Abstract
Purpose
The efficacy and safety of four-factor prothrombin complex concentrate (4F-PCC) in managing bleeding during non-cardiac surgery are unclear. We investigated the associations of 4F-PCC with postoperative RBC transfusion and adverse events in non-cardiac surgery patients with massive intraoperative bleeding.
Methods
This retrospective cohort study consecutively included non-cardiac surgery patients with massive intraoperative bleeding at a tertiary hospital (2014–2020). Administration of 4F-PCC was categorized into dose groups based on quartiles: 0 (reference), 2.8–6.7, 6.7–11.5, 11.5–19.4, and 19.4–87.5 IU/kg. Outcomes included postoperative RBC transfusion, major thromboembolic events, severe acute kidney injury, and lengths of ICU and hospital stay.
Results
Of 137 patients, 89 (65.0%) received 4F-PCC. The 6.7–11.5 IU/kg group were significantly associated with reduced postoperative RBC transfusion compared to the non–4F-PCC group (adjusted mean difference, −1.29 units; 95%CI, −2.55 to −0.04 units, P = 0.044). Such findings were not observed in other dose groups. Notably, the benefits were particularly significant in patients with preoperative platelet count ≥150 × 109/l (P = 0.031), and fibrinogen ≥3 g/l (P = 0.025). The 6.7–11.5 IU/kg group exhibited comparable incidences of major thromboembolic events (13.0% vs 10.4%) and severe acute kidney injury (8.7% vs 8.3%) compared to the non–4F-PCC group. The lengths of ICU and hospital stay were similar across groups.
Conclusion
4F-PCC may be associated with decreased postoperative RBC transfusion in non-cardiac surgery patients experiencing massive intraoperative bleeding, without a significant increase in the risk of major thromboembolic events. Randomized trials with stratified dosing are warranted to confirm efficacy, safety, and determine optimal doses.
Clinical Trial Number
ChiCTR2500096573.
Keywords
Introduction
Every year, more than 300 million individuals worldwide undergo non-cardiac surgeries. 1 Approximately 17% of postoperative deaths are attributed to bleeding. 2 Massive bleeding is frequently accompanied by coagulopathy, which develops as result of consumption and loss of coagulation factors and hemodilution. 3 Coagulopathy exacerbates bleeding severity and increases transfusion requirement.4–6 Amidst persistent blood shortages and concerns about transfusion-related adverse events,7,8 interventions aimed at improving coagulation status have been introduced for surgical patients facing massive bleeding.9,10 Four factor-prothrombin complex concentrate (4F-PCC) has demonstrated efficacy in enhancing coagulation status by providing coagulation factors II, VII (inactivated or activated), IX, and X. 9
Evidence suggests that 4F-PCC can effectively decrease bleeding and red blood cell (RBC) transfusion in cardiac surgery. In randomized controlled trials of cardiac surgeries, 4F-PCC administration reduced intraoperative RBC transfusion 11 and RBC transfusion within 24 h post-surgery 10 compared to plasma transfusion. Currently, randomized controlled trials regarding the efficacy of 4F-PCC in patients undergoing non-cardiac surgery are lacking. Retrospective studies of patients who underwent liver transplantation showed that 4F-PCC administration was associated with decreased intraoperative RBC transfusion 12 and a lower rate of perioperative RBC transfusion. 13 The mechanism by which 4F-PCC decreases RBC transfusion is thought to improve the coagulation status and reduce microvascular bleeding. Thus, evidence suggest that 4F-PCC potentially confers hemostatic benefits to non-cardiac surgery patients. 14
In this retrospective cohort study of non-cardiac surgery patients, we aimed to determine whether 4F-PCC administration is associated with decreased postoperative RBC transfusion in patients with massive intraoperative bleeding. Notably, we assessed whether the therapeutic dose of 4F-PCC and the baseline coagulation status influenced 4F-PCC efficacy. Given previous findings indicating the need for monitoring thromboembolic events associated with the 4F-PCC, 15 we further analyzed the risk of major thromboembolic events in patients to identify candidate populations that could benefit from 4F-PCC administration. We hypothesized that certain dose ranges of 4F-PCC might be associated with improved hemostatic efficacy—reflected by reduced postoperative RBC transfusion—without a significant increase in thromboembolic events.
Material and Methods
Ethics
Ethical approval for this study was provided by the Institutional Review Board of Peking Union Medical College Hospital, Beijing, China on 17 April 2023, and the need for informed consent was waived owing to the retrospective nature (reference number: K3751). This study was registered at chictr.org.cn (trial number: ChiCTR2500096573) and adhered to the STROBE guidelines.
Study Population
We consecutively included all the adult patients who experienced massive intraoperative bleeding during non-cardiac surgery between January 1, 2014 and December 31, 2020 at Peking Union Medical College Hospital. Massive intraoperative bleeding was defined as bleeding necessitating the transfusion of ≥10 units of RBC during surgery.16,17 Patients with congenital or acquired coagulation disorders (n = 3), those who underwent resuscitation for hemorrhagic shock prior to surgery (n = 7), intraoperative mortality (n = 2), hemostatic embolization or reoperation within the initial 24 h post-surgery (n = 6), discharge against medical advice (n = 1), or incomplete transfusion data were excluded (n = 7) (Figure 1).
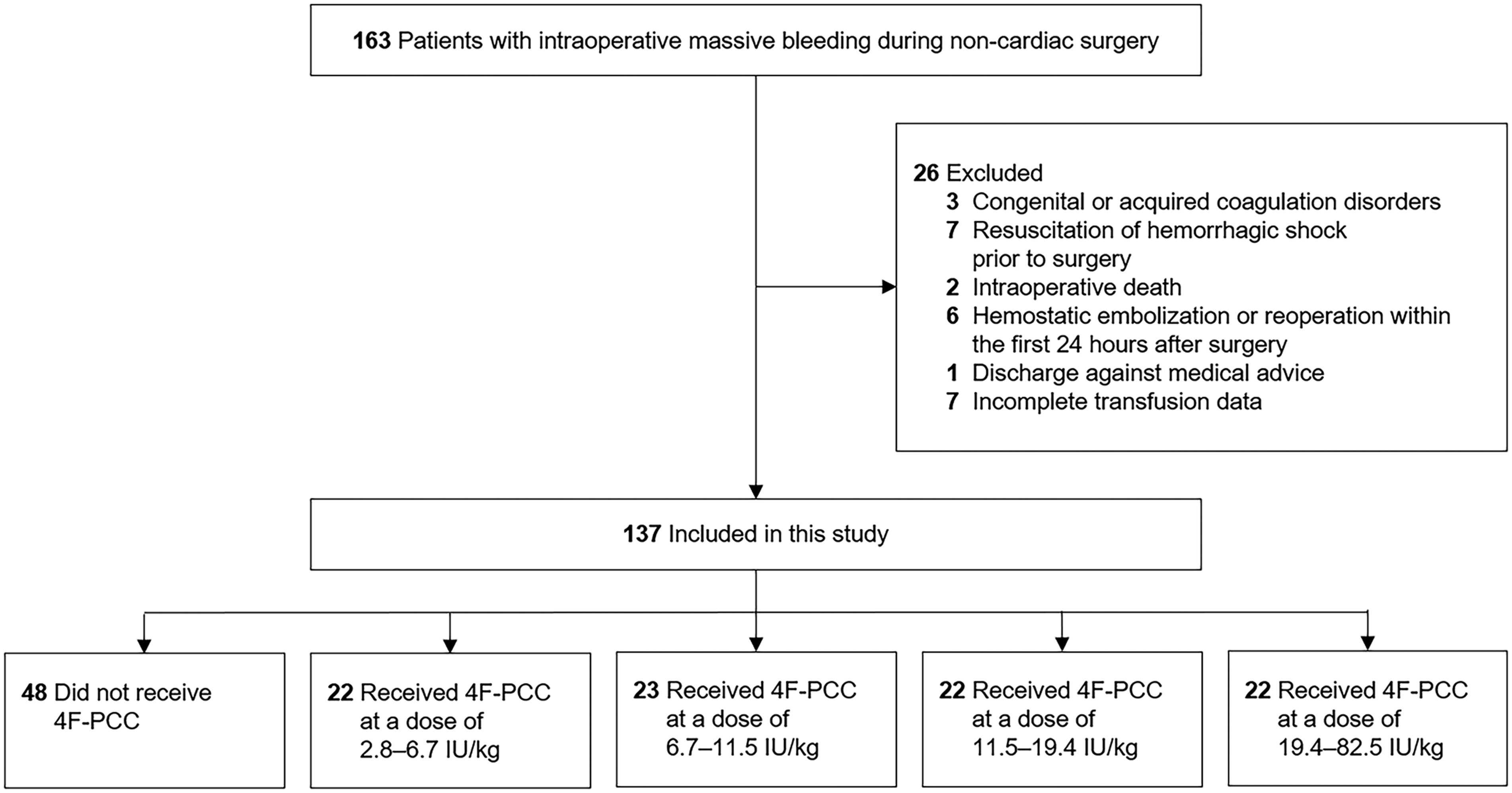
Flowchart of the Study. Abbreviations: 4F-PCC, Four-Factor Prothrombin complex Concentrate.
Variable Definitions
The intervention variable was the administration of 4F-PCC (Hualan Biological Engineering, Inc., KangShuNing, Xinxiang, Henan, China), which contains non-activated coagulation factors II, VII, IX, and X. Administration occurred during surgery and/or within the initial 24 h post-surgery, categorized into five dose groups based on quartiles of Factor IX units per kilogram (0, 2.8–6.7, 6.7–11.5, 11.5–19.4, and 19.4–87.5 IU/kg).
Outcomes encompassed RBC transfusion within the initial 24 h post-surgery (defined as postoperative RBC transfusion), postoperative plasma transfusion, postoperative plasma transfusion, major thromboembolic events (including ischemic stroke, myocardial infarction, pulmonary embolism, peripheral arterial thrombosis, and symptomatic deep venous thrombosis),18–20 severe acute kidney injury (defined as stage 2 or 3 according to the Kidney Disease Improving Global Outcomes criteria 21 ), length of intensive care unit stay, and length of hospital stay during postoperative hospitalization. All postoperative complications were monitored throughout the index hospitalization until discharge.
We evaluated the following clinical characteristics as potential confounders: age, sex, BMI, ASA status, Charlson comorbidity index, preoperative laboratory values (hemoglobin level, platelet count, international normalized ratio [INR], activated partial thromboplastin time [APTT], fibrinogen level, and serum creatinine), surgery type, surgery length, volume of intraoperative blood loss, RBC transfusion, fluid infusion (crystalloids and nonblood colloids), volume of plasma, platelets, and fibrinogen concentrate transfusion, use of other hemostatic agents (tranexamic acid and Hemcoagulase Agkistrodon for Injection [a thrombin-like enzyme] 22 ), and noradrenaline infusion during surgery and/or within the initial 24 h post-surgery.
Perioperative Transfusion Protocol
The protocol for the management of intraoperative massive hemorrhage was adhered to the ‘Perioperative blood transfusion guidelines’ issued by the Chinese Society of Anesthesiology 23 (Supplemental Figure S1). This protocol emphasizes balanced resuscitation with blood components (red blood cells and plasma in a 1:1 ratio) combined with decisions guided by laboratory tests and thromboelastography (TEG) findings when available. The key laboratory parameters (arterial blood gas, complete blood count, INR, APTT, and fibrinogen) are evaluated at 30–60-min intervals to guide transfusion and therapeutic decisions. The TEG is utilized when conditions permit (Supplemental Figure S2). Salvaged blood of 200 mL was considered equivalent to one unit of packed allogeneic RBC, 24 which was factored into the calculation for intraoperative RBC transfusion. If, after two to three cycles of resuscitation, bleeding remains uncontrolled — indicated by persistent surgical field oozing or difficulty achieving hemostasis —, or laboratory tests show prolonged PT/APTT or an extended R time on TEG, clinicians are prompted to consider administer 1–2 vials of 4F-PCC (200–400 IU, approximately 5 IU/kg), with repeat dosing if clinically indicated. Postoperative blood management relied on standard laboratory testing, such as hemoglobin level (a threshold of <70 g/l or <100 g/l for high-risk patients), platelet count (a threshold of 50–100 × 109/l), APTT (a threshold of 1.5 times the midpoint of normal range), INR (a threshold of 2.0), fibrinogen level (a threshold of 1.5 g/l), and TEG testing. The 4F-PCC was used in cases where coagulopathy (INR > 2.0 or R > 8 min) and volume overload (characterized by pulmonary hydrostatic edema 25 ) were concerned. In general, 1000 IU of 4F-PCC is considered a substitute for two units of plasma, 19 a formula used to calculate plasma transfusion. For other hemostatic agents, tranexamic acid is routinely used as a prophylactic measure to reduce perioperative bleeding, particularly in cardiac, major orthopedic, and major gynecologic surgeries. Tranexamic acid is also administered early in cases of postpartum hemorrhage, as well as in cases of severe hemorrhage, with an initial dose of 1 g for adult patients. Hemocoagulase Agkistrodon for Injection is used for the prevention of hemorrhage at surgeons’ discretion.
Statistical Analysis
Variables were compared among the groups using ANOVA or Kruskal-Wallis tests for continuous variables and Pearson's χ² or Fisher's exact test for categorical variables where appropriate. Pairwise comparisons were performed for variables that differed significantly between groups.
Initially, we analyzed 4F-PCC administration as a binary variable to investigate its association with postoperative RBC transfusion. We then categorized the patients into five dose groups based on the quartiles of 4F-PCC dose to explore the effects of 4F-PCC dose on postoperative RBC transfusion. Two regression models were built: Model 1 was an unadjusted model and Model 2 was a multivariate linear regression. Covariates for the multivariable linear regression model were selected based on P < 0.20 in the bivariate analysis, and universal confounders such as age and sex were included.
We used a multivariate-adjusted restricted cubic spline 26 to further characterize the relationship between 4F-PCC dose and postoperative RBC transfusion. The four knots were placed at default positions according to the 4F-PCC dose quartiles (5%, 35%, 65%, and 95%). 27 The identified covariates were adjusted in the restricted cubic spline model. The non-linearity of the dose-response association was assessed using Wald statistics. When there was evidence of non-linearity, a two-line piecewise linear model with a single change point was performed.
Next, we conducted subgroup analyses to explore the association between 4F-PCC and postoperative RBC transfusion where patients’ preoperative coagulation status and intraoperative transfusion volume varied. Analyses were stratified based on preoperative platelet count (≥150 × 109/l vs <150 × 109/l 28 ), fibrinogen level (≥3 g/l vs <3 g/l 29 ), and intraoperative RBC transfusion (≥15 units vs <15 units 30 ), with the associations evaluated within each subgroup.
To investigate the safety of 4F-PCC in non-cardiac surgery patients, we used multivariate logistic regression model to assess the associations of 4F-PCC with major thromboembolic events and severe acute kidney injury. Further, we used multivariate linear regression model to analyze the associations of 4F-PCC with the postoperative plasma and palates transfusion, lengths of intensive care unit and hospital stay. Covariates included age, sex, and the confounders identified by the bivariate analysis.
A two-sided P value of less than 0.05 indicated statistical significance. Restricted cubic splines and regression analyses were performed using the R software, (version 4.2.3; R Foundation for Statistical Computing, Vienna, Austria). All other statistical analyses were performed using SPSS software (version 22.0; SPSS, Chicago, USA).
Results
Patient Characteristics
This study included 137 patients who underwent non-cardiac surgery with massive intraoperative bleeding. The median (IQR) age of the patients was 53.0 (41.5–63.5) years, and 91 (66.4%) were female. Among them, 89 (65.0%) received 4F-PCC in the perioperative period. The median (IQR) 4F-PCC dose was 11.5 (6.7–19.4) IU/kg. The demographic and clinical characteristics of the patients are presented in Table 1. A more specific list of procedures broken down by surgery type is included in Supplemental Table S1. Compared with patients who did not receive 4F-PCC, the patients who received 4F-PCC at a dose ≥ 6.7 IU/kg were accompanied by a lower level of preoperative fibrinogen.
Patients’ Demographic and Clinical Characteristics.

Values are presented as median (interquartile range) or number (proportion); * Indicates statistical significance compared to patients who did not receive 4F-PCC (Pcorrected < 0.05); a Includes liver, vascular, thoracic, obstetric, otolaryngologic, and neurological surgeries.
Abbreviations: APTT, activated partial thromboplastin time; 4F-PCC, four-factor prothrombin complex concentrate; INR, international normalized ratio; RBC, red blood cell.
Intraoperatively, compared with those without 4F-PCC administration, those who received 4F-PCC at a dose ≥ 6.7 IU/kg were administered higher doses of fibrinogen concentrate, while patients receiving ≥ 11.5 IU/kg experienced greater blood loss and required larger volumes of blood product transfusion.
4F-PCC Administration and Postoperative RBC Transfusion
Initially, we investigated whether 4F-PCC administration was associated with a lower volume of postoperative RBC transfusions in the overall population. The median (IQR) volume of postoperative RBC transfusion was 2.0 (0.0–4.0) units. In the unadjusted analysis, we did not observe a significant difference in postoperative RBC transfusion between the patients who received 4F-PCC and those who did not (mean difference [MD] 0.92 units; 95%CI −0.10 to 1.94 units, P = 0.078). Moreover, a multivariate linear regression model did not yield significant results (MD −0.74 units; 95%CI, −1.71 to 0.23 units, P = 0.132; adjusted for age, sex, surgery type, volume of intraoperative RBC transfusion, fluid infusion, and volume of perioperative plasma, platelets, and fibrinogen concentrate transfusion).
Therapeutic Dose of 4F-PCC and Postoperative RBC Transfusion
Given that previous studies have reported that the effect of 4F-PCC is dose-dependent,31–33 we analyzed the hemostatic effect of 4F-PCC by categorizing its therapeutic dose into quartiles as follows: 0 (reference group), 2.8–6.7, 6.7–11.5, 11.5–19.4, and 19.4–87.5 IU/kg. Notably, patients who received 4F-PCC at a dose of 6.7–11.5 IU/kg required significantly a lower volume of postoperative RBC transfusion compared to those who did not receive 4F-PCC (MD −1.29 units; 95%CI, −2.55 to −0.04 units, P = 0.044; adjusted for age, sex, surgery type, volume of intraoperative RBC transfusion, fluid infusion, and volume of perioperative plasma, platelets, and fibrinogen concentrate transfusion) (Figure 2). And patients who received 4F-PCC at a dose of 11.5–19.4 IU/kg showed a non-significant trend toward requiring a lower volume of postoperative RBC transfusion when compared to those without 4F-PCC administration (MD −1.18 units; 95%CI −2.58 to 0.21 units, P = 0.096), though this did not reach statistical significance. Such findings were not observed in other quartiles (2.8–6.7 IU/kg or 19.4–87.5 IU/kg). To further describe the effects of 4F-PCC, we used restricted cubic spline regression to model the relationship between 4F-PCC and postoperative RBC transfusion: a J-shaped relationship was detected in the overall population (Supplemental Figure S3, P = 0.027 for non-linearity). A piecewise linear model showed that the hemostatic effect of 4F-PCC is maximal at the dose of 7.1 IU/kg. Collectively, our results suggest that 4F-PCC administration potentially benefits non-cardiac surgery patients with massive intraoperative bleeding at optimal doses.

Associations of four-factor prothrombin complex concentrate with postoperative red blood cell transfusion and major thromboembolic events. (A) Multivariable linear regression model was used to estimate the association between 4F-PCC and postoperative RBC transfusion, adjusted for age, sex, surgery type, volume of intraoperative RBC transfusion, fluid infusion, and volume of perioperative plasma, platelets, and fibrinogen concentrate transfusions. (B) Multivariate logistic regression model was used to estimate the association between 4FPCC and major thromboembolic events, adjusted for age, sex, ASA status, preoperative international normalized ratio, and volume of perioperative platelet transfusion. Abbreviations: 4F-PCC, four-factor prothrombin complex concentrate; MD, mean difference; NA, not applicable; OR, odds ratio.
Baseline Coagulation status and the Efficacy of 4F-PCC
The baseline coagulation status was heterogeneous among the included patients, which has been reported to potentially influence the effects of hemostatic agents. 34 Here, we investigated whether baseline coagulation parameters influenced the effect of 4F-PCC. In patients with a preoperative platelet count ≥150 × 109/l, 4F-PCC administration (at a dose of 6.7–11.5 IU/kg) was significantly associated with a lower volume of postoperative RBC transfusion compared to those who did not receive 4F-PCC (MD −1.56 units; 95%CI −2.98 to −0.15 units, P = 0.031; adjusted for age, sex, surgery type, volume of intraoperative RBC transfusion, fluid infusion, and volume of perioperative plasma, platelets, and fibrinogen concentrate transfusion). In contrast, such associations were not detected in patients with a preoperative platelet count <150 × 109/l (Supplemental Table S2).
Among patients with a preoperative fibrinogen level ≥3 g/l, 4F-PCC administration (at a dose of 11.5–19.4 IU/kg) was significantly associated with a lower volume of postoperative RBC transfusion compared to those who did not receive 4F-PCC (MD −2.10 units; 95%CI −3.93 to −0.27 units, P = 0.025); further, patients who received 4F-PCC at a dose of 6.7–11.5 IU/kg showed a nonsignificant trend toward requiring a lower volume of postoperative RBC transfusion when compared to those without 4F-PCC administration (MD −1.54 units; 95%CI −3.16 to 0.08 units, P = 0.062) (adjusted for age, sex, surgery type, volume of intraoperative RBC transfusion, fluid infusion, and volume of perioperative plasma, platelets, and fibrinogen concentrate transfusion). Such associations were not detected in patients with a preoperative fibrinogen level <3 g/l (Supplemental Table S2).
In patients who received intraoperative RBC transfusion <15 units, 4F-PCC administration (at a dose of 6.7–11.5 IU/kg) was significantly associated with a lower volume of postoperative RBC transfusion in contrast to the patients without 4F-PCC administration (MD −1.72 units; 95%CI −3.07 to −0.37 units, P = 0.015); also, patients who received 4F-PCC at a dose of 11.5–19.4 IU/kg showed a nonsignificant trend of requiring a lower volume of postoperative RBC transfusion as compared to those who did not receive 4F-PCC (MD −1.57 units; 95%CI −3.11 to −0.02 units, P = 0.051) (adjusted for age, sex, surgery type, volume of intraoperative RBC transfusion, fluid infusion, and volume of perioperative plasma, platelets, and fibrinogen concentrate transfusion), though this did not reach statistical significance. Such associations were not detected in patients with intraoperative RBC transfusion ≥15 units (Supplemental Table S2). Collectively, our results suggest that non-cardiac surgery patients with a relatively intact baseline coagulation status are more likely to benefit from 4F-PCC administration.
4F-PCC Administration and Postoperative Plasma and Platelet Transfusion
The median (IQR) volume of postoperative plasma transfusion was 2.0 (0.0–4.0) units. Compared with the reference group (0 IU/kg), patients receiving the highest dose group (19.4–87.5 IU/kg) had significantly increased plasma transfusion (2.5 [1.5–4.0] vs 2.0 [0.0–2.0] units, P < 0.05), and platelet transfusion (0.0 [0.0–1.0] vs 0.0 [0.0–0.0] units, P < 0.05) in the unadjusted analyses (Supplemental Table S3).
In multivariable regression models adjusting for confounders, no significant reduction in plasma or platelet transfusion was observed across lower or intermediate dose groups. In contrast, the highest dose group (19.4–87.5 IU/kg) was independently associated with increased postoperative platelet transfusion compared with those without 4F-PCC administration (MD 0.56, 95% CI 0.31 to 0.80, P < 0.001) (Supplemental Table S3).
4F-PCC Administration and the Risks of major Thromboembolic Events and Severe Acute Kidney Injury
Major thromboembolic events occurred in 17 patients (12.4%), of whom eight patients (5.8%) developed myocardial infarction, six patients (4.4%) had symptomatic deep vein thrombosis, two patients (1.4%) had pulmonary embolism, one patient (0.7%) had ischemic stroke, and one patient (0.7%) had peripheral arterial thrombosis (Table 2). Logistic regression analysis showed no significant difference in the incidence of major thromboembolic events between the patients who received 4F-PCC at a dose of 6.7–11.5 IU/kg and those who did not receive 4F-PCC (13.0% vs 10.4%; OR 0.51, 95%CI 0.08 to 3.10, P = 0.462; adjusted for age, sex, ASA status, preoperative INR, and volume of perioperative platelets transfusion) (Figure 2, Supplemental Table S3). Notably, the incidence of major thromboembolic events was numerically higher in the patients who received 4F-PCC at a dose of 11.5–19.4 IU/kg than those who did not administer 4F-PCC (22.7% vs 10.4%; OR 1.25, 95%CI 0.23 to 6.92, P = 0.798), though this difference was not statistically significant. The numerical increases may warrant further investigation in larger studies.
Efficacy and Safety Outcomes.
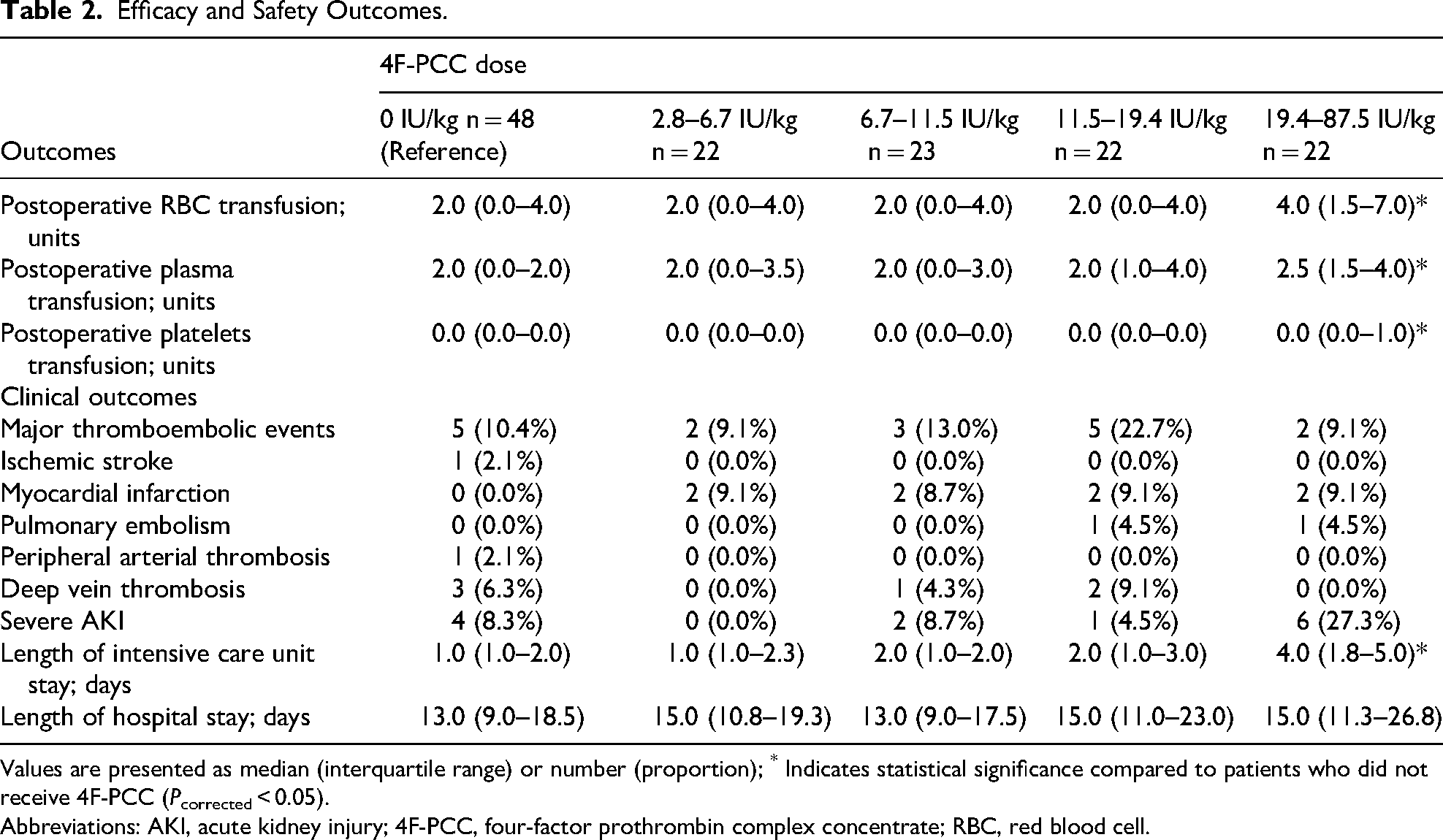
Values are presented as median (interquartile range) or number (proportion); * Indicates statistical significance compared to patients who did not receive 4F-PCC (Pcorrected < 0.05).
Abbreviations: AKI, acute kidney injury; 4F-PCC, four-factor prothrombin complex concentrate; RBC, red blood cell.
Severe acute kidney injury occurred in 13 patients (9.5%). Logistic regression analysis indicated no significant difference in the incidence of severe acute kidney injury between patients who received 4F-PCC at a dose of 6.7–11.5 IU/kg and those without 4F-PCC administration (8.7% vs 8.3%; OR 1.20, 95%CI 0.14 to 10.69, P = 0.867; adjusted for age, sex, ASA status, preoperative serum creatinine, volume of perioperative plasma transfusion). Notably, patients who received 4F-PCC at a dose ≥19.4 IU/kg showed a higher incidence of severe acute kidney injury compared to those who did not receive 4F-PCC (27.3% vs 8.3%; OR 4.12, 95%CI 1.03 to 16.54, P = 0.046). However, this finding lost significance after adjustment (OR 5.00, 95%CI 0.58 to 43.33, P = 0.144; adjusted for age, sex, ASA status, preoperative serum creatinine, and volume of perioperative plasma transfusion) (Supplemental Table S3).
4F-PCC Administration and Postoperative Length of Intensive Care Unit Stay and Hospital Stay
The median (IQR) length of intensive care unit stay was 2.0 (1.0–3.0) days. There was no significant difference in the length of intensive care unit stay between the groups (P = 0.553) (Table 2). In multivariable linear regression analysis adjusted for age, sex, preoperative serum creatinine, surgery duration, and perioperative noradrenaline use, the length of intensive care unit stay did not differ significantly across 4F-PCC dose groups compared to the group without 4F-PCC administration (Supplemental Table S3).
The median (IQR) length of hospital stay was 14.0 (10.0–19.5) days. No significant difference in the length of hospital stay was observed between the groups (P = 0.386) (Table 2). In multivariable linear regression analysis adjusted for age, sex, ASA status, preoperative serum creatinine, platelet count, and intraoperative RBC transfusion volume, no significant association was found between any 4F-PCC dose group and hospital stay duration compared to the group without 4F-PCC administration (Supplemental Table S3).
Discussion
In the present study, non-cardiac surgery patients who received 4F-PCC at a dose of 6.7–11.5 IU/kg for managing massive bleeding were significantly associated with decreased postoperative RBC transfusion compared to those who did not receive 4F-PCC. Subgroup analyses revealed a particularly significant benefit of 4F-PCC in patients with a relatively intact baseline coagulation status (ie, preoperative platelet count ≥150 × 109/l, fibrinogen level ≥3 g/l). We observed comparable incidence rates of major thromboembolic events between patients who received 4F-PCC at a dose of 6.7–11.5 IU/kg and those who did not (13.0% vs 10.4%). Data on the lengths of intensive care unit and hospital stay did not yield significant differences between patients who received 4F-PCC and those who did not. Thus, our study suggests that perioperative 4F-PCC at optimal doses may confer benefits to patients undergoing non-cardiac surgery.
In non-cardiac surgery, the effects of 4F-PCC on decreasing bleeding and/or RBC transfusion are controversial. A retrospective study of 38 non-cardiac surgery patients (ie, general surgery, vascular surgery, etc) reports that 4F-PCC (median dose: 11.5 IU/kg) ceased microvascular bleeding in 96% of the study population. 35 In contrast, a retrospective study of 212 patients who underwent liver transplantation revealed that 4F-PCC (mean dose: 32 IU/kg) was not associated with reduced RBC transfusion. 36 Given the variability in 4F-PCC doses across studies and the influence of therapeutic dose on the efficacy of hemostatic agents,31–33 our study contributes to the understanding of 4F-PCC in non-cardiac surgery by demonstrating that 4F-PCC at optimal doses (specifically, 6.7–11.5 IU/kg) for managing massive intraoperative bleeding is associated with decreased postoperative RBC transfusion. Notably, the detected optimal doses of 4F-PCC for non-cardiac surgery in our study were lower than those recommended for cardiac surgery (ie, 12.5 IU/kg). 14 It is plausible that the coagulation status is less affected in non-cardiac surgery than in cardiac surgery (ie, coagulation factor depletion and hemodilution affect coagulation in cardiac surgery).37,38 Future prospective or randomized studies with stratified dosing are required to determine the optimal doses of 4F-PCC for non-cardiac surgery.
The efficacy of hemostatic agents can be influenced by the coagulation status of surgical patients. An observational study of 18 Canadian centers suggested that adequate circulating coagulation factors are associated with the enhanced efficacy of recombinant activated factor VII in the cardiac surgery. 34 Specifically, a failure of response to treatment is predicted by a set of pretreatment abnormal coagulation tests, such as platelet count <80 × 109/l, and fibrinogen level <1 g/l, among other laboratory tests. 34 Our study indicates that the effect of 4F-PCC in reducing postoperative RBC transfusion is particularly significant in patients with preserved coagulation function (ie, preoperative platelet count ≥150 × 109/l, fibrinogen level ≥3 g/l). These findings are consistent with the notion that successful hemostasis is supported by the coagulation cascade. 39 However, it is important to note that the baseline coagulation status here was only evaluated before surgery; the coagulation tests could easily have changed during surgery, especially in cases of massive bleeding. Baseline coagulation tests could be associated with disease severity, which could potentially influence the effect of 4F-PCC. 40 In patients who received more than 15 units of intraoperative RBCs, no significant benefit of 4F-PCC was observed. This may be explained by the presence of severe, multifactorial coagulopathy in the setting of massive bleeding, which often involves not only coagulation factor depletion but also thrombocytopenia, fibrinolysis, and hypothermia. Under such conditions, 4F-PCC alone may be insufficient to restore hemostasis. These findings underscore the complexity of managing coagulopathy in high-risk surgical patients and highlight the need for future studies to evaluate dynamic coagulation changes and identify the conditions under which 4F-PCC is most effective.
No significant reductions in postoperative plasma or platelet transfusion were observed across 4F-PCC dose groups in adjusted analyses. This may reflect underlying selection bias, as patients receiving 4F-PCC often had more severe coagulopathy and greater intraoperative blood loss, necessitating higher plasma and platelet requirements. Moreover, in our institutional protocol, plasma remains the first-line treatment for coagulopathy; 4F-PCC is administered only when plasma is insufficiently effective, and is not used as a substitute. Therefore, a reduction in plasma transfusion is not necessarily expected. Likewise, platelet transfusion is primarily driven by preoperative levels and intraoperative loss, both of which tend to be higher in patients receiving 4F-PCC.
A major safety concern regarding 4F-PCC has been reported as a risk factor for major thromboembolic events. The risk of thromboembolic events is associated with individual prothrombotic predisposition and 4F-PCC dose. 9 A randomized controlled trial showed that the risk of thromboembolic events was significantly higher in patients with trauma who received 4F-PCC at a dose of 25 IU/kg than in those who received plasma transfusion. 40 Randomized controlled trials of cardiac surgeries report a comparable risk of thromboembolic events when 4F-PCC is administered at doses of 25 IU/kg 10 and 15 IU/kg, 11 compared with plasma transfusion. Notably, our study suggests that 4F-PCC at a dose of 6.7–11.5 IU/kg is potentially effective and safe for non-cardiac surgery patients, as evidenced by a comparable incidence of major thromboembolic events in patients who received 4F-PCC compared to those who did not (13.0% vs 10.4%). However, it is noteworthy that a higher incidence of major thromboembolic events (22.7%) was observed in patients receiving higher doses of 4F-PCC (11.5–19.4 IU/kg). Given the association between history of thromboembolic events and increased thromboembolic risk with 4F-PCC administration, 41 caution is warranted, particularly in at-risk patients. The minimum effective dose of 4F-PCC should be further investigated to lower the thromboembolic risk.
This study has several limitations. First, being an observational study, it could not fully validate the efficacy and safety of 4F-PCC in managing massive bleeding in non-cardiac surgery patients, nor did it determine the optimal doses of 4F-PCC. Randomized controlled trials incorporating multiple dose groups of 4F-PCC for non-cardiac surgery patients are necessary to validate its efficacy, safety, and determine optimal doses. Second, selection and treatment bias may have influenced both the decision to administer 4F-PCC and the dosing strategy. In clinical practice, 4F-PCC doses are often determined by the severity of intraoperative bleeding. Although we adjusted for confounders such as intraoperative RBC transfusion and fluid volume, residual confounding is likely. This may explain why a potential transfusion-sparing effect was observed in moderate-dose groups but not in high-dose recipients, who generally had more severe bleeding. Furthermore, despite standardized protocols, the decision to administer 4F-PCC was ultimately made by the attending clinicians based on intraoperative judgment, introducing potential variability in treatment practice. Clinicians may have used a lower threshold to initiate 4F-PCC rather than plasma in patients with anticipated volume overload or in cases requiring rapid correction of coagulopathy due to immediate availability of 4F-PCC, while being more conservative in those with high thrombotic risk. These factors limit the interpretability of dose–response relationships. Third, there was a lack of systematic evaluation regarding the influence of coagulation status on 4F-PCC efficacy. While the findings suggest that a relatively preserved coagulation function is associated with improved 4F-PCC efficacy in non-cardiac surgery patients, coagulation status was only evaluated before surgery. Further studies should evaluate coagulation status before 4F-PCC administration. Another concern was selection bias. Most patients in this study underwent gynecological surgery (37.2%), and the effects of 4F-PCC may differ among different types of surgeries. Randomized control trials in specialty-specific surgery (eg orthopedic or gynecologic only) are needed to ascertain outcomes and efficacy of 4F-PCC. Presently, the results of this single-center study can only be extrapolated to limited patients. Caution should be exercised regarding the generalizability to a broader population.
Conclusion
Our study suggests that perioperative 4F-PCC administration at optimal doses is associated with decreased postoperative RBC transfusion in patients with massive intraoperative bleeding during non-cardiac surgery. The benefits of 4F-PCC are particularly significant in patients with a relatively intact baseline coagulation status. The incidence of major thromboembolic events did not significantly increase in patients who received 4F-PCC at optimal doses. Our study supports the efficacy and safety of 4F-PCC at optimal doses in patients undergoing non-cardiac surgery.
Supplemental Material
sj-pdf-1-cat-10.1177_10760296251356202 - Supplemental material for Efficacy and Safety of Prothrombin complex Concentrate in Patients with Massive Intraoperative Bleeding During non-Cardiac Surgery: A Retrospective Cohort Study
Supplemental material, sj-pdf-1-cat-10.1177_10760296251356202 for Efficacy and Safety of Prothrombin complex Concentrate in Patients with Massive Intraoperative Bleeding During non-Cardiac Surgery: A Retrospective Cohort Study by Bo Tang, Yuelun Zhang, Jia Gan, Lulu Ma and Yuguang Huang in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Ethical Approval and Informed Consent Statements
Ethical approval for this study was provided by the Institutional Review Board of Peking Union Medical College Hospital, Beijing, China on 17 April 2023, and the need for informed consent was waived owing to the retrospective nature (reference number: K3751). This study was registered at chictr.org.cn (trial number: ChiCTR2500096573).
Author Contributions
B.T.: Conceptualization, Investigation, Methodology, Data curation, Formal analysis, Validation, Visualization, Writing – original draft. Y.L.Z.: Methodology, Formal analysis, Writing – review & editing. GJ.: Resources, Data curation. L.L.M.: Conceptualization, Writing – review & editing, Supervision. Y.G.H.: Supervision, Writing – review & editing, Resources. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by National High Level Hospital Clinical Research Funding 2022-PUMCH-B-006 and 2022-PUMCH-C-011. The funding source had no role in the design of the study, data collection and analysis or preparation of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data underlying this article will be shared on reasonable request to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
