Abstract
In 35 patients with 116 severe premature cardiovascular disease (CVD) events (median age: 48 years), 14 having worsening CVD despite maximal intervention, we evaluated thrombophilia and speculated that anticoagulation might arrest–reverse progressive thrombophilic–atherothrombotic CVD. Thrombophilia–hypofibrinolysis in the 35 patients was compared to 110 patients with venous thromboembolism (VTE) without CVD and to 110 healthy normal controls. Efficacy–safety of anticoagulation was prospectively assessed in 14 of the 35 patients whose CVD worsened over 2 years despite maximal medical–surgical intervention. At entry on maximally tolerated lipid-lowering therapy, median low-density lipoprotein was 88 mg/dL. Measures of thrombophilia–hypofibrinolysis in the 35 cases differed from 110 VTE controls only for the lupus anticoagulant, present in 6 (21%) of 28 cases versus 4 (4%) of 91 VTE controls (P = .01), and for high anticardiolipin antibodies (ACLAs) immunoglobulin G, 5 (14%) of 35 cases versus 4 of 108 VTE controls (4%), P = .04. The 14 patients who were anticoagulated differed from 110 VTE controls only for the lupus anticoagulant, 38% versus 4%, P = .001, and for high lipoprotein (a), 46% versus 17%, P = .028, respectively. The 14 patients with atherothrombosis having inexorably worsening CAD despite maximal medical–surgical therapy were anticoagulated for 6.5 years (median), with clinical CVD progression arrested in 12 (86%), and all 12 became asymptomatic. In the 35 patients with premature CVD, thrombophilia was pervasive, comparable to or more severe than in VTE controls without CVD. When CVD progressively worsens despite maximal intervention, thrombophilia and atherosclerosis (atherothrombosis) are commonly concurrent, and the downhill course of CVD may be arrested–stabilized by anticoagulation.
Keywords
Introduction
Despite conventional intervention therapy with dual platelet inhibition, antihypertensives, β-blockers, lipid-lowering drugs, angioplasty, stents, and coronary artery bypass grafts (CABGs), patients with symptomatic atherothrombosis (thrombus formation on atherosclerotic plaque) 1 have high rates of recurrent vascular events and rehospitalization. 2 –5 Although thrombophilia is thought to be primarily associated with venous thrombosis, atherothrombotic coronary artery disease (CAD) shares risk factors and pathophysiology with venous thromboembolism (VTE), deep vein thrombosis (DVT), and pulmonary embolism (PE). 6 Factor V Leiden heterozygosity is more common in diabetic patients with peripheral artery disease who had unsuccessful percutaneous transluminal angioplasty than in those with a successful procedure. 7 Factor V Leiden, prothrombin G20210A, and methylenetetrahydrofolate reductase (MTHFR) C677T mutations increase the risk of myocardial infarction (MI) and ischemic stroke. 8 Given the development of the Xa inhibitors, 9 –11 combined antiplatelet and anticoagulant therapy, particularly for secondary prevention, is now more commonly being used in patients with atherothrombosis and worsening CAD. The atherothrombotic interaction of hyperlipidemia and thrombophilia is heightened by exogenous estrogen therapy. 12 –14
Our specific aim was to assess atherothrombosis in 35 patients with hyperlipidemia–thrombophilia with 116 atherosclerotic cardiovascular disease (ASCVD) events at a median age of 48 years, 14 of whom had progressively worsening CAD despite maximal medical–surgical intervention. Our hypothesis was that thrombophilia interacted with atherosclerosis (atherothrombosis) leading to severe premature CAD and that anticoagulation might arrest, stabilize, or reverse symptomatic CAD in 14 patients with relentless downhill progression of CAD despite maximal medical–surgical therapy.
Patients and Methods
Patients: Study Design
Studies were carried out following a protocol approved by the institutional review board with signed informed consent. The 35 patients (25 men and 10 women), comprising a sequential case series, were serially referred by their cardiologists because of premature and/or rapidly worsening CAD. Of these 35 patients, 14 (11 men and 3 women) were prospectively anticoagulated because of rapidly progressive CAD worsening despite maximal medical–surgical treatment including, angioplasty, stenting, CABG, antihypertensives, antiplatelet therapy, β-blockers, and maximal lipid-lowering therapy.
Our 14 thrombophilic patients also received either warfarin or off-label novel oral anticoagulant drugs (NOACs; apixaban, rivaroxaban, or dabigatran), 15 since they had thrombophilia comparable to or more severe than our VTE control group and because they had progressive CAD worsening over weeks to months despite maximal medical and surgical therapy. The NOACs have been studied along with dual platelet therapy in the treatment of acute coronary syndrome (ACS), but only rivaroxaban has been approved in Europe and none yet approved in the United States. 16 –18 However, in the Pioneer-AF-ACS study, 19 with PCI-stent placement in atrial fibrillation, either low-dose rivaroxaban (15 mg/d) plus a P2Y12 inhibitor for 12 months or very-low-dose rivaroxaban (2.5 mg/d) plus dual antiplatelet therapy (DAPT) for 1, 6, or 12 months was associated with a lower rate of clinically significant bleeding than was standard therapy with a vitamin K antagonist plus DAPT for 1, 6, or 12 months. Our use of warfarin or NOACs in our 14 thrombophilic–hypofibrinolytic patients allowed assessment of whether anticoagulation at levels conventionally used to reduce VTE events would slow the progression of or stop the patients’ otherwise inexorable downhill cardiac courses.
Controls
Thrombophilia–hypofibrinolysis measures in the 35 cases were compared to 110 thrombophilic controls with VTE, but free of CAD (53 men and 57 women), 20 and were compared to 110 previously reported healthy, normal controls 21 (48 men and 62 women) free of coronary artery, cerebrovascular, or peripheral vascular diseases and free of VTE.
Measures of Thrombophilia and Hypofibrinolysis
Polymerase chain reaction measures of the factor V Leiden, prothrombin G2010A, MTHFR, and plasminogen activator inhibitor 4G4G (PAI-1 gene) mutations were done along with serologic measures of homocysteine, factors VIII and XI, antigenic proteins C, total S, free S, and antithrombin III, lipoprotein (a) [Lp(a)], lupus anticoagulant, and anticardiolipin antibodies (ACLAs) immunoglobulin (Ig) G and IgM, using previously reported methods. 13,21 Serologic tests were done before anticoagulation. Plasminogen activator inhibitor 1 activity was not measured.
Platelet function tests were not done because all 14 patients with inexorable progression of angina and CAD were receiving antiplatelet therapy at the time of referral, and we could not stop such therapy to subsequently carry out platelet function tests.
Laboratory and Clinical Follow-Up During Anticoagulant Therapy
While on anticoagulant therapy, patients were examined every 2 to 3 months in our center, with interval history and physical examination and measurement of complete blood count, platelet count, fasting glucose, renal function, liver function, and lipid profiles. Cholesterol and triglyceride (TG) values measured in mg/dL. To convert cholesterol to mmol/L, multiply by 0.0259. To convert TG to mmol/L, multiply by 0.0113. Patients on warfarin had separate follow-up of their international normalized ratio (INR) levels every 2 months with an INR goal of 2.0 to 3.0.
Statistical Methods
Distributions of coagulation measures in cases versus controls were compared using Fisher exact test. Changes in lipids and lipoprotein cholesterols during treatment in the 35 patients were assessed using paired Wilcoxon tests.
Power and sample size calculations for comparisons of thrombophilia between cases and controls were carried out based on factor V Leiden heterozygosity in 31% of cases versus 2% of normal controls, versus 12% of controls who had sustained VTE. 22 To compare cases with atherothrombosis versus normal controls, there would need to be ≥25 patients in each group to detect differences at a significance level of .05 with power 80%. To compare factor V Leiden heterozygosity in cases with atherothrombosis versus VTE controls, there would need to be ≥73 in each group to detect differences at a significance level of 0.05 with power 80%.
Results
Patients With Atherothrombotic Events
At study entry, the median age of 35 patients was 51, total cholesterol (TC) 169 mg/dL, TG 125 mg/dL, high-density lipoprotein cholesterol (HDLC) 45 mg/dL, and low-density lipoprotein cholesterol (LDLC) 88 mg/dL, reflecting aggressive diet and drug therapy anteceding referral to our center (Table 1). At the end of the study period, with median treatment duration of 26 months, median TC was 133 mg/dL (P < .0001), TG 115 mg/dL, HDLC 45 mg/dL, and LDLC 65 mg/dL (P < .01; Table 1). When compared to the 110 VTE controls, the 35 patients differed from VTE controls only for lower HDLC (Table 1). The comparable total and LDLC in patients and VTE controls reflected the standard-of-care lipid-lowering therapy given to the 35 patients at study entry (Table 1). In the 35 cases, at the end of our treatment period, median TC was lowered from 169 to 133 mg/dL (P < .001) and median LDLC from 88 to 65 mg/dL (P < .01).
Lipids and Lipoprotein Cholesterols in 35 Patients With Atherothrombosis at Study Entry and at the End of the Treatment Periods.a
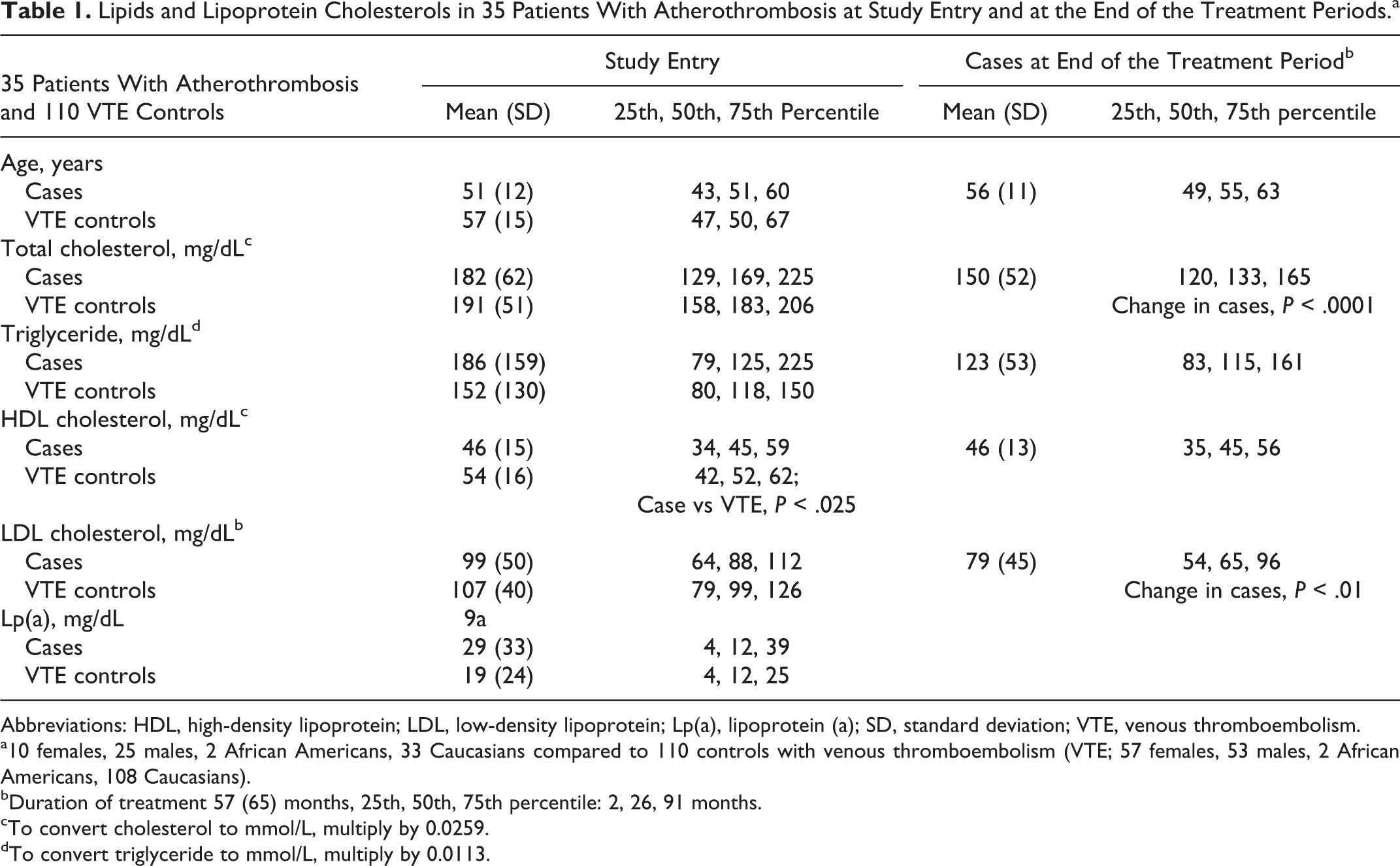
Abbreviations: HDL, high-density lipoprotein; LDL, low-density lipoprotein; Lp(a), lipoprotein (a); SD, standard deviation; VTE, venous thromboembolism.
a10 females, 25 males, 2 African Americans, 33 Caucasians compared to 110 controls with venous thromboembolism (VTE; 57 females, 53 males, 2 African Americans, 108 Caucasians).
bDuration of treatment 57 (65) months, 25th, 50th, 75th percentile: 2, 26, 91 months.
cTo convert cholesterol to mmol/L, multiply by 0.0259.
dTo convert triglyceride to mmol/L, multiply by 0.0113.
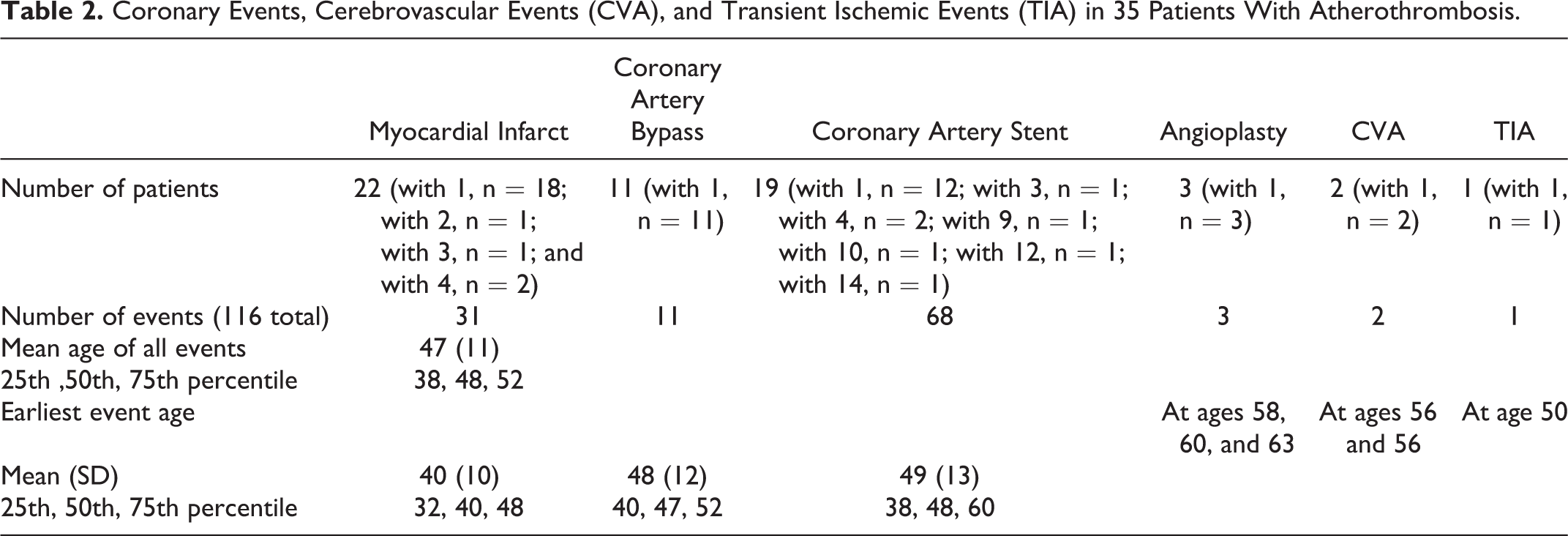
The 35 patients had sustained 116 ASCVD events, with events occurring at a median age of 48 years (Table 2). Twenty-two patients had sustained 31 MIs, 11 had 11 CABG surgeries, and 19 had 68 stents placed (Table 2). The cohort had severe premature CAD, with the median age of 40 for MI, 47 for CABG, and 48 for coronary artery stenting (Table 2).
Coronary Events, Cerebrovascular Events (CVA), and Transient Ischemic Events (TIA) in 35 Patients With Atherothrombosis.
The 35 patients were comparable to 110 thrombosis controls without CAD who had developed VTE for measures of thrombophilia–hypofibrinolysis but were more likely to have high ACLA IgG (14% vs 4%, P = .04) and the lupus anticoagulant (21% vs 4%, P = .01; Table 3).
Coagulation Disorders in 35 Patients With Atherothrombosis (25 Men and 10 Women), Compared to 110 Patients With Venous Thromboembolism (VTE; 53 Men and 57 Women) and With 110 Healthy Normal Controls (48 Men and 62 Women).
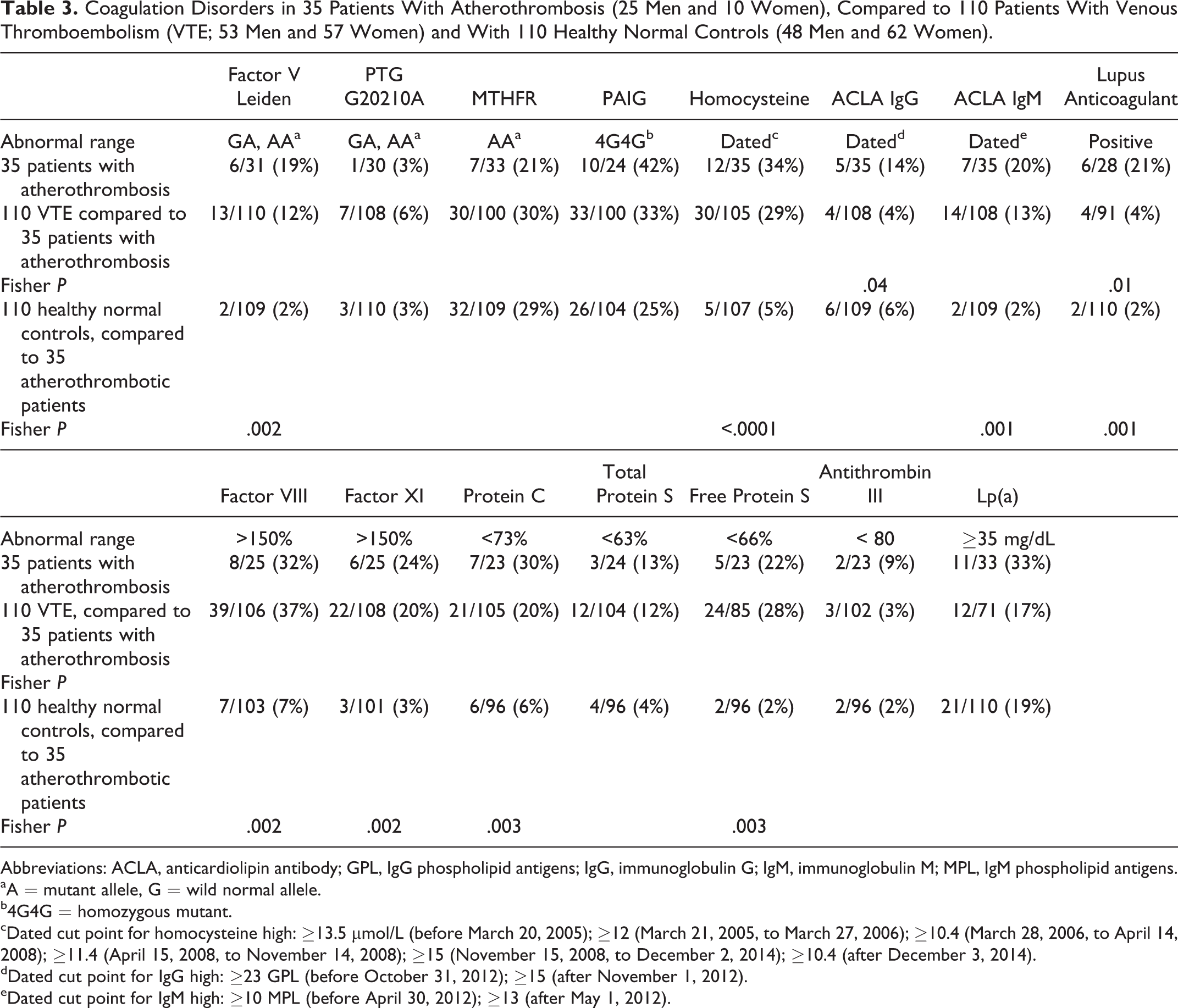
Abbreviations: ACLA, anticardiolipin antibody; GPL, IgG phospholipid antigens; IgG, immunoglobulin G; IgM, immunoglobulin M; MPL, IgM phospholipid antigens.
aA = mutant allele, G = wild normal allele.
b4G4G = homozygous mutant.
cDated cut point for homocysteine high: ≥13.5 µmol/L (before March 20, 2005); ≥12 (March 21, 2005, to March 27, 2006); ≥10.4 (March 28, 2006, to April 14, 2008); ≥11.4 (April 15, 2008, to November 14, 2008); ≥15 (November 15, 2008, to December 2, 2014); ≥10.4 (after December 3, 2014).
dDated cut point for IgG high: ≥23 GPL (before October 31, 2012); ≥15 (after November 1, 2012).
eDated cut point for IgM high: ≥10 MPL (before April 30, 2012); ≥13 (after May 1, 2012).
The 35 patients were more likely than 110 healthy normal controls to have factor V Leiden (19% vs 2%, P = .002), high homocysteine (34% vs 5%, P < .0001), high ACLA IgM (20% vs 2%, P = .001), lupus anticoagulant (21% vs 2%, P = .001), high factor VIII (32% vs 7%, P = .002), high factor XI (24% vs 3%, P = .002), low protein C (30% vs 6%, P = .003), and low free protein S (22% vs 2%, P = .003; Table 3).
Of the 35 patients, there were 14 with progressively worsening CAD despite maximal medical–surgical therapy, 13 of whom had ≥1 premature MI and 1 (case 12; Table 4) who had right coronary artery occlusion without MI at age 55. These 14 patients’ first MI occurred at median age 47 (25th-75th percentile, 37-52 years). Their current median age was 60. Of the 14 patients, 7 had multiple stents, 8 had CABG, and 6 had previous VTE. Despite antihypertensive, antiplatelet, β-blocker, maximal tolerated lipid-lowering therapy, angioplasty, multiple cardiac stents, and CABG, over a median 2-year period (25th-75th percentile, 1-9 years), CAD was steadily progressive in the 14 patients (Table 4).
Duration of Unstable Coronary Artery Disease (CAD) Before Anticoagulation, Duration of Anticoagulation, and Clinical Outcome on Anticoagulation in 14 Patients With Atherothrombosis and Inexorably Progressing CAD Despite Maximal Medical–Surgical Therapy.
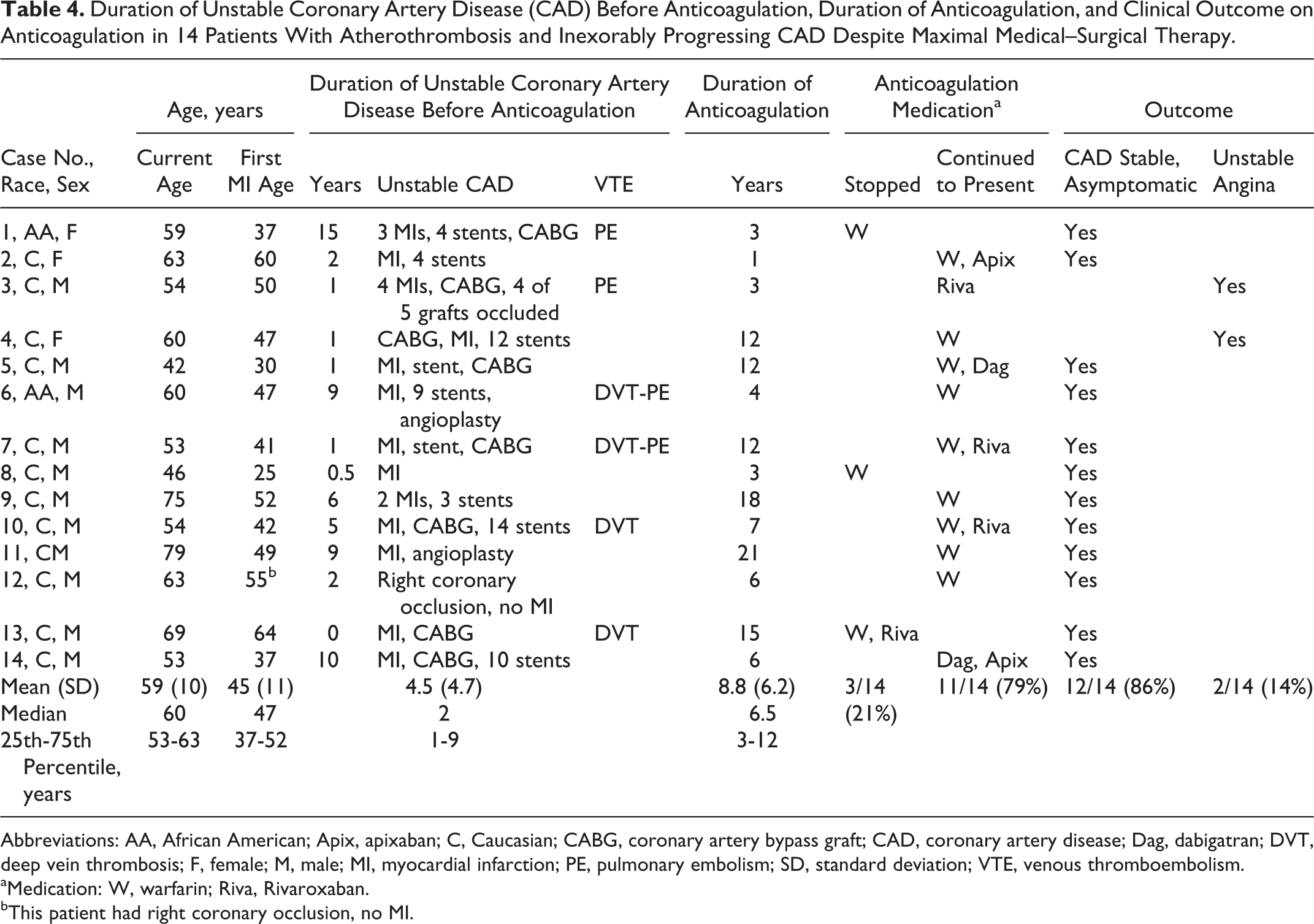
Abbreviations: AA, African American; Apix, apixaban; C, Caucasian; CABG, coronary artery bypass graft; CAD, coronary artery disease; Dag, dabigatran; DVT, deep vein thrombosis; F, female; M, male; MI, myocardial infarction; PE, pulmonary embolism; SD, standard deviation; VTE, venous thromboembolism.
aMedication: W, warfarin; Riva, Rivaroxaban.
bThis patient had right coronary occlusion, no MI.
None of the 14 patients were current cigarette smokers; however, 2 (cases 2 and 6) had >10-year antecedent smoking history of at least 1 pack per day. On maximum tolerated lipid-lowering therapy during our follow-up, the 14 patients’ blood lipids were lowered to and below targeted goals 23 ; median LDLC was 59 mg/dL, TG 99 mg/dL, and HDLC 47 mg/dL.
All 14 patients with progressive CAD were found to have ≥1 thrombophilia–hypofibrinolysis. Three (21%) patients had 1 coagulation disorder, 2 (14%) had 2 disorders, and 9 (64%) had ≥3 disorders (Table 5). The 14 patients were more likely than 110 VTE controls to have the lupus anticoagulant, 38% versus 4%, P = .001, and to have high Lp(a), 46% versus 17%, P = .028 (Table 5).
Coagulation Disorders in 14 Patients With Atherothrombosis, Compared to 110 Controls With Venous Thromboembolism.
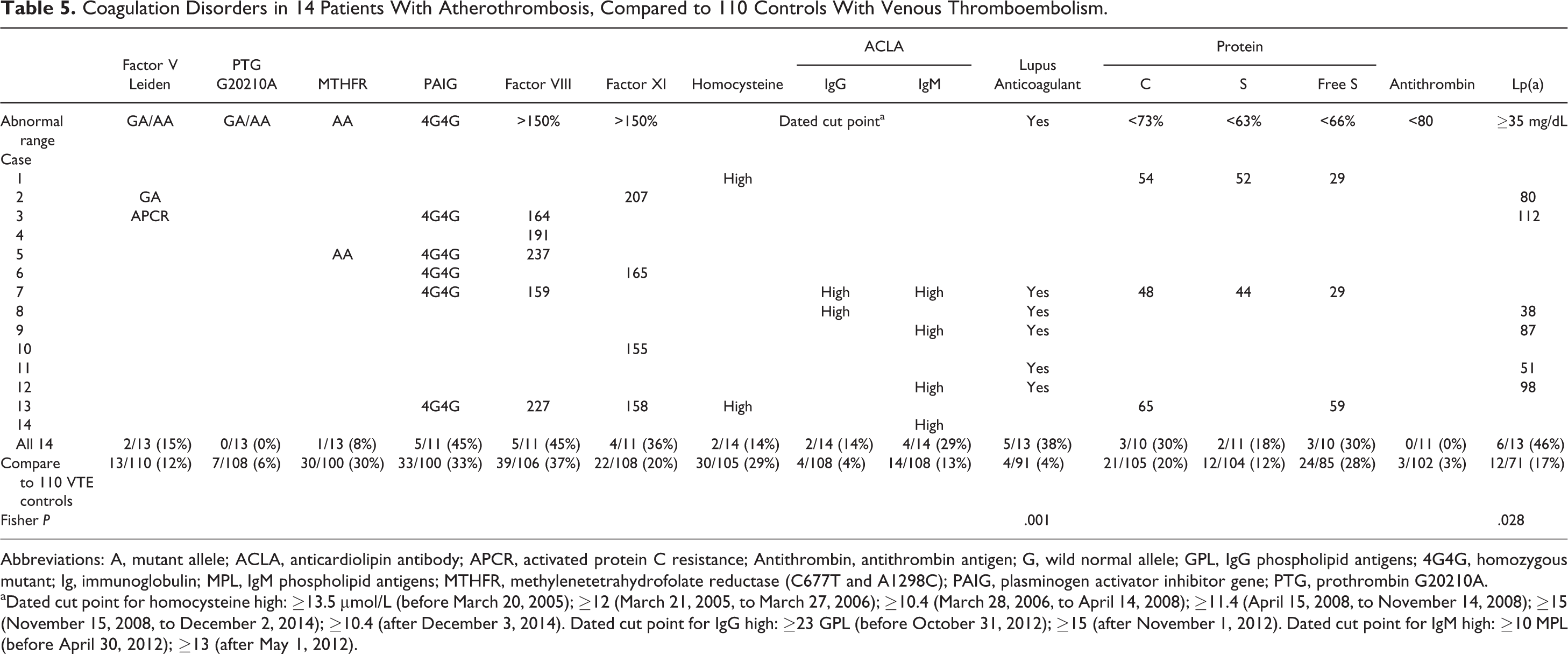
Abbreviations: A, mutant allele; ACLA, anticardiolipin antibody; APCR, activated protein C resistance; Antithrombin, antithrombin antigen; G, wild normal allele; GPL, IgG phospholipid antigens; 4G4G, homozygous mutant; Ig, immunoglobulin; MPL, IgM phospholipid antigens; MTHFR, methylenetetrahydrofolate reductase (C677T and A1298C); PAIG, plasminogen activator inhibitor gene; PTG, prothrombin G20210A.
aDated cut point for homocysteine high: ≥13.5 µmol/L (before March 20, 2005); ≥12 (March 21, 2005, to March 27, 2006); ≥10.4 (March 28, 2006, to April 14, 2008); ≥11.4 (April 15, 2008, to November 14, 2008); ≥15 (November 15, 2008, to December 2, 2014); ≥10.4 (after December 3, 2014). Dated cut point for IgG high: ≥23 GPL (before October 31, 2012); ≥15 (after November 1, 2012). Dated cut point for IgM high: ≥10 MPL (before April 30, 2012); ≥13 (after May 1, 2012).
Response to Anticoagulant Therapy
Before anticoagulation, the 14 patients’ CAD had been symptomatic and worsening for a median of 2 years, with the 25th to 75th percentile range 1 to 9 years (Table 4). Before anticoagulation, recurrence and worsening of symptoms in the 14 patients who were ultimately anticoagulated was in the form of thrombotic arterial occlusion.
Of the 14 patients, 11 had been treated with dual platelet therapy. Anticoagulation was added to the DAPT in these 11 patients. In 6 of the 11 patients, the cardiologist stopped the clopidogrel due to higher bleeding risk. Patients 8 and 11 were on aspirin only as no DAPT was recommended at that time, and they were doing well on aspirin and anticoagulation. Patient 13 was already on warfarin (though compliance was an issue) and thus only aspirin was added. In these 3 patients, no stents were placed.
While on anticoagulation, in 12 of the 14 patients, their previous progressive downhill cardiac course was arrested, then stabilized, and they became asymptomatic, while CAD persisted in 2 patients (patients 3 and 4; Table 4). The median duration of anticoagulation was 6.5 years. The 14 patients treated with anticoagulants are summarized subsequently and in Tables 4 and 5.
Patient 1
This 59-year-old African American female had a PE at age 41, sustained 3 MIs (at ages 37, 38, and 42), had angioplasties with 4 stents placed at ages 37, 38, 42, and 48, and despite being on aspirin and clopidogrel, required CABG at age 52. She was found to have high homocysteine, low protein C, and low total and free protein S (Table 5). After enoxaparin lead-in, she was treated with warfarin starting at age 52, and her progressive angina improved and resolved during a 3-year period of anticoagulation. Anticoagulation was stopped at age 55, and she has remained asymptomatic (Table 4).
Patient 2
This 63-year-old Caucasian female had an MI at age 60, with 4 stents (3 at age 60 and 1 at age 61), but had intractable angina despite aspirin–clopidogrel therapy. One of her pregnancy was complicated by hypertension and preeclampsia. She was found to be heterozygous for factor V Leiden mutation, had high factor XI, and had high Lp(a) (Table 5). After starting warfarin at age 62, she became asymptomatic 6 months later and continues to be asymptomatic after switching to apixaban 2.5 mg twice daily in early 2016 (Table 4).
Patient 3
This 54-year-old Caucasian male had a PE in 2006 and an MI showing multivessel disease requiring 5-vessel CABG at age 50. Following CABG, he had another 3 MIs. While on aspirin–clopidogrel, coronary angiography a few months after the CABG revealed that 3 of the 5 grafts were occluded with aspiration thrombectomy and thrombolysis of a fourth graft. He had angina with minimal exertion and was chair-bound because of angina. He was found to have activated protein C resistance, high factor VIII, 4G4G homozygosity for the PAI-1 gene, and high Lp(a) (Table 5). At age 51, he was started on rivaroxaban 20 mg daily with aspirin only. After 6 months on rivaroxaban, he was able to climb stairs without chest pain and went back to work as a roofer. Currently, he is working full time as a welder and roofer. He continues to have mild intermittent angina pain as of 2016 but has refused further testing as pain is milder and resolves rather quickly (Table 4).
Patient 4
This 60-year-old Caucasian female had a 5-way CABG in 2003 (age 47) but then had an MI 2 weeks later despite being on aspirin. Seven stents were placed from age 47 to 48, and clopidogrel was started during that time. She was found to have high factor VIII (Table 5). Warfarin was started at age 48. She had angina on exertion and orthopnea as of August 2015, with 3 stents placed in right coronary artery. Her symptoms were not completely resolved, with additional 2 stents placed in the left anterior descending artery (LAD) in December 2015. Since then orthopnea and angina pain resolved with minimal dyspnea on exertion (Table 4).
Patient 5
This 42-year-old Caucasian male army ranger had an MI in 2004 (age 30) with stent placement and subsequent CABG. During that time, while on aspirin and clopidogrel, he was found to have high factor VIII and was homozygous for the MTHFR C677T mutation and the PAI-1 4G4G mutation (Table 5). With clopidogrel discontinuation, he was then put on warfarin from 2004 to 2012 and later on was switched to dabigatran 150 mg twice daily. He became asymptomatic after 1 month on warfarin and remains asymptomatic (Table 4).
Patient 6
This 60-year-old African American male had an MI and angioplasty with 9 stents between age 47 and 49. He sustained an unprovoked venous blood clot in his left arm at age 47 and was treated temporarily with warfarin. Despite aspirin–clopidogrel therapy, during that 2-year period, he had angina on minimal exertion. Clopidogrel was discontinued at age 53. He then had DVT-PE at age 56. He was found to have high factor XI and PAI-1 4G4G homozygosity (Table 5). Warfarin was started at that time indefinitely, and as of the present, he is asymptomatic except for an episode of superficial vein thrombosis in right leg with INR of 1.0 two years ago (Table 4).
Patient 7
This 53-year-old Caucasian male had DVT-PE and an MI with stent placement in 2003 (age 41) and was placed on clopidogrel and aspirin. He was found to have the lupus anticoagulant, PAI-1 4G4G homozygosity, high factor VIII, high ACLA IgG and IgM, and low proteins C, S, and free S (Table 5). After enoxaparin lead-in, he started warfarin in 2004 at age 42 and was taken off of clopidogrel at that time. He had progressive angina over the next 10 years and CABG in 2014. Due to persistent angina, 1 month after CABG, he was switched to rivaroxaban (20 mg/d) at age 52. He has been asymptomatic since being on rivaroxaban (Table 4).
Patient 8
This 46-year-old white male had an MI at age 25, without stent placement, placed on aspirin, but continued to have persistent angina on exertion after the MI. He was found to have the lupus anticoagulant and high ACLA IgG (antiphospholipid antibody syndrome [APL]; Table 5). From 1995 (age 25) to 1998 (age 28) on warfarin, he became asymptomatic. From age 29 to the present, without further anticoagulation, he remains asymptomatic (Table 4).
Patient 9
This 75-year-old white male had his first MI at age 52 (1992) at which time he was placed on aspirin. A second MI with 2 stents occurred in 2004 after which clopidogrel was added. He was found to have the lupus anticoagulant at age 52 at the time of first MI, high ACLA IgM, and high Lp(a) (Table 5). Asymptomatic on warfarin, in 2014, he reduced his dose of warfarin, INR dropped to 1.03, and he required 1 new stent. On warfarin for 18 years since 1998, he has been asymptomatic in the past 2 years (Table 4).
Patient 10
This 54-year-old white male had an MI in 2004 with 4 stents placed between 2004 and 2008 while on aspirin–clopidogrel therapy. He had a 5-way CABG at age 46. Two months after surgery, he had 5 stents placed due to progressive angina. He was found to have high factor XI (Table 5) and was put on warfarin. Chest pain continued and he required more stents, with the last 2 placed on July 2014 (total of 14 stents) at age 52, at which time he also sustained a DVT while being off warfarin. He was then switched to rivaroxaban 20 mg/d in late 2014 and has been asymptomatic since (Table 4).
Patient 11
This 79-year-old white male sustained an MI at age 49 (1986), was placed on aspirin, and had angioplasty 9 years later at age 58 (1995) without stent placement. Unstable angina persisted after the angioplasty. The lupus anticoagulant and high Lp(a) were found at age 58 (Table 5), warfarin was started, and he became asymptomatic. He has been on warfarin since and remains asymptomatic (Table 4).
Patient 12
This 63-year-old white male was found to have 60% right coronary artery occlusion in 2008 (age 55) with persistent angina. At that time, he was placed on aspirin–clopidogrel. He was found to have high Lp(a), the lupus anticoagulant, and high ACLA IgM (APL; Table 4). He was placed on warfarin at age 57 (clopidogrel discontinued) and has been asymptomatic on anticoagulation for 6 years (Table 5).
Patient 13
This 69-year-old white male had a DVT 15 years ago at age 54 after an abdominal surgery and was placed on warfarin at that time. He developed another DVT once warfarin was discontinued, and so it was resumed indefinitely. He had 2 silent MIs and 4-way CABG in 2011 at age 64, at which time aspirin was started. Warfarin compliance was an issue due to mild epistaxis and rectal bleeding. He was found to have 4G4G homozygosity for the PAI-1 gene, high factor VIII, high factor XI, low protein C, and low free S (Table 5). On warfarin until late 2014, he then was switched to rivaroxaban (10 mg/d) with resolution of anginal symptoms (Table 4). At age 67, he took himself off rivaroxaban due to recurrence of nose bleeds. A repeat angiogram showed patent grafts.
Patient 14
This 53-year-old Caucasian male had an MI at age 37 (2000) and, after 9 stents while on aspirin and clopidogrel, required CABG at age 46. He was found to have high ACLA IgM (Table 5). Dabigatran, 150 mg twice daily, was started in 2010 (age 47) because of atrial fibrillation and had his last stent placed at age 50. At that time, he was placed on
Anticoagulant Safety
Of the 14 patients, 11 had been treated with dual platelet therapy. Five patients had concurrent DAPT plus anticoagulation and 9 had aspirin plus anticoagulation. The duration of anticoagulation was for a median of 6.5 years, 25th to 75th percentile range of 3 to 12 years (Table 4). None of the 14 patients had major bleeding episodes, and there was only 1 patient with minor bleeds requiring intermittent cessation of anticoagulation. There were no systematic changes in liver or renal function during anticoagulation treatment.
Discussion
Atherothrombosis in the form of CAD, cardiovascular disease (CVD), and peripheral artery disease is the leading factor in mortality on a global scale. 24 Prior atherothrombotic events confer an increased risk of subsequent events, and patients with atherothrombosis have the highest risk of repeat events when compared to patients with stable disease and those with risk factors without atherothrombotic events. 5 Patients with prior arterial disease have more than double the risk of CVD death, MI, stroke, or hospitalization for atherothrombotic events within 1 year when compared to a population with multiple risk factors only, 14.41% versus 5.31%, respectively. 3 Additionally, even in the total patient population with previous arterial disease, within 1 year, 12.81% had a major event (CVD death, MI, and stroke) or was hospitalized for CVD event or revascularization procedure. 3
Increasingly, it is being realized that thrombophilic procoagulants associated with VTE can also be associated with arterial thrombi. 6 –8,12 –14 In young patients (age ≤50 male or ≤55 female), particularly with no apparent cause of arterial occlusion, with early cerebral, coronary, and peripheral vascular ischemia, thrombophilia may play a predisposing role. 25 –28 Arterial thrombotic events have been associated with factor V Leiden, prothrombin (G20210A), and MTHFR C677T mutations. 29 Atherothrombosis has also been associated with primary antiphospholipid syndrome, 30 reduced protein C activity, 31 and hyperhomocysteinemia. 32 –34 Additionally, premature atherothrombosis has been linked to elevated factor VIII. 35 The clinical importance of thrombophilia in arterial thrombosis is emphasized by the superiority of oral anticoagulants versus aspirin alone in secondary prevention of acute arterial ischemia. 36,37 In carriers of hemophilia, a mild decrease in coagulability has a significant protective effect against fatal ischemic heart disease. 38
There is growing literature on antithrombotic therapy in the acute setting of CAD (ACS), 39 including studies of apixaban 40 and rivaroxaban. 41 In the ATLAS-ACS2-TIMI 151 trial, 18 addition of rivaroxaban 2.5 mg/d to DAPT provided significant reduction in cardiovascular events as well as total mortality. Warfarin has been the mainstay of therapy in patients sustaining thrombosis and having the lupus anticoagulant-APL syndrome 42 –44 and was used successfully in the current study in 4 of the 5 cases who had the lupus anticoagulant. There is no controlled clinical trial data yet available 45 to determine whether the new Xa or thrombin inhibitors will be noninferior or superior to warfarin in long-term anticoagulant treatment of atherothrombosis. However, in the Pioneer-AF-ACS study 19 of stent placement in patients with atrial fibrillation, low-dose rivaroxaban (15 mg/d) plus a P2Y12 inhibitor for 12 months or very-low-dose rivaroxaban (2.5 mg/d) plus DAPT for 1, 6, or 12 months had lower rates of clinically significant bleeding than a vitamin K antagonist plus DAPT for 1, 6, or 12 months. However, dabigatran and rivaroxaban failed to prevent thromboembolism in 3 previously reported cases having the APL syndrome. 46
In 14 hyperlipidemic–atherothrombotic patients having severe premature CAD (first MI at median age 47 years) with progressive downhill cardiac status despite maximal medical–surgical therapy, concurrent thrombophilia and atherothrombosis were common. Consistent with the sharing of atherosclerotic and thrombotic risk factors, 6 the 14 atherothrombotic cases were equally or more severely thrombophilic than 110 VTE controls free of CAD and, in addition, were even more likely to have the lupus anticoagulant (38% vs 4%, P = .001) and high Lp(a) (46% vs 17%, P = .028). Moreover, 6 of the 14 atherothrombotic patients with progressively worsening CAD had separately sustained VTE, further testimony to their thrombophilia being associated with both venous and arterial thrombosis. 7,8,12 –14,47 In the current pilot study, the otherwise relentless downhill clinical course of CAD could be safely arrested, stabilized, and, in 12 of 14 patients, reversed by anticoagulant therapy in concert with continued maximal medical cardiac–surgical therapy.
Of note, 3 patients (7, 10, and 13) had progressing coronary ischemia symptoms despite being on warfarin but became asymptomatic once they were switched to rivaroxaban. The improvement in rivaroxaban with consistent anticoagulation effect
48
may reflect previous failure for these 3 patients to remain in a therapeutic INR range, which is commonly seen with warfarin.
49
We also observed resolution of coronary ischemic symptoms in patient 14 when dabigatran was switched to apixaban. However, shortly beforehand,
Despite concurrent use of aspirin or DAPT, and either warfarin or novel oral anticoagulants, there were no major bleeds over a median of 6.5 years of follow-up in the 14 patients.
Two questions arising from this pilot study are (1) how to interpret the lack of bleeding events and (2) how to interpret the improvement in patients on rivaroxaban as opposed to conventional warfarin treatment. The small sample size means that the lack of bleeding events has to be interpreted cautiously, especially considering large clinical trials (ATLAS ACS 2-TIMI 51 and APPRAISE-2) revealing increased bleeding events. 51 –53 Although again hampered by the small sample size, we speculate that the improvement in 3 patients switched from warfarin to rivaroxaban and 1 switched from warfarin to dabigatran contrasted to conventional warfarin reflects the benefits of much more stable anticoagulation delivered by rivaroxaban or dabigatran than warfarin. 49 Enthusiasm for the overall outcome of this pilot study, improvement in CAD symptoms and stable CAD after anticoagulation in 12 of 14 patients, is reduced by the small sample size. Despite arresting, stabilizing, or reversing progressively worsening symptomatic CAD, another limitation is that the major outcomes measured were symptoms and clinical course as opposed to repetitive coronary artery imaging.
Our study was limited by being “open label,” rather than an optimal double-blind design with randomization to anticoagulation or placebo. However, it would be extremely difficult to match anticoagulant versus placebo cases by the specific thrombophilias, by the nature of the antecedent CAD, and by the multiple additional variables (hypertension, diabetes mellitus, etc) contributing to progressive worsening of CAD. A multicenter approach would be needed to accumulate enough patients to provide adequate power and sample size to examine efficacy and safety.
Conclusion
In young individuals, median age 48 in our study, with inexorably progressing CAD despite maximal medical–surgical interventions, investigation into familial and acquired thrombophilias is important. Those who are found to have thrombophilia–hypofibrinolysis may benefit from anticoagulation therapy in addition to maximal conventional medical–cardiac–surgical therapy, as seen in 12 of our 14 atherothrombotic patients where anticoagulation safely arrested, stabilized, and reversed previously worsening symptomatic CAD.
Footnotes
Authors’ Note
Matan Rothschild, Vybhav Jetty, Ping Wang, Marloe Prince, Naila Goldenberg, and Charles J. Glueck contributed to study design, data accumulation, editing, and writing. Christopher Mahida contributed to data accumulation, editing, and writing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for the study came from the Lipoprotein Research Endowment Fund of the Jewish Hospital of Cincinnati, which played no role in study design, collection, analysis and interpretation of the data, in the writing of the report, and in the decision to submit for publication.
