Abstract
Background:
The incidence of neonatal hypoxic–ischemic encephalopathy (HIE) is reportedly high in countries with limited resources. Its pathogenesis is multifactorial. A role for thrombophilia has been described in different patterns of preterm and full-term perinatal brain injury.
Aim:
This study aims to identify risk factors associated with neonatal HIE and also to determine the contributions of genetic thrombophilia in the development of neonatal HIE.
Methods:
Sixty-seven neonates with HIE and 67 controls were enrolled in the study. Clinical history and examination were undertaken. Patients and controls were tested for the presence of factor V G1691A and prothrombin G20210A mutations. In addition, protein S, protein C, and antithrombin III levels were assessed.
Results:
Parental consanguinity and performing emergency cesarean section (CS) were significant risk factors for neonatal HIE (odds ratio [OR] 6.5, 95% confidence interval [CI] 2.6-15.3, P < .001, OR 12.6, 95% CI 2.52-63.3, P = .002, respectively). No significant difference was found regarding maternal age and parity. About 33% of cases and 6% of controls were found to have at least 1 thrombophilic factor (P < .001). Factor V G1691A mutation significantly increased the risk of neonatal HIE (OR 4.5, 95% CI 1.4-14.5, P = .012), while prothrombin G 20210A mutation and protein C deficiency were not.
Conclusion:
Parental consanguinity, emergency CS, and factor V mutation may contribute to the higher risk of developing neonatal HIE.
Introduction
Neonatal encephalopathy (NE) is a serious condition occurring in 1 to 2 of every 1000 live term babies. 1 Neonatal encephalopathy was defined as a clinical picture, occurring during the first 72 hours after birth, consisting of one or more of the following symptoms: altered consciousness, irritability, abnormal tone pattern, convulsions, or lethargy. Neonatal encephalopathy was considered to be secondary to perinatal asphyxia on the basis of at least 2 of the following criteria: (1) late decelerations on fetal monitoring or meconium-stained liquor, (2) delayed onset of respiration, (3) 5-minute Apgar score <7, (4) arterial umbilical cord pH <7.10, and (5) multiorgan failure. 2
It is mostly encountered following perinatal hypoxia–ischemia (HI). 3 The incidence of neonatal hypoxic–ischemic encephalopathy (HIE) is reportedly high in countries with limited resources; however, precise figures are not available. Birth asphyxia is the cause of 23% of all neonatal deaths worldwide. It is one of the top 20 leading causes of burden of disease in all age-groups by the World Health Organization and is the fifth largest cause of death in children younger than 5 years. Although data are limited, birth asphyxia is estimated to account for 920 000 neonatal deaths every year and is associated with another 1.1 million intrapartum stillbirths. More than a million children who survive birth asphyxia develop problems such as cerebral palsy, mental retardation, learning difficulties, and other disabilities. 4,5
There are multifactorial risk factors for neonatal HIE including prepregnancy factors (socioeconomic status, family history of seizures or neurological disease, maternal thyroid disease or obesity, and fertility treatment), antenatal factors (bleeding, hypertensive disease, infection, and coagulation disorders), fetal factors (multiple pregnancy, chromosomal or congenital anomalies, fetal growth restriction, prematurity, coagulation disorders, postmaturity, malpresentation, and male sex), and intrapartum factors (infection, bleeding, uterine rupture, cord accident, prolonged labor, oxytocin induction, meconium stained liquor, and fetal heart rate deceleration). 6 –8
Thrombophilia is not a disease by itself, it means systemic predisposition to thrombosis. 9 The role of genetic and acquired thrombophilias in the pathogenesis of perinatal arterial stroke is quite controversial and not completely understood. 10 –12
The role of congenital thrombophilic states such as activated protein C resistance 13 in the majority of cases due to the factor V (FV) G1691A gene mutation 14,15 ; the 20210A allele within the 3-untranslated region of the prothrombin gene 16 ; antithrombin III, protein C, or protein S deficiencies 17 ; and an increased lipoprotein level have been discussed as common risk factors for venous thrombosis in children and adolescents. 18 A role for thrombophilia has been described in different patterns of preterm and full-term perinatal brain injury such as perinatal arterial ischemic stroke, intraventricular hemorrhage, and HI injuries. 19 –21 Previous studies have shown an association of thrombophilia in mothers of neonates with neonatal brain damage. 19,22 The pathogenesis of brain injury after HI is complex. Recent research has focused on several mechanisms, including excitotoxicity, oxidative stress, inflammation, and coagulation. There has been increasing interest in the contribution of genetic factors interfering with these mechanisms. 23 Previous studies have focused on genes related to inflammatory or coagulation pathways. 24
Although neonatal HIE is not uncommon, much remains to be learned about its risk factors on which depend prevention or treatment strategies.
Aim of the Study
This study aimed to identify maternal, intrapartum, neonatal factors associated with neonatal HIE, and to determine the contribution of genetic thrombophilia in the development of neonatal HIE.
Patients and Methods
A case–control study was conducted at the Neonatology unit of Assiut University Children Hospital during the period from December 2012 to November 2013. Sixty seven neonates diagnosed with HIE according to Cowan et al, 2 confirmed by brain computed tomography (CT) and or magnetic resonance imaging (MRI), were enrolled in the study.
Inclusion Criteria
Full-term neonates who were delivered in Woman Health Center of Assiut University Hospital with clinical diagnosis of HIE.
Exclusion Criteria
Preterm infants (less than 37 weeks of gestation) or those affected by stroke or encephalopathy associated with arterial catheterization, surgery, metabolic disorders, or congenital heart disease.
Control Group
Sixty-seven healthy neonates from the same geographic area served as control group. Controls were recruited from vaccination clinic, where full-term neonates were vaccinated with BCG vaccine and zero dose of polio vaccine, controls were allocated into males and females then a comparable number with cases was selected randomly.
Ethics
The present study was performed in accordance with the ethical standards laid down in the updated Declaration of Helsinki and approved by the medical ethics committee of Faculty of Medicine, Assiut University. Informed consent was taken from the parents of patients and controls.
Methods
For patients and controls, full clinical history was taken, and complete physical examination was done. The newborn was considered to be small for gestational age (SGA) when his birth weight is less than 10th percentile. 25 Laboratory work-up including complete blood count, prothrombin time (PT), partial thromboplastin time (PTT), thrombophilic factors assay including factor V (FV), factor II mutations and protein S, protein C, and antithrombin III levels. Neuroimaging and blood gas analysis were done only for patients.
Blood Sampling
Venous blood of 4 mL was withdrawn from each individual participating in the study. The blood sample was taken from patients at days 2 to 3 of life, also sample taken from controls who presented to vaccination clinic within first week of life. The samples were divided as follows: 1.8 mL were mixed with 0.2 mL trisodium citrate in a screw capped plastic tube and were centrifuged within 30 minutes of blood sampling in a cooling centrifuge for 10 minutes at 3000 rpm. Prothrombin and activated PTT were measured immediately, the rest of the separated plasma was divided into aliquots and stored at −80° C (for not more than 2 months) for protein C, protein S, and antithrombin III assay, which was performed using the automated coagulameter Sysmex CA 1500 (Siemens GmbH, Germany) according to the manufacturer’s instructions. Protein C and antithrombin deficiency was diagnosed when the functional plasma activity and immunological antigen concentration of a protein were below the lower age-related limit (protein C, 20% and antithrombin, 30%). 26 The diagnosis of protein S deficiency was based on reduced free protein S antigen level below 30% combined with a decreased or normal total protein S antigen concentrations. 26 Factor deficiency was only considered after a confirmatory second blood sample.
DNA Extraction
Venous blood of 2 mL was transferred into EDTA tube (separation tube for DNA; Zhejiang Kangshi Medical Devices Co, Ltd, Hangzhou, China). Genomic DNA was extracted from EDTA-anticoagulated blood samples using QIAamp DNA Mini Blood kit (Qiagen GmbH, Cat. No. 29104; Hilden, Germany).
Analysis of FV G1691A
FV-G1691A was assessed by polymerase chain reaction (PCR). 15 A typical PCR mixture comprised genomic DNA (50-200 ng), MgCl2 (1.5 mmol/L), sense and antisense oligonucleotide primers (20 pmol, Midland, Texas), deoxynucleotide mixture 0.2 mmol/L concentration per each deoxynucleotide (Promega, Madison, WI, USA), and Taq DNA polymerase (2.5 U/reaction; Promega). 5′-GGA ACA ACA CCA TGA TCA GAG CA-3′ and 5′-TAG CCA GGA GAC CTA ACA TGT TC-3′ are the primer sequences for FVL according to what was previously described. 15 After final extension at 72° C for 10 minutes, the amplification product for FV G1691A was digested with MnlI restriction enzyme (New England Biolabs, Ipswich, MA, USA). The electrophoretic separated fragments were visualized by gel electrophoresis (2% agarose gel containing 100 pg/mL ethidium bromide) applying PCR marker provided by (Promega, Cat No G3161) under a UV lamp. Normal alleles with G/G at nucleotide 1691 yielded the following 3 fragments: 37, 93, and 157 bp. As FVL mutation abolishes the MnlI restriction site, a homozygous A/A yields only 130 and 157 bp fragments. In heterozygous G/A, all 37, 93, 130, and 157 bp fragments are produced.
Analysis of Prothrombin G20210A Mutation
Similarly, prothrombin G20210A mutation was assessed by PCR. 16 The PCR conditions were similar to those utilized for FV-G1691A analysis. The PCR products were digested by HindIII (Invitrogen Cat No 15207-012). 5′-CAAGCCTGATGAAGGGAAAC-3′ and 5′-ATAGCACTGGGAGCATTGC-3′ are the primer sequences for prothrombin G20210A according to what was previously described. 16 The normal sequence of the prothrombin gene displayed a band of 345 bp after HindIII digestion. The mutant factor II gene heterozygous genotype is characterized by 2 fragments of 345 and 322 bp; the absence of the fragment of 345 bp and the presence of only the fragment of 322 bp are distinctive of the homozygous genotype.
Statistical Analysis
Comparison of risk factors in patients and controls was calculated by Chi-square test or, where relevant, by Fischer exact test. In addition, odds ratios (ORs) and 95% confidence intervals (CIs) were calculated. A 2-tailed P < .05 was considered statistically significant. The effect of the variables, which showed a significant difference in the univariate analysis, was assessed using a multivariate logistic regression model. T test was used to compare mother’s age of patients and controls, while Mann-Whitney U test was used to compare nonparametric measures (proteins C, S, and antithrombin III). All statistical analyses were performed using the SPSS software program (version 16).
Results
Of 637 full-term neonates admitted to intensive care unit during the research period, 67 (10.5%) full-term neonates had a confirmed diagnosis of HIE. Forty-five (67.2%) patients were males. Antenatal and/or intrapartum fetal heart rate deceleration was detected in 23 (34%) cases. Failure to achieve spontaneous breathing and resuscitation with positive pressure ventilation was needed in 34 (50.7%) neonates. Low Apgar score (<7 at 5 minutes) was recorded in 26 (38.8%) neonates. Metabolic acidosis (pH < 7.1 within 60 minutes after birth) was detected in 37 (55%) neonates. The clinical presentations of the studied patients were as follows: Altered consciousness was detected in 15 (22.4%) patients, irritability in 31 (46.3%) patients, lethargy in 26 (38.8%) patients, convulsions in 33 (49.3%) patients, hypertonia in 20 (29.9%) patients, and hypotonia in 17 (25.4%) patients. CT brain and/or MRI revealed evidence of HI injury in all patients (except for 5 [7%] patients, who died before doing CT brain). Brain edema was detected in 48 (77.4%) patients, and cerebral hypodensities were detected in 14 (22.5%) patients. Of 67 patients with HIE, 18 (26.9%) died during hospital admission. Main leading causes of death were respiratory failure (12 cases), renal shutdown (2 cases), and shock mostly due to adrenal failure (4 cases).
Table 1 shows maternal, intrapartum, and neonatal risk factors in patients and controls. There was no significant difference between patients and controls regarding maternal age and parity. Two (3%) patients’ mothers had rheumatic heart disease, 3 (4.4%) had diabetes mellitus, 2 (3%) showed hypertension before pregnancy, and 2 (3%) had antepartum hemorrhage. History of previous abortion was reported in 29.9% and 25.4% of patients’ and controls’ mothers, respectively, with no significant difference between them. There was insignificant increase in the number of mothers of patients with preeclampsia than mothers of controls. Receiving antenatal care was insignificantly lower in patients’ than controls’ mothers (88.1% vs 92.5%). Parental consanguinity and performing emergency cesarean section (CS) were detected as a significant risk factors for neonatal HIE (OR 8.1, 95% CI 3.7-17.7, P < .001) and (OR 11.78, 95% CI 2.6-53.34, P < .001), respectively. Prolonged second stage of labor, perinatal sepsis, cord accident, fetal heart rate deceleration, and malpresentation were reported in cases only, and their rates were 16%, 9%, 3%, 34%, and 18%, respectively. Accurate data were not available for controls. Four patients and 1 controls were twin sets. There was no significant difference between patients and controls regarding birth order of the newborn. Being SGA was a significant risk factor for neonatal HIE (OR 5.7, 95% CI 1.2-27.1, P = .03). Positive family history of similar condition was reported in 6% of patients and not in controls.
Maternal, Intrapartum, and Neonatal Risk Factors for Neonatal HIE.
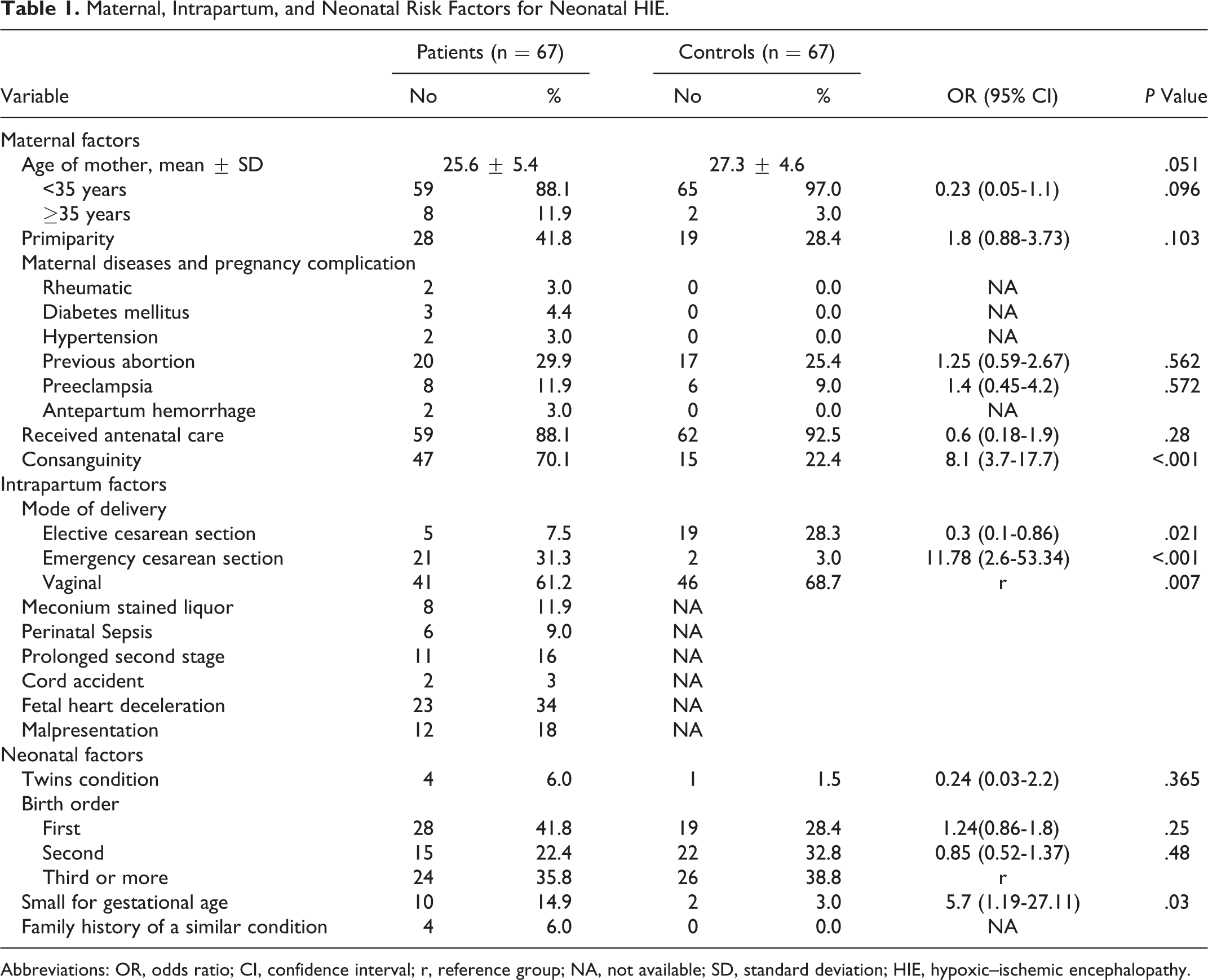
Abbreviations: OR, odds ratio; CI, confidence interval; r, reference group; NA, not available; SD, standard deviation; HIE, hypoxic–ischemic encephalopathy.
The means of protein C% and protein S% were significantly lower in patients than in controls (65.28 ± 32.8 vs 87.04 ± 23.14 and 75.5 ± 19 vs 85 ± 14.4, respectively) with P < .001 for each. Thrombophilic risk factors in patients and controls are shown in Table 2. 2 Twenty-two patients (32.8%) have at least 1 thrombophilic factor compared to 4 (6%) controls (OR 7.7; 95% CI 2.5-23.9, P < .001). Factor V G1691A mutation significantly increased the risk of neonatal HIE (OR 4.5, 95% CI 1.4-14.5, P = .012). Eleven (16.4%) patients and 3 (4.4%) controls were homozygous for FV mutation, and parental consanguinity was reported in 3 patients and 1 control of those homozygous for FV mutation. The percentage of prothrombin G 20210A mutation was not statistically significantly higher in patients than controls (8.9% vs 2.9%, P = .274). Five (7.4%) patients and 1 (1.4%) control were homozygous for factor II mutation, and parental consanguinity was reported in 2 (2.9%) patients. Eight patients and 2 controls (11.9% and 3.0%, respectively) have protein C deficiency, with insignificant difference between them. Neither patients nor controls had protein S or antithrombin III deficiency. Table 3 shows that multivariate logistic analysis clarifies that performing emergency CS, consanguinity and genetic thrombophilia were independent significant risk factors for neonatal HIE. However, being SGA was not persistently significant factor.
Univariate Analysis of Thrombophilic Risk Factors in Patients With Neonatal HIE.
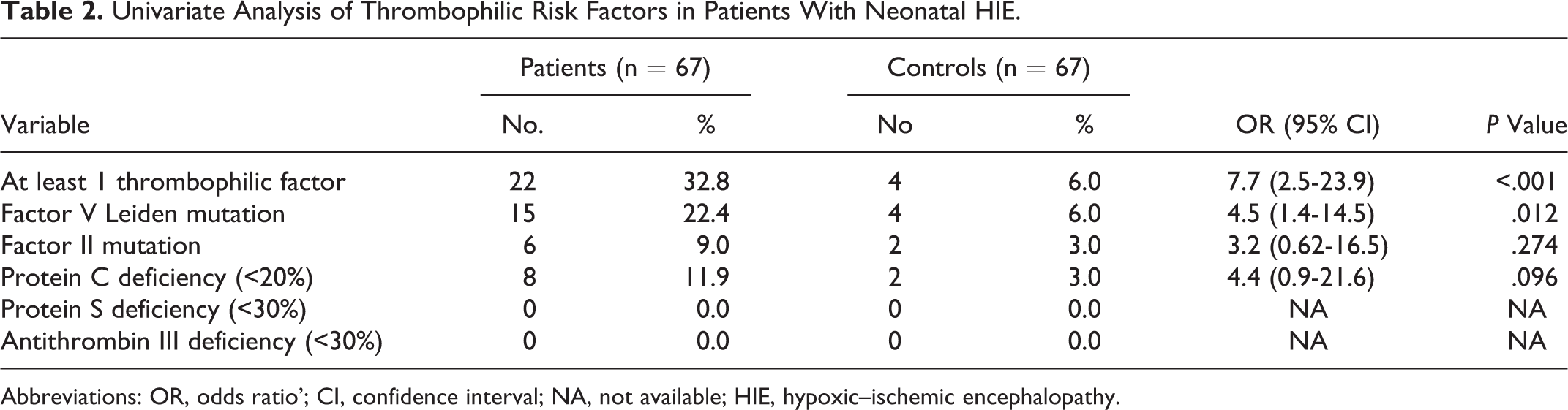
Abbreviations: OR, odds ratio’; CI, confidence interval; NA, not available; HIE, hypoxic–ischemic encephalopathy.
Risk Factors of Neonatal HIE by Multivariate Logistic Regression.
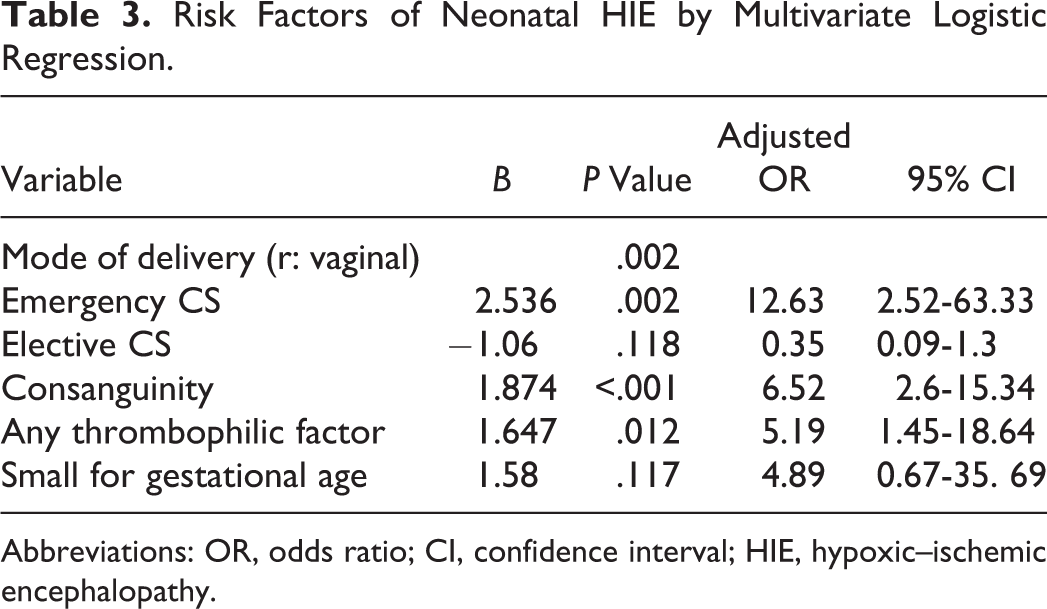
Abbreviations: OR, odds ratio; CI, confidence interval; HIE, hypoxic–ischemic encephalopathy.
Discussion
In this study, 67 neonates diagnosed with HIE underwent evaluation for their maternal, intrapartum, and neonatal factors together with their levels and mutations of some thrombophilic factors as risks for developing HIE. In agreement with Butt et al, 27 no significant difference was detected between patients and controls regarding maternal age and parity. On the contrary, Martinez et al 28 reported that primiparity and maternal age ≤ 20 are significant risks for neonatal HIE and Berglund et al 29 mentioned that maternal age ≥ 30 is associated with that risk. Rheumatic heart disease and diabetes mellitus were reported in patients’ mothers (3% and 4.4% respectively). Rheumatic heart disease may have a role in occurrence of HIE through maternal collapse. 6 Diabetes mellitus may increase the risk of HIE by increasing risk of macrosomia, hypoglycemia, hypocalcemia, respiratory distress syndrome, polycythemia, hyperbilirubinemia, and cardiomyopathy. 30 In agreement with Martinez et al, 28 there was a insignificantly higher percentage of previous abortion in patients’ mothers than controls’. Also, preeclampsia was insignificantly higher in patients’ mothers than controls’, this was comparable with the results of Curry et al. 31 Endothelial disruption associated with preeclampsia could function as prothrombotic stimulus. 32 Going with Ellis et al, 33 maternal antepartum bleeding was reported in 3% of patients’ mothers.
Antenatal care aims to screen women to detect risk factors for adverse outcomes and to minimize their effects. 34 In the present study, there was no significant difference between patients’ mothers and controls’ as most of them received antenatal care. Butt et al 27 reported that absence of antenatal care was a significant risk factor for HIE.
Consanguinity was found to be a statistically significant risk factor for neonatal HIE (OR 6.52, 95% CI 2.6-15.34, P < .001). This could be explained by possibility of underlying genetic susceptibility. 35,36 Strømme et al 37 found an 11-fold increased risk of progressive encephalopathy in consanguineous Pakistanis.
Regarding the mode of delivery, in univariate analysis, emergency CS was a significant risk factor for neonatal HIE (OR 11.78, 95% CI 2.6-53.34, P < .001), while elective CS was found to be significantly protector factor for HIE (OR 0.3, 95% CI 0.1-0.86, P = .021). However, the multivariate analysis showed that elective CS was not persistently significant. Daniel et al 38 and Elvedi et al 39 reported that incidence of birth asphyxia was significantly higher in emergency CS than elective CS mostly because of the indications for emergency CS as fetal distress, failure of progress of normal labor, and cephalopelvic disproportion which carry higher risk on the neonates. 40
Prolonged second stage of labor, 27 cord accident, 7,40 malpresentation, 8,40 fetal heart rate deceleration, 6,40,41 meconium stained liquor, 7,8,40 and perinatal infection 8,31,41 were found to be significant risk factors for neonatal HIE. In our study, these factors were reported in the patients only, as accurate data were not available for controls.
In our study, twinning was not a significant risk factor for HIE, keeping with Curry et al. 31 On the contrary, Martinez et al 28 found that twin gestation was significantly associated with higher risk of HIE. Also, birth order was not a significant risk for HIE; however, Milsom et al 40 observed more neonatal asphyxia with the first baby.
In this study, being SGA was a significantly risk factor in univariate analysis (OR 5.7, 95% CI 1.2-27.1, P = .03) but it was not persistently significant in multivariate analysis. Previous studies reported that SGA baby is more at risk of HI injury. 34,41 A family history of similar condition was positive in 6% of our studied patients, Okereafor et al 7 considered family history of convulsions as a risk factor for HIE.
A role of thrombophilia has been described in different patterns of preterm and full-term perinatal brain injury such as perinatal arterial ischemic stroke, intraventricular hemorrhage, and HIE. 19 –21 Mutations of FV G1691A and prothrombin gene reported to be associated with hypercoagulable state. 15,16,42,43 Gurgey et al 44 reported that congenital prothrombotic factors (FV and factor II mutations) showed a trend toward a higher frequency in neonates with thrombosis. In this study, patients and controls underwent thrombophilic factors assay including FV and factor II mutation and protein S, protein C, and antithrombin III deficiencies. About 33% of cases were found to have at least 1 thrombophilic factor compared to 6% of controls (OR 5.2, 95% CI 1.4-18.6, P = .012). Other researchers found at least 1 thrombophilic factor in higher percentages (more than 60%) of infants 10,22 or children 45 with stroke. These higher percentages could be due to the larger number of factors they searched for. Similarly, Curry et al 31 studied the prothrombotic factors in infants with perinatal arterial stroke and their mothers and found that 68% had at least one abnormality in mother, infant, or both. The current study revealed that FV G1691A mutation significantly increased the risk of neonatal HIE (OR 4.5, 95% CI 1.4-14.5, P = .012), since it was detected in 22.4% of patients compared to 6% of controls. Of these patients, 11 (16.4%) patients and 3 (4.4%) controls were homozygous, and parental consanguinity was reported in 3 patients and 1 control of those homozygous for FV mutation which is not a significant percentage. Prothrombin G 20210A was insignificantly higher in patients than controls. Similar findings were found in infants 10,22 or children 45 with stroke. Gawish 46 reported that mutation in factor II and FV might be important risk factors for stroke in Saudi neonates. In the contrary, Akman et al 47 found normal values of prothrombotic factors in full-term neonates with cerebral infarcts. Simchen et al 22 recorded protein C deficiency as a significant risk factor for perinatal arterial stroke (relative risk 12.2, 95% CI 2.5-59.9). This study revealed higher frequency of protein C deficiency in patients than controls but the difference was not significant, this is in agreement with the results of Kenet et al. 48 Gunther et al 10 found protein C deficiency only in full-term neonates with stroke but not in controls, while Lynch et al 45 reported protein C deficiency in children with stroke, just in one case out of 59 cases.
In our study, neither patients nor controls had protein S or antithrombin III deficiency, this is in agreement with Akman et al 47 and Gunther et al 10 who found similar results. On the other hand, Simchen et al 22 recorded protein S deficiency in infants with perinatal stroke and not in controls. At the same time, antithrombin III deficiency was not recorded in their studied groups. 22 Lynch et al 45 recorded antithrombin III deficiency in children with stroke, only in one case out of 59 cases.
Conclusion
In conclusion, indications for emergency CS, consanguinity, and FV mutation may contribute to the higher risk of developing neonatal HIE.
Limitations of this Study
In addition to selection bias of the case–control design, the relatively small number of cases and controls limited the ability to estimate accurately the magnitude of the problem of genetic thrombophilia and its burden as risk for neonatal HIE. There was some missing data about intrapartum risk factors in the control group, and the inability to assess thrombophilic factors in mothers of neonates was also another limitation of the study.
Recommendations
We recommend: - a screening for thrombophilia should be done in all neonates with HIE. Identify patients with thrombophilic factors could be useful in order to administer antithrombotic prophylaxis in the presence of risk factors; - to have long follow up studies for HIE neonates, specially those proved to have thrombophilic abnormalities to study the long-term impact of thrombophilia on their outcome; and - to have further studies to evaluate the impact of acquired thrombophilia on the risk of HIE.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
