Abstract
The article’s aim was to determine predictors for short- and long-term prognosis of patients with pulmonary embolism (PE). Cohort prospective study based on the National registry on venous thromboembolism. Eighty-four patients with PE, on age 60.3 + 12.5 years, were selected and followed up in a prospective study. Pulmonary embolism was confirmed by computed tomography angiography in all the patients, while deep venous thrombosis was confirmed by ultrasound in 21 patients. Study population was followed up for 6.7 months. Multivariate regression analysis was done where right ventricular (RV) diameter (mean 37.5 mm), systolic pulmonary artery pressure (68 ± 23 mm Hg) measured by echocardiography,
Introduction
Pulmonary embolism (PE) is a fatal condition, especially in the first month. Mortality is between 3% in those patients with isolated deep venous thrombosis (DVT), up to 11% in patients presented clinical PE. The long-term prognosis (from 1-12 months) is determined with rate of recurrence and bleeding. Many models included comorbidities and risk factors to determine the probability of occurrence (Wels, Geneve), and prognosis of this condition (Pulmonary Embolism Severity Index score and Computerized registry of patients with venous thromboembolism [RIETE]).
Patients and Methods
Eighty-four patients with PE, from the National registry on VTE (age 60.3 + 12.5 years), were selected in a prospective study. The National registry is a multicenter nonrandomized survey of patients with PE with or without DVT. Pulmonary embolism was confirmed by computed tomography (CT) angiography in all of them, and DVT was confirmed by ultrasound in 21 patients.
Inclusion Criteria
Consecutive patients with symptomatic PE, with or without DVT were included. Venous thromboembolism was confirmed by compression ultrasound (for DVT) and multislice CT scan (PE). All patients (or their relatives) provided written or oral consent for participation in the registry, in accordance with local ethics committee requirements.
The following baseline data were collected at the time of inclusion in the study: age; gender; body weight; clinical presentation (DVT or PE as the first presentation); presence of comorbidities including chronic heart or lung disease; recent major bleeding (<30 days prior to VTE); concomitant medications, including antiplatelet drugs, statins, or steroids; presence of major provoking risk factors for VTE including active cancer (defined as newly diagnosed cancer, metastatic cancer, or cancer undergoing treatment), recent immobility (defined as nonsurgical patients assigned to bed rest with bathroom privileges for ≥4 days 2 months prior to VTE), surgery (2 months prior to VTE), leg trauma or fracture, use of hormonal therapy, pregnancy, or puerperium; and laboratory results including full blood count and serum creatinine levels (Table 1). Information on thrombophilia testing, when available, was also documented.
Basic Characteristics of Study Population.
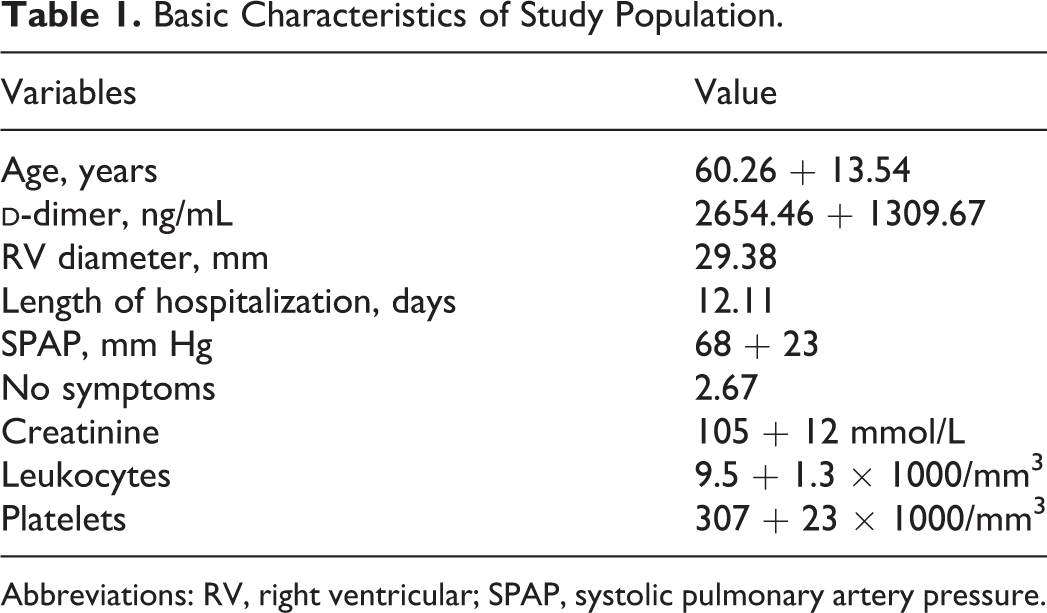
Abbreviations: RV, right ventricular; SPAP, systolic pulmonary artery pressure.
Treatment and Follow-Up
Eighty-four consequent patients with PE, from the National registry on VTE (age 60.3 + 12.5 years), were selected in a prospective study. The study population was followed up for 6.7 months.
Patients were managed according to the clinical practice of each participating hospital (ie, there was no standardization of treatment). The type, dose, and duration of anticoagulant therapy were recorded. Nine (10.71%) patients were treated with novel anticoagulants (NOAC) (rivaroxaban), 49 (58.33%) patients with unfractionated heparin, and 26 (30.96%) patients with low-molecular heparin, in the acute phase. Treatment was observed as NOAC in 19% (16 patients) and vitamin K antagonists in 81% (68 patients).
Patients were followed up for the first month and up to 6 months in the outpatient clinic. During each visit, any signs or symptoms suggesting PE recurrences or bleeding and complications were noted. Each episode of clinically suspected recurrent PE was evaluated by repeat compression ultrasonography, lung scans, helical-CT scan, or pulmonary angiography as appropriate. Length of hospitalization during the acute phase and total duration of anticoagulation were followed up by the physician, according to clinical stability of patient and level of the
Multivariate regression analysis was done, where variables from Table 1, right ventricular (RV) diameter (mean 37.5 mm, where 39 patients were with RV diameter >30 mm), systolic pulmonary artery pressure (68 ± 23 mm Hg, 25 patients with value >40 mm Hg), as measured by echocardiography,
Results
During the 6 months, 3 adverse events were observed (2 of major bleeding or 2.4%, and 1 of recurrent PE or 1.2%).
Predictive Model for Pulmonary Embolism Recurrence and Major Bleeding.a
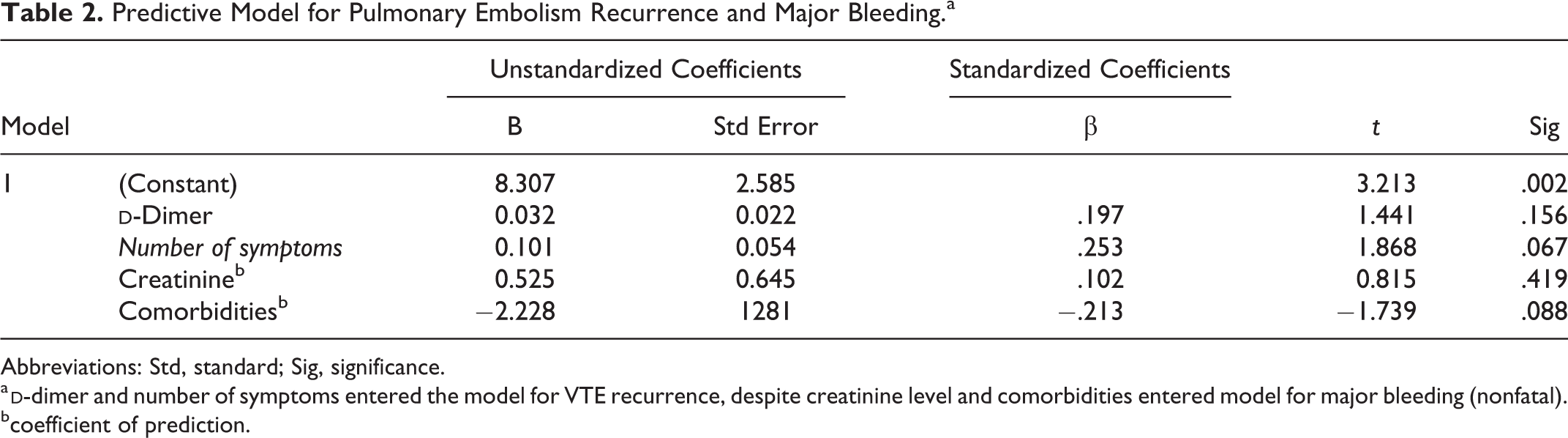
Abbreviations: Std, standard; Sig, significance.
a
bcoefficient of prediction.
Predictive Model for 6 Months Follow-Up of Nonfatal Pulmonary Embolism in a Term for Length of Hospitalization and Duration of Anticoagulation.a
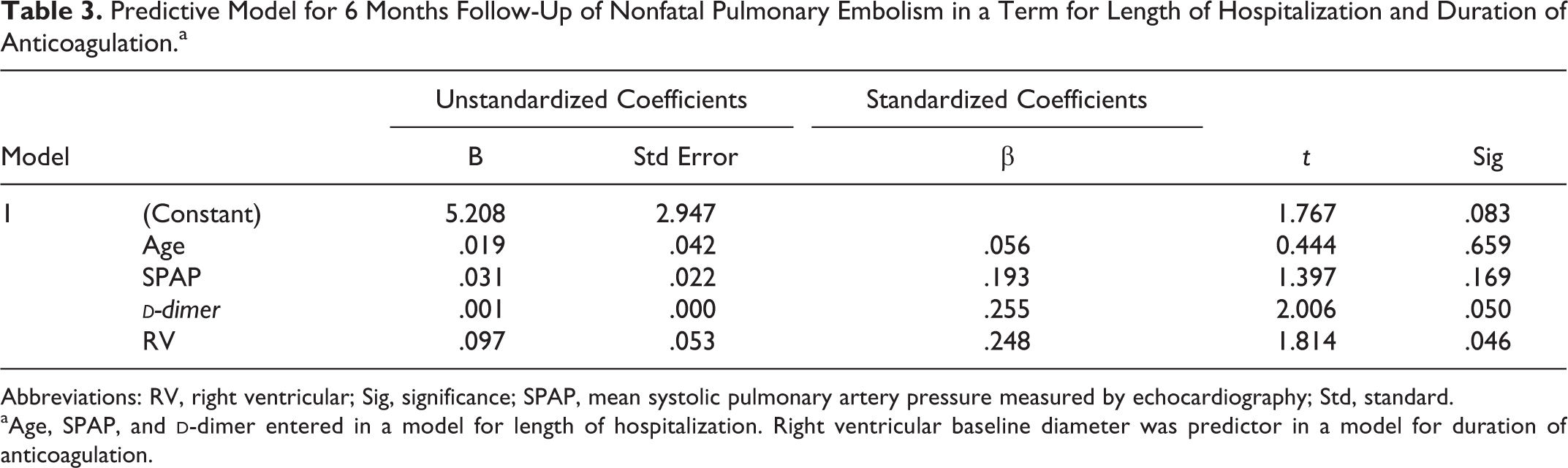
Abbreviations: RV, right ventricular; Sig, significance; SPAP, mean systolic pulmonary artery pressure measured by echocardiography; Std, standard.
aAge, SPAP, and
Predictive Model for 6 Months Follow-Up of Nonfatal Pulmonary Embolism.
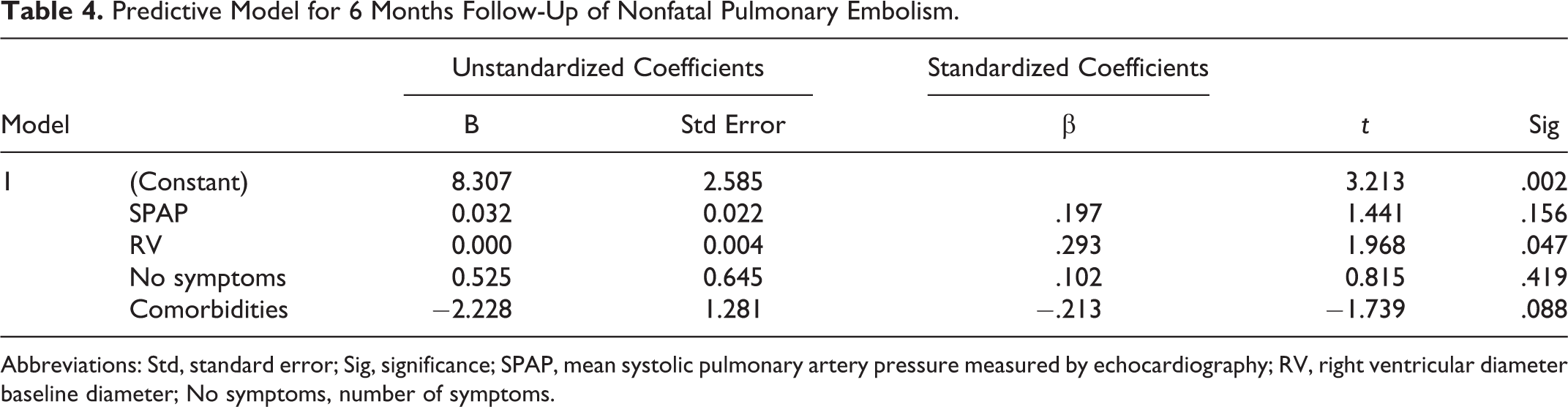
Abbreviations: Std, standard error; Sig, significance; SPAP, mean systolic pulmonary artery pressure measured by echocardiography; RV, right ventricular diameter baseline diameter; No symptoms, number of symptoms.
Discussion
In real clinical practice, more than half of patients with VTE and unprovoked events and more than third of these patients with a transient risk factor receive anticoagulant treatment for more than 12 months. The American College of Chest Physicians (ACCP) guidelines recommend a 3-month treatment duration for patients with VTE, secondary to transient risk factors, and to recommend considering lifelong treatment duration for patients with unprovoked VTE or with cancer. 1 Thus, we found that the duration of anticoagulant therapy often exceeded the recommendations, particularly in patients with transient risk factors for VTE. These ACCP guidelines remain firm in the revisited recommendations of 2016. 2 Our data suggest that in real life, physicians appear to be more concerned about the risk of bleeding than about the risk of recurrent PE episode after discontinuing therapy. Yet, in our study population, the rate of PE recurrences and the rate of major bleeding during the course of anticoagulation were not significantly different: major (but nonfatal) bleeds clearly was 2.4% and recurrent embolism 1.2%.
The European Society of Cardiology (ESC) in 2014 hoped to cement the concept of intermediate-risk PE, with their 2014 guidelines critically reviewing the combinations of imaging (echocardiographic or CT angiography) parameters and laboratory biomarkers that can be used to detect right ventricular dysfunction and/or myocardial injury.
3
Taking into account that both imaging and laboratory tests have shown consistent prognostic value, individually and in concordance with each other,
4,5
the ESC guidelines aimed to discourage uncritical time and resource-consuming laboratory and echocardiographic testing in every confirmed patient with PE with no prior triage and instead suggested a stepwise classification of early risk.
3
Lankeit et al in 201, specifically took the risk stratification value of the biomarker N-terminal probrain natriuretic peptide together with a clinical score and the echocardiographic measurement of right ventricular dysfunction to optimize their model of risk stratification.
4
Jiménez et al in 2013 used a derivation and validation cohort to design a final model to use multiple biomarkers along with a clinical score and lower limb ultrasound testing.
5
In this regard, it was proposed that
Our present study has several limitations. A clear limitation present is in relation to the treatment approach. Namely, patients were not treated with a similar approach. 22 There were clear variations present in the treatment plan in accordance with the presence of silent PE at baseline, and this is likely to have been influenced by a physician’s assessment of a patient’s risk of death. Although treatment varied by the physician’s assessment of a patient’s risk, it is not likely that local treatment practices exist, as the multiple centers from which the data originates follow the same guidelines. Another possible limitation may be that although the centers included in the study are overall typical centers that would treat PE and VTE patients, any centers involved with the national registry are probably at least partially more inclined to evidence-based management of PE and VTE as opposed to other treatment centers in the country. Similar to the previously cited article, for the same reasons, our results may not be fully replicable in all hospitals in the nation. 13 Since the analysis is done from registry, potential future research can aim to include different imaging parameters in potential diagnostic management studies and consequent follow-up of the patients.
Conclusion
Our results confirmed the predictive value of
Footnotes
Acknowledgments
We'd like to thank Dr Jovanova and Dr Al Baraqoni for their participation, and Dr Monreal for his intelectual support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
