Abstract
Endothelial damage has been implicated in the pathogenesis of vascular complications in β-thalassemia intermedia (β-TI). Soluble fms-like tyrosine kinase 1 (sFLT-1) is a member of the vascular endothelial growth factor receptor (VEGFR) family. Soluble fms-like tyrosine kinase 1 is an antiangiogenic protein that induces endothelial dysfunction by adhering to and inhibiting VEGF and placenta growth factor. The aim of this study was to assess the level of sFLT-1 in 35 children and adolescents with β-TI, correlating it with markers of hemolysis and iron overload as well as cardiopulmonary complications. Patients were studied focusing on the history of cardiac disease, splenectomy, transfusion, chelation/hydroxyurea therapy, serum ferritin, and sFLT-1 levels. Echocardiography and measurement of carotid intima–media thickness (CIMT) were done for all participants. Soluble fms-like tyrosine kinase 1 was significantly higher in TI patients compared to the control group (median [interquartile range], 110 [80-155] pg/mL versus 70 [60-90] pg/mL; P < .001). Splenectomized patients and those who had pulmonary hypertension risk or heart disease had higher sFLT-1 levels than those without (P < .001). The sFLT-1 cutoff value that differentiates patients with and without pulmonary hypertension risk or heart disease was determined. Soluble fms-like tyrosine kinase 1 was lower among patients who received chelation therapy and/or hydroxyurea. Significant positive relations were observed between sFLT-1 and lactate dehydrogenase, serum ferritin, liver iron concentration, tricuspid regurgitant jet velocity, and CIMT. We suggest that sFLT-1 represents a link between angiogenesis, endothelial dysfunction, and subclinical atherosclerosis. Measurement of sFLT-1 as a marker of vascular dysfunction in β-TI may provide utility for early identification of patients at increased risk of cardiopulmonary complications.
Keywords
Introduction
β-Thalassemia intermedia (β-TI) is a term developed to describe patients with manifestations that are neither mild enough nor severe enough to be classified in the spectrum’s extremes of β-thalassemia. Three main factors are responsible for the clinical sequelae of TI: ineffective erythropoiesis, chronic anemia, and iron overload. 1
Transfusional iron overload can affect heart function by directly damaging tissue through iron deposition or via iron-mediated effects at other sites. The main cardiac abnormalities reported in patients with thalassemia and iron overload are left ventricular systolic and diastolic dysfunction, pulmonary hypertension, valvulopathies, arrhythmias, and pericarditis. Even though patients with β-TI require fewer transfusions than those with β-thalassemia major (β-TM), they are still at high risk for cardiac complications. 2
Vascular endothelial growth factor (VEGF), a potent hypoxia-regulated factor, plays several roles in angiogenesis, which is a crucial process in the pathogenesis of several inflammatory, autoimmune, and malignant diseases. 3 Soluble fms-like tyrosine kinase-1 (sFLT-1) is a member of the VEGF receptor (VEGFR) family. 4 Soluble fms-like tyrosine kinase-1 is an antiangiogenic protein that inhibits VEGF and placental growth factor (PGF) signaling, attenuating the formation of new blood vessels and promoting maturation of those vessels that have already been created. 5 By adhering to and inhibiting VEGF and PGF, it induces endothelial dysfunction. 4
Endothelial damage has been implicated in the pathogenesis of vascular complications in β-thalassemia. 3 Endothelial dysfunction generally leads to vascular remodeling, 6 namely, arterial structural alterations and, consequently, potential changes in mechanical properties. 7 An important link between endothelial dysfunction and angiogenesis has been shown in patients with TI. 8 We sought to assess the potential value of serum sFLT-1 levels as a biomarker of vascular dysfunction in children and adolescents with β-TI, correlating it with markers of hemolysis and iron overload as well as cardiopulmonary complications and carotid intima–media thickness (CIMT) as the index of subclinical atherosclerosis.
Materials and Methods
This cross-sectional study included 35 patients with β-TI ≤18 years recruited from the regular attendants of the Pediatric Hematology Clinic, Pediatric Hospital, Ain Shams University. Thirty-five age- and sex-matched healthy participants were enrolled as a control group. An informed consent was obtained from the guardian of each patient or control before participation. The procedures applied in this study were approved by the Ethical Committee of Human Experimentation of Ain Shams University and are in accordance with the Helsinki Declaration of 1975.
All patients were diagnosed with TI based on criteria previously described. 9,10 Exclusion criteria were symptomatic heart disease, infection, active hepatitis (serum transaminases >3 times above upper limit of normal), renal impairment, diabetes mellitus, rheumatoid arthritis, or other autoimmune disease. Patients received hydroxyurea as an oral daily dose ranging from 15 to 25 mg/kg/d. Chelation therapy was in the form of deferoxamine infused subcutaneously in a dose that ranged from 30 to 45 mg/kg/d given 5 d/wk or oral deferasirox (DFX) at a dose of 20 to 30 mg/kg/d once daily. For combined chelation therapy, deferoxamine was given for 5 d/wk with daily DFX.
Clinical Assessment
All included patients were subjected to detailed medical history and thorough clinical examination with special emphasis on disease duration, cardiac disease, and spleen status. The main indications for splenectomy were growth retardation or poor health, leukopenia, thrombocytopenia, increased transfusion demand, or symptomatic splenomegaly. 11 For transfusion status, patients were classified as follows: transfusion dependent (patients on regular-interval transfusion protocols [once every 1-3 months for a pretransfusion hemoglobin of ≥8 g/dL] initiated mainly for failure to thrive in childhood, bone deformities, progressive splenic enlargement, persistent worsening anemia, or development of complications during the course of the disease) and transfusion independent (patients who required incidental transfusions for transient severe anemia secondary to infections or surgery). The transfusion received was calculated as the transfusion index: volume of transfused packed red cells in mL per kg body weight per year (expressed as the mean value in the last 3 years).
Sample Collection
Peripheral blood samples were collected on potassium ethylenediaminetetraacetic acid (1.2 mg/mL) for complete blood count (CBC) and hemoglobin analysis. For chemical analysis and enzyme-linked immunosorbent assay (ELISA), clotted samples were obtained, and serum was separated by centrifugation for 15 minutes at 1000g and then stored at −20°C till subsequent use in ELISA.
Laboratory Analysis
Laboratory investigations included CBC using Sysmex XT-1800i (Sysmex, Kobe, Japan) with the assessment of mean pretransfusion hemoglobin, examination of Leishman-stained smears for differential white blood cell count, hemoglobin analysis by high-performance liquid chromatography using D-10 (Bio-Rad, Marnes-la-Coquette, France), liver and kidney function tests, markers of hemolysis (lactate dehydrogenase [LDH] and indirect bilirubin) and high-sensitivity C-reactive protein (hs-CRP), as well as serum ferritin on Cobas Integra 800 (Roche Diagnostics, Mannheim, Germany). Patients with any clinical evidence of infection or hs-CRP >10 mg/L were excluded. The serum ferritin level was measured on this autoanalyzer based on a particle-enhanced immunoturbidimetric assay at the start of the study with the calculation of the mean value of the last year prior to the study in order to know the ferritin trend.
Determination of serum levels of sFLT-1 was done by ELISA using Quantikine Human sVEGF R1/Flt-1 Immunoassay (R&D systems, Minneapolis, Minnesota). This assay employs the quantitative sandwich enzyme immunoassay technique. A monoclonal antibody specific for VEGFR-1 has been precoated onto a microplate. In brief, 100 μL of Assay Diluent RD1-68 was added to each well, then 100 μL of standard, control, and samples was added, and wells were covered followed by 2-hour incubation at room temperature on a horizontal orbital microplate shaker. After washing away any unbound substances, 200 μL of VEGFR-1 conjugate (an enzyme-linked monoclonal antibody specific for VEGFR-1) was added to each well, and wells were covered followed by 2-hour incubation at room temperature. Following a wash to remove any unbound antibody–enzyme reagent, 200 μL of substrate solution was added to the wells and incubated for 30 minutes at room temperature away from light. The color developed in proportion to the amount of VEGFR-1 bound in the initial step and then 50 μL of stop solution was added to each well. The optical density of each well was determined within 30 minutes, using a microplate reader set at 450 nm. Intra- and interassay coefficients of variation were 2.9% and 7.8%, respectively. The lower limit of detection of sFLT-1 ranged from 1.5 to 13.3 pg/mL with a mean of 3.5 pg/mL.
Echocardiography
All studied patients were clinically asymptomatic for pulmonary hypertension and cardiovascular abnormalities. Screening for pulmonary hypertension was performed by noninvasive Doppler echocardiography with different modalities (Doppler, 2-D imaging, and M-mode) using Vivid 7 (GE Healthcare, Horten, Norway) to evaluate right and left ventricle (LV) function, pulmonary artery pressure, and tricuspid regurgitant jet velocity (TRV). A TRV ≥2.5 m/s was used as a proxy for patients at risk for pulmonary hypertension. 12,13 Heart disease was defined by systolic LV dysfunction (LV shortening fraction <30% or LV ejection fraction <55%). 14
Measurement of CIMT
All of the carotid scans were performed by a single investigator who was blinded to the clinical data of patients using carotid Doppler ultrasound scanner (Vivid 7; GE Healthcare) with a 7.5-MHz linear array transducer following a predetermined standardized scanning protocol. 15 All ultrasound scans were performed by an experienced vascular operator who was unaware of children’s clinical details. Patients were placed in the supine position having the neck in hyperextension. The location of measurement was standardized in every study at the common carotid artery at 1 cm distance from the carotid bulb. The 3 best quality images for each of the carotid arteries were selected and analyzed. Best quality was defined with those images that produced the most number of points for analysis. For each image, the greatest distance between the lumen–intima interface and media–adventitia interface (intima–media thickness [IMT]) was measured at a minimum of 100 points. The mean and maximum IMT of each image were then averaged to give the final result for each participant.
Assessment of Liver Iron Concentration
Magnetic resonance imaging (MRI) assessment of liver iron concentration (LIC) was done by MRI R2* for transfusion-dependent TI patients (n = 20) in whom repeated transfusions lead to iron accumulation in the liver. It was determined using a single 10-mm slice through the center of the liver scanned at 12 different echo times. 16,17
Statistical Analysis
Analysis of data was done using Statistical Program for Social Science, version 21 (SPSS Inc, Chicago, Illinois). Quantitative variables were described in the form of mean and standard deviation (SD) or median and interquartile range (IQR; 75th and 25th percentiles). Qualitative variables were described as number and percentage. Kolmogorov-Smirnov test was used for testing the distribution of normality. In order to compare parametric quantitative variables between 2 groups, Student t test was applied. For comparison of nonparametric quantitative variables between 2 groups, Mann-Whitney U test was used. Qualitative variables were compared using χ2 test or Fischer exact test when frequencies were below 5. Pearson correlation coefficients were used to assess the association between the 2 normally distributed variables. When a variable was not normally distributed, a Spearman correlation test was performed. Multivariable linear regression analysis was employed to determine the relation between sFLT-1 as the dependent variable and clinical, laboratory, and radiological variables. Receiver operating characteristic curve was used to determine the best cutoff value of sFLT-1 that best combined sensitivity and specificity. The area under the curve (AUC) was calculated for each plot. A P value <.05 was considered significant in all analyses.
Results
Clinical Characteristics of Patients With β-TI
The clinical, laboratory, and radiological data of the studied patients are shown in Table 1. β-Thalassemia intermedia patients included 19 males and 16 females. The median age of TI patients was 11.6 years. Twenty (57.1%) patients were transfusion dependent, 28.6% were splenectomized, 40% had pulmonary hypertension risk (elevated TRV ≥2.5 m/s), and 37.1% had heart disease. Carotid intima–media thickness was significantly increased among patients compared to the control group (0.51 ± 0.12 mm vs 0.32 ± 0.1 mm; P < .001). The mean LIC among transfusion-dependent TI patients was 6.7 ± 1.8 mg/g liver dry weight.
Clinicopathological and Radiological Characteristics of the Studied Patients With β-Thalassemia Intermedia.

Abbreviations: CIMT, carotid intima–media thickness; HbF, hemoglobin F; IQR, interquartile range; LIC, liver iron concentration; SD, standard deviation; SDS, standard deviation score; sFLT-1, soluble fms-like tyrosine kinase 1; TI, Thalassemia intermedia; WBC, white blood cell.
aThe mean LIC was calculated among transfusion-dependent TI patients.
Soluble fms-Like Tyrosine Kinase 1 Levels Among β-TI Patients and Healthy Controls
Soluble fms-like tyrosine kinase 1 was significantly higher in TI patients compared to the control group (median [IQR], 110 (80-155) pg/mL vs 70 (60-90) pg/mL; P < .001; Figure 1). Soluble fms-like tyrosine kinase 1 levels were significantly higher in transfusion-dependent TI patients compared to nontransfusion-dependent patients (P < .001). Patients who had splenectomy, pulmonary hypertension, or heart disease showed higher sFLT-1 levels than those without (P < .001; Table 2). Receiver operating characteristic curve analysis revealed that the cutoff value of sFLT-1 at 110 pg/mL could differentiate patients with and without pulmonary hypertension with a sensitivity of 86.9% and a specificity of 100%, AUC: 0.944, P < .001. Hydroxyurea-treated patients or those who received chelation therapy had lower sFLT-1 levels than untreated patients (Table 2).
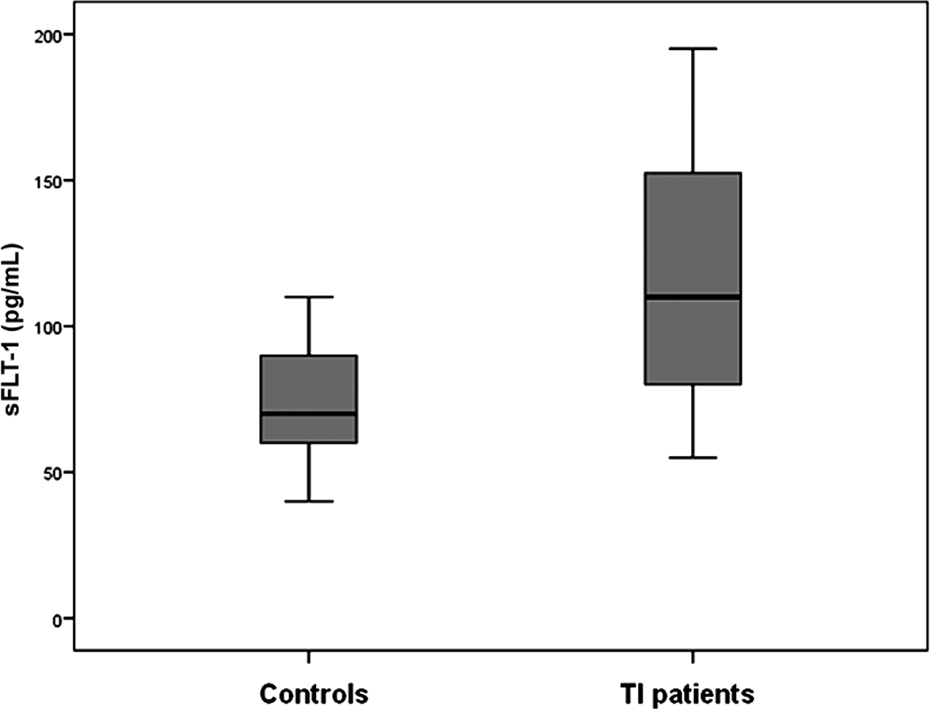
Soluble fms-like tyrosine kinase 1 (sFLT-1) in thalassemia intermedia (TI) patients compared to healthy controls.
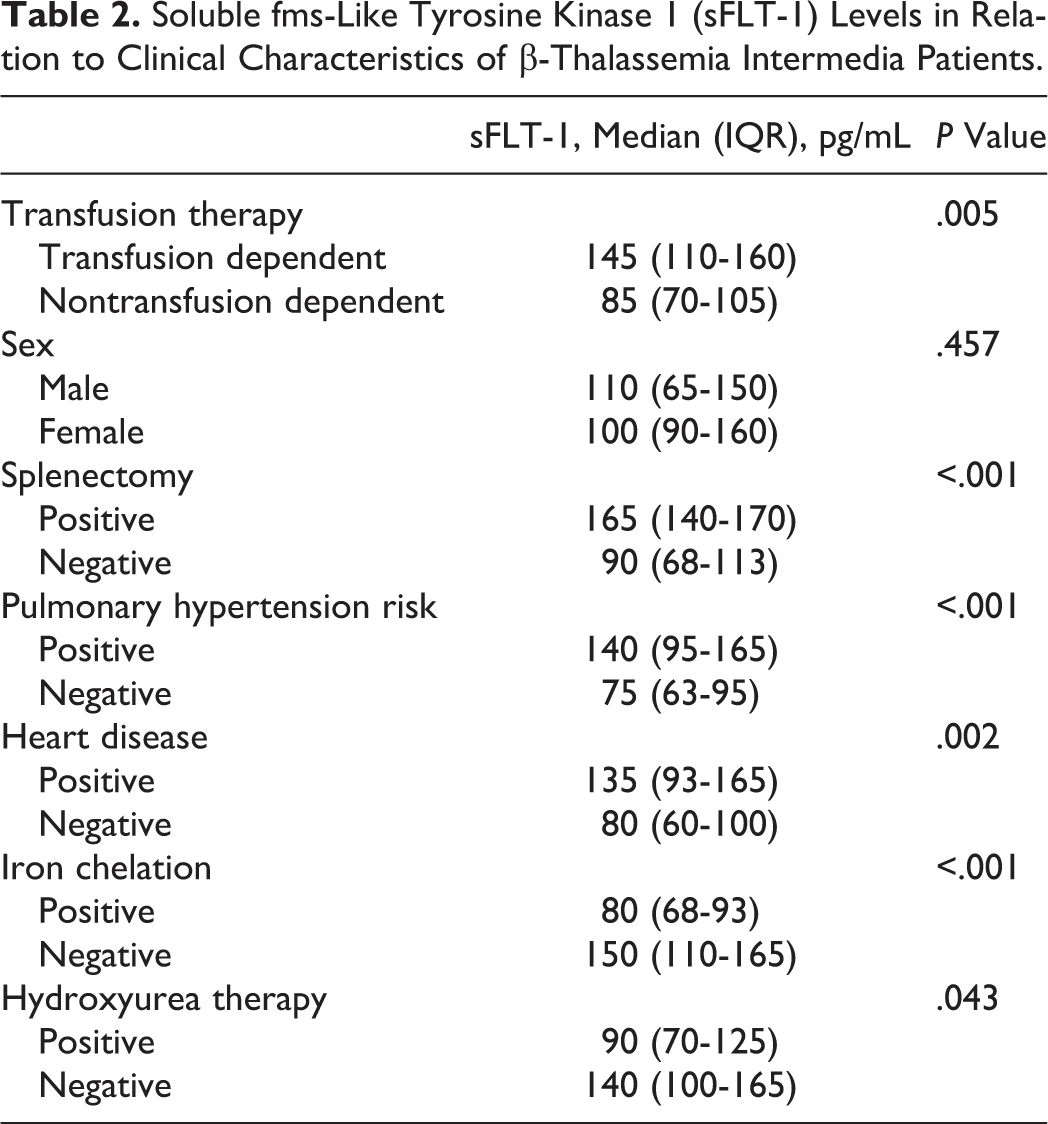
Soluble fms-Like Tyrosine Kinase 1 (sFLT-1) Levels in Relation to Clinical Characteristics of β-Thalassemia Intermedia Patients.
Significantly positive relations were found between sFLT-1 and transfusion index (r = .417, P = .017), indirect bilirubin (r = .357, P = .035), LDH (r = .716, P < .001), serum ferritin (r = .879, P < .001), TRV (r = .635, P < .001), and CIMT (r = .785, P < .001; Figure 2), whereas levels were negatively correlated with hemoglobin (r = −.398, P = .018). Of note, sFLT-1 levels were positively correlated with LIC (r = .742, P < .001) in the group of patients who performed MRI assessment. Multivariable linear regression analysis (Table 3) revealed that LDH, serum ferritin, TRV, and CIMT were independently related to sFLT-1 levels in TI patients (r2 = .637, P < .001).
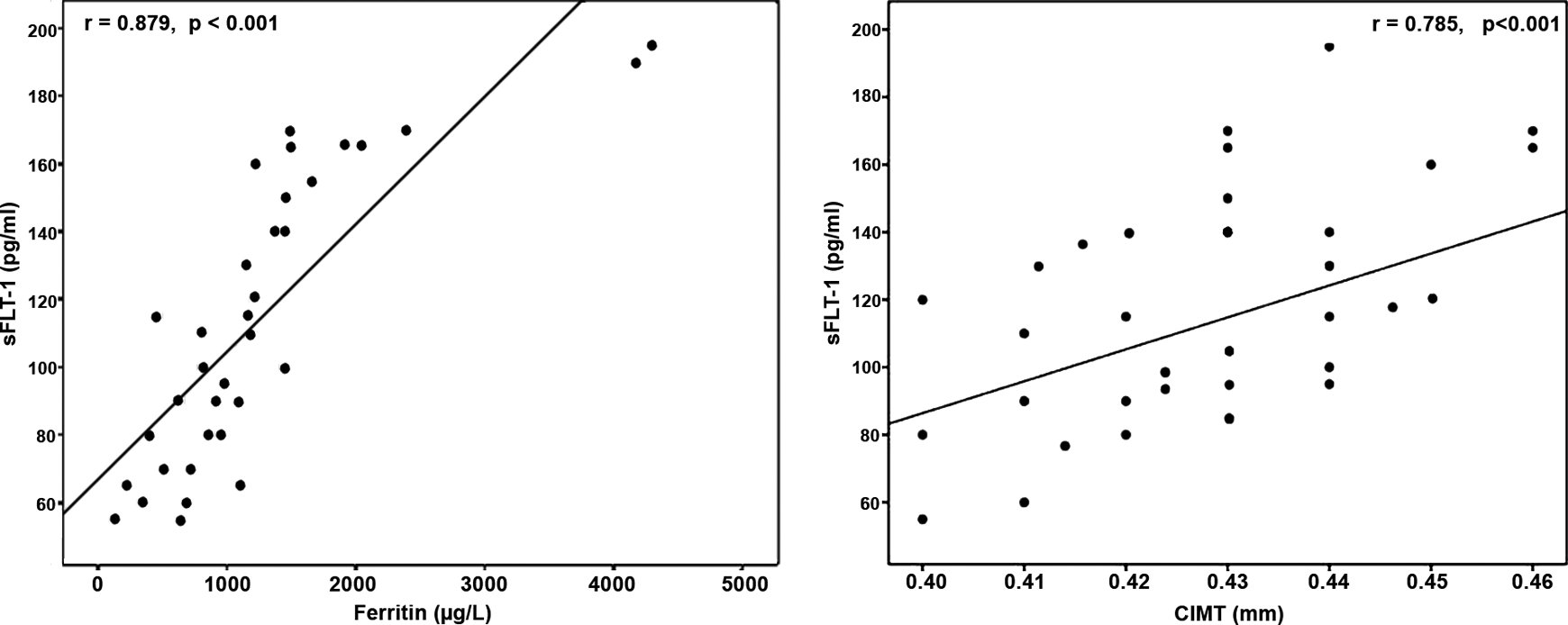
Positive correlations between soluble fms-like tyrosine kinase 1 (sFLT-1) and serum ferritin and carotid intima–media thickness (CIMT) among the studied thalassemia intermedia patients.
Significant Variables Related to Increased Soluble fms-Like Tyrosine Kinase 1 Levels in Patients With β-Thalassemia Intermedia as Shown by Multivariable Linear Regression Analysis.
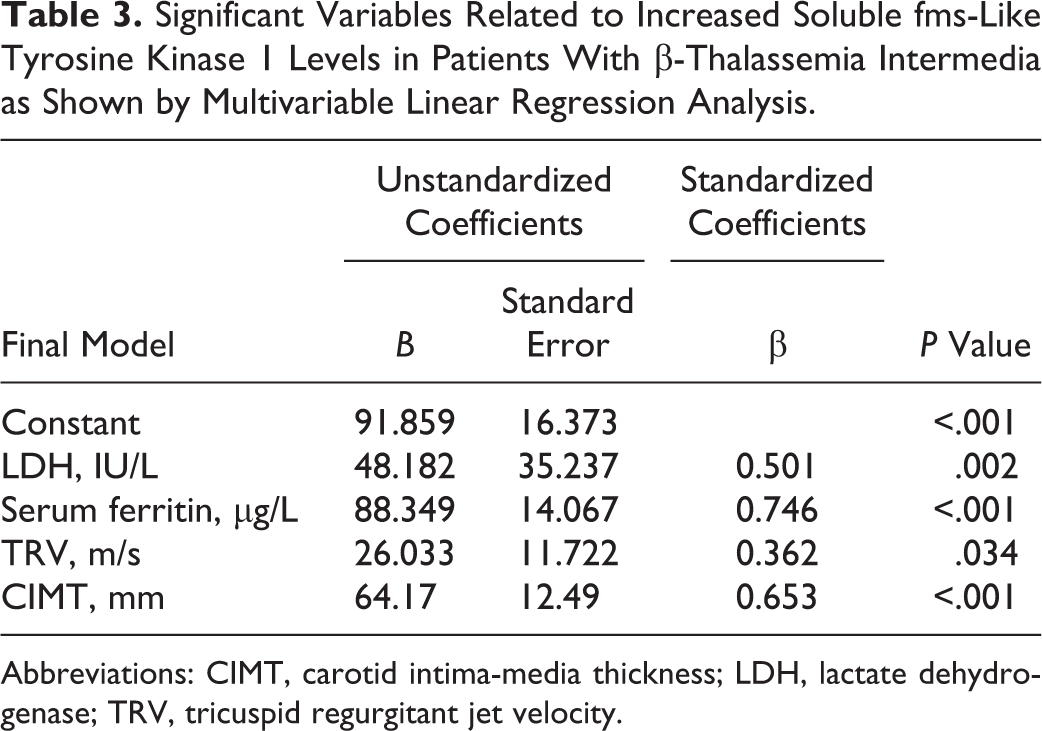
Abbreviations: CIMT, carotid intima-media thickness; LDH, lactate dehydrogenase; TRV, tricuspid regurgitant jet velocity.
Discussion
Angiogenesis has been investigated in hemolytic anemias; sickle cell disease (SCD), 18,19 and β-TM. 3 Soluble fms-like tyrosine kinase 1 is a shortened splice variant of the VEGFR-1 that signals angiogenesis. 20 Soluble fms-like tyrosine kinase 1 has been implicated in the pathogenesis of sickle cell vasculopathy, constituting a potential biomarker for pulmonary hypertension 21 and sickle cell nephropathy. 22 To our knowledge, no previous studies analyzed sFLT-1 levels in TI or its role in vascular complications and relation to CIMT. We found elevated sFLT-1 levels in patients with TI compared to healthy controls. In particular, sFLT-1 was increased among regularly transfused TI patients compared to occasionally transfused patients. Moreover, patients with cardiopulmonary complications, whether pulmonary hypertension and/or heart disease, had higher sFLT-1 levels than those without and levels were positively correlated with transfusion index, markers of hemolysis, serum ferritin, and LIC. Our findings suggest that sFLT-1 represents a link between angiogenesis, hemolysis, iron overload, and endothelial dysfunction among TI patients.
It has been reported that at the heart of complications among TI patients lies the triad of chronic anemia, ineffective erythropoiesis, and iron overload. 23 –26 Iron overload in β-TI results from repeated blood transfusions, increased iron absorption, or chronic hemolysis. 27 Tissue hypoxia has been proposed as a major stimulus for the upregulation of VEGF, and patients with anemia have elevated levels of VEGF. This suggests that anemia might impact on the progression of angiogenesis. 3,28 The increased sFLT-1 levels are associated with decreased circulating levels of VEGF and placental growth factor (PlGF), resulting in endothelial dysfunction, 20,21 which plays a critical role in the pathogenesis of vascular complications in β-thalassemia. 3 These data may explain elevated sFLT-1 levels among our TI patients, particularly those with cardiopulmonary complications.
In this regard, Voskaridou et al 29 reported that patients with β-TM had increased levels of angiogenic cytokines such as VEGF, basic fibroblast growth factor, angiogenin, and angiopoietin compared to healthy controls. Also, Butthep et al 30 reported that patients with thalassemia are characterized by increased levels of VEGF and tumor necrosis factor (TNF). Mohan et al 18 reported elevated VEGF plasma levels in clinically asymptomatic SCD patients. On the other hand, Duits et al 19 determined serum levels of important angiogenic growth factors including VEGF in SCD patients. They detected only free or unbound VEGF serum levels, perhaps indicating a methodological basis for discrepancies between reported VEGF levels in SCD. Nevertheless, the authors indicated a pro-angiogenic state in SCD, mainly because of elevated angiopoietin-2 levels.
In agreement with our findings, Ataga et al 21 observed significant correlations between sFLT-1 and various measures of hemolysis including LDH, total bilirubin, indirect bilirubin, and reticulocyte count among patients with SCD. This suggests that sFLT-1 may be induced by erythropoiesis such as its ligand, PlGF, which is known to be upregulated by erythropoietin. 31 Furthermore, Youssry et al 22 found a significant positive correlation between serum sFLT-1 and microalbuminuria, LDH, and indirect bilirubin among SCD patients.
In this study, sFLT-1 levels were higher in our TI patients with pulmonary hypertension and a positive correlation with TRV. A recent study by Derchi et al 32 established for the first time the presence of pulmonary hypertension by right heart catheterization in patients with thalassemia. It also showed that the prevalence was higher in TI patients compared to those with TM. Autopsies of a large series of patients with TI revealed thrombotic lesions in the pulmonary arteries, which may have been due to circulating platelet aggregates. 33 A high rate of pulmonary hypertension in splenectomized TI patients has been documented and attributed to a chronic thromboembolic state. 34,35 Hemolytic anemia-associated pulmonary hypertension results from endothelial dysfunction, increased vascular tone, inflammation, hypercoagulability, vascular remodeling, and destruction of the pulmonary vasculature. 1,36,37 All these factors lead to elevated sFLT-1 levels among TI patients with pulmonary hypertension and those with splenectomy. Receiver operating characteristic curve analysis also showed that the cutoff value of sFLT-1 at 110 pg/mL could differentiate patients with and without pulmonary hypertension with high sensitivity and specificity.
Ataga et al 21 found that the level of sFLT-1 was significantly higher in SCD patients with pulmonary hypertension and suggested that sFLT-1 may contribute to the pathogenesis of SCD-associated pulmonary hypertension by inducing endothelial dysfunction. This is because sFLT-1 is known to negatively regulate the action of VEGF on 2 levels: (1) it binds and sequesters VEGF in the circulation and (2) it occupies the VEGFR and prevents VEGF occupancy and subsequent signal transduction. 38 Vascular endothelial growth factor, a well-known promoter of angiogenesis and an endogenous regulator of endothelial integrity, has been described to stimulate production of NO in endothelial cells. 39 Vascular endothelial growth factor promotes endothelial cell survival and angiogenesis through activation of the protein kinase, Akt, which in turn activates endothelial NO synthase and increases NO generation. 38 By antagonizing VEGF action and inhibiting Akt phosphorylation, sFLT-1 decreases NO bioavailability, thus leading to endothelial dysfunction. 21
Two main factors determine cardiac disease in β-TI; one is the high output state that results from chronic tissue hypoxia and the other is the vascular involvement that leads to an increased pulmonary vascular resistance and an increased systemic vascular stiffness. 40 Circulating sFLT-1 is generated as a result of myocardial injury and subsequent development of heart failure. Elevated levels of sFLT-1 have been associated with adverse outcomes in stable patients with heart failure. 41 Therefore, it was expected to find elevated sFLT-1 levels among our TI patients with cardiac abnormalities even before clinically evident disease. Li et al 42 reported that in patients with β-thalassemia who do not receive early regular transfusion and iron chelation therapy, iron deposition may occur at an early age. Important organs and tissue functional lesions and related complications also result. Mohammad 43 demonstrated that echo-derived right and left side cardiac complications are not uncommon in TI patients. Tricuspid regurgitant jet velocity as a continuous predictor was associated positively with age, cardiac volumes, and pulmonary regurgitation, though negatively associated with ejection fraction.
To study the relation between sFLT-1 and vascular wall structure among TI patients, we assessed CIMT as an early index for subclinical atherosclerosis compared to healthy controls. Carotid intima–media thickness was significantly increased in patients compared to controls and was positively correlated with sFLT-1. Shin et al 44 reported that a high sFLT-1 level is predictive of CIMT progression in patients with hypertension and thus indicative of progression of atherosclerosis. Vascular endothelial growth factor receptor 1 is expressed on endothelial cells, monocytes, and macrophages. Stimulation of the receptor triggers intracellular tyrosine kinase activity, thus regulating monocyte/macrophage-mediated processes that have been implicated in the pathogenesis of atherogenesis. 45
The association between the carotid atherosclerosis and VEGF and sFLT-1 levels was also investigated in 909 healthy middle-aged participants of a cross-sectional substudy of the “Salzburg Atherosclerosis Prevention Program in Subjects at High Individual Risk (SAPHIR)” study. 46 Only a weak correlation was established between the plasma VEGF, sFLT-1 levels, and CIMT. 46 However, this substudy of the SAPHIR trial was conducted on healthy participants and had no long-term follow-up results. The study by Shin et al 44 was the first prospective follow-up study that assessed the potential relationship between CIMT progression (a widely accepted marker of atherosclerosis) and sFLT-1. Treatment with sFLT-1 in an animal model of atherosclerosis results in a prominent suppression of the atherogenic process. 47
Thalassemia intermedia individuals may develop iron overload from increased gastrointestinal absorption of iron even without transfusion. 48 Iron-chelating agents are necessary to prevent or decrease the consequences of hemosiderosis. 49 Deferasirox iron-chelation therapy can act as an antioxidant by decreasing intra- and extracellular toxic iron species and reducing oxidative stress in patients with thalassemia. 50 –52 Reactivation or augmentation of hemoglobin F (HbF) using hydroxyurea is also a possible therapeutic approach for patients with β-TI. 53,54 Other actions of hydroxyurea, in particular, its effects on vascular endothelium, adhesion molecule expression, and cytokine production, may also play a role in the final therapeutic outcome. 55 These data explain lower levels of sFLT-1 among TI patients who received chelation or hydroxyurea therapy.
Although the median age of our enrolled patients was 11.6 years and sFLT-1 levels measured in this cross-sectional study was not correlated with age or weight of the studied patients, it is worth to note that sFLT-1 is a marker of placental insufficiency studied in utero and may be high in small for gestational age neonates. 56 Low birth weight, caused by intrauterine growth restriction (IUGR), has been recently known to be associated with increased rates of cardiovascular disease. 57 Moreover, aortic wall thickening in fetuses and children with IUGR shows differences with respect to those who were appropriate for gestational age. 58,59 This may reflect a correlation between impaired growth in utero, Doppler abnormalities, low birth weight, and early signs of vascular dysfunction. 59 These points should be taken into consideration when investigating sFLT-1 levels and CIMT in a longitudinal study including infants who are small for gestational age.
Study Limitations
One limitation of this study is the relatively small number of SCD patients included in this study. This necessitates further large prospective studies to verify our results. Longer periods of follow-up are also required to establish the effect of elevated sFLT-1 levels on the development of cardiovascular morbidity and mortality among these patients. Whether therapeutic options based on these data could improve the cardiovascular outcome of SCD patients represents an interesting area for future research.
In conclusion, elevated sFLT-1 levels in children and adolescents with β-TI represent a link between angiogenesis, hemolysis, iron overload, endothelial dysfunction, and subclinical atherosclerosis. Measurement of sFLT-1 as a marker of vascular dysfunction in β-TI may provide utility for early identification of patients at increased risk of cardiopulmonary complications. Future studies are required to shed further light on the regulation of antiangiogenic processes in hemolytic anemia and establish approaches to their therapeutic manipulation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
