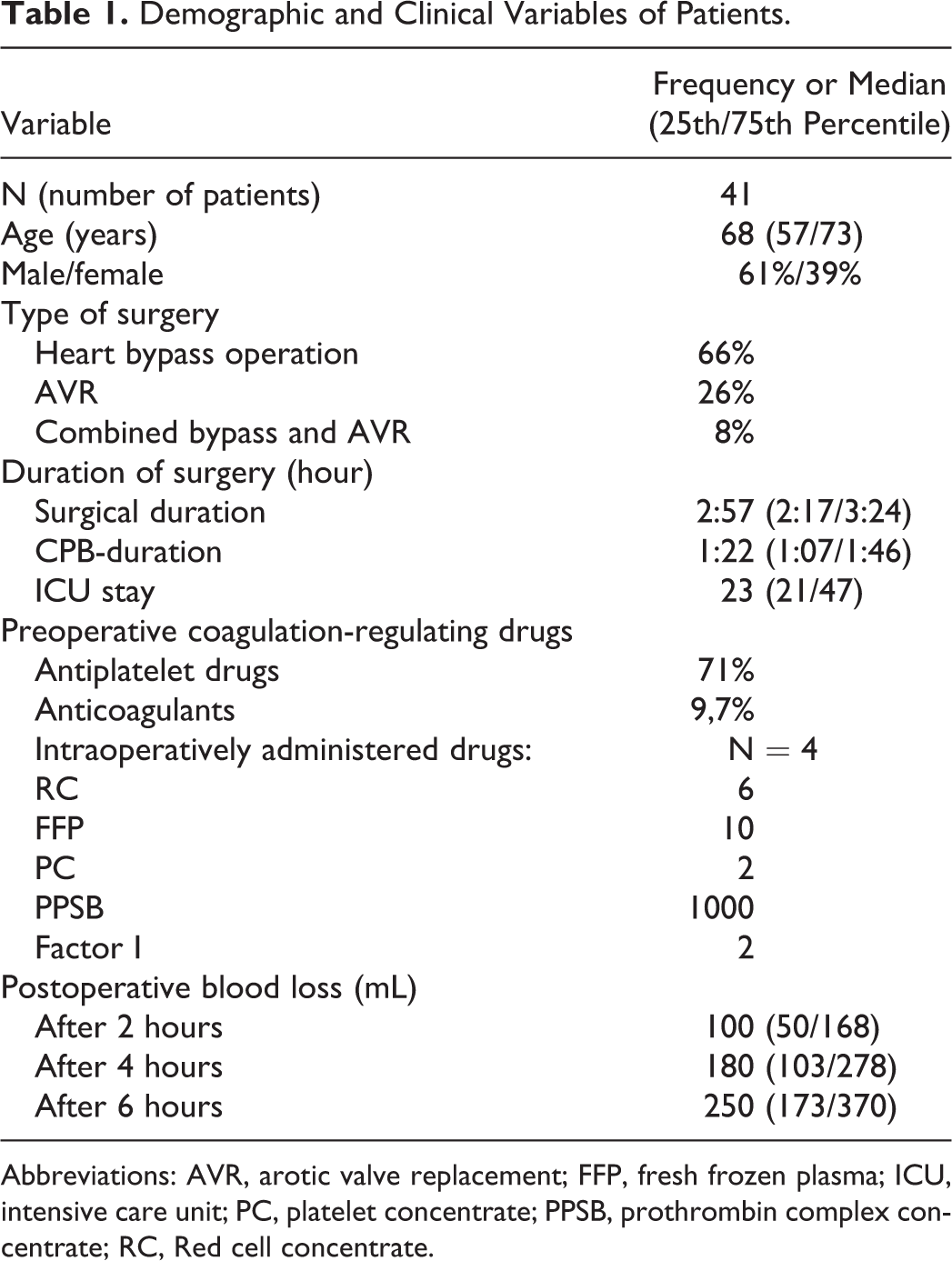
Abbreviations: AVR, arotic valve replacement; FFP, fresh frozen plasma; ICU, intensive care unit; PC, platelet concentrate; PPSB, prothrombin complex concentrate; RC, Red cell concentrate.
Preoperative Coagulation-Regulating Drugs.

Abbreviation: ASS, acetylsalicylic acid.
Comparison of point-of-care (POC) Values Recorded at Measuring Point 1-4 (MP1-4) in Patients with (Bleeder) and without Blood Loss (Nonbleeder) Above and Below 90th Percentile.
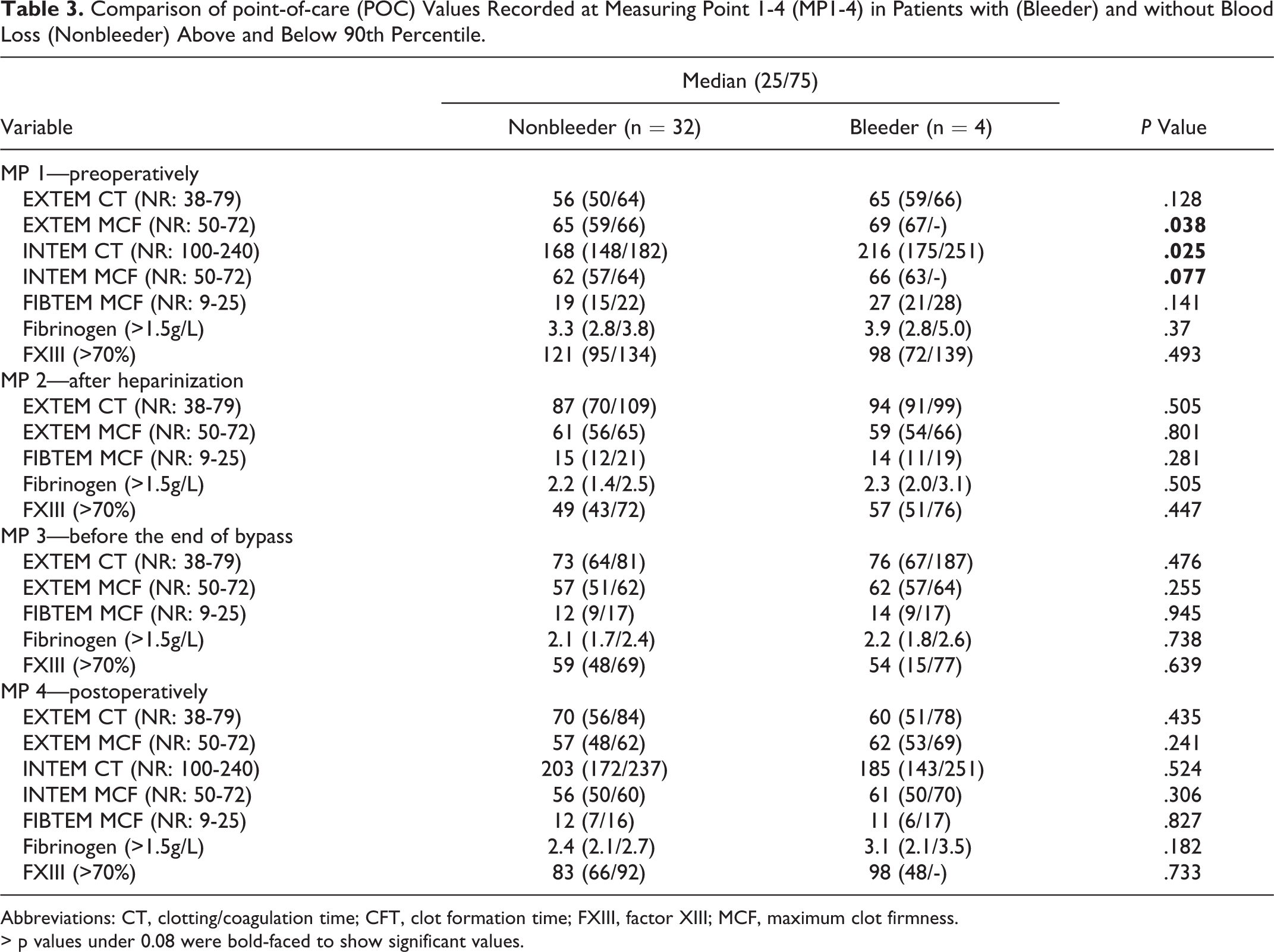
Abbreviations: CT, clotting/coagulation time; CFT, clot formation time; FXIII, factor XIII; MCF, maximum clot firmness.
> p values under 0.08 were bold-faced to show significant values.
Correlation of Laboratory and MCF FIBTEM Fibrinogen With factor XIII levels.
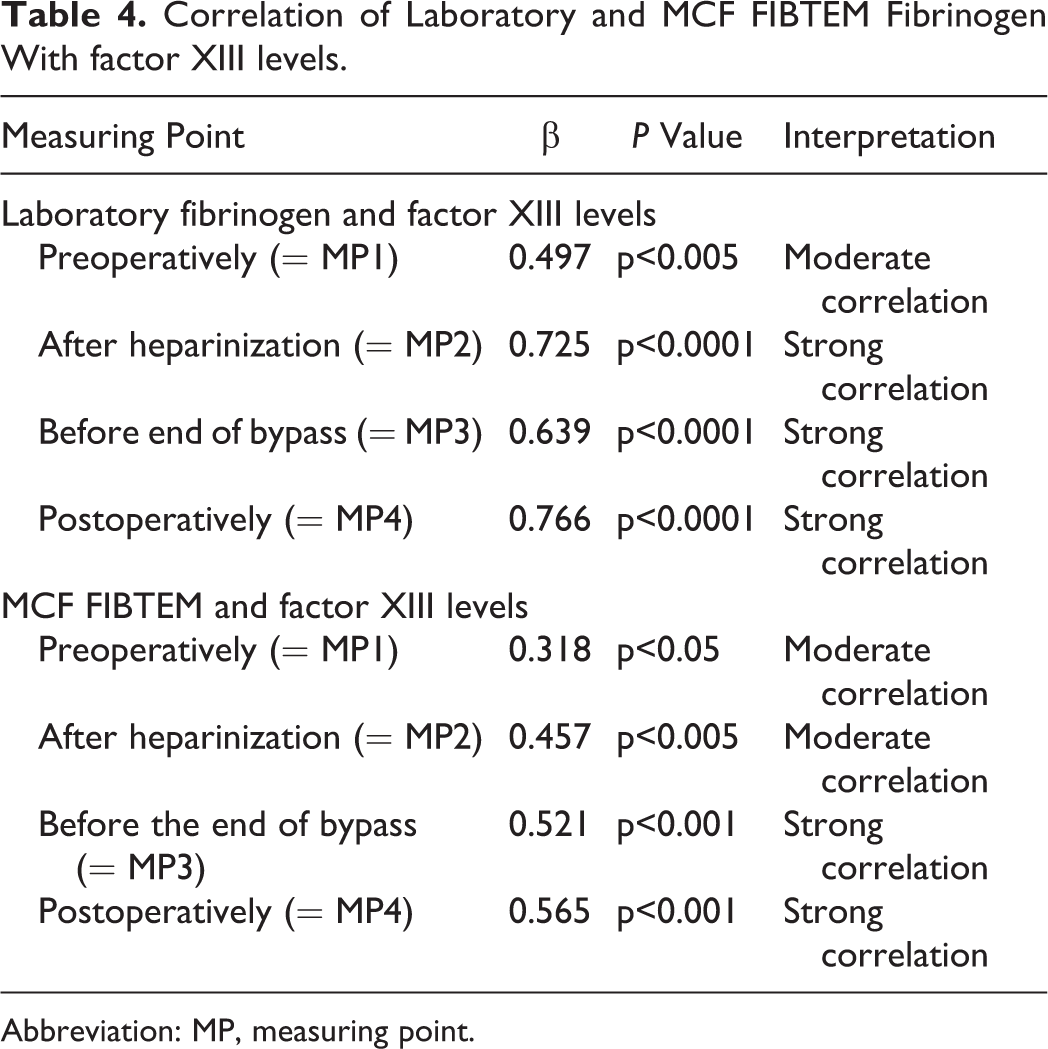
Abbreviation: MP, measuring point.
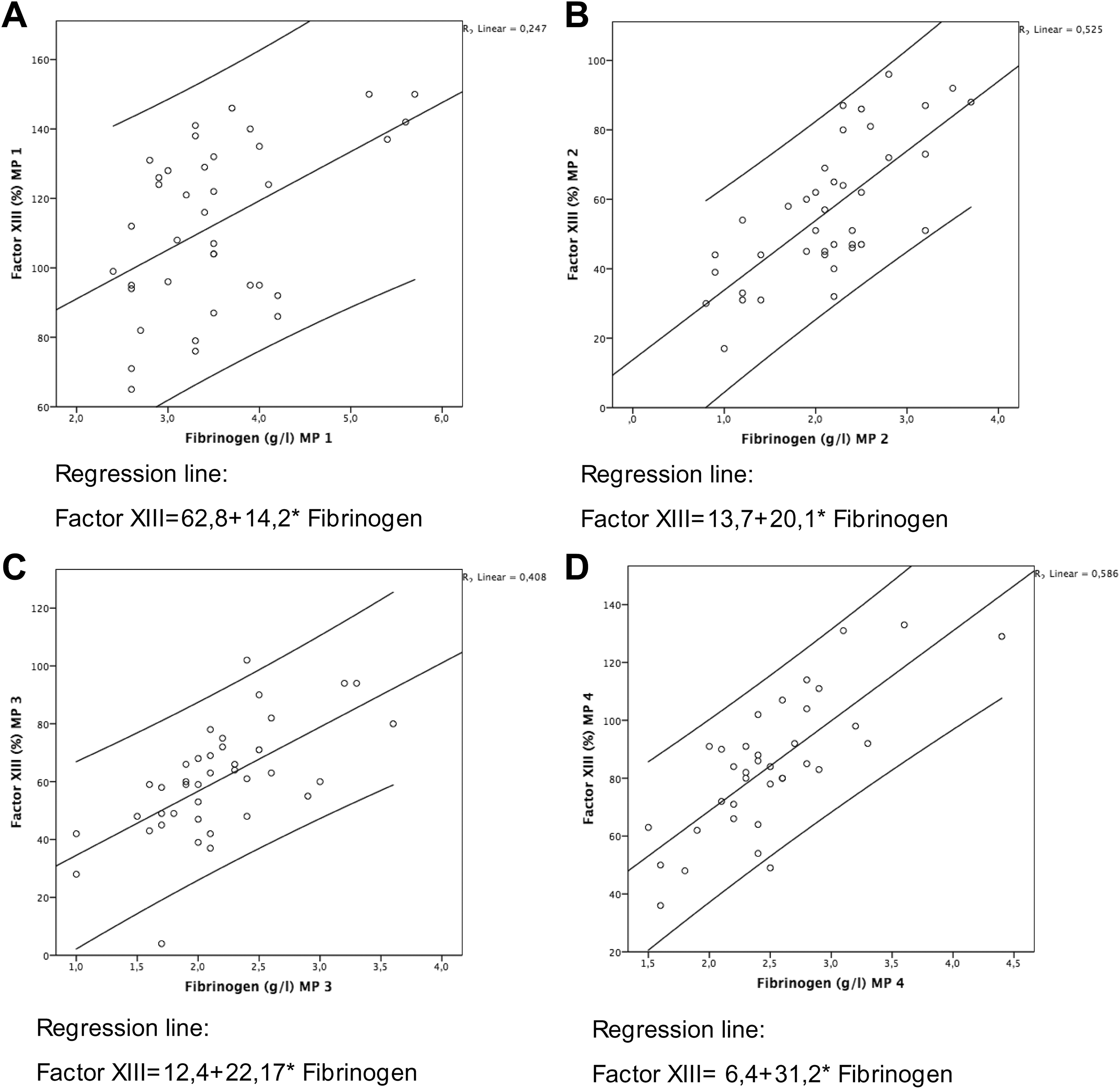
A-D, Linear regression analysis of plasma fibrinogen and factor XIII (FXIII).
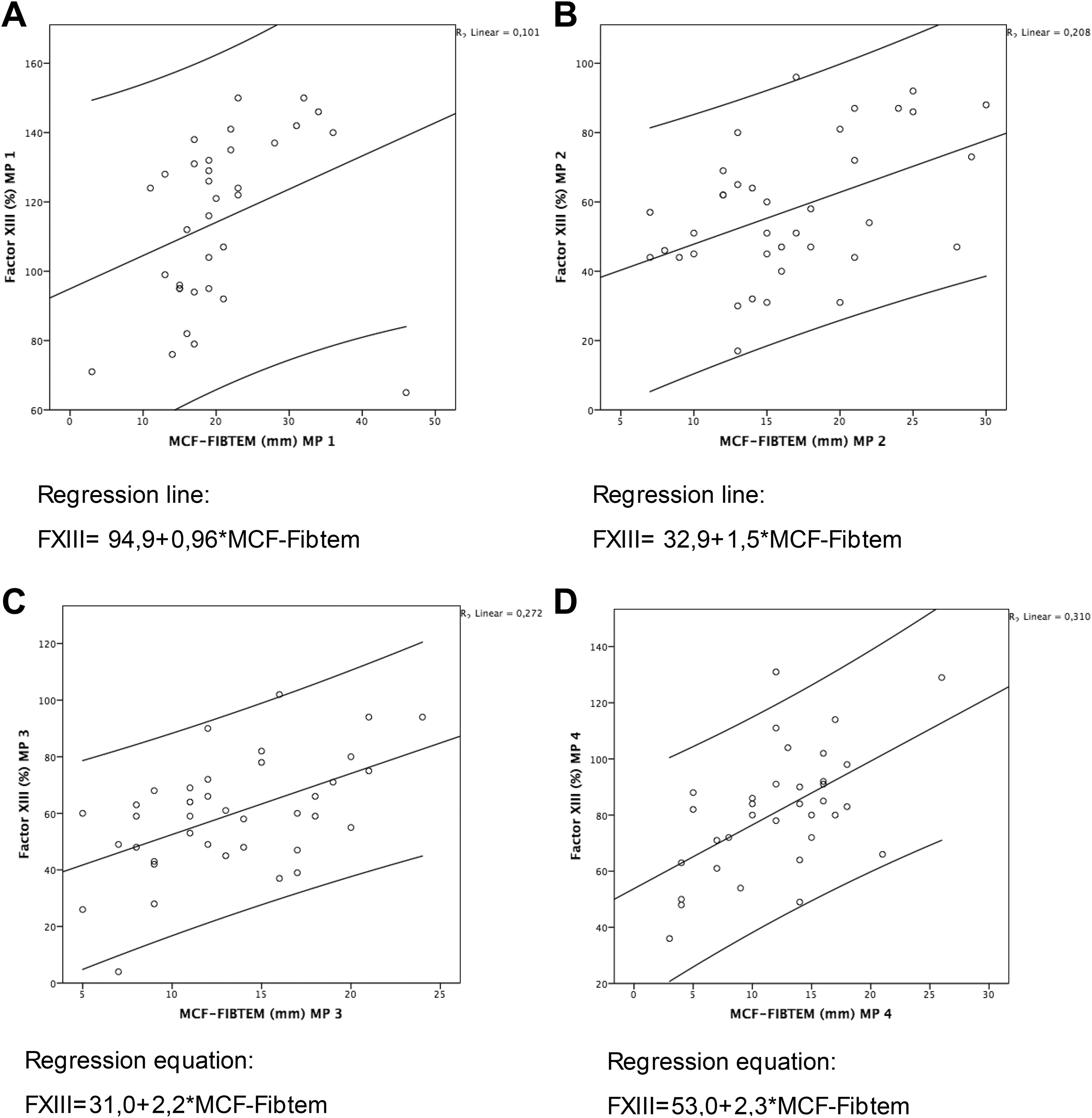
A-D, Linear regression analysis of mean clotting firmness in FIBTEM and factor XIII (FXIII).
Statistics
The statistical analysis was performed using SPSS 20. To find out differences between the group of patients with moderate blood loss and those with excessive blood loss, the Mann-Whitney U test was used. To examine the relationships between variables, the Pearson correlation coefficient r was tested. For nonnormally distributed variables, the relationship was examined using Spearman rank correlation coefficient. More precisely, we examined whether statistically significant coefficients were obtained (P < .05). Besides, we investigated the correlations between the decrease of FI and FXIII over the measuring points 1 to 4 and the suffered blood loss using linear regression. Furthermore, a regression line was calculated to study the development of the FI and FXIII levels. This regression line was calculated based on the 2 regression coefficients that have been estimated on the basis of our regression model (i) for the straight line (= intercept of the straight line) and (ii) for the influencing factor (= slope of the straight line).
Results
Patient Characteristics
Table 1 demonstrates patient characteristics. Preoperatively administered coagulation-regulating drugs are shown in Table 2.
Rotational Thromboelastometry
Changes between the measuring points (MP1/MP4) were P < .005 for CTINTEM and highly significant (P < .0001). The clotting time (CT) in INTEM and EXTEM increased from pre- to postoperatively, whereas the maximum clot firmness (MCF) decreased highly significantly in all tests. Compared with the preoperative values, postoperative CT increased 20% in INTEM and 19% EXTEM. The MCF resulted in a median of 9.7% in INTEM and 13.6% in EXTEM. With a reduction of 36.8%, the MCF showed the greatest operation-related decrease in FIBTEM, which argues for a drastic decrease in hemostatic activity. Also the conventional laboratory parameters (Hb, INR, aPTT, and FI) showed a surgery-related reduction.
Impedance Aggregometry
Prior to operation, no lack of platelets was evident. During surgery, a mild drop was recorded. The platelet count of 4 patients decreased below 100 000/µL, without reaching the critical limit of 50 000/µL. The decrease in the area under the curve (AUC) was highly significant in ASP, TRAP, and ADP tests, without the need for interventions. It must therefore be deduced that the functionality of platelets always lowers in the course of surgery time, although no value indicating the necessity of an intervention could be detected by means of impedance aggregometry. Thus, and according to Multiplate analysis, no platelet dysfunction was detectable among our patient collective. In 29 out of 41 patients, antiplatelet drugs were not discontinued preoperatively, which was reflected in the Multiplate by significantly reduced, yet sufficiently stimulating properties before surgery. (Figure 3).
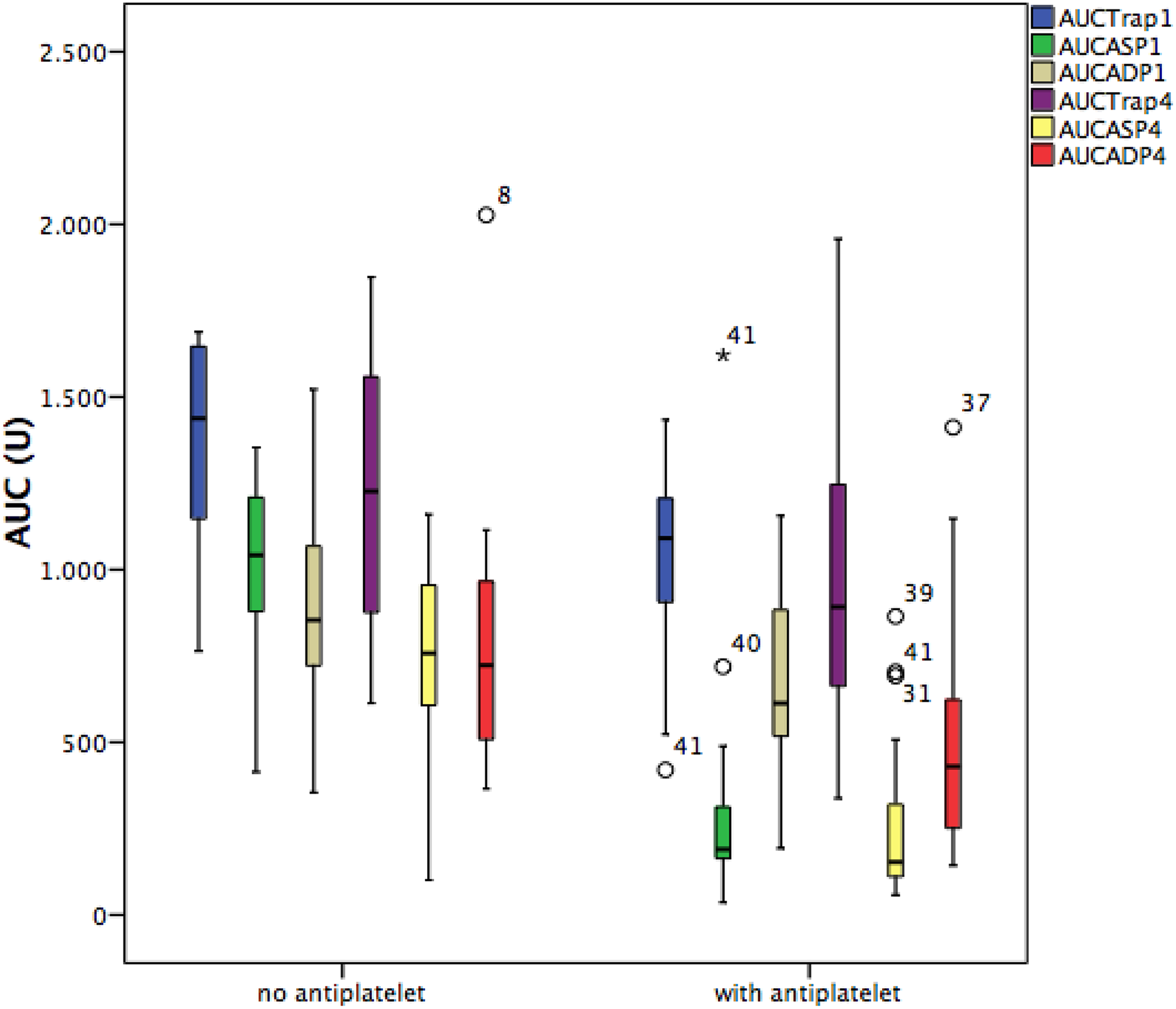
Multiplate analysis in patients with/without ADP-antagonists.
Loss of Blood
In the whole group of patients, a moderate blood loss was observed. Only 1 patient had an excessive blood loss of more than 200 mL/h. Except this, no cases of coagulopathy were observed. The continuous blood loss was reflected in the regression analysis as a function of the preoperative CTINTEM with r = 0.378 (P < .05; confidence interval 95% [CI]: 0.25-2.83) and MCFINTEM with r = 0.305 (P < .071; 95% CI: 0.31-7.66). For further analysis, the patient population was divided into 2 groups based on the distribution of drainage loss: patients with a blood loss >90th percentile or above 293 mL/2 hours were defined as “increased” (= bleeder). Differences in the preoperative MCF-EXTEM/-INTEM and CT-INTEM compared to the nonbleeding group are given in Table 3. More precisely, the MCF of the bleeder group was 4 mm higher than that in the nonbleeder group, and the CT-INTEM was correspondingly lengthened to 48 seconds. Based on the available data, we were not able to derive an estimate of blood loss.
Discussion
Clotting Activity and Blood Loss
The study showed a drastic surgery-related decrease in clotting factors, platelet count and platelet function. Already at the application of heparin (MP2), a significant compromise could be recorded. This implies that an hemostatic disturbance already existed at an early stage of surgery and just continued during the further course of the operation.
Regarding intraoperative coagulation dysfunction, due to denaturation and activation at the blood–air interface of the oxygenator, coagulation factors will also be reduced, 17 and platelets and endothelium activation lead to a continuous diffuse thrombin production, which in turn contributes to other consumption of coagulation. 17 Additionally, the extracorporeal circulation fosters inflammatory processes leading to an increased expression of tissue factor on monocytes contributing to an activation of the extrinsic pathway. 18,19 In the end, these processes are leading to an uninterrupted production of thrombin.
The serine protease thrombin (factor IIa) catalyses fibrin to FI and activates, among others, FXIII and platelets. The result of this continuously deferred activation is that fewer potential components of clot formation and wound healing are available after CPB. All these changes are characterized by a further decrease in hemostatic activity up to the measuring time after arrival in the ICU. 18,20 Consistently, perioperatively, the clotting activity recorded a decline of 10% to 36% in our study. Yet, it was especially the CT and clot formation that did reach pathological norm limits. Out of the postoperative parameters, a total of 28% of the measurements gave a result outside the standard value limits.
As expected, a strong correlation between FIBTEM measurement in ROTEM and FI concentration in standard laboratory analysis was confirmed in this study. However, it was striking that the FI concentration measured in standard laboratory testing showed considerably less critical readings than in case of the MCF-FIBTEM measurements. Yet, this diagnostic discrepancy is exclusively due to methodological differences. As opposed to the laboratory method according to Clauss et al, 21 MCF-FIBTEM does not determine the plasma levels of FI and fibrin dimer but is able to prove on the basis of clot formation whether there is a relevant lack of functional FI for clot formation. 22,23
The platelet aggregation (ADP, ASP, and TRAP assay) yielded almost exclusively regular measurement results. The platelets of patients undergoing platelet aggregation therapy were indeed less stimulated, yet sufficiently able to aggregate. In summary, it should be noted that the coagulation analysis performed in this study revealed a predominant decrease in plasma coagulation, which may foster coagulopathy or impair sufficient hemostasis under certain conditions such as massive blood loss, hypothermia, or systemic inflammatory response syndrome (SIRS). Platelet functionality had no significant reduction due to surgery and CPB.
Patients with excessive blood loss defined as loss of thoracic drainage >90th percentile were predominantly male and slightly younger on average. It is generally considered that the blood loss is dependent on the intake of platelet aggregation drugs, the preoperative hematocrit value, and the bypass time. 1,15,17 In this study, however, we were able to approve this dependence.
Another finding of the present study was the observation of a correlation between continuous drainage loss and the initial intrinsic coagulation measured before the start of surgery. On average, the CTINTEM was 14.4% longer, and the MCF-INTEM was slightly higher in groups of patients with excessive blood loss. As observed in similar studies, the preoperative MCF-EXTEM also showed higher values in patients with elevated blood loss and likewise the FI levels and MCF-FIBTEM in this group showed higher averages. High FI levels have so far been only observed in the context of increased intraoperative or postoperative blood loss and transfusion. 24 –26 In a previous study, which also included inflammatory parameters, an increased hemostatic activity was associated with increased FI, leukocytes, and CRP, suggesting an acute phase response that had already become evident prior to surgery. 26
Both, low FI and FXIII levels affect blood loss negatively. 24,27,28 In accordance with the evaluated patients, the measured concentrations of FI and FXIII revealed a weak correlation with postoperative blood loss.
Some studies show that critically low Multiplate results such as ADP test <31 U are associated with increased blood loss. 15 Thus, a previous case–control study carried out by Mishra et al, which included 81 cardiac surgical patients, showed that uninterrupted antiplatelet intake led to higher blood and platelet concentrate transfusion rates, greater blood loss, and more rethoracotomies comparing with the results of the patient group whose medication had been interrupted 5 days preoperatively. Furthermore, Mishra et al demonstrated for the analyzed patients that perioperative aggregometry and, in particular, the postoperative ASP test allow the researcher to estimate the future blood loss and transfusion necessity. 15 These interdependencies could not be confirmed in the present study. Instead, the platelets of patients with uninterrupted antiplatelet medication had, as expected, decreased stimulating properties before surgery, but no significant differences in blood loss were observed. Under surgery and CPB, all platelet receptors were affected roughly to the same extent but without reaching abnormal reference ranges or inducing abnormal blood loss. Thus, among the evaluated patients, no drawback caused by continuous antiplatelet drug intake could be detected.
In summary, except from preoperative intrinsic clot formation, the observed dependence of blood loss on ROTEM, Multiplate, factor I, and FXIII concentration was only weak. The used POC methods therefore appear unsuitable as screening method. As reference values, however, we used values obtained from healthy volunteers, whose comorbidity profiles differed significantly from cardiac surgery patients. As a further consequence of these findings, it will also be necessary to adapt existing reference ranges to the special circumstances of cardiac surgical procedures. For the future, it would therefore also be desirable to have a universal diagnostic scheme in which the clinical situation of the patient (eg, bypass time, SIRS, comorbidities, transfusion, blood loss) is taken into consideration when defining the reference values relevant for automated diagnostics.
Fibrinogen and FXIII
Another new finding of the present study was the observation that FI and FXIII levels of all measured time points correlated with each other and that, based on the measured FI, an assessment of FXIII levels is likely possible. Consequently, also an indirect near-patient determination of FXIII would be feasible. So far, photometric FXIII determination has prevailed as common laboratory method of quantitative analysis. 29 Nevertheless, this technique is not considered a routine laboratory procedure as it is rarely used and takes too much time for dynamic treatment planning. Several studies that attempted to measure FXIII either by adding FXIII to EXTEM (TeenTem) or by correlating it with APTEM point into the direction that thromboelastometry is not suitable to determine FXIII levels satisfactorily. 11,14
This seems to be a relevant issue as FXIII plays an essential role in the blood coagulation process by cross-linking fibrin molecules to form an insoluble clot and rendering the clot resistant against fibrinolysis. Moreover, several authors described FXIII deficiency in cardiac surgery patients due to adverse effects of extracorporeal circulation on hemostasis. 12,13 Factor XIII plasma levels after CPB are inversely correlated with postoperative blood loss. Patients who were administered FXIII in the postoperative period showed reduced drainage blood loss and therefore reduced transfusion requirements. 13 These data are not in contrast to a recent study by Karkouti et al 1 demonstrating no effect on transfusion avoidance, transfusion requirements, or reoperation in moderate-risk cardiac surgery patients by replenishment of FXIII levels after CPB (Karkouti et al 1 ), as this study only included patients with moderate decrease in FXIII levels. In this context, it should be accentuated that only significantly depressed FXIII concentrations result in clinically relevant bleeding disorders. Therefore, the new possibility to estimate the FXIII concentration on the basis of MCF-FIBTEM is a further step toward more targeted bedside monitoring and can consequently contribute to the optimization of time, staff, and money resources.
Summary
Diagnostic and clinical relevant grey areas in routine coagulation tests can be compensated by the use of POC technology in cardiac surgery. However, since no method is currently available giving a complete overview of the hemostasis, it seems favourable to add complementary methods such as rotational thromboelastometry and impedance aggregometry or their combination.
In our investigation in cardiac surgery patients, a well-known predominant decrease in plasma coagulation was observed. The subsequent analysis of coagulation tests and the observed drainage quantities showed a correlation of the preoperative intrinsic coagulation and blood loss. Apart from deficiencies of coagulation factors, we additionally were able to demonstrate that an increased hemostatic activity was observed in patients with increased blood loss.
As expected, platelet function in patients with a preoperative platelet inhibition was weakened. Nevertheless, after surgery, there was sufficient aggregation without apparent adverse effects on blood loss.
Finally, we’re able to show for the first time that FI levels, whether determined by standard laboratory surveillance or means of FIBTEM, correlate with the FXIII levels in plasma. Based on these findings, it would be possible for the clinical staff to make an assessment of FXIII based on the FI values and would allow an integration of FXIII into the coagulation monitoring in cardiac surgery.
Footnotes
Authors’ Note
Ethical approval to report this study was obtained from local ethics committee of the University of Halle (approval number: HAL 213). Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Acknowledgments
Ma.B. carried out the studies and data analyses and drafted the manuscript. Mi.B. and C.R. carried out the sample analyses. Ma.B. participated in the design of the study and performed the statistical analysis. L.F., Mi.B., H.T., F.R., and E.C. conceived the study, and participated in its design and coordination. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
