Abstract
The beneficial effect of autologous peripheral blood stem cell transplantation (APBSCT) may be compromised by acute vascular complications related to hypercoagulability. We studied the impact of graft product on thrombin generation of normal plasma and the expression of tissue factor (TF) and procoagulant platelet-derived procoagulant microparticles (Pd-MPs) in samples of graft products. Graft products from 10 patients eligible for APBSCT were mixed with platelet-poor plasma (PPP) or platelet-rich plasma (PRP) from healthy volunteers and assessed for in vitro thrombin generation. In control experiments, thrombin generation was assessed in (1) PPP and PRP without any exogenous TF and/or procoagulant phospholipids, (2) PPP with the addition of TF (5 pM) and procoagulant phospholipids (4 μM), (3) in PRP with the addition of TF (5 pM). Graft products were assessed with Western blot assay for TF expression, with a specific clotting assay for TF activity and with flow cytometry assay for Pd-MPs. The graft product enhanced thrombin generation and its procoagulant activity was related to the presence of Pd-MPs and TF. The concentration of Pd-MPs in the graft product was characterized by a significant interindividual variability. The present study reveals the need for a thorough quality control of the graft products regarding their procoagulant potential.
Introduction
Autologous peripheral blood stem cell transplantation (APBSCT) is among the treatments of choice in patients with malignancies who undergo chemotherapy. The beneficial effect of APBSCT risks to be compromised by acute vascular complications related to hypercoagulability. 1 –4 In patients receiving APBSCT, the presence of hypercoagulable state has been documented. 5,6 The graft product consists of white blood cells in a vast majority—a few percentages of which are CD34+ hematopoietic stem cells (HSCs)—macromolecule solution, and remaining cryopreservative agents. However, the mechanism by which the graft product contributes to blood hypercoagulability is undefined.
Whether hypercoagulability related to APBSCT is the result of direct activation of hosts’ blood coagulation by the transfused material or is the consequence of cellular reactions resulting in an inflammatory response of the host is a matter of research. The potential transient activation of host blood coagulation by the cryopreservative agent dimethyl sulfoxide (DMSO) has been recently proposed. 7 The cellular components of the graft product could also possess inherent procoagulant properties contributing to blood hypercoagulability of host patients. However, this issue has been inadequately explored.
Thrombin generation, assessed with clotting-based assays or chromogenic substrates, is an established tool in blood coagulation research. 8 –13 Calibrated automated thrombogram (CAT) is the most recent tool for thrombin generation assessment. This assay, using a fluorogenic substrate, allows to study thrombin generation after triggering coagulation in plasma in the presence of cells and while fibrin formation occurs. Thrombogram is informative for the initiation, propagation, and inhibition of thrombin generation. 14 Hence, the assessment of thrombin generation in human plasma could contribute to the understanding of the interplay between cellular components and blood coagulation.
The present study was designed to investigate the inherent procoagulant properties of the graft product. To this aim, normal plasma from healthy volunteers was supplemented with graft product designated to be given to patient eligible for APBSCT. Thrombin generation in the presence of blood product was compared with that induced by physiologically relevant concentrations of tissue factor (TF) and procoagulant phospholipids (PL). We also assessed the expression of TF and the TF activity (TFa) as well as the presence of procoagulant platelet-derived microparticles (Pd-MPs) and erythrocyte-derived microparticles (Ed-MPs) in the samples of graft product.
Patients and Methods
Autologous Peripheral Blood Stem Cells
Samples of graft product were obtained from 10 patients with ovarian cancer, eligible for autologous transplantation. All patients provided written informed consent. The study protocol was in conformity with the Declaration of Helsinki and was performed after approval by the local institutional ethics committee.
Autologous peripheral blood stem cells were harvested by apheresis after the mobilization of HSCs following a cycle of chemotherapy and growth factor (granulocyte colony-stimulating factor [G-CSF]) administration (5 µg/kg/d for 7 to 12 days). Apheresis was initiated as soon as the peripheral blood CD34+ cell count reached 10 to 20/mL. The target for stem cell collection was to obtain at least 3 × 106 CD34+ cells/kg of recipient body weight. After standard quality controls, leukapheresis products were preserved in a cryopreservation solution (Voluven/10% DMSO) and kept frozen in vapor nitrogen at −160°C until transplantation. On the transplantation day, the bags were thawed in a water bath at 37°C, washed with a washing solution (0.2% glucose, 0.9% NaCl, and 10% anticoagulant citrate dextrose [ACD]), and finally suspended in a solution of 5% glucose, and then the material was transfused to the patient. The residual material in the bag was used in order to carry out the in vitro experiments.
Normal human plasma
Plasma samples for thrombin generation experiments were obtained from 15 healthy volunteers, students or members of the laboratory staff, who had not taken any medication during the last 30 days before venipuncture. Blood samples were taken by atraumatic antecubital venipuncture and collected in siliconized Vacutainer tubes (Becton Dickinson, Meylan, France) containing buffered 0.13 M trisodium citrate (9 parts blood to 1 part citrate solution; 3.8%). Platelet-poor plasma (PPP) was prepared after centrifugation of citrated whole blood for 30 minutes at 2000g at room temperature. Platelet-rich plasma (PRP) was prepared after centrifugation of citrated whole blood for 10 minutes at 150g at room temperature. After centrifugation, the supernatant PRP was removed and the platelet count was adjusted at 150 × 109/L by dilution with autologous PPP. The PPP and PRP samples were used for mixture experiments with the graft product.
Experimental design
Graft product samples were added in PPP or PRP from the healthy individuals in a ratio 1:10 (vol/vol) and then assessed for thrombin generation. Preliminary experiments showed that at this ratio, the effect of the graft product on thrombin generation in both PPP and PRP reached a plateau (data not shown). To evaluate the inherent procoagulant properties of the graft product, thrombin generation was assessed in the following experimental conditions:
Platelet-poor plasma/saline: PPP without any addition of TF and/or procoagulant phospholipids or graft product. With this experiment, the basic thrombin generation capacity of recalcified PPP was assessed.
Platelet-poor plasma/graft: PPP spiked with the graft product. With this experiment, the capacity of the graft product to enhance thrombin generation of recalcified PPP was evaluated.
Platelet-rich plasma/saline: PRP without any exogenous addition of TF and/or procoagulant phospholipids or graft product. With this experiment, the basic thrombin generation capacity of recalcified PRP was evaluated.
Platelet-rich plasma/graft: PRP spiked with the graft product. With this experiment, the capacity of the graft product to enhance thrombin generation of recalcified PRP was evaluated.
Platelet-rich plasma/TF-PL: PPP spiked with physiologically relevant concentrations of TF (5 pM) and procoagulant phospholipids (4 μM; PPP-Reagent).
Platelet-rich plasma/TF: PRP spiked with 5 pM TF without any addition of exogenous procoagulant phospholipids (PRP-Reagent) since procoagulant phospholipids stem from platelets.
The PPP-Reagent and PRP-Reagent were purchased from Diagnostica Stago (Gennevilliers, France).
Thrombin generation assessment
Thrombin generation was assessed according to the method described by Hemker et al using the CAT and the respective software (Thrombinoscope b.v., Maastricht, the Netherlands). Thrombin generation was initiated by adding 20 µL triggering solution containing CaCl2 (16.7 mM final concentration) and fluorogenic substrate (Z-Gly-Gly-Arg-AMC, 417 µM final concentration). 14,15 The following parameters of thrombogram were analyzed: (1) lag time of thrombin generation (lag time), (2) time to reach maximum concentration of thrombin (ttPeak), (3) maximum concentration of thrombin (Peak), (4) mean rate index (MRI) of the propagation phase of thrombin generation calculated by the formula: Peak/(ttPeak − lag time) and expressed in nM/min, and (5) endogenous thrombin potential (ETP).
Immunoprecipitation and Western Blot Analysis for TF Expression
The cells of the graft product were washed in phosphate-buffered solution and cells were subsequently suspended in a lysing buffer (Tris–HCl pH 8.2, 20 mM Tris, 150 mM NaCl) containing proteases inhibitors (1% Brig 97, 5 mM iodoacetamide, 25 mM phenantrolyne, 20 μg/mL aprotinin, 10 mM dithiothreitol (DTT) [all from Sigma-Aldrich, Saint-Quentin-Fallavier, France], and 10 mM phenylmethylsulfonyl fluoride (PMSF) [from Amersham Biosciences Europe, Orsay, France] and incubated for 30 minutes at 4°C. After centrifugation at 1000g for 30 minutes at 4°C, the supernatant containing all cellular components except nuclei was collected. A protein G-Sepharose column (from Amersham Biosciences Europe) was washed 3 times with the lysis buffer, was incubated for 2 hours at room temperature with 2 µg of mouse monoclonal antibody against human TF (American Diagnostica, Neuville-sur-Oise, France, product number 4503), and then washed 3 times with lysis solution to eliminate unbound anti-TF antibody. The cell lysate mixture was added to the anti-TF/protein G-Sepharose complex column and incubated at 4°C overnight. After 6 washes with the lysis solution, the proteins were eluted with 100 to 250 µL of 2× Laemmli sample buffer, in nonreducing conditions (125 mM Tris–HCl pH 8, 10 mM EDTA, 0.1% SDS) or in reducing conditions (125 mM Tris–HCl pH 8, 10 mM EDTA, 0.1% SDS–b-mercaptoethanol 0.5%). 16 Human pancreatic primary adenocarcinoma cell line BXPC63 (lot F-11067, from American Type Culture Collection, Rockville, Maryland) was used as a positive control for TF expression. Western blot immunoelectrophoresis was performed as described elsewhere. 17
Flow Cytometry Analysis for Markers of Hypercoagulability
Phosphatidylserine and TF expression
Graft samples from 10 patients were assessed for phosphatidylserine (PS) expression. For the detection of PS expression, 5 µL of graft product was incubated with 10 µL of fluorescein isothiocyanate–labeled recombinant human annexin V (AnV-FITC; ABCys, Paris, France). Analyses were performed on Cytomics FC500 flow cytometer (Beckman-Coulter, Villepinte, France).
Microparticles labeling and quantification
Platelet-derived microparticles and Ed-MPs as well as Pd-MPs and Ed-MPs expressing PS (Pd-MP/PS+ and Ed-MP/PS+, respectively) were measured in the graft product using flow cytometry assay on the Cytomics FC500 instrument (Beckman Coulter, Paris, France) as described elsewhere. 18 A limitation of the assay is that this instrument is not sensitive to identify ultra-small microparticles (< 0.5 μm).
Specific TFa
Tissue factor activity (TFa) was measured in thawed graft samples and in normal PPP samples, the same which were used for thrombin generation experiments. Tissue factor activity was assessed with an in-house method. This assay measures the ability of TF-FVIIa to activate factor X using a 1-stage kinetic chromogenic method as previously described. 19 –21 The proteolytic activity of the complex TF-FVIIa was quantified by the amount of FXa produced and measured amidolytically with a specific FXa substrate as described elsewhere. 19 –21
Statistical Analysis
The number of patient “donors” of graft samples (n = 10) was calculated taking into consideration that the intra-assay variability of CAT performed in PPP or PRP was less than 2%. Comparisons between the examined samples and the control groups were performed using nonparametric Mann-Whitney test since the values did not show normal distribution. Two-sided values of P < .05 were considered statistically significant. SPSS statistical software package (Chicago, Illinois) was used for statistical analysis. Values are means (standard deviation [SD]).
Results
Procoagulant Capacity of the Graft Product
When thrombin generation was assessed in PPP/saline, the lag time was 16.6 ± 8.2 minutes, the ttPeak was 22.2 ± 10.1 minutes, the Peak was 123.3 ± 107.7 nM, the ETP was 1237 ± 887 nM/min, and the MRI was 33.0 ± 38.4 nM/min. In PPP/graft, the lag time and the ttPeak decreased by about 2-fold as compared to the PPP/saline. In PPP/graft, the Peak and the MRI significantly increased as compared to the PPP/saline. The ETP was not significantly different between PPP/graft product and PPP/saline (Table 1).
Thrombin Generation in PPP and PRP in the Presence of Graft Product and in Control Experiments (PPP or PRP/Saline, PPP/TF-PPL, and PRP/TF).a
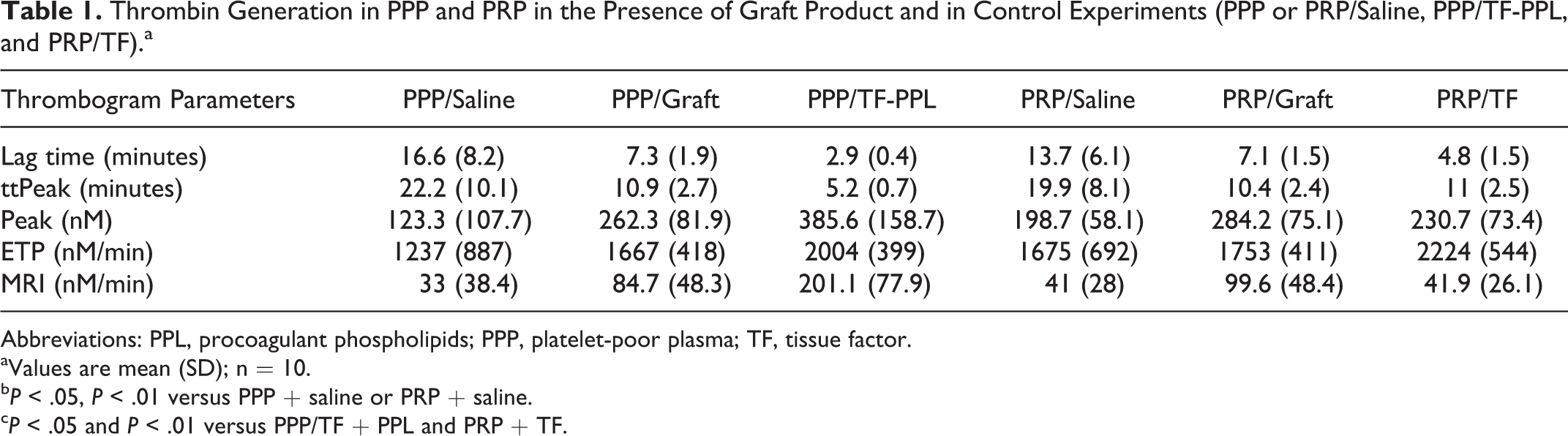
Abbreviations: PPL, procoagulant phospholipids; PPP, platelet-poor plasma; TF, tissue factor.
aValues are mean (SD); n = 10.
b P < .05, P < .01 versus PPP + saline or PRP + saline.
c P < .05 and P < .01 versus PPP/TF + PPL and PRP + TF.
In PRP/saline, the lag time was 13.7 ± 6.1 minutes, the ttPeak was 19.9 ± 8.1 minutes, the Peak was 198.7 ± 58.1 nM, the ETP was 1675 ± 692 nM/min, and the MRI was 41 ± 28 nM/min. In PRP/graft, the lag time and the ttPeak decreased by about 2-fold as compared to PRP/saline. In PRP/graft, the Peak and the MRI also significantly increased as compared to PRP/saline. The ETP was not significantly different between PRP/graft product and PRP/saline. Thrombogram parameters were not significantly different between PPP/graft product and PRP/graft product (Table 1).
In PPP/TF-PL, the lag time was 2.9 ± 0.4 minutes, the ttPeak was 5.2 ± 0.7 minutes, the Peak was 385.6 ± 158.7 nM, the ETP was 2004 ± 399 nM/min, and the MRI was 201.1 ± 77.9 nM/min. In PPP/graft, the lag time and the ttPeak were significantly longer as compared to PPP/TF-PL. The Peak and the MRI were significantly lower in PPP/graft as compared to PPP/TF-PL. The ETP was not significantly different between PPP/graft product and PPP/TF-PL (Table 1).
In PRP/TF, the lag time was 4.8 ± 1.5 minutes, the ttPeak was 11 ± 2.5 minutes, the Peak was 230.7 ± 73.4 nM, the ETP was 2224 ± 544 nM/min, and the MRI was 41.9 ± 26.1 nM/min. In PRP/graft, the lag time and ttPeak were significantly prolonged as compared to PRP/TF. The Peak and the MRI were significantly higher in PRP/graft as compared to PRP/TF. The ETP was not significantly different between PRP/graft product and PRP/TF (Table 1). Representative thrombin generation curves obtained from PPP and PRP in the presence of graft product, saline, or TF are depicted in Figure 1.
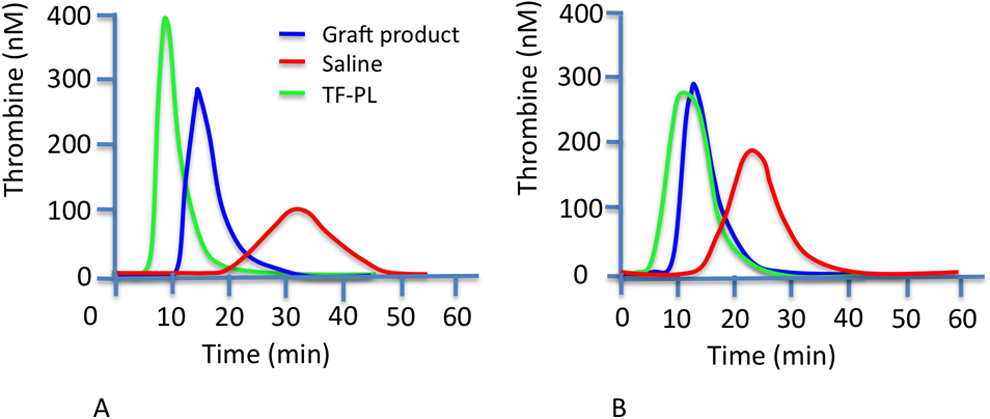
Effect of the graft product on thrombin generation in (PPP; frame A) and in (PRP; frame B) compared to thrombin generation triggered in the presence of TF and procoagulant phospholipids or saline. Representative thrombograms from 1 of 10 experiments. PPL indicates procoagulant phospholipids; PPP, platlet-poor plasma; PRP, platlet-rich plasma; TF, tissue factor.
Expression of Procoagulant Elements in Graft Product
Western blot analysis using a specific anti-TF antibody revealed a faint band at the level of 50 kDa in cell lysates from the graft product. This band was less intense as compared to the TF expressed by pancreatic cancer cells BXPC3, which was the positive control of the experiment (Figure 2). The concentration of TFa in the graft product was significantly higher as compared to normal PPP (2.96 ± 1.13 pM vs 0.25 ± 0.54 pM, respectively; P < .05).
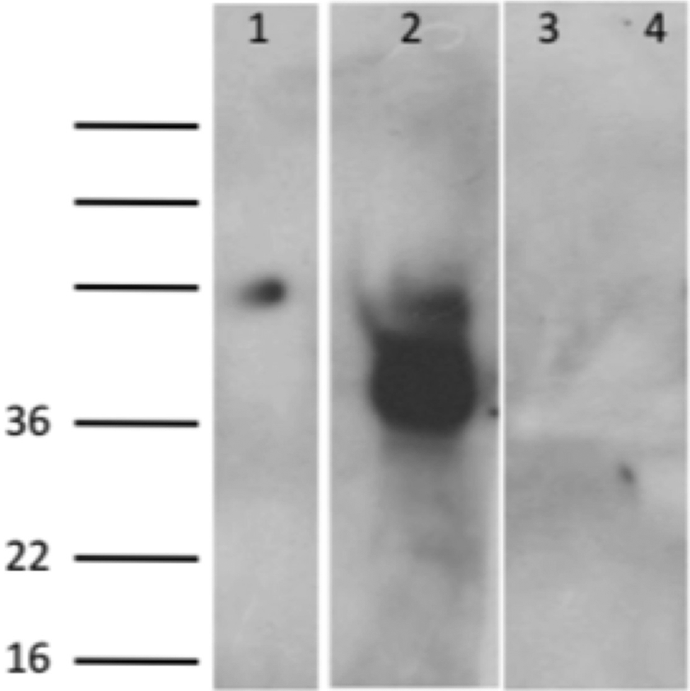
Tissue factor expression in the graft product (line 1) compared to pancreatic cancer cells BXPC3 (line 2; positive control). Western blotting was revealed with the murine IgG1 antihuman tissue factor (TF) monoclonal antibody as described in Materials and Methods. Negative results after incubation with the control isotype were shown for the graft product and BXPC3 cells (lines 3 and 4, respectively). A faint expression of TF was observed in the graft product sample. The BXPC3 cells expressed, as it was expected, abundant amounts of TF, situated at the levels of 47 kDa. Representative Western blot from 1 of 10 experiments.
The expression of PS in the graft product (25 ± 6 MIF U) was significantly higher as compared to that in plasma from healthy individuals (9 ± 4 MIF U; P < .05).
The concentration of Pd-MP in the graft product (26 457 ± 29 839 MP/μL) was significantly higher as compared to that measure in plasma from healthy individuals (control group: 752 ± 432 MP/μL; P < .05). The concentration of Pd-MP/PS+ in the graft product (683 ± 858 MP/μL) was not significantly different as compared to that in the control group (700 ± 200 MP/μL). The concentration of Ed-MPs and Ed-MP/PS+ was significantly lower in the graft product samples as compared to that measured in the plasma from healthy individuals (Table 2).
Concentration of Procoagulant Elements in the Graft Product Samples and in Normal PPP.a
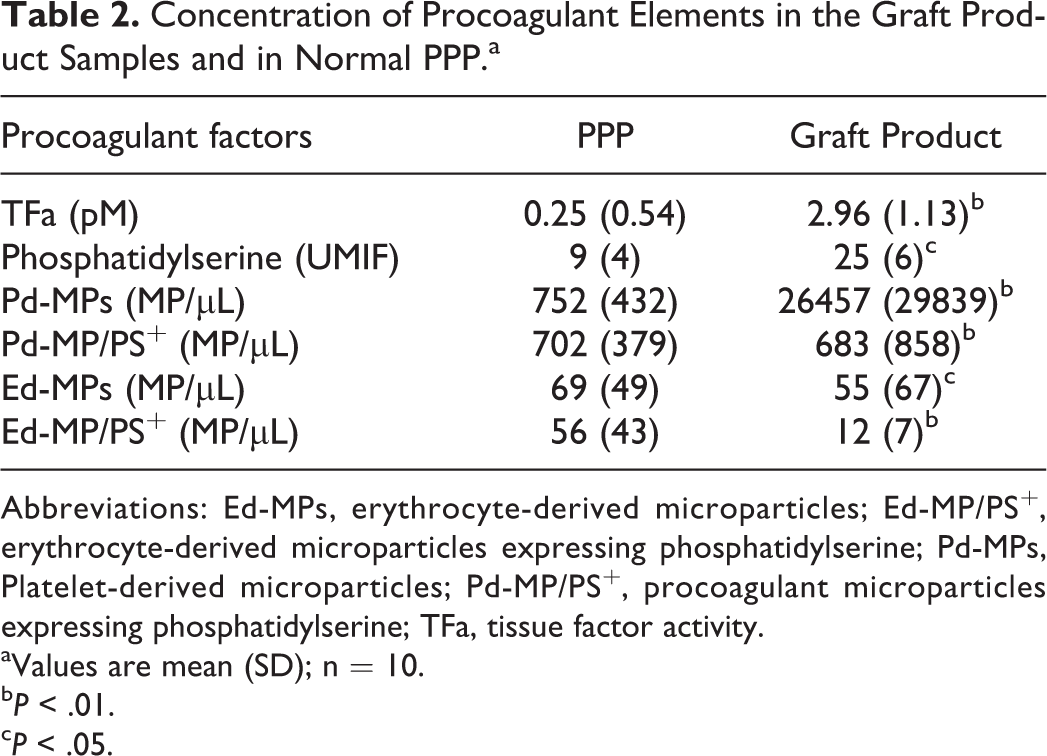
Abbreviations: Ed-MPs, erythrocyte-derived microparticles; Ed-MP/PS+, erythrocyte-derived microparticles expressing phosphatidylserine; Pd-MPs, Platelet-derived microparticles; Pd-MP/PS+, procoagulant microparticles expressing phosphatidylserine; TFa, tissue factor activity.
aValues are mean (SD); n = 10.
b P < .01.
c P < .05.
A significant interindividual variability of Pd-MP expression was observed in graft product samples. The distribution of the individual values of Pd-MP and Pd-MP/PS+ is depicted in Figure 3.
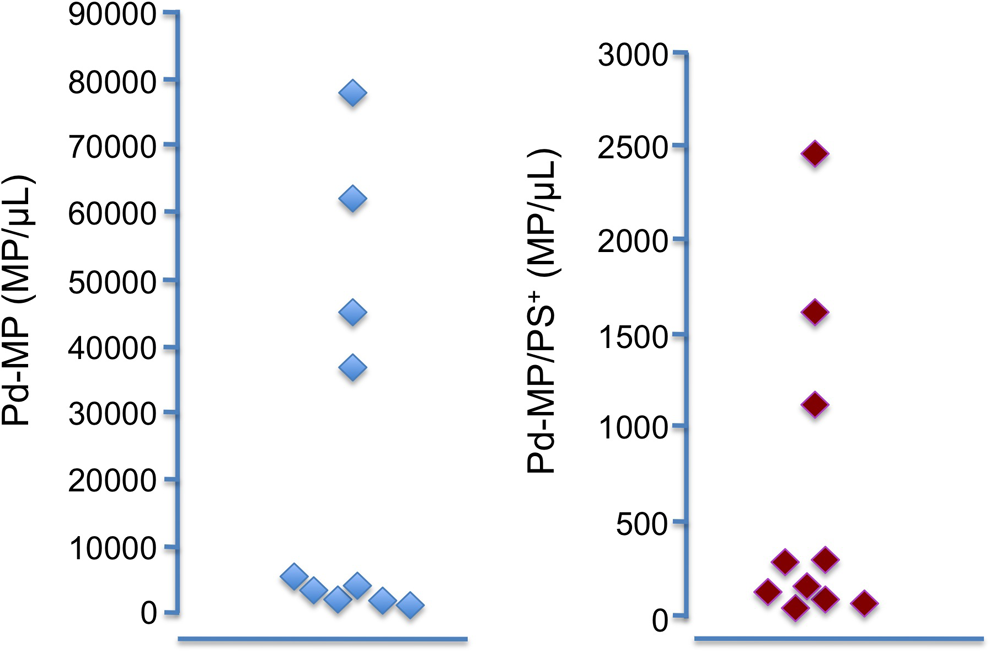
Interindividual variability of procoagulant microparticles (Pd-MP) and procoagulant microparticles expressing phosphatidylserine (Pd-MP/PS+) in graft product samples (n = 10).
Discussion
The present study demonstrates that the graft product that is used for APBSCT in patients with solid tumors possesses inherent procoagulant activity which is principally characterized by the presence of Pd-MPs and secondly by the presence of TF. The addition of graft product in normal human PPP or PRP, in a dilution similar to that used in clinical practice, resulted in the enhancement of thrombin generation. In PPP, the graft product induced the acceleration of the initiation and the propagation phases of thrombin generation and increased the concentration of generated thrombin but it did not significantly affect the ETP. According to the mathematic model proposed by Mann, the acceleration of the initiation phase of thrombin generation mainly results from TF pathway activation. In contrast, the ETP represents the integral enzymatic work done by thrombin. The enhancement of the propagation phase is the consequence of abundant formation of prothrombinase and intrinsic tenase. 22 According to the same model, increased concentration of clotting factors (ie, factors VII, VIII, or IX) or procoagulant phospholipids and/or decreased concentration of the natural coagulation inhibitors results in the increase of the integral amount of thrombin. In the experimental system presented herein, the levels of the clotting factors and natural coagulation inhibitors were within the normal range since the PPP and PRP samples were from healthy volunteers. This explains the absence of any significant difference regarding ETP in the presence or absence of the graft product. Thus, the observed enhancement of thrombin generation should stem from the presence of TF and procoagulant phospholipids in the samples of the graft product. To control this hypothesis, we investigated with Western blot electrophoresis the expression of TF in the graft product samples using a monoclonal antibody specific for TF. We also assessed the specific TFa in the studied samples. The graft product expressed some TF and also possessed significantly increased TFa as compared to the normal PPP. The presence of high concentrations of procoagulant phospholipids (PS) and high amounts of procoagulant Pd-MP were also documented.
To the best of our knowledge, this is the first study demonstrating that graft product used for APBSCT possesses inherent procoagulant activity, which is characterized by the coexistence of TF and Pd-MPs. In steady-state conditions, a limited amount of TF is expressed in the circulation, and the amount of circulating procoagulant phospholipids and PD-MP is very low. 23,24 According to our data, the transfusion of the graft product represents a procoagulant stimulus. Recently, Holbro et al reported a reversible increase of thrombin generation and procoagulant microparticles in vivo following infusion of stem cells containing DMSO. The authors attributed the transient hypercoagulability to an activating effect of DMSO on cells. 7 In our study, preliminary experiments did not demonstrate any significant effect of DMSO on thrombin generation of normal plasma (data not shown). In addition, the standardized preparation procedure of the graft product warrants the elimination of the DMSO, because the samples were systematically washed after thawing. The present study provides evidence showing that the enhancement of thrombin generation is related to some procoagulant elements of cellular origin (ie, TF and procoagulant phospholipids), which are present in the graft product.
In the next step of the study, we evaluated the degree of enhancement of thrombin generation induced by the graft product. We compared thrombin generation in normal PPP spiked with graft product to that of normal PPP in which physiologically relevant concentrations of TF (5 pM) and procoagulant phospholipids (4 μM) were added. This comparison showed that graft product induced a weaker thrombin generation as compared to that induced by a physiologically relevant TF concentration and procoagulant phospholipids. These results are in accordance with the findings of the Western blot analysis, which showed a faint expression of TF in the samples of the graft product. Thus, we document that although graft product possessed some procoagulant activity, its intensity was not sufficient to induce hypercoagulablity in normal human plasma. We assume that graft product transfusion could induce hypercoagulablity in the presence of a microenvironment, which already offers some additional procoagulant elements.
Then, we compared the enhancement of thrombin generation induced by graft product to that produced after triggering thrombin generation in PRP in the presence of physiologically relevant TF concentration (5 pM). This experiment showed that the impact of the graft product on the initiation phase of thrombin generation was weaker than that produced by physiologically relevant TF concentration. In contrast, the addition of the graft product in plasma led to significantly enhanced propagation phase of thrombin generation and consequently higher concentration of generated thrombin as compared to that generated by normal platelets and 5 pM of TF.
The ensemble of the data presented herein allow to propose that the cellular elements present in the graft product represent a potent stimulus of thrombin generation. Enhancement of thrombin generation is supported by the presence of procoagulant microparticles. 25 Procoagulant microparticles in the graft product might be an alternative source of procoagulant phospholipids that contribute to the enhancement of thrombin generation. The presence of TF in the graft product has a moderate impact on the initiation of thrombin generation, whereas the procoagulant Pd-MP might have an important amplifying impact on thrombin generation.
Afterward, we analyzed the concentration of Pd-MP and Ed-MP in the graft product samples. This approach showed an increased concentration of Pd-MP as compared to the normal plasma. Very low levels of Ed-MP were identified in the samples, verifying that the procedure for graft product preparation did not allow any contamination with erythrocyte elements. Further analysis of the data on individual basis demonstrated a significant interindividual variability regarding the concentration of Pd-MP/PS+. Taking into consideration that the method for the preparation of the graft product from the source material is highly standardized and reproducible, our data lead to the hypothesis that the high interindividual variability of the Pd-MP concentration could stem either from the source material or from blood collection conditions of handling of the APBSCT before the transfusion. However, the design of the present study does not allow to explore whether the high interindividual variability of Pd-MP concentration in the graft product preparations is related to patients’ underlying diseases, the characteristics of the cancer, or the chemotherapy they received. This issue deserves to be investigated in a future study that will include a higher number of patients, well defined according to the type of cancer and chemotherapy. The mobilization treatments, the leukapheresis process, and the freezing and thawing procedure of the graft product samples could also have some impact on the formation of the procoagulant microparticles. The effect of these procedures on the acquisition of the procoagulant properties of the graft product has to be thoroughly evaluated in a prospective study. Since the cellular elements present in the graft product (ie, TF and Pd-MP) have a procoagulant activity, our study reveals the need of a thorough quality control of the stem cell preparations regarding their procoagulant potential. Finally, the data presented herein allow to hypothesize that transfusion of APBSCT to patients may induce an in vivo hypercoagulable state. This hypothesis was not investigated since the present study was designed to modelize the procoagulant potency of the APBSCT and not the reaction of blood coagulation system of the recipient patients. The clinical relevance of the procoagulant properties of the graft product and the potential implication in the risk of acute vascular complications after APBSCT have to be prospectively studied in patients undergoing this treatment without any prophylactic heparin administration.
In conclusion, the present study demonstrates for the first time that graft products used for autologous transplantation enhance in vitro thrombin generation of the host plasma and express TFa and procoagulant Pd-MP. These data raise the question about the need for a quality control and standardization of the procedures used along the graft manipulation regarding their procoagulant potential.
Footnotes
Authors’ Note
F.S. and A.S. have made substantial contribution to the study organization, did the experiments, participated in the analysis, and interpretation of the data. V.V. has been involved in the preparation of the graft product samples. E.M. performed the Western blot analysis. J.L. has made substantial contribution to the design of the study and critically revised the manuscript. P.V.D. participated in the interpretation of the data and contributed essential reagents and tools. J.P.L. participated in the design of the study. I.E. participated in the design of the study. A.L. participated in the design of the study and the interpretation of the data. G.T.G. designed and directed the study and wrote and edited the paper.
Acknowledgment
We thank Hayat Mokrani for her skillful technical assistance.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: P. Van Dreden is an employee in Stago.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Association de Recherche sur la Thrombose et l’Évaluation de son Risque.
