Abstract
Obesity is associated with a hypercoagulable state. Thrombin generation test (TGT) and microparticle levels were not studied in obese children extensively. It is aimed to determine whether any differences in the coagulation system between obese and normal weighed children exist with the use of TGT and microparticles release. A total of 120 obese and 38 healthy children were included to the study. An increase of thrombin generation and microparticles levels were found in obese children. Hyperinsulinism could not find a risk factor for hypercoagulability in our obese children. None of the parameters of TGT has been shown to be related to metabolic parameters and metabolic syndrome. Microparticles release time is found to correlate only to body mass index (BMI) Standard deviation score (SDS) in obese children. Hypercoagulability is associated with childhood obesity. Significant correlation between degree of obesity and microparticles release suggested that high adipokine levels secreted from adipose tissue can stimulate procoagulant status-independent metabolic dearrangements.
Introduction
The prevalence of obesity is increasing among children and adolescents worldwide. Obesity is associated with several problems including glucose intolerance, hyperinsulinism, diabetes, dyslipidemia, cardiovascular diseases, and hypertension. Arterial and venous thromboembolic risks have been shown to increase in obese participants.1,2
It was shown that obesity is associated with a hypercoagulable state either with increased levels of clotting factors or inhibition of the fibrinolytic pathways. 2 Insulin resistance is settled in the center of obesity. It is not known exactly how insulin resistance affects coagulation system.
Traditional coagulation tests, such as the prothrombin time and activated thromboplastin time, cannot evaluate the whole coagulation system, and coagulation factor assays can only identify specific deficiencies. 3 Thrombin is one of the central enzymes in blood coagulation and measurement of a participant’s capacity to generate thrombin is very useful as a reflection of a thrombotic phenotype compared to conventional coagulation tests.3,4
Microparticules are another important elements that act on inflammation, coagulation, and vascular biology as related to vascular diseases. They are considered to be membrane nanofragments with procoagulant and proinflammatory properties. 5 They are derived from various cells, mostly platelets, erythrocytes, leucocytes, and endothelial cells. 6 Elevated number of circulating microparticules has been found in patients with vascular involvement and hypercoagulability. 7
Thrombin generation test and microparticle levels were not studied in obese children extensively. It is aimed to determine whether any differences in the coagulation system between obese and normal weight children exist with the use of TGT and microparticles release; if there is any difference, what are the contributing factors on the changes of coagulation system in obesity? Is hypercoagulability related to metabolic dearrangenments (hyperinsulinism, dyslipidemia), with occurrence of metabolic syndrome, or degree of obesity?
Participants and Methods
A total of 120 obese children aged 5 to 18 years were admitted to our department of Pediatric Endocrinology between March 2010 and June 2010 consecutively. As a control group, 38 normal weight, healthy, children with the same age and sex were included to the study. Body weight was measured using calibrated electronic weighing scales (Seca). Stature was determined using a stadiometer as the maximum distance from the floor to the vertex of the head. Obesity was defined according to body mass index (BMI). After weight and height measurements were taken, BMI was calculated as weight (kg)/height (m2). Participants with BMI ≥95 were defined as obese. 8 To compare BMI across different ages and between genders, BMI SDS was calculated. Z-scores of waist circumference were calculated from reference data using the LMS method. 9 Inclusion criteria were (1) Age ≥5 and ≤18 years; (2) Excluding secondary obesity; (3) No history of drugs (anticoagulant, NSAI, etc.); and (4) No hematological disorders. Waist circumference was measured at the smallest circumference between the ribs and the iliac crest.
Pubertal Development Was Determined According to Tanner Staging
Systolic and diastolic blood pressure of each participant was taken with appropriate cuff size after 20 minutes of rest. The average of 3 measurements was defined as hypertension, if it was >95th percentile according to their age and height percentiles.
10
Blood samples were taken only of obese participants during the morning between 08
Microparticle release
Peripheral venous samples were collected in 1 ml 0.109 mol/L Trisodium citrate-containing tubes. They were centrifuged for 15 minutes at 2500g and plasma samples stored at −20°C. Frozen samples were thawed in a 37°C water bath for 5 minutes and vortexed. Plasma samples were studied by using STA-PROCOAG-PPL Kit (Diagnostica Stago SAS, France). Samples added to PPL/MP free plasma, and incubated. Activated reagent than added to serum samples. Coagulation time was measured by Start4 (France).
Thrombin Generation Test
Plasma samples measured by Thrombin Generation Kits including Thrombin Calibrator, PPP-Reagent 5pM, FluCa-Kit. Thrombin Calibrator: 1 mL distilled water was added. PPP-Reagent 5pM: Diluted 1 mL distilled water. FluCa Kit: Includes Fluo-Buffer, Fluo_Substrate solutions. Fluo-Buffer kept in 37°C water bath before starting the study. Fluo-Buffer and Fluo_Substrate solutions used together.
In all, 20 µL PPP-Reagent 5pM (5pM tissue factor/4 mcM phospholipid, Stago, US) was added to polypropylene 96-well microtiter plate (Immulon 2Hb, US). Then, citrated platelet poor plasma (80 mcl) added and plate was warmed to 37°C for 5 minutes. For each plasma sample, a calibrator assay was included in which the tissue factor/phospholipid (Thrombin calibrator, Stago). The calibrator corrects for inner filter effects and quenching variation among individual plasma. Each sample and calibrator was assayed Calibrated Automated Thrombinogram Assay (CAT Assay, Thrombinoscope BV, Maastricht, the Netherlands).
Blood glucose, insulin, lipids levels, liver function tests, and thyroid function tests were measured by routine methods. Identification of metabolic syndrome among children was based on the adult criteria defined by the National Cholesterol Education Program (NCEP). 11 These criteria have been modified for children. Metabolic syndrome was defined in presence of ≥3 of the following 4 criteria: obesity (BMI >95th percentile adjusted for age and sex), hypertension (elevated blood pressure as systolic or diastolic BP >95th percentile for age and sex), dyslipidemia (high-density lipoprotein [HDL] concentration <5th percentile, triglyceride concentration >95th for age and sex), and glucose intolerance.
Abnormal glucose homeostasis was determined according to criteria recommended by the American Diabetes Association (ADA). 12 Impaired fasting glucose (IFG) was defined as fasting plasma glucose ≥100 mg/dL. Glucose at 120 minutes during the standard OGTT between 140 and 200 mg/dL was defined as impaired glucose tolerance. Hyperinsulinism was accepted as high (above 3.5) homeostasis model assessment of Insulin resistance (HOMA-IR). HOMA-IR was calculated using the following formula: HOMA-IR = [fasting insulin (μIU/mL) × fasting glucose (nmol/L)]/22,5. The value above 3,5 for HOMA-IR was accepted as reflecting hyperinsulinism. 13 Abnormal fasting lipids were defined based on normative data for children across sex and age groups. Dyslipidemia was determined if the level of triglyceride and/or total cholesterol was greater than reference values and/or HDL level was <35 mg/dL. 14
In obese children, the relationship between TGT parameters, microparticles relapse, and metabolic parameters were analyzed.
Informed consent was obtained from the parents of all children and from children when possible.
Statistical Analysis
Data are presented as mean ± standard deviation. Parameters of TGT and microparticles release were correlated to metabolic and clinic parameters by Pearson correlation test. Patients and control group were compared by Student t and chi-square tests. A P value >.05 was considered significant.
Results
A total of 120 obese children (86 female, 34 male) and 38 normal-weight children (were evaluated. The mean age of obese group was 11.4 ± 3.2 years (5 to 18 years of age) and control group was 11.2 ± 4.2 years. Of the obese children, 70% were pubertal. The mean BMI of obese group was 26.87 ± 4.35 kg/m2, and BMI SDS was 1.98 ± 0.51 (Table 1 ).
Clinical and Laboratory Characteristics of Obese Group
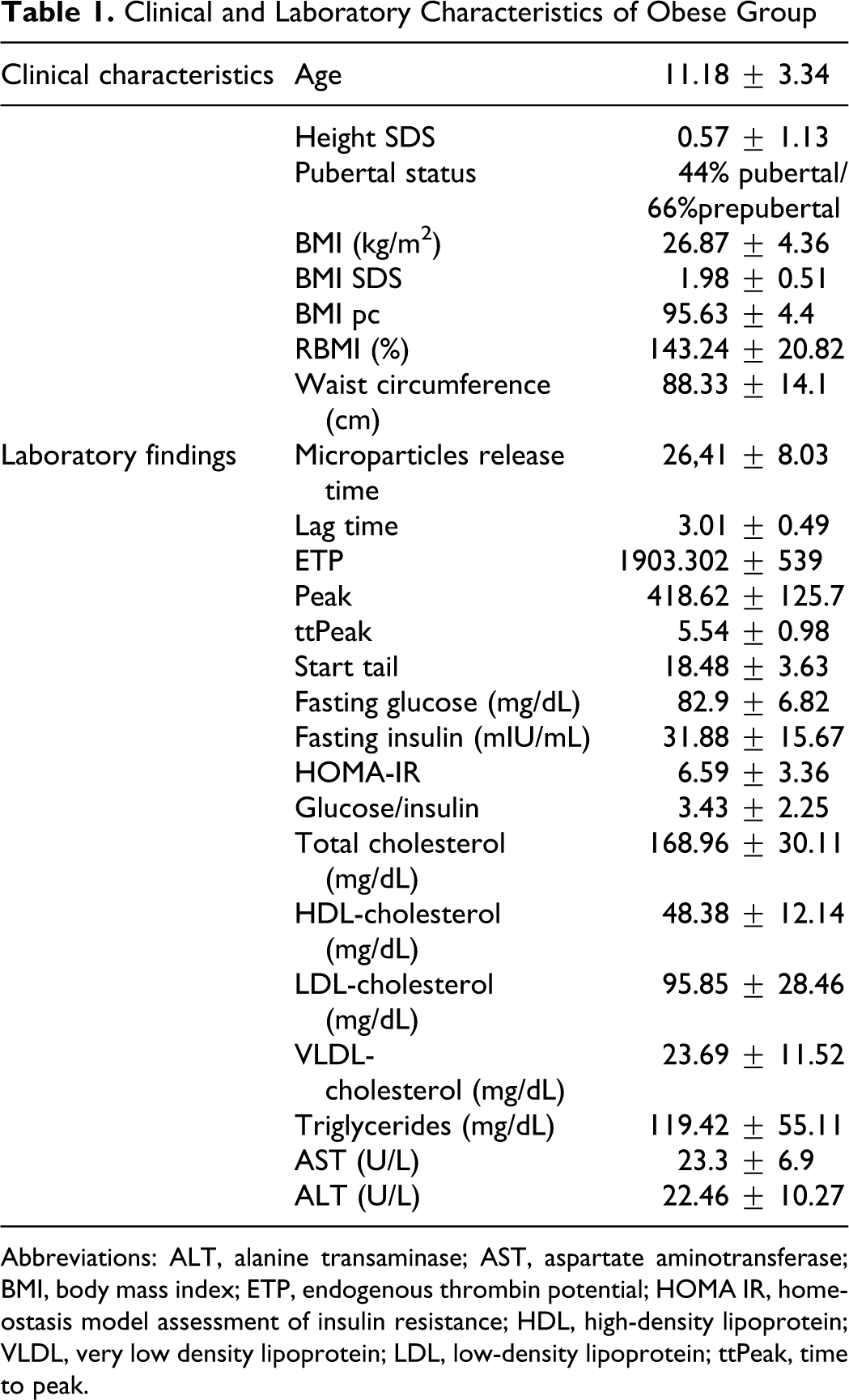
Abbreviations: ALT, alanine transaminase; AST, aspartate aminotransferase; BMI, body mass index; ETP, endogenous thrombin potential; HOMA IR, homeostasis model assessment of insulin resistance; HDL, high-density lipoprotein; VLDL, very low density lipoprotein; LDL, low-density lipoprotein; ttPeak, time to peak.
An increase of thrombin generation and microparticles levels were found in obese children. The obese children showed significantly higher endogenous thrombin potential (ETP) and Peak than control participants. Lag time, time to peak (ttPeak), and start tail were shorter than in control group. Microparticles release time was also significantly shorter in obese children than normal weight children (Table 2 ).
Comparison of Microparticles Release and Thrombin Generation Test Parameters Between Obese and Control Group
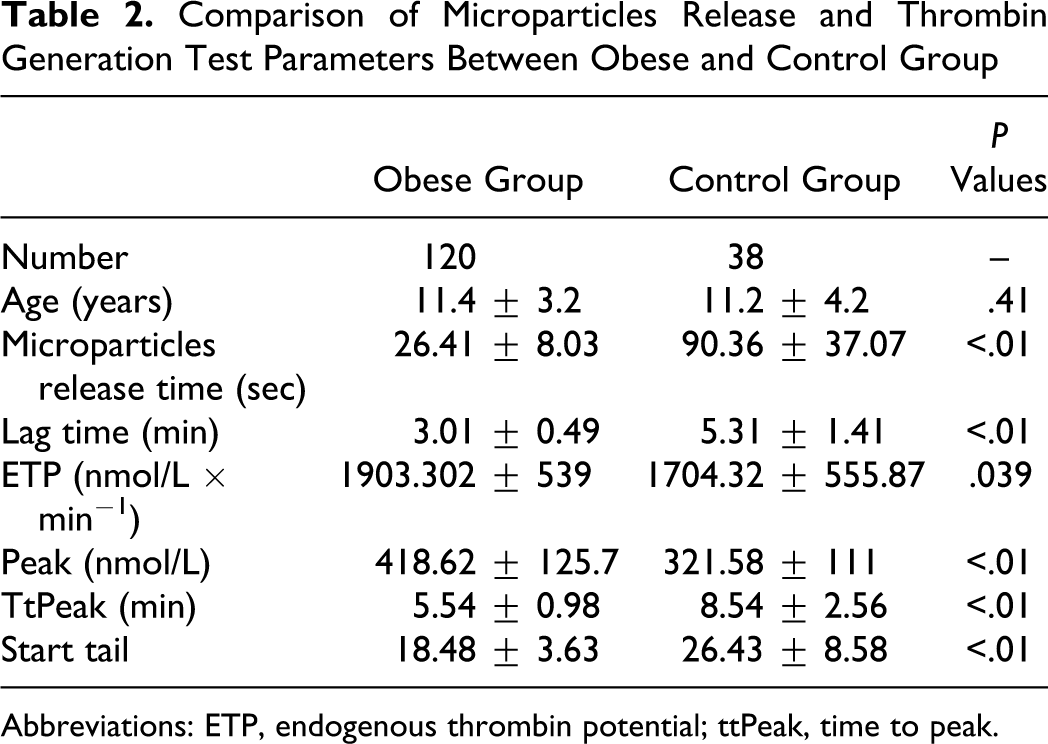
Abbreviations: ETP, endogenous thrombin potential; ttPeak, time to peak.
The correlation between thrombin generation test parameters and metabolic parameters are shown in Table 3 . There is no correlation between all the thrombin generation test parameters with fasting insulin, fasting glucose, triglyceride, total cholesterol, HDL cholesterol, low-density lipoprotein (LDL) cholesterol, and very low density lipoprotein (VLDL) cholesterol levels. Thrombin generation test parameters were also not correlated to HOMA-IR.
Pearson Correlation Coefficient (r) Calculated Between Microparticles Release, Thrombin Generation Test Parameters, and Metabolic Parameters in Obese Children
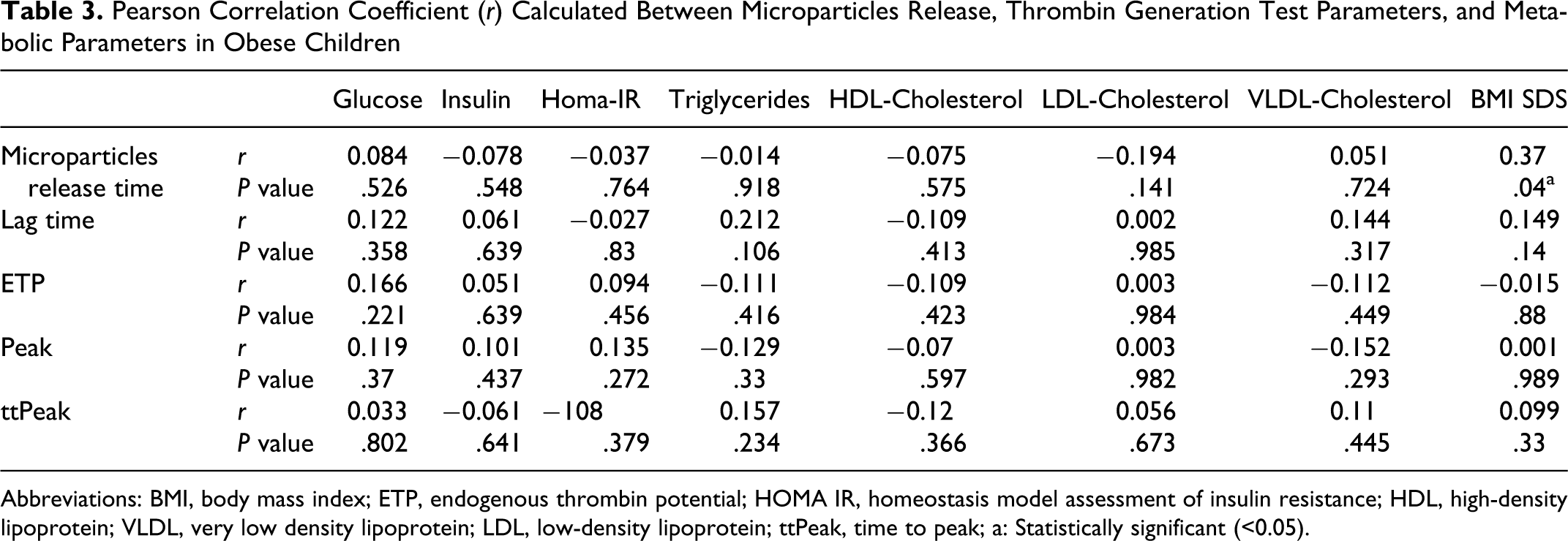
Abbreviations: BMI, body mass index; ETP, endogenous thrombin potential; HOMA IR, homeostasis model assessment of insulin resistance; HDL, high-density lipoprotein; VLDL, very low density lipoprotein; LDL, low-density lipoprotein; ttPeak, time to peak; a: Statistically significant (<0.05).
Obesity degree is demonstrated as an affecting factor for microparticles release. A significant negative correlation was found between microparticles release time and BMI SDS in obese children. But neither of the TGT parameters were correlated to BMI SDS.
Metabolic syndrome was detected in 27% of obese children. No statistically significant differences were detected between either TGT parameters or microparticle release time and the occurrence of metabolic syndrome in obese children.
Discussion
The childhood obesity is being recognized as a major health problem and is associated with increased risks of serious medical problems such as glucose intolerance, hypertension, cardiovascular disorders, and dyslipidemia. Several studies demonstrated that elevated levels of coagulation factors such as PAI-1, prothrombin, factors VII and VIII, and fibrinogen were found in obese participants. 15 Hypercoagulable state in obesity is an additive factor on vascular diseases development.
We measured TGT and demonstrated that thrombin generation is increased in obese children. Classical clotting tests such as a PTT and PT do not reflect thrombin generation entirely. 16 It was concluded that TGT measured by “Calibrated Automated Thrombogram” that we used is superior to the traditional coagulation tests for the detection of the coagulopathy. 17 Thrombin generation assays are provide the measurement both the rate of thrombin formation and the total amount of thrombin formed, so they reflect the potential coagulation activity in plasma. 18 The measurement of in-vitro thrombin generation is defined as a very promising tool to detect an individual’s coagulation potential. 19
Four major parameters can be assessed in this test: The lag time, the ttPeak, the peak height, and ETP. 17 In this study, we showed that obese children had shorter lag time, shorter ttPeak, higher peak height, and more ETP in TGT than normal weight children.
Microparticles are small (<0.1 mm) membrane vesicles that promote coagulation. 20 Elevated number of circulating microparticles have been found in patients with vascular involvement and hypercoagulability. 7 Apoptosis and vascular cell activation are main contributors for microparticles release and deleterious effect to atherothrombosis. Circulating microparticles provide an additional procoagulant phospholipid surface for accumulate of the enzyme complexes of the coagulation cascade. Microparticles act as a surrogate marker of endothelial dysfunction, and they are also able to augment preexisting vascular dysfunction. 21
We found that “microparticles release time” is shorter in obese children than in control group. This means that the more microparticles are generated in obese children, the faster coagulation cascade begin.
All these findings support the outcome that hypercoagulability, defined as an increased capacity to form thrombin, and microparticles formation of the blood are found in obese children. In other words, a prothrombotic state begins in childhood with obesity.
It is very well known that prothrombotic state will accelerate cardiovascular diseases. To decrease the cardiovascular disease prevalence, preventive measures against obesity should start in childhood period.
Interestingly, none of the parameters of TGT has been showed to be related to metabolic parameters and developing metabolic syndrome. Hyperinsulinism could not find a risk factor for hypercoagulability in our obese children either as basal insulin or HOMA-IR index. This result is in discordance with some of the recent reports. Frithsch et al reported that ETP and Peak were correlated significantly to insulin resistance (HOMA-IR) but not to basal insulin level. 1 One of the difference from our study is analyzed patients' obesity degree. Their study group consist of normal and overweight children but not obese children. In addition none of our obese children have glucose intolerance. Hyperglycemia or glucose intolerance could affect coagulation status in obese children.
A recent study reported an association of increasing of coagulability with hyperglycemia and hyperinsulinemia in type 2 adult diabetic patients. 22 Type 2 diabetic patients in this study were also obese. The differences in the degree of obesity or diabetic status of patients would be causative factors to explain this discrepancy within all those studies.
Microparticles release time is found to correlate with BMI SDS in obese children but not other metabolic parameters. Only the degree of obesity seems to be the affecting factor for increased level of microparticles.
These findings suggest that hypercoagulable state seen in obese children starts independent of hyperinsulinism, glucose intolerance, dyslipidemia, or overall metabolic syndrome.
The cause of shortening the microparticles release time with increasing BMI SDS will be attributed to the amount of adipose tissue itself. It is well-known that obesity is associated with a chronic inflammatory state predisposing to atherosclerosis. 23 Adipose tissue produces adipokines such as tumor necrosis factor (TNF) and interleukin-6 (IL-6) that contribute to systemic and vascular inflammation. 24 It was inevitable that the levels of adipokines augments with increasing adipose tissue. Release of endothelial microparticles (EMPs) has been observed after in-vitro stimulation of endothelial cells with TNF-alpha, PAI-1, thrombin, uremic toxins, reactive oxygen species. 25
As a conclusion, hypercoagulability showed with thrombin generation test and microparticle release is associated with childhood obesity. We could not find any relation to the metabolic parameters. Significant correlation between degree of obesity and microparticles release suggested that high adipokine levels secreted from adipose tissue can stimulate procoagulant status independent metabolic dearrangements.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.F
The author(s) received no financial support for the research and/or authorship of this article.
