Abstract
We assessed contributions of thrombophilia to premature cardiovascular disease (CVD) events (≤ age 45) in 153 patients. Test results of thrombophilia–hypofibrinolysis were obtained in 153 patients with CVD ≤ age 45, 110 healthy normal controls, and 110 patients who had venous thromboembolism (VTE) without CVD. Of the 153 patients with CVD, 121 (79%) had sustained myocardial infarction, 70 (46%) had coronary artery stenting, and 53 (35%) had coronary artery bypass grafts. The first CVD events occurred at ages >20 to 35 in 47 patients and at ages >35 to 45 in 106 patients. At study entry, median low-density lipoprotein cholesterol was 126 mg/dL, 56 (37%) smoked, 56 (37%) had hypertension, and 56 (37%) were diabetic. Cases differed from normal controls for high factor VIII (10 [22%] of 45 vs 7 of 103 [7%], P = .007), high homocysteine (32 [21%] of 151 vs 5 [5%] of 107, P = .0002), low free protein S (5 [11%] of 44 vs 2 [2%] of 96, P = .032), high anticardiolipin antibodies (ACLA) IgM (11 [9%] of 129 vs 2 [2%] of 109, P = .024), high lipoprotein (a) [Lp(a)] (46 [30%] of 151 vs 21 [19%] of 110, P = .038), and the lupus anticoagulant (4 [11%] of 37 vs 2 [2%] of 110, P = .035). There were no differences (P > .05) between cases and VTE controls except free protein S and Lp(a). Free protein S was more often low in VTE controls (24 [28%] of 85 vs 5 [11%] of 44, P = .03) and Lp(a) was more often high in cases (46 [30%] of 151, VTE controls 12 [17%] of 71, P = .032). In 153 patients with premature CVD ≤ age 45, thrombophilia was pervasive (high factor VIII, homocysteine, ACLA IgM, low free protein S, high Lp(a), and lupus anticoagulant), evidencing thrombotic contribution to premature CVD. Moreover, thrombophilia in patients with premature CVD was comparable to VTE controls, emphasizing the pervasive nature of thrombophilia in premature CVD.
Introduction
In patients who have developed early arterial thrombosis (age ≤ 50 [male], ≤ 55 [female]), particularly with no apparent cause of arterial occlusion, evaluation for thrombophilia is important as thrombophilia may play a predisposing role in patients with cerebral, coronary, and peripheral vascular ischemia. 1 –4 Arterial thrombotic events have been associated with factor V Leiden, prothrombin G20210A, and methylenetetrahydrofolate reductase (MTHFR C677 T) mutations. 5 Atherothrombosis has also been associated with primary antiphospholipid syndrome, 6 reduced protein C activity, 7 and hyperhomocysteinemia, 8,9 among other conditions. 10 Additionally, a case of premature atherothrombosis has been linked to elevated factor VIII. 11 In carriers of hemophilia, a mild decrease in coagulability has a significant protective effect against fatal ischemic heart disease. 12
The clinical importance of thrombophilia in arterial thrombosis is emphasized by the superiority of oral anticoagulants versus aspirin alone in secondary prevention of acute arterial ischemia. 13,14 Since the development of the Xa inhibitors, 15 –17 combined antiplatelet and anticoagulant therapy, particularly for secondary prevention, is now more commonly being used in patients with atherothrombosis and worsening cardiovascular disease (CVD).
Patients with symptomatic atherothrombosis (thrombus formation on atherosclerotic plaque) 18 have high rates of recurrent vascular events and rehospitalization. 19 –22 Thrombophilia is thought to be primarily associated with venous thrombosis; however, atherothrombotic coronary artery disease (CAD) shares risk factors and pathophysiology with venous thromboembolism (VTE), deep vein thrombosis, and pulmonary embolism. 23 Factor V Leiden heterozygosity is more common in diabetic patients with peripheral artery disease who had unsuccessful percutaneous transluminal angioplasty than in those with a successful procedure. 24 Factor V Leiden, prothrombin G20210A, and MTHFR C677 T mutations increase the risk of myocardial infarction (MI) and ischemic stroke. 5 The link between atherothrombosis and thrombophilia remains a topic of major focus. 1,25,26
Atherothrombosis in the form of CVD and peripheral artery disease is the leading factor of mortality in the United States, compromising over a quarter of all-cause mortality in 2002. 27 Prior atherothrombotic events confer an increased risk of subsequent events, and patients with atherothrombosis have the highest risk of repeat events when compared to patients with stable disease and those with risk factors but without atherothrombotic events. 22 Steg et al found that patients with prior arterial disease have nearly triple the risk of CVD death, MI, stroke, or hospitalization for atherothrombotic events within 1 year when compared to a population with multiple risk factors only, 14.41% versus 5.31%. 20 Additionally, in the total patient population with previous arterial disease, 12.81% had a major event (CVD death, MI, and stroke) or were hospitalized for CVD or revascularization procedure within 1 year. 20 In 142 patients with MI ≤ age 45 compared to 95 controls, Pineda et al reported that lipoprotein (a) (Lp[a]), fibrinogen, and D-dimer levels were independently associated with cases. 28
Our specific aim was to examine the role of thrombophilia and atherothrombosis in 153 patients referred because of CVD ≤ age 45, with CVD events at ages 20 to 30 in 23 patients, ages >30 to 35 in 24, and ages >35 to 45 in 106.
Materials and Methods
Patients
Studies were carried out following a protocol approved by the institutional review board with signed informed consent. The 153 patients (113 men, 40 women), comprising a sequential case series, were serially referred by their cardiologists because of premature and/or rapidly worsening CVD (Table 1). The patients were studied between January 3, 1994, and May 3, 2016.
Lipids at Study Entry in 153 Patients With CVD and 110 Normal Controls.
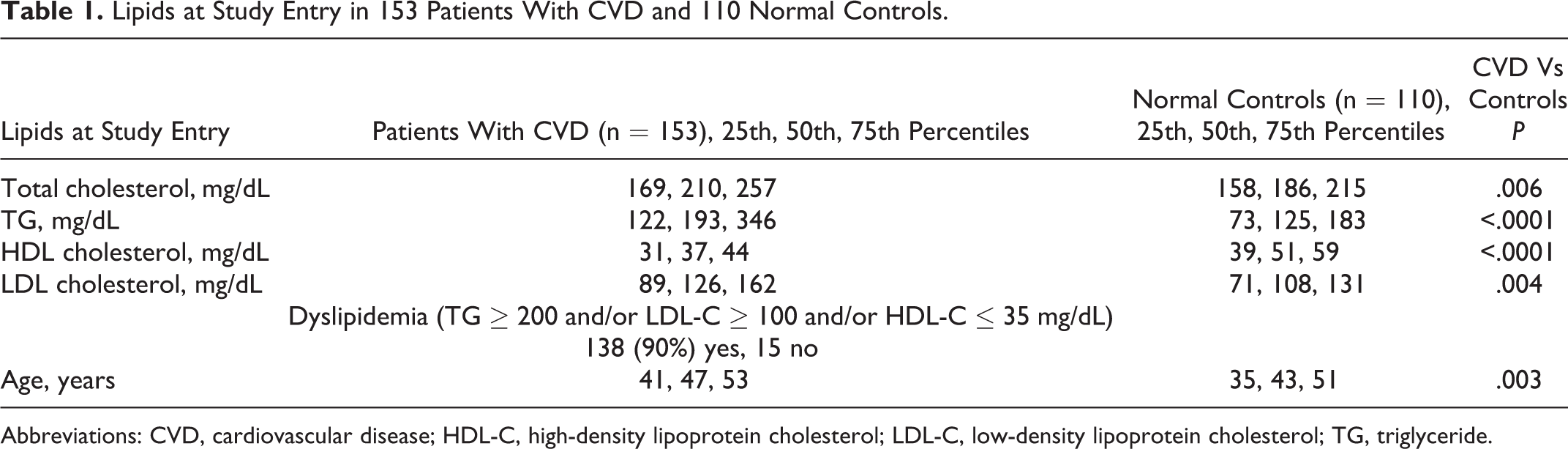
Abbreviations: CVD, cardiovascular disease; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; TG, triglyceride.
Controls
Thrombophilia–hypofibrinolysis results in the 153 cases were compared to 110 previously reported healthy, normal controls 29 (48 men, 62 women) free of coronary artery, cerebrovascular, peripheral vascular diseases and VTE. Cases were also compared to 110 thrombosis controls with VTE but free of CVD (53 men, 57 women). 30
Test Results for Thrombophilia and Hypofibrinolysis
Using previously reported methods, 29,31 test results for thrombophilia and hypofibrinolysis were obtained by the Colorado Coagulation Laboratory of LabCorp and are summarized in Appendix A. Fasting lipids and lipoproteins were measured by LabCorp.
Polymerase chain reaction (PCR) measures of the factor V Leiden, prothrombin G20210A, MTHFR, and plasminogen activator inhibitor (PAI-1 gene) mutations were done along with serologic measures of homocysteine; factors VIII and XI; antigenic proteins C, total S, free S, and antithrombin; lupus anticoagulant; anticardiolipin antibodies (ACLAs) IgG and IgM; and Lp(a).
Serologic tests were done before anticoagulation. Plasminogen activator inhibitor 1 activity was not measured.
Antigenic total and free protein S rather than protein S activity (functional assay) was done as the initial test for detecting congenital protein S deficiency, because of the greater variety of patient conditions that can interfere with the accuracy of functional protein S testing as compared to antigen testing. 32 Measurement of antigenic free protein S, protein C, and antithrombin would identify type 1 defects but would miss type 2 deficiency in proteins S, protein C, and antithrombin.
Homocysteine reference ranges were changed over time (Table 2) based on reagent lots and performance of the assay on new platforms but always using the same methodology (nephelometry).
One Hundred Fifty-Three Patients With Premature CVD (≤ age 45): MI, Coronary Artery Stent, or CABG, and 110 Normal Controls.
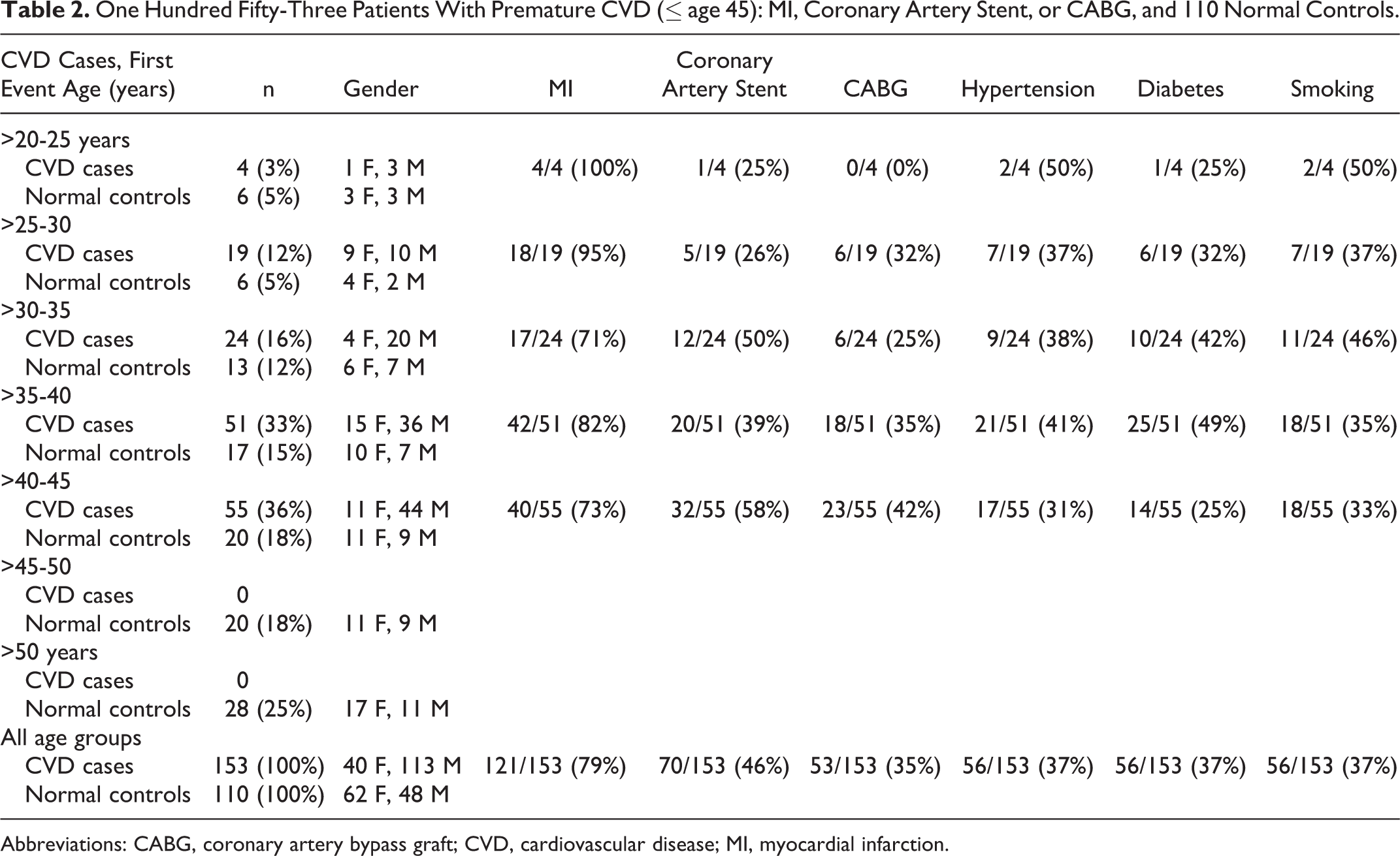
Abbreviations: CABG, coronary artery bypass graft; CVD, cardiovascular disease; MI, myocardial infarction.
Anticardiolipin antibody IgG and IgM reference ranges were changed over time (Table 2) based on changes in reagent manufacturer. The population 95th percentile was used by the Colorado Coagulation Laboratory to establish the reference range.
Due to limitations of third-party medical insurance coverage for thrombophilia evaluation, the thrombophilia workup was not complete in all patients, particularly for factor VIII, antigenic proteins C, S, antithrombin, and the lupus anticoagulant.
Statistical Methods
Statistical analyses were done using SAS 9.4. Distributions of coagulation test results in cases versus controls were compared using χ2 tests. When cells in the 2 × 2 table had expected counts <5, Fisher exact test was used. Lipids and lipoprotein cholesterol levels in cases versus controls were compared by Mann-Whitney U test.
Power and sample size calculations for comparisons of thrombophilia between cases and controls were carried out based on factor V Leiden heterozygosity in 31% of cases versus 2% of normal controls as well as 12% of controls who had sustained VTE. 33 To compare cases with atherothrombosis versus normal controls, there would need to be ≥25 participants in each group to detect differences at a significance level of 0.05 with 80% power. To compare factor V Leiden heterozygosity in cases with atherothrombosis versus VTE controls, there would need to be ≥73 in each group to detect differences at a significance level of 0.05 with 80% power.
Spearman correlation coefficients between the total number of abnormalities in 11 serologic coagulation test results, 4 PCR test results, 15 test results of thrombophilia, and the total number of 4 CVD risk factors (hypertension, diabetes, smoking, and male gender) were calculated.
Logistic regression analysis was done with the dependent variables being 0, 1, or ≥2 abnormal test results of 11 serologic tests and explanatory variables such as age, smoking, hypertension, diabetes, and gender. A second logistic regression analysis was done with the dependent variables being 0, 1, or ≥2 abnormal coagulation test results of 15 tests taken (11 serologic, 4 PCR) and explanatory variables such as age, smoking, hypertension, diabetes, and gender.
Results
Cases had higher total and low-density lipoprotein (LDL) cholesterol, higher triglycerides, and lower HDL cholesterol than normal controls (Table 1). At study entry, median LDL cholesterol in cases was 126 mg/dL and median triglyceride 193 mg/dL (Table 1). At entry, dyslipidemia was present in 90% of cases (Table 1).
Of the 153 patients, 56 (37%) smoked, 56 (37%) had hypertension, and 56 (37%) were diabetic (Table 2). Of the 153 patients, 49 (32%) had none of these 3 CVD risk factors, 56 (37%) had 1, 32 (21%) had 2, and 16 (10%) had all 3. Of the 153 patients with CVD, 121 (79%) had sustained MI, 70 (46%) had coronary artery stenting, and 53 (35%) had coronary artery bypass grafts (Table 2). The first CVD events occurred at ages 20 to 30 in 23 patients, at ages >30 to 35 in 24 patients, and at ages >35 to 45 in 106 (Table 2).
Test results of thrombophilia–hypofibrinolysis were obtained from 153 patients with CVD ≤ age 45, 110 healthy normal controls, and 110 patients who had venous thromboembolism (VTE) without CVD (Tables 3 and 4).
Coagulation Disorders in 153 Young Patients With CVD (113 Men, 40 Women), Compared with 110 Healthy Normal Controls (48 Men, 62 Women) and with 110 VTE Controls (53 Men, 57 Women).a
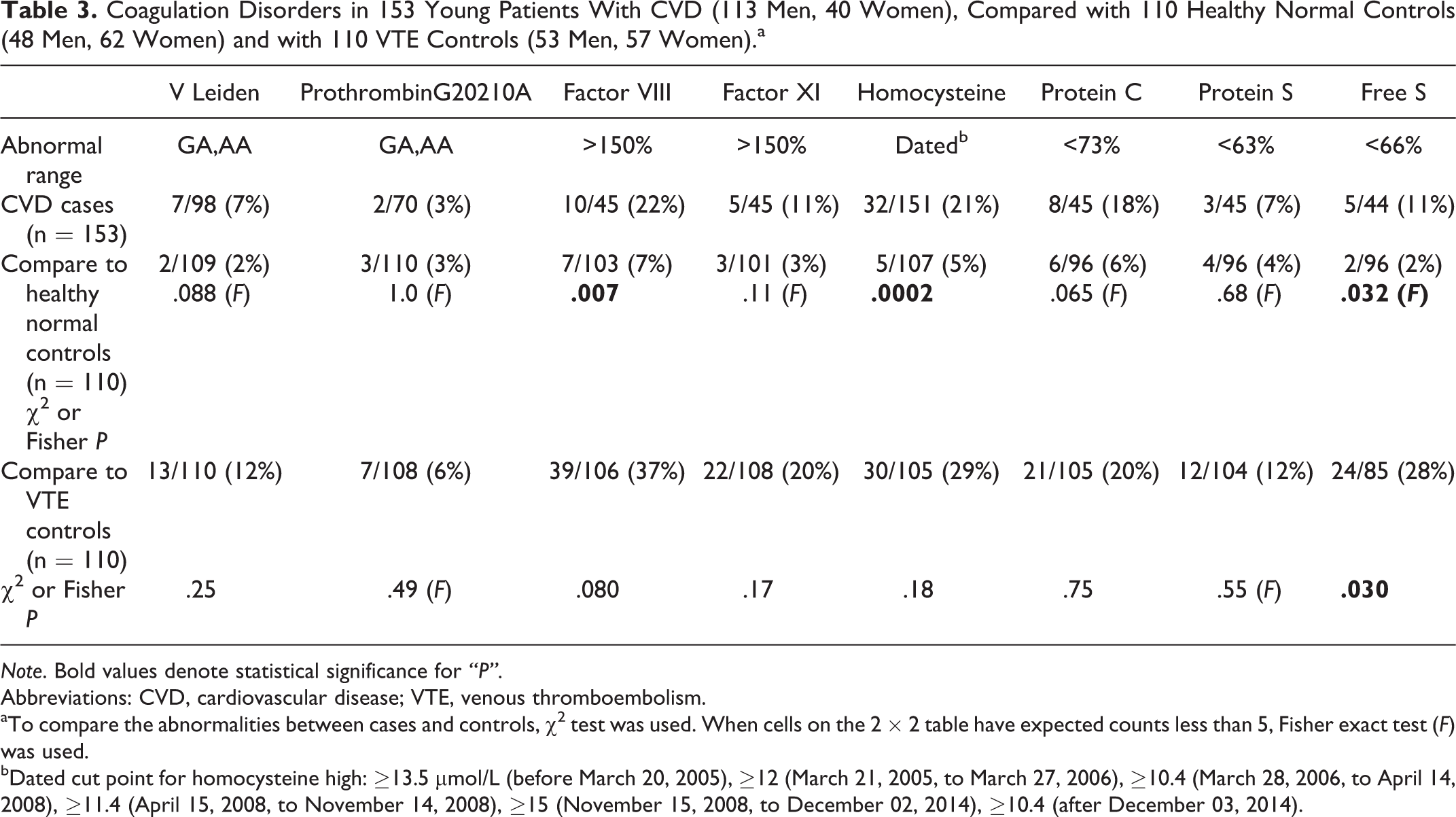
Note. Bold values denote statistical significance for “P”.
Abbreviations: CVD, cardiovascular disease; VTE, venous thromboembolism.
aTo compare the abnormalities between cases and controls, χ2 test was used. When cells on the 2 × 2 table have expected counts less than 5, Fisher exact test (F) was used.
bDated cut point for homocysteine high: ≥13.5 μmol/L (before March 20, 2005), ≥12 (March 21, 2005, to March 27, 2006), ≥10.4 (March 28, 2006, to April 14, 2008), ≥11.4 (April 15, 2008, to November 14, 2008), ≥15 (November 15, 2008, to December 02, 2014), ≥10.4 (after December 03, 2014).
Coagulation Disorders in 153 Young Patients with CVD (113 Men, 40 Women), Compared with 110 Healthy Normal Controls (48 Men, 62 Women) and With 110 VTE Controls (53 Men, 57 Women).a
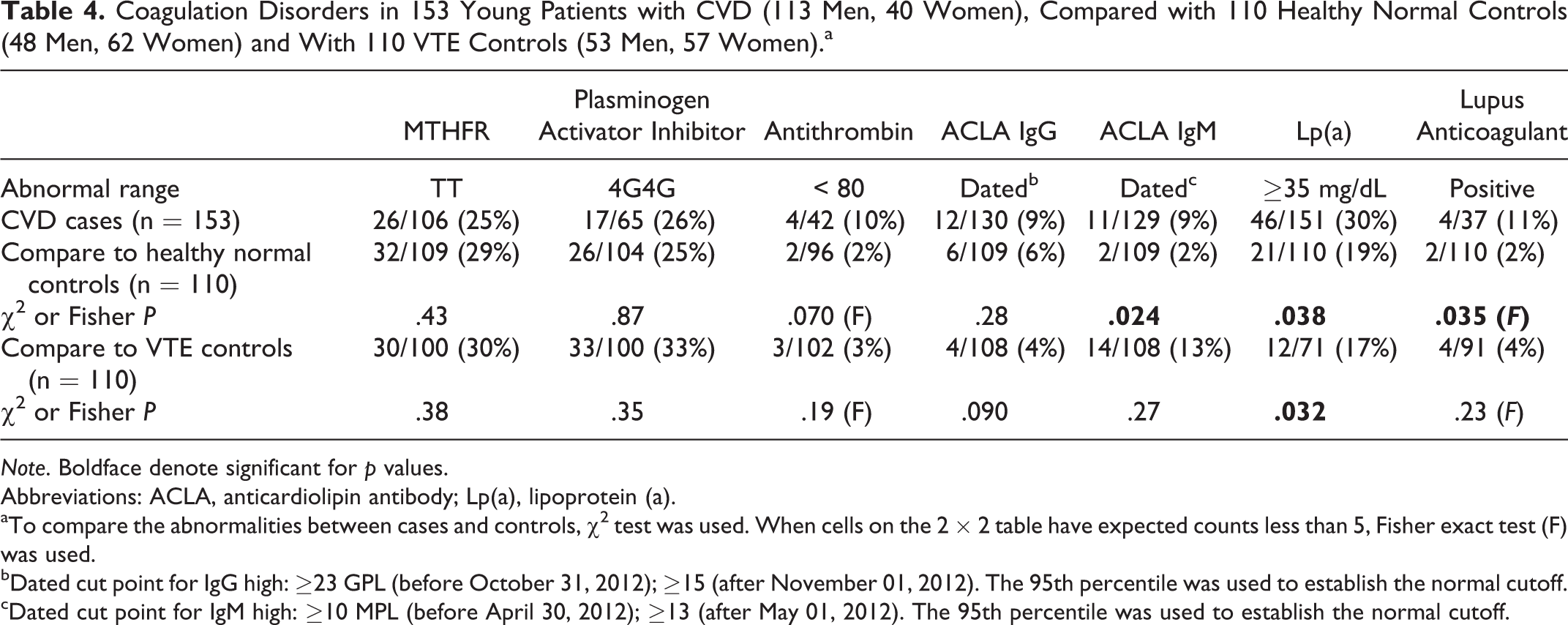
Note. Boldface denote significant for p values.
Abbreviations: ACLA, anticardiolipin antibody; Lp(a), lipoprotein (a).
aTo compare the abnormalities between cases and controls, χ2 test was used. When cells on the 2 × 2 table have expected counts less than 5, Fisher exact test (F) was used.
bDated cut point for IgG high: ≥23 GPL (before October 31, 2012); ≥15 (after November 01, 2012). The 95th percentile was used to establish the normal cutoff.
cDated cut point for IgM high: ≥10 MPL (before April 30, 2012); ≥13 (after May 01, 2012). The 95th percentile was used to establish the normal cutoff.
Cases differed from normal controls for high factor VIII (10 [22%] of 45 vs 7 [7%] of 103, P = .007), high homocysteine (32 [21%] of 151 vs 5 [5%] of 107, P = .0002), low free protein S (5 [11%] of 44 vs 2 [2%] of 96, P = .032), high ACLA IgM (11 [9%] of 129 vs 2 [2%] of 109, P = .024), high Lp(a) (46 [30%] of 151 vs 21 [19%] of 110, P = .038), and the lupus anticoagulant (4 [11%] of 37 vs 2 [2%] of 110, P = .035; Table 3 and 4).
There were no differences (P > .05) between cases and VTE controls except free protein S, more often low in VTE controls (24 [28%] of 85 vs 5 [11%] of 44, P = .03), and Lp(a), more often high in cases (46 [30%] of 151 vs 12 [17%] of 71, P = .032; Table 3 and 4).
The total number of abnormalities in 11 serologic coagulation test results and the total number of 4 CVD risk factors (hypertension, smoking, diabetes, and male gender) were inversely correlated (Spearman correlation coefficient r = −.19, P = .019).
The total number of abnormalities in 4 genes (factor V, prothrombin, MTHFR, and PAI-1 4G4G mutation) was not significantly correlated with the total number of 4 risk factors (r = −.066, P = .42). The total number of abnormalities in 15 coagulation test results (4 genes and 11 serologic) was inversely correlated with the 4 CVD risk factors (r = −0.18, P = .028).
As displayed in Table 5, only gender and hypertension were significantly associated with procoagulant test results. When male gender or hypertension was absent, there were a higher number of thrombophilic abnormalities, both serologic alone and serologic plus PCR abnormalities (Table 5).
Logistic Regression Models for Total Number of Abnormalities in 11 Serologic Coagulation Measures, and in 4 PCR Coagulation Measures.a

Abbreviations: AUC, area under the curve; SLE, significant level of entry; SLS, significant level of stay.
aStepwise selection from conventional risk factors for coronary artery disease: age, hypertension, smoking, diabetes, and male gender (SLE = .15, SLS = .05).
Discussion
In premature and advanced atherosclerosis, both systemic and vascular wall–driven coagulation have increasing importance. 25 Patients with severe premature CVD may have both conventional atherosclerosis and concomitant thrombophilia-driven arterial thrombosis. 18,19 –23,34 –36 Premature arterial thrombotic occlusion, especially in young women, is associated with a higher prevalence of thrombophilic gene polymorphisms and thrombophilia overall compared to controls, highlighting a biologically plausible link with arterial thrombosis. 1,2,5,37 –39 The atherothrombotic interaction of hyperlipidemia and thrombophilia is heightened by exogenous estrogens. 31,40,41
Beyond conventional risk factors for premature CVD, our current study documents the significant and pervasive presence of thrombophilia, with high levels of factor VIII, homocysteine, low free protein S, high ACLA IgM, and high Lp(a) and increased presence of the lupus anticoagulant in cases compared to healthy normal controls without CVD. Moreover, when compared to 110 controls with VTE but without CVD, our cases with thrombophilia contributing to premature CVD differed from the VTE controls only for low free protein S, more common in VTE controls than CVD cases, and for high Lp(a), more common in cases, indicating that patients with very early CVD share prothrombotic factors with patients characterized by thrombosis (VTE). 23
The findings of our current study in patients with CVD events before age 45 are congruent with those of Andreotti and Becker 1 who focused attention on young patients who developed arterial thromboses and were noticed to have a significantly higher prevalence of thrombophilic gene polymorphisms compared to controls. The overlap between lipid–lipoprotein and prothrombotic risk factors for CVD in our study where 90% of 153 patients had dyslipidemia at entry has also been emphasized by Prandoni et al, 42 Voetsch and Loscalzo, 4 Botto et al, 43 Jurcut et al, 40 and by our studies of the relationships of high homocysteine to atherothrombosis. 44 In 145 patients with MI ≤ age 45, Pineda et al 28 reported that variables independently and positively associated with cases were Lp(a), fibrinogen, and D-dimer.
The total number of coagulation abnormalities (among the 15 test results taken) was inversely corrected with the total number of other CVD risk factors (hypertension, smoking, diabetes, male gender), P = .028, emphasizing the contributory role of thrombophilia to premature CVD when conventional risk factors are not present. When male gender or hypertension was absent, there were a higher number of thrombophilic abnormalities in the cohort selected by premature CVD, where hypertension, diabetes, and smoking were present in 37%, 37%, and 37% of cases. This emphasizes the important contributory role of thrombophilia to premature CVD when conventional risk factors are absent.
There is growing literature on antithrombotic therapy in the acute setting of CAD (acute coronary syndrome). 45 These investigations include studies of apixaban 46 and rivaroxaban. 47 Warfarin has been the mainstay of therapy in patients sustaining thrombosis and having the antiphospholipid syndrome 48 –50 and was used successfully in the current study in 3 of the 4 cases who had the lupus anticoagulant. There is no controlled clinical trial data yet available 51 to determine whether the new anti-Xa or thrombin inhibitors will be noninferior or superior to warfarin in long-term anticoagulant treatment of atherothrombosis. However, dabigatran and rivaroxaban failed to prevent thromboembolism in 3 previously reported cases having the antiphospholipid syndrome. 52
Strengths of our study include a large cohort with very early CVD, further characterized by a detailed study of thrombophilia. A limitation of our study was that factors VIII, XI, and antigenic proteins C, S, and antithrombin could be measured in only ∼30% of our patients and the lupus anticoagulant in only 24%, limited by third-party insurance coverage for thrombophilia evaluation. Nevertheless, despite the lack of power related to these small numbers, cases with premature CVD were more likely than normal controls to have high factor VIII (P = .007), low antigenic free protein S (P = .032), and the lupus anticoagulant (P = .035).
Our current data suggest that thrombophilia is a pervasive and important contributory risk factor for premature CVD ≤ age 45 and, speculatively, offers a therapeutic target if premature CVD progresses despite maximal tolerated conventional medical–surgical therapy.
Footnotes
Appendix A
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
