Abstract
The purpose of the study was to assess whether the occurrence of restenosis is associated with CD45+ platelet count and neutrophil to lymphocyte ratio in patients with type 2 diabetes mellitus (DM) after drug-eluting stent (DES) implantation for stable coronary artery disease (CAD). The study comprised 126 patients, including 55 patients with type 2 DM and stable CAD who underwent elective coronary artery stenting with DES and follow-up angiography within 6 to 12 months. Blood samples were collected from each patient on the morning of the coronary angiography procedure. The variables related to in-stent restenosis were selected by logistic regression analysis. The logistic regression analysis showed that 2 inflammatory factors, CD45+ platelet count (odds ratio [OR] = 4.51, 95% confidence interval [CI]: 1.50-13.50, P = .007) and neutrophil to lymphocyte ratio (OR = 3.09, 95% CI: 1.05-9.10, P = .04), were significantly associated with the risk of in-stent restenosis after stenting with DES in patients with stable CAD and type 2 DM. A receiver operator characteristic curve analysis indicated that the area under the curve was 0.83% (0.05%; P < .001), which showed that the logistic model had good predictive accuracy (based on CD45+ platelet count and neutrophil to lymphocyte ratio) for the risk of in-stent restenosis development in DES in patients with CAD and type 2 DM. Two novel biomarkers of restenosis, CD45+ platelet count and neutrophil to lymphocyte ratio, may be effectively used to predict in-stent restenosis after DES implantation in patients with CAD and type 2 DM.
Keywords
Introduction
Restenosis continues to be a major obstacle to successful coronary artery stenting, and the ability to predict the likelihood of restenosis occurrence is poor. The attempt to define patients who benefit most from coronary artery stenting led to a search for biomarkers for restenosis development. When drug-eluting stents (DESs) were introduced in clinical practice, the mechanism for the development of in-stent restenosis changed. The DESs are designed in a way that acute inflammatory reactions to vessel wall injury and stent struts are attenuated by an antiproliferative drug coating. This results in delayed arterial wall recovery and the persistence of low-grade chronic inflammation at the place of injury. 1 Although the rate of restenosis decreased significantly after DESs were introduced, a small percentage of patients still develop in-stent restenosis during the 6- to 12-month follow-up period. Some studies even suggested a possibility of a late catch-up phenomenon, raising the question whether DESs may simply delay restenosis development. 2 –4 This prompted the need to discriminate patients with a higher risk of restenosis in DES, giving rise to a search for novel biomarkers. To date, many biomarkers of restenosis in DES are under investigation, but the data are controversial. Among suggested biomarkers are myeloperoxidase, monocyte chemotactic protein-1, and cardiotrophin-1. 5 Eosinophil cationic protein was identified as a significant predictor of restenosis in the first-generation DESs; they were coated with sirolimus or paclitaxel, which stimulated hypersensitivity and an allergic response. 6,7 Data on the most studied inflammatory biomarker, high-sensitive C-reactive protein (hs-CRP), are also controversial; in some studies, it failed to predict restenosis in DES, 8 –10 while it was significant in another study. 11 Recently, several novel biomarkers of restenosis were introduced. Among them are plasma phospholipids and sphingolipids 12 as well as processed B-type natriuretic peptide. 13 In our recent work, we revealed that CD45+ platelet count and neutrophil to lymphocyte ratio (NLR) predict the development of restenosis in DES. 14 In the present work, we further investigated the predictive ability of these biomarkers in patients with coronary artery disease (CAD) and type 2 diabetes mellitus (DM).
This study provides evidence that CD45+ platelet count and NLR are significant biomarkers for restenosis occurrence after DES implantation for stable CAD in patients with type 2 DM.
Materials and Methods
Study Patients
The study comprised 126 patients of both genders with stable CAD, including 55 patients with type 2 DM, consecutively admitted to the National Medical Research Centre of Cardiology, Moscow, Russia, between January 2012 and December 2015 for follow-up angiography, which was 6 to 12 months after successful elective percutaneous coronary angioplasty with DES. In all patients, follow-up angiography was conditioned by clinical symptoms. Exclusion criteria were as follows: age more than 75 or less than 40 years, unstable angina, myocardial infarction within 2 months prior to stenting, previous coronary artery bypass grafting or percutaneous coronary intervention, heart failure, abnormal liver or renal function, active infection, and inflammation. All patients provided written informed consent, and this study was approved by the ethics committee of the National Medical Research Centre of Cardiology.
All patients received DES implantation according to standard guidelines. Implanted stents were 18 to 33 mm in length and 2.25 to 3.5 mm in diameter. During the procedure, patients received a bolus injection of heparin. All patients received aspirin (100 mg/d) and clopidogrel (300 mg loading dose, followed by 75 mg/d for at least 12 months). No patients had major procedure-related complications. During the observation period, all patients received statins and, if required, antianginal and antihypertensive therapies. Patients’ demographic and clinical characteristics, as well as their coronary angiographic information, were collected carefully at baseline prior to percutaneous coronary intervention and follow-up angiography. In-stent restenosis was defined as a stenosis diameter of ≥50% occurring in the segment inside the stent or 5 mm proximal or distal to the stent at follow-up angiography. Type 2 DM was defined as either a previous diagnosis of DM treated with diet, oral agents, or insulin or a new diagnosis of DM if fasting blood glucose is ≥7.0 mmol/L on 2 occasions.
Laboratory Analysis
Blood samples were collected from each patient on the morning of the coronary angiography procedure. Blood was taken from the patients versus cubitalis in vials containing EDTA. Blood measurements, including neutrophils, monocytes, lymphocytes, basophils, eosinophils, and platelets, were performed on whole blood. Blood for laboratory analysis was centrifuged for 20 minutes at 2000g. The supernatant was stored at −38°C. Plasma levels of total cholesterol, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, triglyceride, glucose, creatinine, and hs-CRP were measured in all patients.
Identification of CD45+ Platelet
Patients’ blood was collected by cubital venipuncture and anticoagulated with 3.8% sodium citrate at an anticoagulant to blood ratio of 1:9. Platelet-rich plasma (PRP) was separated by centrifugation at 150g for 10 minutes at room temperature. The supernatant was aspirated and stored at room temperature no longer than 4 hours. Platelet-poor plasma was prepared by centrifugation of the blood at 2000 g for 15 minutes. We identified platelet-specific CD41 antigen glycoprotein IIb (GP IIb) in the GP IIb–IIIa complex and a leukocyte-specific CD45 antigen expressed by all leukocyte types using monoclonal antibodies conjugated with phycoerythrin (PE) and CY5-PE, respectively. Mouse-conjugated immunoglobulins of similar isotypes were used as a control. All antibodies were from “Becton Dickinson.” For the analysis of antigen expression, 100 µL of 2 mM EDTA in 0.1 M phosphate-buffered saline (rN 7.4) was added to 100 µL PRP, and the mixture was fixed with 200 µL of 2% paraformaldehyde in a phosphate buffer and stored at 4oS for no longer than 4 days. Antibodies (10 µL) were incubated with fixed PRP (5 µL) at room temperature for 40 minutes in the dark; then, a 500-µL phosphate buffer was added, and the samples were analyzed in an FACS Calibur flow cytofluorometer (Becton Dickinson).
Statistical Analysis
Values displaying normal distribution were expressed as the mean (standard deviation), and values with skewed distribution were expressed as the median (lower quartile; upper quartile). To check hypotheses related to the type of distribution, the Shapiro-Wilk W test was used. Patients were compared using a nonparametric 2-tailed Fisher exact test or Mann-Whitney U test for comparing 2 unmatched samples and a Kruskal-Wallis analysis of variance by ranks test for comparing 3 or more samples.
To construct the logistic model and subsequently determine the independent risk factors for stent restenosis, the essential variables derived from binary comparisons were assessed via logistic regression analysis. A receiver-operator characteristic (ROC) curve was constructed to determine the logistic model’s predictive value. Differences were considered to be statistically significant if the null hypothesis could be rejected with >95% confidence interval (CI). The statistical analysis was carried out using SPSS Statistics software version 17.0 (SPSS Inc.: Chicago, USA).
Results
The study comprised 126 patients with stable CAD in whom 217 DES have been implanted. Patients were divided into 2 groups according to the presence of type 2 DM. The first group comprised 55 patients with type 2 DM, and the second group consisted of 71 patients without type 2 DM. Clinical characteristics of patients of both groups at the time of stenting are presented in Table 1. The distribution of patients between the groups did not differ significantly in age, gender, or tobacco smoking status, as well as incidence of hyperlipidemia, arterial hypertension, or previous myocardial infarction. Restenosis occurred more frequently in patients with DM: 30 (54.5%) of restenosis cases in patients with type 2 DM versus 23 (32.4%) of restenosis cases in patients without type 2 DM (P = .01).
Baseline Clinical Characteristics.a
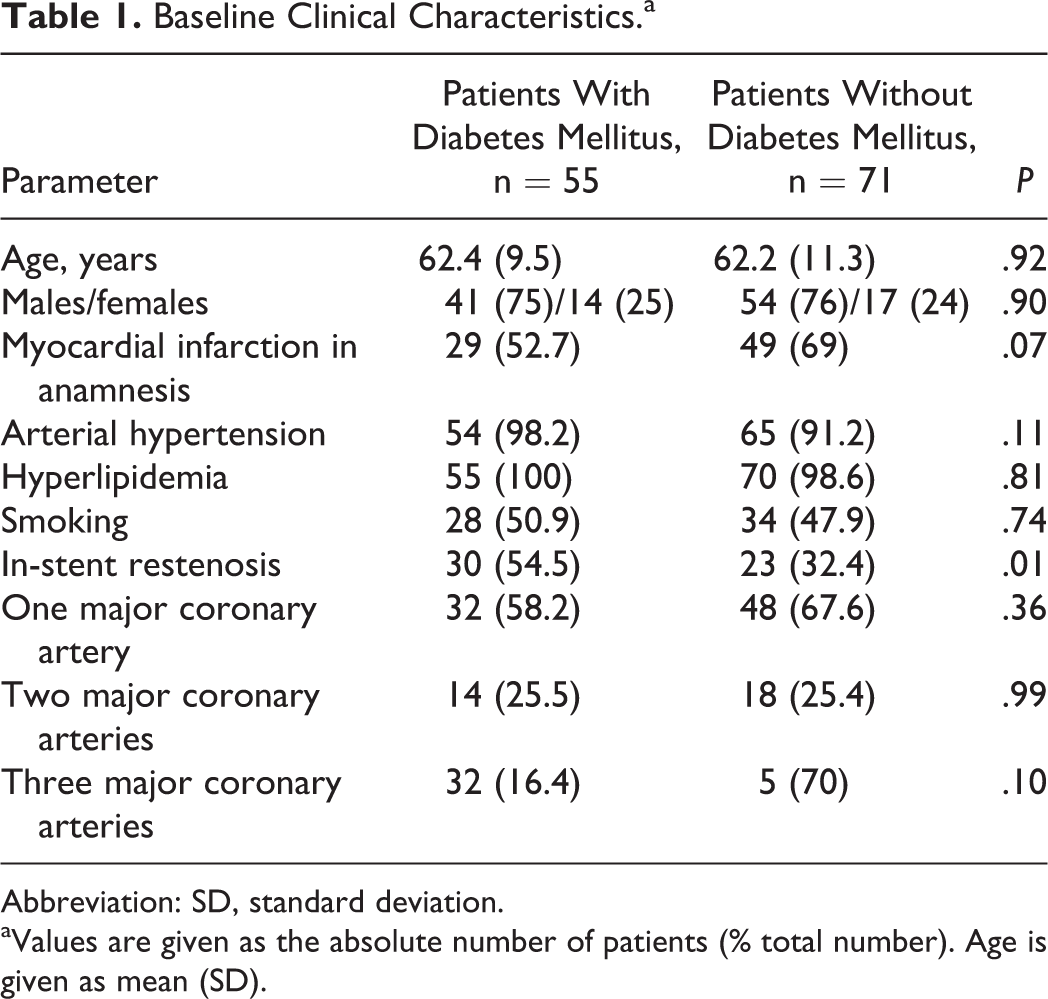
Abbreviation: SD, standard deviation.
aValues are given as the absolute number of patients (% total number). Age is given as mean (SD).
Angiographic characteristics of patients at the time of stenting are presented in Table 2. The average number of implanted stents, stenting of left anterior descending, circumflex, and right coronary arteries, as well as stenting of occlusions and diffuse lesions, did not differ between the groups. Patients with type 2 DM underwent stenting of arteries with small (≤2.75 mm) diameter DESs more frequently than patients without type 2 DM in 27 (49.1%) and 23 (32.4%) cases, respectively (P = .09).
Baseline Angiographic Characteristics.a
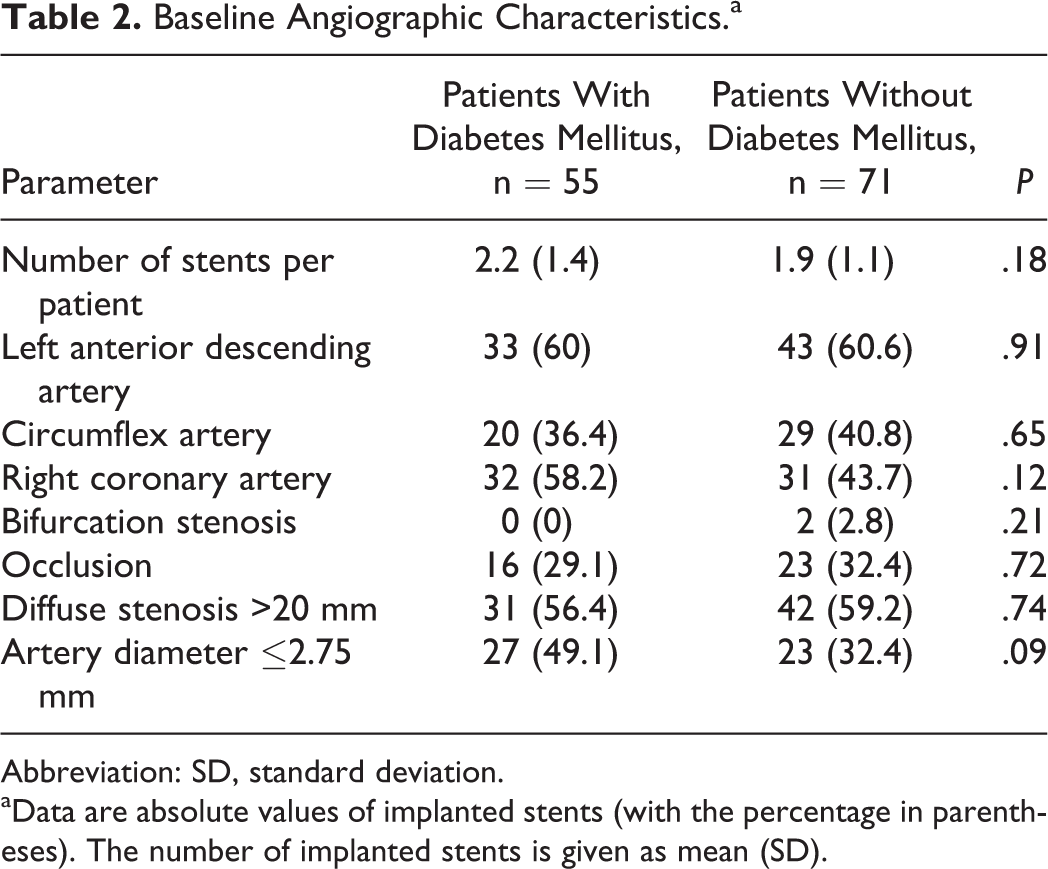
Abbreviation: SD, standard deviation.
aData are absolute values of implanted stents (with the percentage in parentheses). The number of implanted stents is given as mean (SD).
Risk Factors for Restenosis Development
We applied a binary logistic regression analysis to obtain the binary comparisons of more than 35 different clinical, laboratory, and angiographic variables and define significant risk factors for restenosis development. Five factors were identified as the most significant (see Table 3). In patients with type 2 DM, CD45+ platelet count (odds ratio [OR] = 4.51, 95% CI: 1.50-13.50, P = .007) and NLR (OR = 3.09, 95% CI: 1.05-9.10, P = .04) were significant. Angiographic factors were not significant in this group (Figure 1A). These factors were stenting of arteries with small diameter (<2.75 mm; OR = 1.08, 95% CI: 0.38-3.13, P = .88) and the number of simultaneously implanted stents (OR = 1.19, 95% CI: 0.80-1.79, P = .39).
Risk Factors for Restenosis Occurrence in Patients With Type 2 DM and CAD, Identified With Binary Logistic Regression Analysis.a
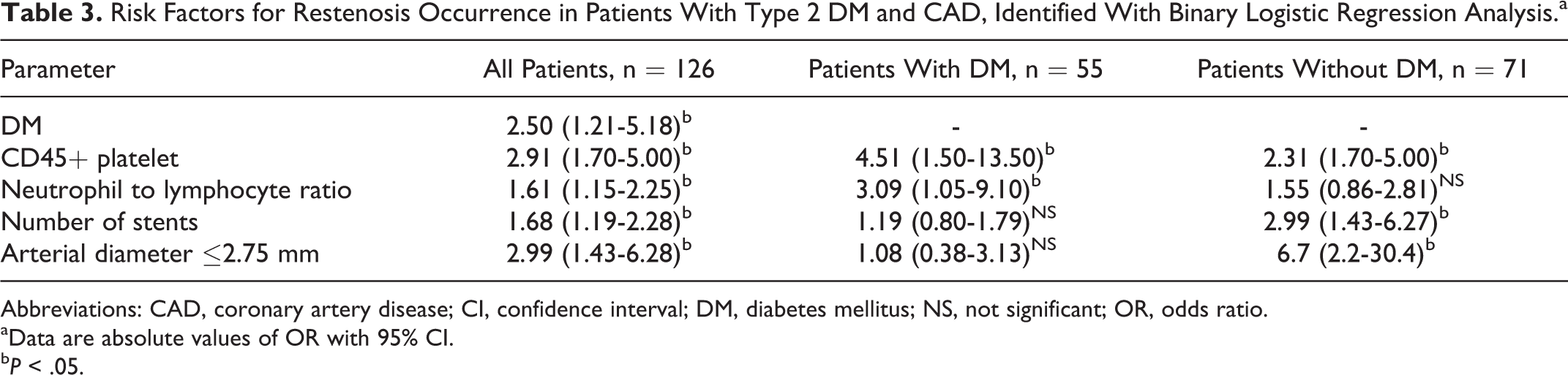
Abbreviations: CAD, coronary artery disease; CI, confidence interval; DM, diabetes mellitus; NS, not significant; OR, odds ratio.
aData are absolute values of OR with 95% CI.
bP < .05.
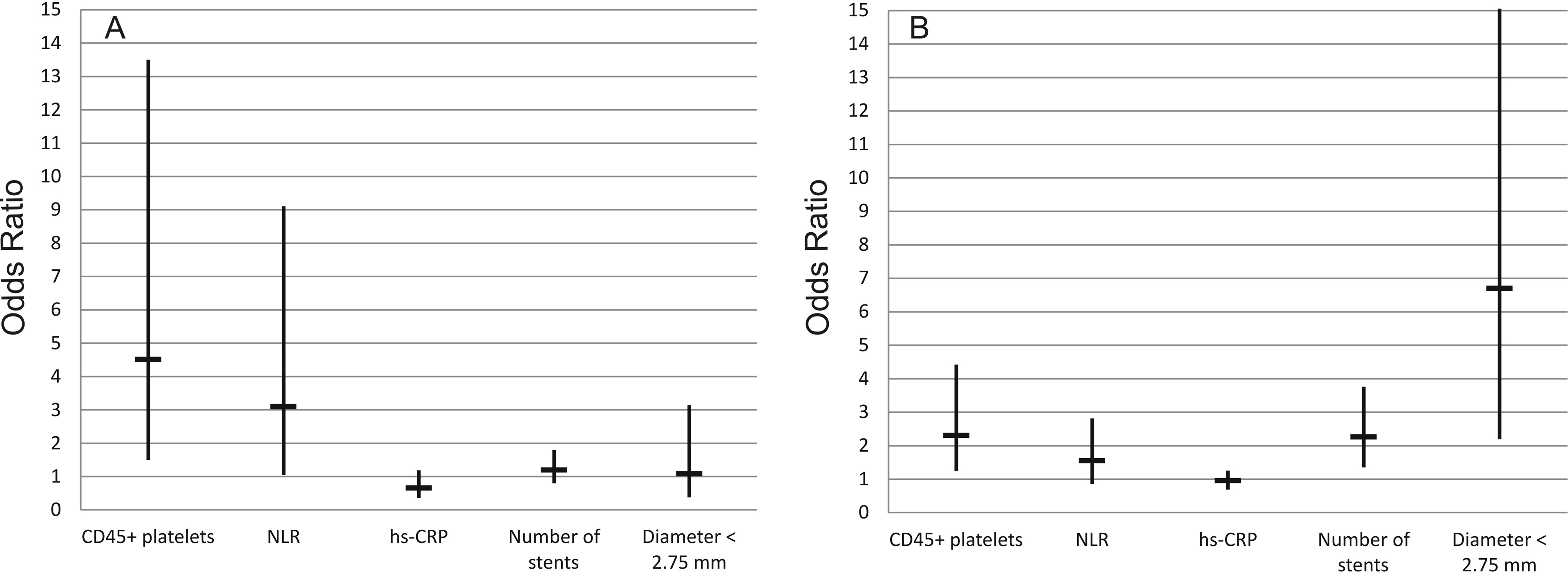
Risk variables for restenosis identified using binary logistic regression analysis. A, Patients with coronary artery disease and type 2 diabetes mellitus; B, patients with coronary artery disease but without type 2 diabetes mellitus. Data are absolute values of OR with 95% CI. CI indicates confidence interval; hs-CRP, high-sensitive C-reactive protein; NLR, neutrophil to lymphocyte ratio; OR, odds ratio.
The results were virtually the opposite in patients without type 2 DM. In this group, stenting of arteries with small diameter (OR = 6.7, 95% CI: 2.2-30.4, P = 0.001) and the number of implanted stents (OR = 2.99, 95% CI: 1.43-6.27, P = .002) were significant factors associated with restenosis. In the group without type 2 DM, inflammatory biomarkers’ significance was diminished (Figure 1B). CD45+ platelet count had OR = 2.31 (95% CI: 1.70-5.00, P = .007), and NLR was not significant (OR = 1.55, 95% CI: 0.86-2.81, P = .15). We found that the hs-CRP level was not a reliable predictor of restenosis in both groups: OR = 0.95 (95% CI: 0.69-1.25, P = .71) for the group with type 2 DM and OR = 0.65 (95% CI: 0.36-1.18, P = .45) for the group without type 2 DM.
We performed a multiple logistic regression analysis to construct the logistic model and identify independent predictors of restenosis development in patients with type 2 DM. The choice of the strongest combination of independent variables was based on the highest predictive potential and the highest percentage of findings, which were precisely predicted by the model. The logistic model includes 3 independent predictors: CD45+ platelet count, NLR, and type 2 DM. Other variables were excluded from the model because they were insignificant. The logistic regression equation for prognosis of restenosis in DESs in patients with CAD and type 2 DM was formulated as follows:
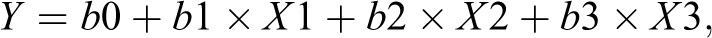
where X1 is content of CD45+ platelet, X2 is NLR, and X3 is type 2 DM. Coefficient values b0 to b3 of the logistic model and their ODs, 95% CI, and statistical significance of each coefficient are given in Table 4. The probability of restenosis development was calculated as follows:
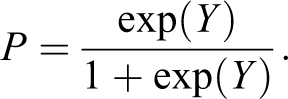
Independent Predictors of Stent Restenosis in the Logistic Equation.a

Abbreviations: CI, confidential interval; OR, odds ratio; SE, standard error.
aVariables: X1 represents CD45+ platelet, X2 represents neutrophil to lymphocyte ratio, and X3 represents type 2 diabetes mellitus.
The model possesses a high-efficiency index of predictive ability with 83% diagnostic precision. The model demonstrates a high-level OD, which equals 15.1 (P < .01). To determine the predictive power of the logistic model with the results of the logistic regression analysis of findings (n = 126), we built a ROC curve. The area under the curve was 0.83% (0.05%; P < .001). Thus, the power of the logistic model for predicting restenosis development in patients with CAD and type 2 DM should be considered very high.
Sample Size Calculation
We investigated 126 patients in which 53 cases of restenosis occurred and 3 variables were cogent predictors of restenosis in the logistic model. The number of events per predictive variable was 53/3 = 17.6 for the full sample. The logistic regression analysis demands at least 10 events for 1 variable to verify the statistical significance of a variable. 15 Thus, the sample size was sufficient to verify the significance of acquired variables.
Discussion
We found that CD45+ platelet counts are increased in the blood of patients with CAD and type 2 DM. The CD45 antigen has a leukocyte origin. Its appearance on platelets is strong evidence of interactions between leukocytes and platelets. However, the mechanisms through which these cells interact remain unknown. They could involve direct cell-to-cell contact or microparticle-mediated contact. In the latter case, microparticles transfer the membrane proteins of parent cells to the target cells. It has been proven that leukocytes generate microparticles. 16 More specifically, they shed microparticles under conditions of acute injury and inflammation. There are experimental data demonstrating that the interaction and binding of microparticles to target cells may be facilitated by adhesion receptors, 17 –19 and such interactions may affect the functional activity of cells. Microparticle-facilitated platelet–leukocyte interactions are well established. 19
Platelets with a CD45 leukocyte antigen appear in circulation probably due to the capture of leukocyte microparticles. They were recently detected in the blood of patients with myocardial infarction. 20 In our recent work, we showed that increased counts of platelets with CD45 leukocyte antigens are characteristic for patients with restenosis in DES. 14 In the present article, we demonstrate that patients with type 2 DM and in-stent restenosis in DES have higher counts of CD45+ platelet in circulation than patients with in-stent restenosis in DES but without type 2 DM (OR: 4.51 vs 2.31, respectively). Considering the opinion that chronic inflammation underlies restenosis development after DES, the increased CD45+ platelet count may reflect a higher level of platelet–leukocyte interactions. It is not clear whether this interaction is a result of local inflammatory activation of cells near the stent placement, or it just reflects the propensity for a more active inflammatory response. The possible corroboration of the latter statement comes from the fact that full recovery of the endothelium and neointima takes no more than several months, and our findings were made 6 to 12 months after the stents were implanted. This may imply that increased CD45+ platelet counts are due to increased inflammatory activity rather local cell activation near the stent.
Another finding is that elevated NLR may serve as an independent predictor of in-stent restenosis in DES in patients with type 2 DM. Elevated NLR is a characteristic of inflammation. It has emerged in recent years as an independent predictor of major adverse cardiovascular events and mortality, 21,22 as well as a predictor of in-stent restenosis in bare-metal stents. 23 –25 In our recent work, we have already reported elevated NLR in persons with restenosis in DES. 14 This was found in the general population. In the present work, we demonstrate that in patients with type 2 DM and in-stent restenosis in DES, NLR is elevated even more than in the general population with restenosis (OR: 3.09 vs 2.68, respectively).
In this study, hs-CRP was not a significant predictor of restenosis in DES in patients with type 2 DM. This finding is in line with the results of previous studies, where hs-CRP failed to predict restenosis in DES. 8 –10 There was no apparent methodological error. We suggest that there might be some other reason. Intriguingly, there are data showing that CRP has 2 isoforms. 26 It is synthesized in the liver under inflammatory conditions as a pentameric form, which is putatively inactive. The following conformational rearrangement in the area of local inflammation leads to the dissociation of pentameric CRP (pCRP) into 5 monomeric parts, which are not detectable by hs-CRP assay. These parts, named monomeric CRP (mCRP), are reportedly accountable for CRP’s pro-inflammatory activity. 26 There are data demonstrating that mCRP but not pCRP can activate the endothelium 27,28 in vitro. It has also been found in vivo on circulating microparticles in persons with acute myocardial infarction. 29 Also, mCRP deposits were described in autopsy samples of carotid atheroslerotic plaques, without pCRP traces in the same plaques. 30 These novel data may provide the answer to discrepancies in results in hs-CRP measurements in patients with cardiovascular disease.
We shall also consider the fact that angiographic characteristics were not significant as predictors of restenosis in DES in patients with type 2 DM. It could be said that in patients with DES implantation and a strong pro-inflammatory condition such as DM, the inflammatory biomarkers should be preferred over noninflammatory. It is worth mentioning that in patients without type 2 DM, noninflammatory characteristics were the best predictors of restenosis in DES. In this group, stenting of small diameter arteries and the number of implanted stents provides impressive 6.7 and 2.99 OR, while inflammatory characteristics lose significance (Figure 1).
The results of this study make it possible to suggest that the further search for biomarkers of restenosis in DES could benefit from directing efforts into studying different biomarkers for specific conditions. Thus, we may strengthen the inherent predictive power of biomarkers by choosing inflammatory biomarkers to predict in-stent restenosis in patients with pro-inflammatory conditions such as type 2 DM or noninflammatory biomarkers to predict restenosis in patients without the propensity to develop chronic inflammation.
Conclusions
Two novel biomarkers of restenosis, CD45+ platelet count and NLR, may be effectively used to predict in-stent restenosis after DES implantation in patients with CAD and type 2 DM.
Study Limitations
This is a retrospective study. Coronary angiography was not routinely performed in every patient after stent implantation. Follow-up coronary angiography was performed predominantly in patients presenting with recurrent angina pectoris. Many factors are associated with restenosis development after coronary stenting, so it cannot be excluded that additional factors which have not been assessed in our study could have a similar influence on restenosis development. We do not know the baseline CD45+ platelet levels in patients who participated in this investigation, whether the patients with restenosis had changes in the levels of CD45+ platelet at baseline or whether this difference is a consequence of stent implantation.
Footnotes
Authors' Note
I strongly declare that Brothers of Mercy Hospital provided all neccesary support for this paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Russian Science Foundation (project #16-15-10098).
