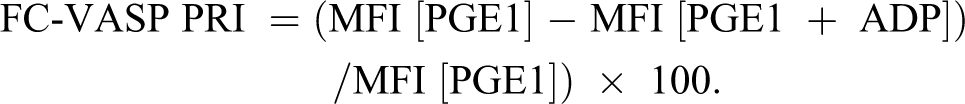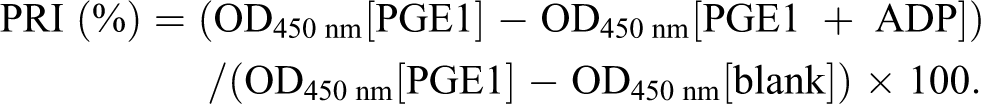Abstract
The level of platelet reactivity during P2Y12-adenosine diphosphate receptor antagonist is associated with ischemic and bleeding risks following percutaneous coronary intervention in acute coronary syndrome. Determining platelet reactivity inhibition may be valuable for confirming effective platelet inhibition for individual patients and identifying patients at risk of bleeding. The enzyme-linked immunosorbent assay (ELISA)-based vasodilator-stimulated phosphoprotein (VASP) assay offers unique advantages over other methods and has not been used in the Chinese population. We enrolled 10 healthy volunteers and 54 patients with acute coronary syndrome. The volunteers received no treatment, and patients were administered a loading dose of clopidogrel or ticagrelor. The platelet reactivity index (PRI) was measured using flow cytometry (FC)-VASP and ELISA-VASP at baseline and 8-hour postloading dose. Blood samples of healthy volunteers and clopidogrel- or ticagrelor-treated patients were frozen and stored for 1, 2, and 4 weeks after initial activation. All frozen samples were tested using ELISA-VASP. The PRI assessed by FC-VASP and ELISA-VASP correlated well showing a high degree of consistency in identifying high or low on-treatment platelet reactivity. No significant time-dependent changes in PRI results were observed in frozen samples stored up to 4 weeks compared to nonfrozen samples. The PRI of ticagrelor-treated patients was lower than that of clopidogrel-treated patients. The ELISA-VASP effectively assesses the PRI, and results obtained from frozen specimens are unaffected by storage and shipment prior to assay. Ticagrelor was superior to clopidogrel in decreasing the PRI.
Introduction
Antiplatelet therapy after percutaneous coronary intervention (PCI) is important for the prevention of major adverse cardiac events. 1 In order to assess the efficacy of antiplatelet therapy and guide the application of antiplatelet drugs, a variety of platelet function tests have been developed and are widely used in clinical practice. 2,3
Vasodilator-stimulated phosphoprotein (VASP) measurement is the most specific approach to evaluate the extent to which P2Y12 receptors are functionally blocked by a P2Y12 antagonist. 4 Vasodilator-stimulated phosphoprotein is independent of all other factors that interfere with platelet reactivity (PR), particularly glycoprotein GPIIb/IIIa inhibitors that block the final pathway of platelet aggregation. 5 At present, VASP measurement is performed by flow cytometry (FC) and is a time-consuming and costly assay available only in some research laboratories. The FC-based technique is also limited because blood samples have to be processed within 48 hours of collection. In addition, blood samples must be maintained at room temperature (RT) before analysis and cannot be stored in a frozen state for transportation or later analysis. Recently, a new enzyme-linked immunosorbent assay (ELISA)-based approach has been developed (BioCytex, Marseille, France) to overcome these limitations and has been validated in some clinical trials. 6 This new ELISA technique has been used to test PR inhibition in patients with acute coronary syndrome (ACS) and healthy volunteers in small-scale studies, 7 but it has not been evaluated in the Chinese population or in ticagrelor-treated patients with ACS. We measured platelet reactivity index (PRI) in 10 healthy volunteers and 54 patients with ACS treated with clopidogrel or ticagrelor using the FC-VASP and ELISA-VASP methods. We also froze and stored blood samples for 1, 2, and 4 weeks and tested the stability of frozen samples from patients treated with clopidogrel or ticagrelor analyzed by ELISA. The present study evaluated the accuracy of ELISA-VASP PRI measurement for the identification of high on-treatment platelet reactivity (HPR) and low on-treatment platelet reactivity (LPR) in patients with ACS treated with clopidogrel or ticagrelor.
Methods
Study Design
The study received approval number 201403 from the Ethics Committee of the General Hospital of Chinese Armed Police Forces (CAPF, Beijing, China) and was conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent for sample use.
We enrolled 54 patients with ACS who scheduled to undergo PCI at the General Hospital of CAPF and 10 healthy volunteers. All the patients were randomly administered clopidogrel or ticagrelor. Randomization was achieved using sequentially numbered, opaque, sealed envelopes to ensure allocation concealment. Randomization took place immediately after eligibility had been confirmed on the basis of the inclusion and exclusion criteria. Half of the patients (27) were treated with a loading dose (LD) of 300 mg clopidogrel, followed by a 75 mg daily maintenance dose, and half received an LD of 180 mg ticagrelor followed by a 90 mg twice daily maintenance dose. All patients received acetylsalicylic acid (aspirin) 100 mg qd until they could tolerate. Glycoprotein IIb/IIIa receptor antagonists, low-molecular-weight heparin, and other additional medications were administered as directed by the treating cardiologist. The volunteers did not receive any treatment. The PRIs of patients with ACS were measured by FC-VASP and ELISA-VASP before the LD of ticagrelor or clopidogrel and 8-hour post-LD. The PRIs of healthy volunteers were also measured using FC-VASP and ELISA-VASP. The initial blood drawn was discarded to avoid measuring platelet activation induced by needle puncture. Blood was collected into a vacutainer containing 3.8% trisodium citrate. The vacutainer was filled to capacity, inverted 3 to 5 times for gentle mixing, and sent immediately to the laboratory or stored at RT for up to 48 hours. After activation and lysis (see below), some sample aliquots were stored at –20°C for periods of 1, 2, and 4 weeks. Fresh samples were assayed by FC within 2 hours. Frozen samples were assayed by ELISA-VASP measurement 1, 2, and 4 weeks later. All samples were shipped to the central core laboratory, and the staff performing the platelet function assays were blinded to clinical data, randomization, and results of other assays. The patients were followed up for a 30-day period. Events including death, myocardial infarction, stroke, target vessel or target lesion revascularization, and bleeding events according to the Bleeding Academic Research Consortium (BARC) criteria were recorded.
Study Population
Healthy volunteers were randomly selected from patients presenting to our hospital for routine analysis and were ≥18 years of age. Patients with ACS were selected from those admitted to our hospital for an ST-segment elevation myocardial infarction (STEMI) or a non–ST-segment elevation acute coronary syndrome (NSTE-ACS) from May 2014 to October 2015. Patients with ACS were randomized to an LD of 300 mg clopidogrel or 180 mg ticagrelor before PCI.
Inclusion criteria were (1) provision of informed consent prior to any study-specific procedures, (2) male or female participants aged 18 to 80 years, and (3) patient with ACS scheduled to undergo PCI.
Exclusion criteria were (1) hypersensitivity to the active substance or to any of the excipients, (2) active bleeding or bleeding diathesis, (3) previous transient ischemic attack, (4) antiplatelet (clopidogrel, prasugrel, and ticagrelor) administration in the week before the index event, (5) platelet count <100 000/mL, hematocrit <30%, (6) left ventricular ejection fraction ≤30%, (7) renal failure with creatinine ≥3 mg/dL, (8) history of liver disease, (9) increased risk of bradycardia, (10) concomitant therapy with drugs known to interfere with CYP3A4 metabolism, and (11) concomitant therapy with drugs that interfere with P-glycoprotein metabolism.
Sample Size Calculation
We hypothesized that the correlation between FC and ELISA was 0.9 and chose a power of 90% and a 2-sided α level of .05. Based on these assumptions, at least 8 healthy volunteers were required to reach statistical significance. Regarding FC-VASP, we assumed the area under the receiver operating characteristic (ROC) curve for identifying HPR was 0.92, and the null hypothesis value was 0.7. We chose a power of 90% and a 2-sided alpha level of .05. Based on these assumptions, we needed at least 50 patients with ACS to reach statistical significance.
Assessment of VASP Phosphorylation by FC
The FC assay was performed within 48 hours of sample collection following the instructions of the test kit and as described in previous studies. 8,9 Whole blood samples (10 μL) were first incubated with prostaglandin E1 (PGE1) or PGE1 + adenosine diphosphate (ADP) at RT for 10 minutes and then fixed and permeabilized. Samples were immunolabeled by incubation with a serine 239-phosphorylated VASP-specific mouse monoclonal antibody (clone 16C2) followed by staining with a fluorescein isothiocyanate-labeled anti-mouse polyclonal antibody. Flow cytometric analysis was performed using an FACSCalibur (BD Biosciences, San Jose, California). The platelet population was identified from its forward and side scatter distribution, and 5000 platelet events were gated and analyzed for mean fluorescence intensity (MFI) using BD CellQuest software (BD Biosciences). The PRI was calculated from the MFI of samples incubated with PGE1 or PGE1 and ADP (PGE1 + ADP) according to the formula:
Assessment of VASP Phosphorylation by ELISA
The assessment of VASP phosphorylation by ELISA was performed following the instructions of the ELISA kit (CY-QUANT VASP/P2Y12). Repeated samples were analyzed or frozen and stored within 48 hours of sample collection and after activation and lysis. Fresh blood samples (50 μL) were added to a pair of freeze-proof microtubes containing PGE1 or PGE1 + ADP. Following incubation (at RT for 10 minutes) and lysis, 1 group of fresh blood samples were analyzed immediately while others were vortexed, frozen, and stored at −20°C for future testing. Frozen samples were thawed at RT before analysis. All procedures of the assay were performed directly in the 96-well strip coated with a mouse anti-human VASP monoclonal antibody. The PGE1-activated and corresponding PGE1+ADP-activated samples were loaded into the wells. A dilution buffer was also loaded as a blank. The wells were covered, incubated for 30 minutes at RT, and washed 3 times, each with 300 μL of washing solution. The specific mouse antihuman phosphorylated VASP monoclonal antibody coupled with peroxidase was immediately added. The wells were covered, incubated for 30 minutes at RT, and again washed as described above. Tetramethylbenzidine was added for color development and incubated for 5 minutes at RT. The reaction was stopped by acidification with sulfuric acid, and the absorbance of the reaction product was measured at 450 nm within 4 hours after stopping the reaction. Optical density at 450 nm (OD450 nm) was directly related to the phosphorylated VASP present in the sample after PGE1 or PGE1 + ADP activation. The PRI was calculated using optical density (cOD450 nm) in the presence of PGE1 alone or PGE1 + ADP simultaneously, as follows:
Statistical Analysis
All statistical analyses were performed using SPSS Statistics for Windows, version 20.0 (IBM Corp., Armonk, New York). Continuous variables were compared with a t test or Wilcoxon rank-sum test and are expressed as mean ± standard deviation (SD) or median. Categorical variables were compared with χ2 statistics or Fisher exact test and are expressed as frequencies and percentages. The PRI (%) are expressed as mean ± SD. The relation between FC- and ELISA-VASP assays was assessed by the Pearson correlation coefficient (r). Intermethod reliability comparing the ELISA-VASP and FC-VASP was assessed using the Bland-Altman 95% limits of agreement. The difference between fresh blood samples and frozen samples at different time points was evaluated with repeated measures analysis of variance tests. Intertest correlation for HPR or LPR with treatment was determined using contingency tables and χ2 tests.
Results
Baseline Characteristics
A total of 54 eligible patients with ACS (STEMI, n = 9; NSTE-ACS, n = 45) and 10 healthy volunteers were enrolled. The baseline characteristics of the patients and volunteers are listed in Table 1.
Baseline Characteristics of the Patients and volunteers, According to Treatment Group.
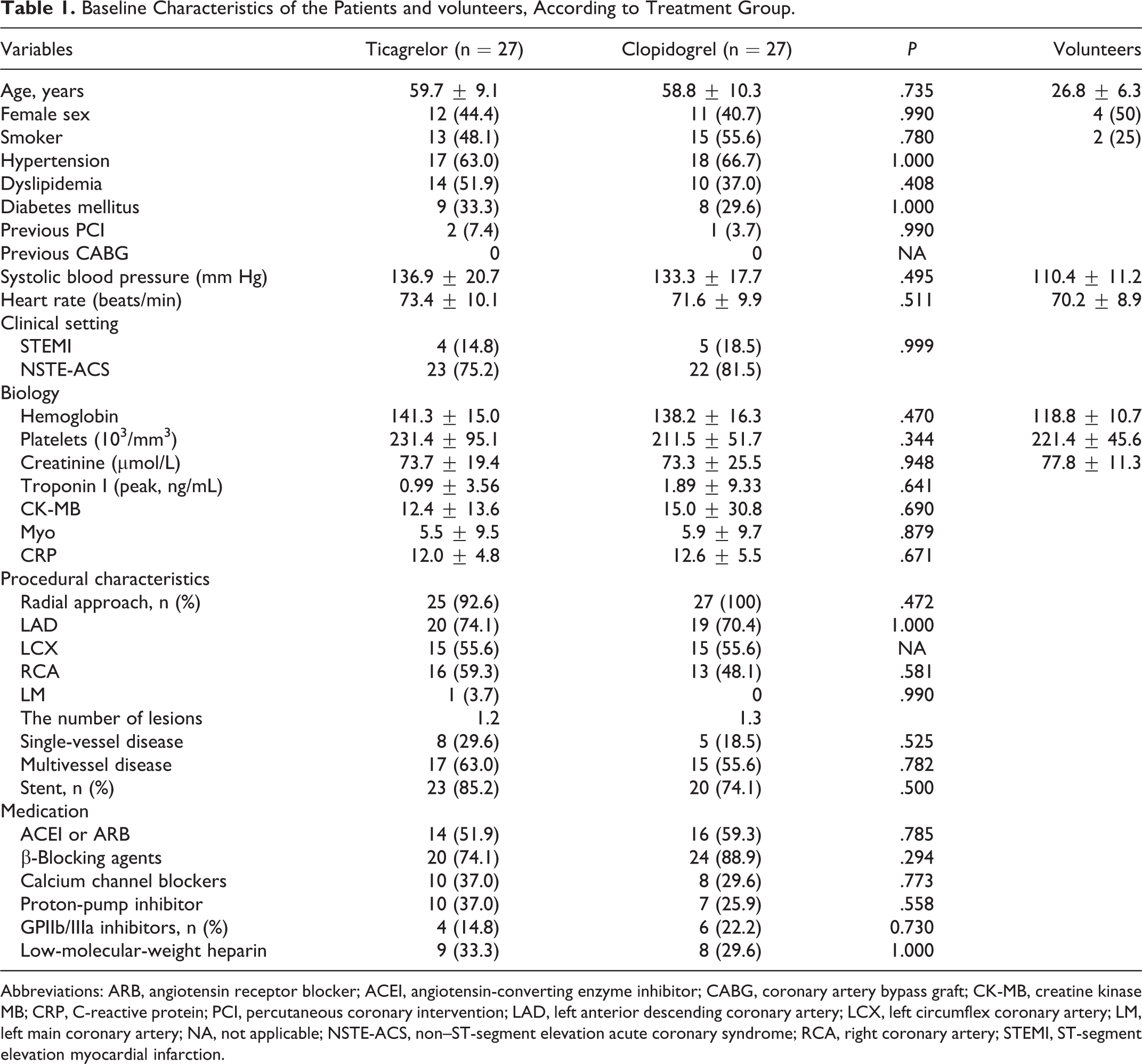
Abbreviations: ARB, angiotensin receptor blocker; ACEI, angiotensin-converting enzyme inhibitor; CABG, coronary artery bypass graft; CK-MB, creatine kinase MB; CRP, C-reactive protein; PCI, percutaneous coronary intervention; LAD, left anterior descending coronary artery; LCX, left circumflex coronary artery; LM, left main coronary artery; NA, not applicable; NSTE-ACS, non–ST-segment elevation acute coronary syndrome; RCA, right coronary artery; STEMI, ST-segment elevation myocardial infarction.
Comparison of the Flow Cytometric and ELISA-Based VASP Assay in Healthy Volunteers
In healthy volunteers, the mean FC-PRI was 80.6% ± 8.4% (range, 69.5%-93.4%) and the mean ELISA-PRI was 82.3% ± 10.9% (range, 60.3%-97.5%). The PRIs determined by ELISA and FC correlated well (r = 0.89, P < .001; Figure 1A). Bland-Altman analysis was performed to evaluate the agreement between the 2 methods. The scatter plots for the 2 approaches are shown in Figure 2A.
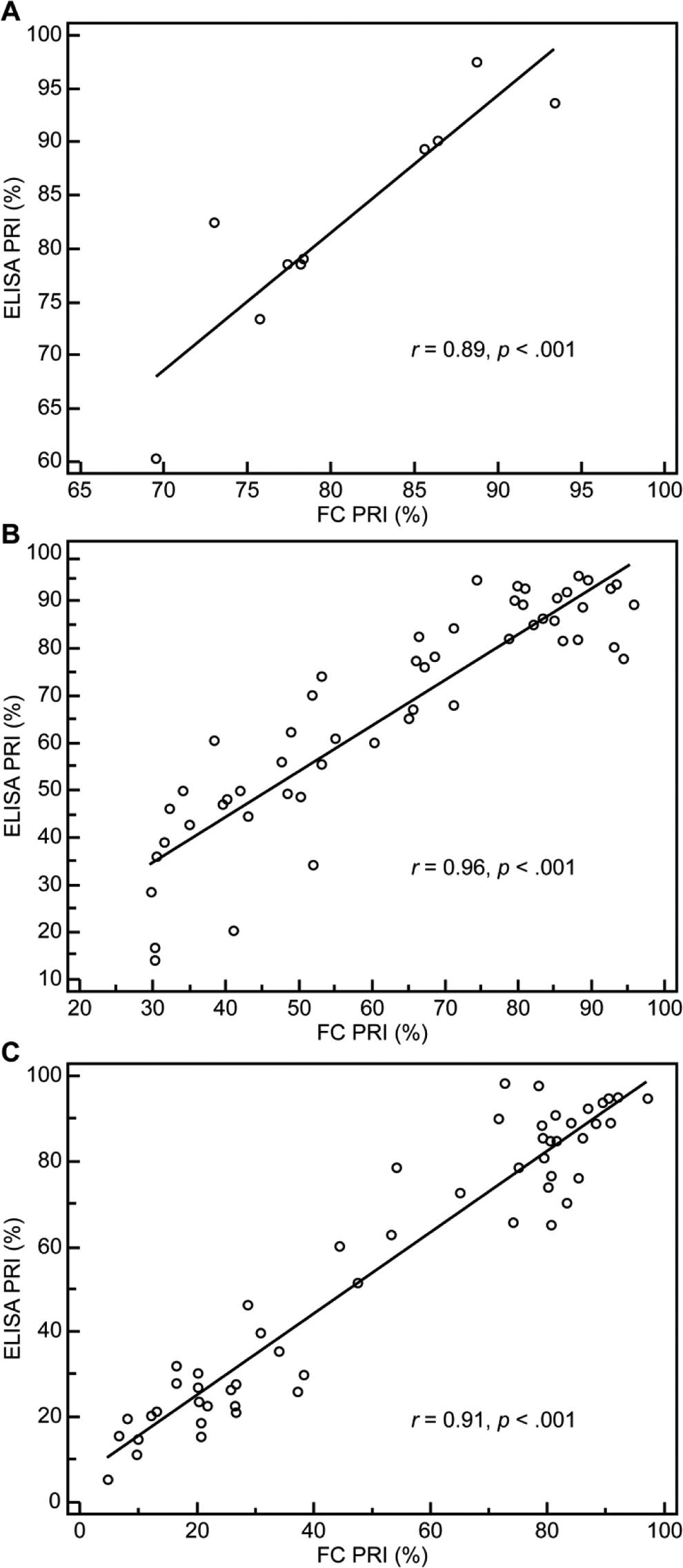
Correlation of the platelet reactivity index (PRI, %) between enzyme-linked immunosorbent assay (ELISA)-based and flow cytometric vasodilator-stimulated phosphoprotein (VASP) phosphorylation assay in (A) healthy volunteers (r = 0.89; P < .001), (B) clopidogrel-treated patients (r = 0.96; P < .001), and (c) ticagrelor-treated patients (r = 0.91; P < .001).
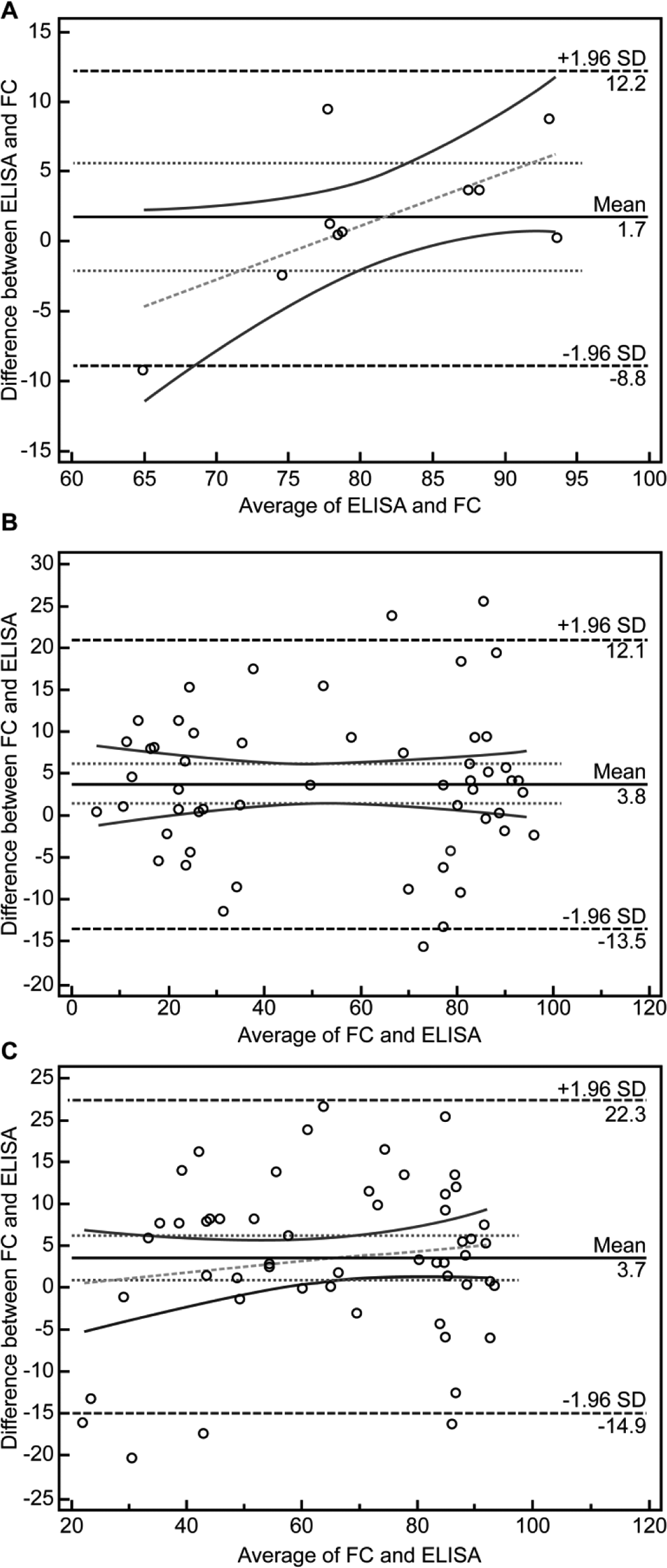
Bland-Altman plot comparing the enzyme-linked immunosorbent assay (ELISA) and flow cytometric assays in healthy volunteers (A), clopidogrel-treated patients (B), and ticagrelor-treated patients (C).
Comparison of the Flow Cytometric and ELISA-Based VASP Assay in Patients With ACS
In patients with ACS, the mean FC-PRI before clopidogrel administration was 82.1% ± 10.0% (ELISA: 86.4% ± 6.9%). Similarly, the mean FC-PRI before ticagrelor dosing was 81.1% ± 8.8% (ELISA: 84.6% ± 9.6%). The PRI determined by FC-VASP 8 hours after drug administration was 47.2% ± 14.3% in patients randomized to clopidogrel, where the value in the ticagrelor group was 23.8% ± 12.7%. In both clopidogrel-treated and ticagrelor-treated patients, the mean ELISA-PRI showed a very high correlation with the FC-PRI (Figure 1B and C). We used Bland-Altman analysis to evaluate the correlation between the 2 methods. The mean difference was 3.8% and 3.7%, respectively, in the clopidogrel-treated and ticagrelor-treated groups (Figure 2B and C). However, the results also indicated that the ELISA-PRI was a little higher than the FC-PRI.
Stability of Frozen Samples From Volunteers Analyzed by ELISA-VASP
The PRIs of frozen samples stored for 1, 2, and 4 weeks were very stable. There was no significant difference in the PRIs of volunteer samples following 4 weeks of storage at −20°C (P > .05; Figure 3A). Compared to the PRI of fresh blood, mean differences of 3.2% (−8.2% to 2.0%), 12.6% (−15.2% to 40.4%), and 0.6% (−5.2% to 6.3%) were found for samples stored frozen for 1, 2, and 4 weeks, respectively (95% confidence interval [CI]). However, due to repeated freezing and thawing of the samples, the PRIs of the 2 samples noticeably decreased in week 2. The 2 samples were thawed in the second week and then stored at −20°C for 2 weeks (Figure 3B and C).
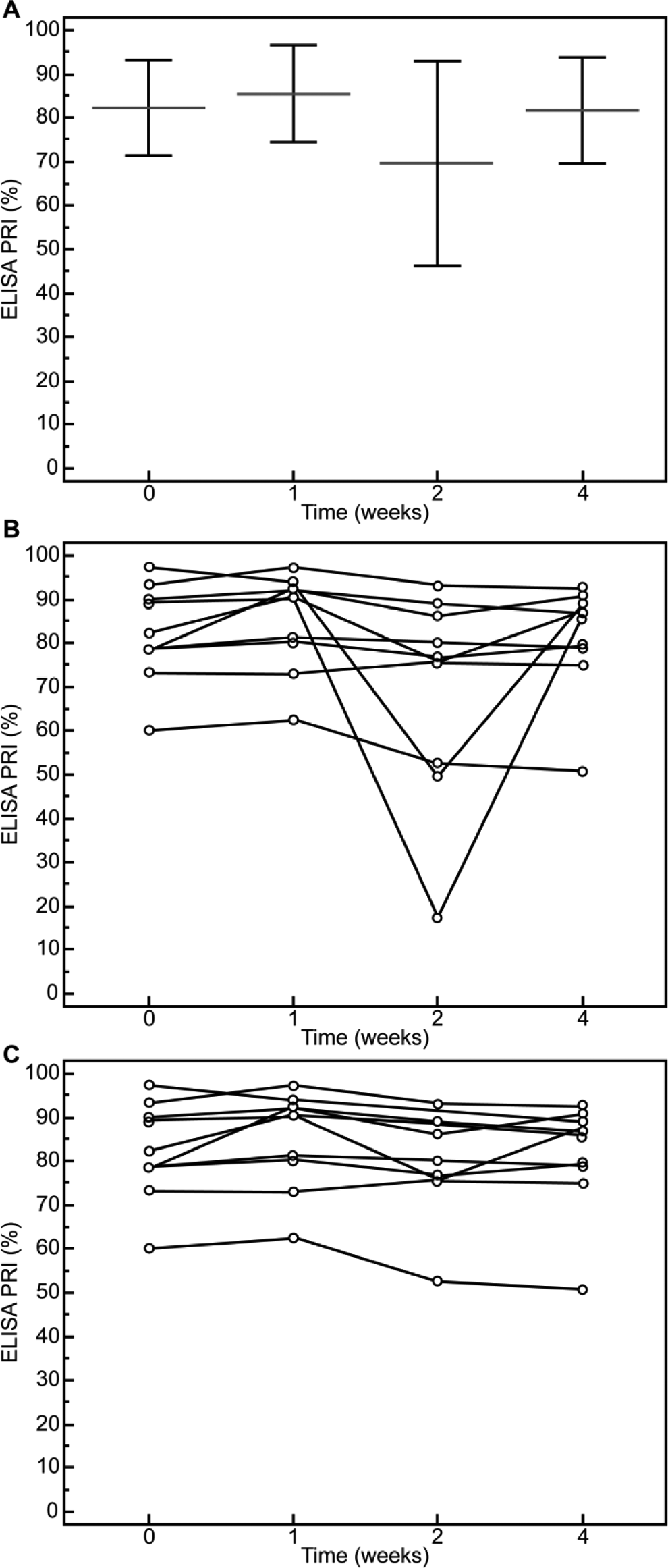
(A) No significant change occurred during 4 weeks of −20°C storage of the blood samples of healthy volunteers (P > .05). (B) Due to repeated freezing and thawing of the samples, the platelet reactivity index (PRI) of 2 samples markedly decreased in week 2. The 2 samples were thawed in the second week and then stored at −20°C for 2 weeks. Excluding the 2 samples in (B), there was no significant time-dependent change in frozen samples stored for 4 weeks (C).
Stability of Frozen Samples From Patients Treated With Clopidogrel or Ticagrelor Analyzed by ELISA-VASP
The blood samples of patients with ACS treated with clopidogrel or ticagrelor were also frozen and stored for 1, 2, and 4 weeks. The ELISA-VASP was performed to measure the PRIs at 1, 2, and 4 weeks. Compared to fresh samples, there was no obvious time-varying change in the PRI of frozen samples stored for 4 weeks. The PRI values reflecting patient response to clopidogrel or ticagrelor were stable throughout the period of storage. Compared to the PRI values of fresh samples, we found the mean differences in the PRI of samples stored for 1, 2, 4 weeks were 2.3% (−0.5% to 5.1%), 0.5% (−2.1% to 3.0%), and 1.4% (−1.8% to 4.7%), respectively, in ticagrelor-treated patients (Figure 4A), and 0.9% (−6.0% to 4.2%), 1.3% (−3.8% to 6.5%), and 2.2% (−7.7% to 3.4%), respectively, in clopidogrel-treated patients (Figure 4B; 95% CI). There was no significant difference between fresh blood samples and frozen samples.
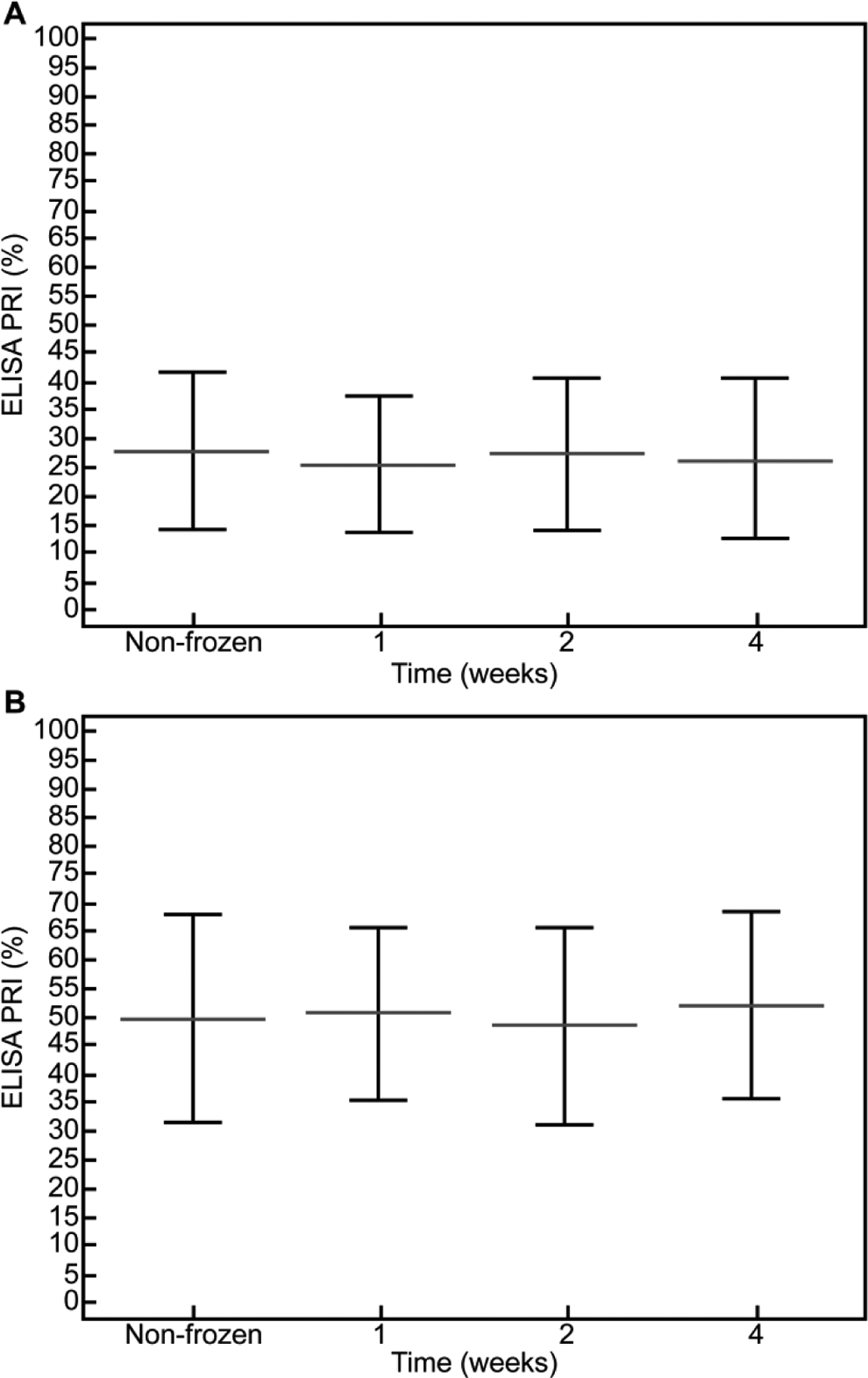
(A) Compared samples from ticagrelor-treated patients analyzed by enzyme-linked immunosorbent assay (ELISA) immediately or after frozen storage for 1, 2, or 4 weeks according to repeated measures analysis of variance (ANOVA) (P > .05). Results expressed as mean ± standard deviation (SD). (B) Compared samples from clopidogrel-treated patients analyzed by ELISA immediately or after frozen storage for 1, 2, or 4 weeks according to repeated measures ANOVA (P > .05).
Identification of Patients With HPR and LPR
Numerous prior studies have linked HPR to a greater risk for ischemic complications, whereas LPR has been associated with bleeding events. 10,11 The HPR was defined as a VASP index >50% and LPR was defined as a VASP index <16% (Figure 5). The rate of patients with HPR (12/54) determined by FC-VASP did not differ from that determined by ELISA-VASP using the same cutoff point (14/54, P = .69). The rate of patients with LPR was 7 of 54 determined by FC-VASP, whereas the rate of patients with LPR was 6 of 54 when determined using ELISA-VASP (P = .78). The agreement between the 2 assays in establishing HPR was high as estimated by a κ coefficient of .697 (95% CI: 0.472-0.921; Table 2). The κ value indicating agreement in the identification of LPR was .738 (95% CI: 0.456-1.000; Table 3). The rate of HPR was higher in clopidogrel-treated patients than in ticagrelor-treated patients regardless of the test used (FC-VASP: P = .007; ELISA-VASP: P = .005). There was no significant difference in the occurrence of LPR between ticagrelor-treated patients and clopidogrel-treated patients (FC-VASP: P = .105; ELISA-VASP: P = .194; Figure 6). This is consistent with a previous report showing that ticagrelor had a stronger antiplatelet effect than clopidogrel. 12
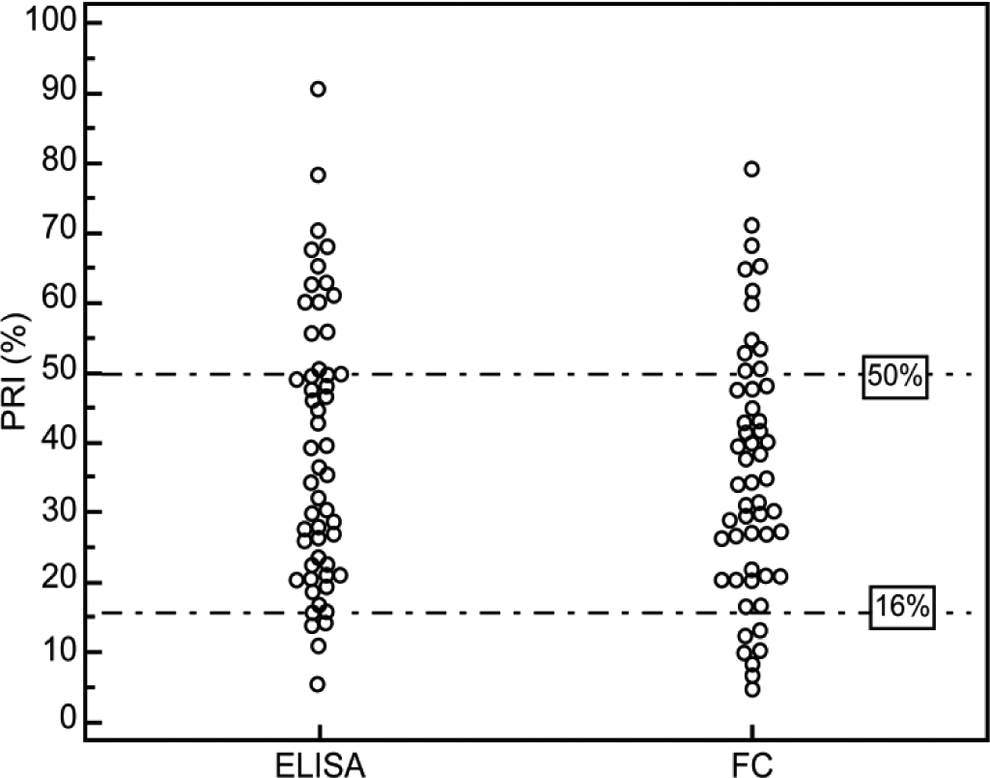
Interindividual variability in platelet reactivity index (PRI) following the loading dose of ticagrelor or clopidogrel by enzyme-linked immunosorbent assay (ELISA) and flow cytometry (FC).
Agreement Between FC and ELISA in Determining HPR.
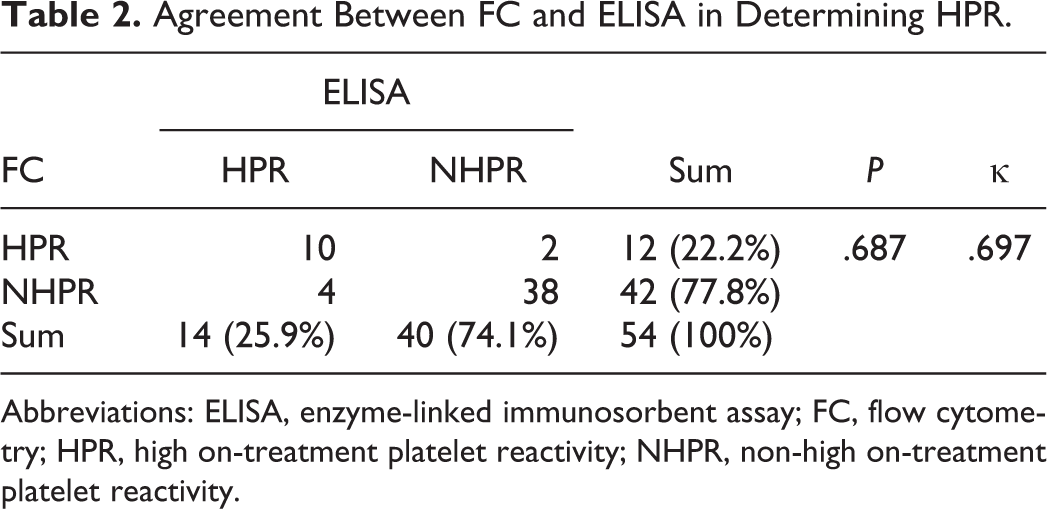
Abbreviations: ELISA, enzyme-linked immunosorbent assay; FC, flow cytometry; HPR, high on-treatment platelet reactivity; NHPR, non-high on-treatment platelet reactivity.
Agreement Between FC and ELISA in Determining LPR.
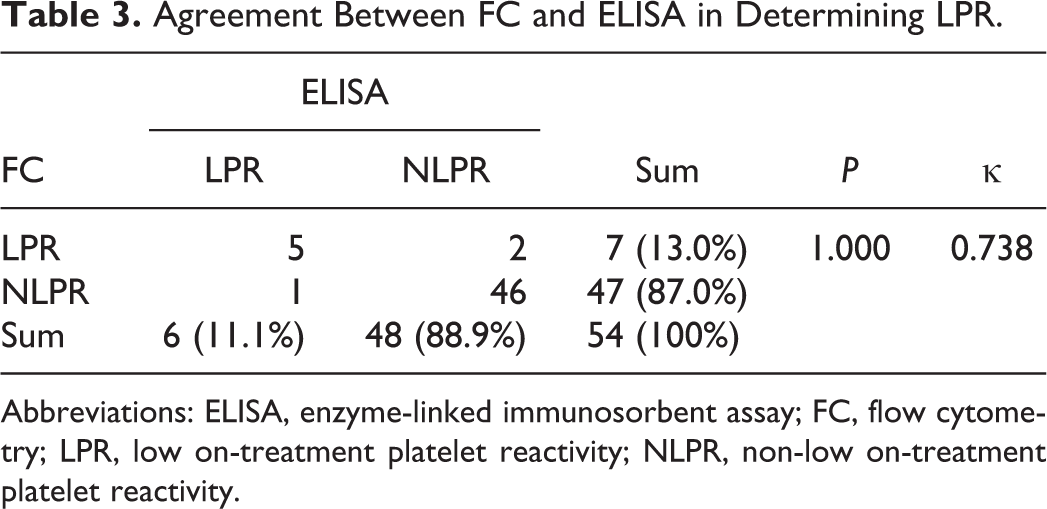
Abbreviations: ELISA, enzyme-linked immunosorbent assay; FC, flow cytometry; LPR, low on-treatment platelet reactivity; NLPR, non-low on-treatment platelet reactivity.
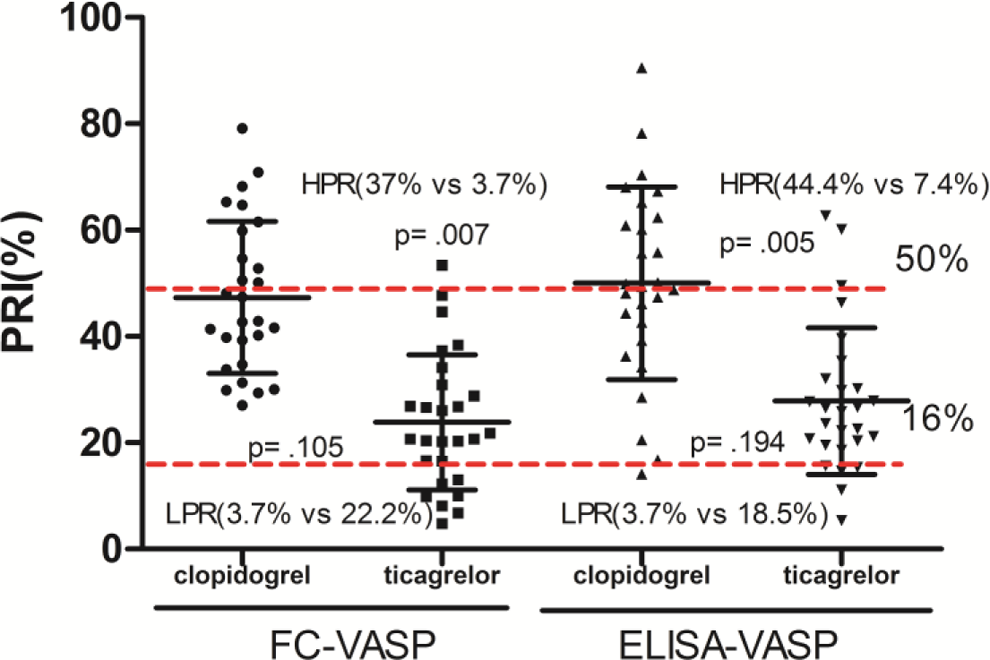
Comparison of high on-treatment platelet reactivity (HPR) and low on-treatment platelet reactivity (LPR) in patients treated with clopidogrel and ticagrelor.
Receiver Operating Characteristic Curves
According to the ROC curve analysis, ELISA-VASP demonstrated good diagnostic performance in identifying HPR and LPR. Compared to FC-VASP, it displayed an area under the curve (AUC) of 0.932 (P < .0001) when identifying patients with HPR using a cutoff of > 50% of PRI (Figure 7A). It showed a sensitivity of 83.3%, specificity of 90.5%, positive predictive value of 71.4%, and negative predictive value of 95.0% for the identification of patients with HPR. The ROC curve analysis showed the optimal cutoff value for ELISA-VASP in determining HPR was 53.1%. Choosing a cutoff value of 53.1% for ELISA-VASP PRI was more precise with a sensitivity of 83.3%, specificity of 92.9%, positive predictive value of 76.9%, and negative predictive value of 95.1%. The AUC was 0.951 (P < .0001) for ELISA-VASP when identifying LPR, and when using a cutoff value of 16%, the sensitivity, specificity, positive and negative predictive values of ELISA-VASP for detection of LPR were 83.33%, 95.83%, 71.43%, and 97.87%, respectively. Based on the ROC analysis, the optimal cutoff point for LPR was 21.2% (Figure 7B). Selecting a threshold of 21.2% for ELISA-VASP PRI resulted in a sensitivity of 100% and a specificity of 87.2%.
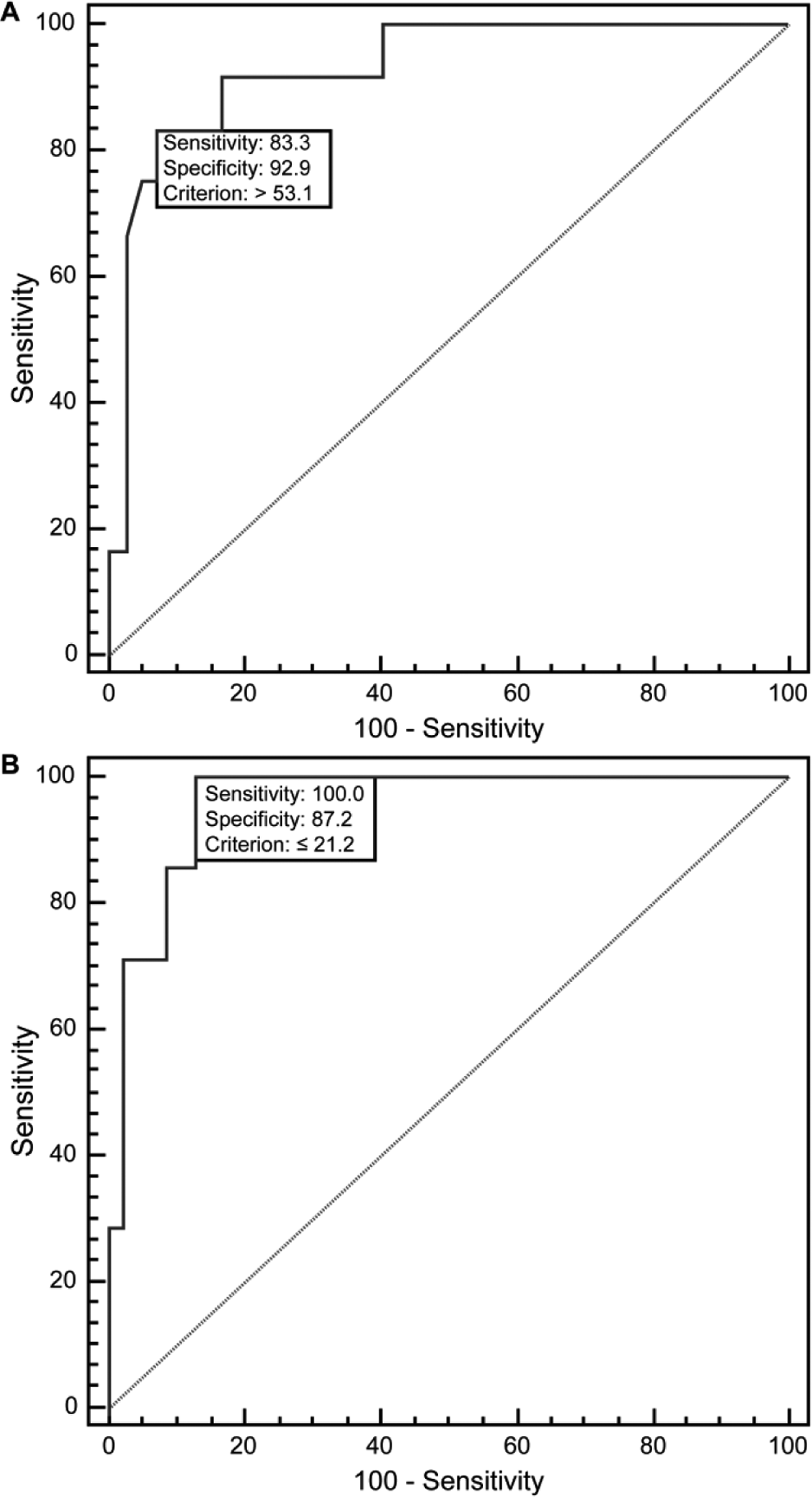
(A) The receiver operating characteristic (ROC) curve analysis comparing the enzyme-linked immunosorbent assay (ELISA)-vasodilator-stimulated phosphoprotein (VASP) to the flow cytometry (FC)-VASP measurement in determining patients with high on-treatment platelet reactivity (HPR). (B) The ROC curve analysis comparing the ELISA-VASP to the flow cytometry (FC)-VASP measurement in determining patients with low on-treatment platelet reactivity (LPR).
Follow-Up
The patients with ACS were seen after 30 days. No major bleeding occurred in either treatment arm. Eight patients (5 allocated to the ticagrelor and 3 to the clopidogrel arm) experienced BARC1 or BARC2 bleeding events. One patient who received ticagrelor was switched to clopidogrel because of bleeding. One patient experienced subacute stent thrombosis on the third day post-PCI (clopidogrel arm). Of these patients who had bleeding events, 6 exhibited LPR. The patient who had subacute stent thrombosis experienced HPR.
Discussion
Dual antiplatelet treatment (DAPT) consisting of aspirin and a P2Y12-inhibitor (clopidogrel, ticagrelor, or prasugrel) has been the cornerstone of antiplatelet therapy for patients undergoing PCI for the past 10 years. 13 The DAPT has greatly reduced the incidence of cardiovascular events after PCI. However, a considerable number of patients demonstrate HPR. 14,15 Recently, the level of PR inhibition measured by platelet assays has been shown to predict the risk of recurrent thrombotic events following PCI. 16,17 Therefore, the calculation of platelet aggregation is useful for guiding the choice of antiplatelet drugs and individualizing treatment. Several platelet function tests have been developed to measure antiplatelet effects during antiplatelet therapy to facilitate an optimal clinical outcome. There are many platelet detection methods including light transmission aggregometry (LTA), VerifyNow (Accriva Diagnostics, San Diego, California), VASP, and thromboelastography (TEG). However, the lack of consensus on which platelet assay to use for PR monitoring is an important concern. The LTA, TEG, and VerifyNow all have shortcomings. The LTA has the limitation that it requires skilled technicians to prepare the platelets, and no standard methodology exists. VerifyNow is very expensive and rarely used in China. The TEG has a lower sensitivity and specificity than similar tests. 18 –20 The VASP assay is the most specific for evaluating the P2Y12-ADP receptor blockade. This test is reproducible, standardized, and not affected by other antiplatelet agents. 4,5,21,22 However, its main limitations are its requirement for a flow cytometer and skilled technicians, and high cost. Reports indicate that the ELISA-based VASP assay may be a better alternative. 6
At present, using the ELISA-VASP assay to detect the PRI of patients with coronary artery disease has rarely been reported, especially in the Chinese population. Researchers comparing FC-VASP with ELISA-VASP have reported contradictory results. In some studies, the ELISA-VASP measured slightly higher PRI values than the FC method while other studies had opposite results. Our study was the first to use this method to detect the PRI in healthy Chinese individuals and those with ACS. It was an attempt to explore the effects of storage time and storage conditions of the treated blood on assay results. Although we found that the ELISA-VASP produced higher PRI values compared to the FC method, there was no significant difference between the PRI measured by FC-VASP and that measured by ELISA-VASP. Further, the correlation between the 2 methods was very high in both healthy volunteers and ADP-receptor antagonist-treated patients. Our study also indicated that after pretreatment of the blood samples, the PRI values of stored frozen samples remained unchanged for up to 4 weeks, and there was no time-dependent trend. Therefore, as an alternative to older tests, VASP-ELISA is easy to perform, produces results quickly, reduces measurement error, and is relatively inexpensive. In addition to these advantages, we found no time limitations for storage and batch shipment prior to assay allowing for greater flexibility during the time between initial blood processing and analysis. Further, up to 96 samples can be processed at once. Finally, it can be used not only for healthy patients but also for those being treated with clopidogrel or ticagrelor.
Currently, it is generally accepted that the VASP assay is able to determine both HPR (PRI > 50%) and LPR (PRI < 16%) for patients receiving P2Y12 receptor antagonist therapy. 23 The rate of patients with HPR using the standard cutoff of FC-VASP, PRI > 50% (12 of 54), did not differ from that of ELISA-VASP using a similar PRI cutoff > 50% (14 of 54). There was also no significant difference in determining LPR between ELISA-VASP and FC-VASP. The ability of ELISA-VASP to distinguish HPR or LPR in patients with ACS was comparable to FC-VASP and the agreement, confirmed by κ value, was high (.697 for HPR and .738 for LPR). Moreover, in the direct pharmacodynamic comparison of ticagrelor with clopidogrel in patients with ACS, this study demonstrated that ticagrelor provides stronger platelet inhibition than clopidogrel in both platelet function tests. Ticagrelor appears to be superior to clopidogrel in reducing PR during the first 24 hours of ACS.
With regard to the evaluation of PR responses to P2Y12 inhibitors, platelet function monitoring predicts not only ischemia but also bleeding events. 6,9 It is widely accepted that HPR is associated with adverse events, whereas some studies have also suggested a possible link between LPR and bleeding. 24 –27 During the 30 days of follow-up in this study, no major bleeding event occurred in either treatment arm. Eight patients (5 allocated to the ticagrelor arm and 3 to the clopidogrel arm) experienced BARC1 or BARC2 bleeding complications. One patient experienced subacute stent thrombosis (clopidogrel arm). Although there were higher rates of bleeding events and lower rates of ischemic events in patients taking ticagrelor than those taking clopidogrel, there was no significant difference in major adverse cardiovascular events or bleeding in 30 days of follow-up between the groups. We speculate that this may be related to the short observation time and the small sample size. Since both ischemic and bleeding events are important factors for patient survival, attempts should be made to minimize both complications in PCI-treated patients. It was reported that patients with PRI values in the on-treatment platelet range (16% < PRI < 50%) had the lowest rates of adverse events, 10 and the switch from ticagrelor to clopidogrel (or clopidogrel to ticagrelor) was common. 28 Thus, platelet function testing may predict the effect of platelet inhibition and facilitate selection of the optimal P2Y12-inhibitor strategy during therapy.
As this study is a single-center study with a small sample size and short follow-up period, we suggest that multiple-center studies with larger sample sizes are needed to overcome these limitations. In addition, this study did not include prasugrel, which has not been used in China. Finally, we used only 1 platelet assay to compare ELISA-VASP.
Notable progress has been made in the development of new antiplatelet medications to decrease adverse cardiovascular events in patients with ACS. Recently, individualized treatment has become the new way of guiding antiplatelet therapy in patients with ACS. Therefore, tailored antiplatelet treatment might bring clinical benefits to patients with ACS. As a fast, easy, and simple method of detecting platelet inhibition, VASP-ELISA has unique advantages over other methods and may provide guidance for clinical therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the General Hospital of Chinese Armed Police Force under a grant (number WZ2014054).