Abstract
Background
Acute coronary syndrome (ACS) remains a leading cause of mortality globally. Dual antiplatelet therapy (DAPT) is standard after percutaneous coronary intervention (PCI). While ticagrelor has demonstrated favorable myocardial effects in preclinical studies, limited clinical data compared its impact on left ventricular (LV) remodeling versus clopidogrel.
Objective
To evaluate and compare the effect of ticagrelor and clopidogrel on LV remodeling among ACS patients post-PCI.
Methods
Eligible participants were adults (≥18 years) with confirmed ACS who were prescribed ticagrelor or clopidogrel for at least 3 months after PCI and had complete echocardiographic data at baseline and within one-year follow-up. Patients with missing follow-up imaging, switching between antiplatelets, contraindications to DAPT, or noncompliance were excluded. Echocardiographic parameters—LV end-diastolic volume (LVEDV), LV end-systolic volume (LVESV), LV ejection fraction (LVEF)—and B-type natriuretic peptide (BNP) were assessed, and multivariable regression models adjusted for confounders.
Results
A total of 137 patients met criteria (87 on ticagrelor, 50 on clopidogrel). Ticagrelor was associated with an adjusted LVEDV reduction of 8.17 mL compared with clopidogrel (95% CI −15.84 to −0.50; P = .039), an LVESV reduction of 8.09 mL (95% CI −13.88 to −2.29; P = .007), and a greater LVEF improvement of 4.05% (95% CI 2.41-5.70; P < .001). The reduction in BNP was also greater with ticagrelor by 73.56 pg/mL (95% CI −144.08 to −3.05; P = .043).
Conclusion
Ticagrelor was associated with more favorable LV remodeling parameters compared with clopidogrel in this cohort. These findings warrant confirmation in randomized trials.
Keywords
Introduction
Acute coronary syndrome (ACS) is a clinical condition characterized by a sudden reduction in coronary blood flow and remains a leading global cause of mortality. 1 Dual antiplatelet therapy (DAPT) is the cornerstone of treatment for patients with ACS undergoing percutaneous coronary intervention (PCI). The most commonly prescribed antiplatelet agents following PCI include aspirin, clopidogrel, ticagrelor, and prasugrel. 2 Clopidogrel is a selective, irreversible P2Y₁₂ receptor inhibitor, whereas ticagrelor is a reversible antagonist of the same receptor. 3 Both agents exert their therapeutic effects by inhibiting adenosine diphosphate–mediated signal transduction pathways, which are critical for platelet activation and aggregation. As platelets play a central role in the pathogenesis of thrombotic events in atherosclerotic cardiovascular disease, inhibition of platelet aggregation has been shown to reduce the incidence of major adverse cardiovascular events, including myocardial infarction (MI) and death. 3
In patients with ACS, MI leads to loss of contractile function in the affected myocardial region, resulting in ventricular dilation and remodeling. Impaired left ventricular (LV) systolic function, a frequent complication following acute MI (AMI), significantly contributes to the development of congestive heart failure and is a strong predictor of long-term prognosis. 4 Notably, ticagrelor has been shown to attenuate LV remodeling in animal models. 5
Recent evidence suggests that the degree of platelet inhibition may influence postinfarction ventricular remodeling. Several studies have demonstrated that higher on-treatment platelet reactivity is associated with adverse changes in LV volumes and ejection fraction (EF) following MI, despite adherence to guideline-directed therapy. For instance, a prospective study by Miyoshi et al found that patients with elevated platelet reactivity immediately after PCI were significantly more likely to develop adverse LV remodeling, as assessed by echocardiography at 8 months, with platelet reactivity emerging as an independent predictor of remodeling severity. Notably, patients who did not exhibit adverse remodeling had significantly lower platelet reactivity (mean P2Y₁₂ reaction units [PRU] ∼213 vs 260), suggesting a clinically relevant threshold around 245 PRU. 6 Similarly, an observational study by Tsuji et al confirmed that high platelet reactivity—even when measured during the chronic phase post-MI—was independently associated with adverse remodeling in multivariate analysis. 7 These findings support a mechanistic link between suboptimal platelet inhibition and pathological myocardial remodeling. Moreover, the HEALING-AMI trial directly compared ticagrelor and clopidogrel in patients with ST-elevation MI (STEMI) and found that ticagrelor was associated with lower LV end-systolic volume (LVESV) and natriuretic peptide levels, as well as a reduced incidence of adverse remodeling over 6 months. 8
While the HEALING-AMI study suggested potential benefits of ticagrelor on LV remodeling, clinical evidence in this area remains limited and inconsistent. Most existing studies have primarily focused on composite cardiovascular outcomes, such as MI and mortality, rather than direct measures of structural cardiac changes—specifically, LV end-diastolic volume (LVEDV), LVESV, and LV ejection fraction (LVEF). The effect of P2Y₁₂ inhibitors on these remodeling parameters—key predictors of heart failure progression—remains inadequately addressed in clinical research. Furthermore, the HEALING-AMI trial had several methodological limitations, including an open-label design, high dropout rate, absence of intention-to-treat analysis, and concerns regarding medication adherence. In addition, it did not consistently control for background therapies known to influence remodeling. 8 To our knowledge, no other clinical studies have specifically examined the impact of P2Y₁₂ inhibitors on LV remodeling in patients with ACS following PCI. Therefore, the present study aims to evaluate and compare the effects of ticagrelor and clopidogrel on LV remodeling in ACS patients undergoing PCI.
Methodology
A retrospective cohort study was conducted involving adult patients (aged ≥18 years) diagnosed with ACS who underwent cardiac catheterization and PCI between 2010 and 2020 at King Fahad Medical City Heart Center. Eligible participants met the following inclusion criteria: (1) age ≥18 years; (2) confirmed diagnosis of ACS, including STEMI or non-STEMI (NSTEMI); (3) prescribed either ticagrelor or clopidogrel for at least 3 months following PCI; and (4) availability of complete echocardiographic data at both baseline and with a one-year of follow-up.
Exclusion criteria included: (1) missing or incomplete follow-up echocardiographic assessments; (2) switching between ticagrelor and clopidogrel during the follow-up period; and (3) documented contraindications to DAPT or known noncompliance.
Echocardiographic parameters were extracted from formal cardiology reports generated during routine clinical care by board-certified cardiologists using standardized institutional protocols aligned with international echocardiographic guidelines. As this was a retrospective study, formal blinding was not applicable; however, reporting cardiologists were not involved in the study design or data analysis thereby minimizing the risk of assessment bias.
The primary study outcomes included LVESV, LVEDV, LVEF, and B-type natriuretic peptide (BNP) levels, measured at baseline and within one year of follow-up. The proportion of patients with an LVEF <40% was also assessed at both time points.
The study protocol was approved by the institutional review board at King Fahad Medical City (Approval No. 21-155) and conducted in accordance with the ethical principles of the 1975 Declaration of Helsinki.
Statistical Methods
Sample size estimation was conducted using G*Power (version 3.1.9.7), applying the Wilcoxon-Mann-Whitney test suitable for comparing 2 independent groups with non-normally distributed data. An effect size of 0.5 was derived from the published findings of the HEALING-AMI trial. 8 Using a one-tailed test, an alpha error probability of .05, and a target power of 80%, the minimum required sample size was calculated to be 114 participants. A 2:1 allocation ratio favoring ticagrelor was prespecified to allow more detailed outcome assessment in the newer treatment group. To enhance the robustness and generalizability of the findings, the final sample was expanded to include 137 patients: 87 who received ticagrelor (all available eligible records) and 50 who received clopidogrel. This adjustment enabled more comprehensive data utilization. Descriptive statistics were used to summarize categorical variables, which were expressed as frequencies and percentages. The χ2 test (or Fisher exact test when applicable) was employed to compare categorical variables between groups. The Shapiro-Wilk test was used to assess normality of continuous variables. Since all continuous variables were found to be non-normally distributed, they were reported as medians with corresponding ranges (minimum to maximum), and comparisons between groups were performed using the Mann-Whitney U test.
To adjust for potential confounders, robust linear regression models were applied to estimate the adjusted differences in changes from baseline to one-year follow-up in LVEDV, LVESV, LVEF, and BNP levels. Covariates included in the models were age, sex, ACS type, comorbidities, and background medication use. Dyslipidemia was defined in accordance with the 2018 AHA/ACC Cholesterol Management Guideline 9 as the presence of elevated low-density lipoprotein cholesterol (≥130 mg/dL), low high-density lipoprotein cholesterol (<40 mg/dL for men or <50 mg/dL for women), elevated triglycerides (≥150 mg/dL), or ongoing use of lipid-lowering therapy.
A significance level of .05 was considered for all statistical tests. Data analysis was performed using R software (version 4.2.2).
Results
A total of 46,055 patients were initially identified as having received either ticagrelor or clopidogrel between 2010 and 2020. From this cohort, 904 patients were screened using a randomly generated sequence to identify those meeting study criteria. Ultimately, 137 patients fulfilled the inclusion criteria and were enrolled in the final analysis. The selection process is illustrated in Figure 1.

Study flow diagram.
Baseline characteristics are presented in Table 1. The median age of the study cohort was 78 years (range: 54-115), and 66% of the patients were male. Both STEMI and NSTEMI were represented, with STEMI comprising 54% of cases. Hypertension and diabetes were prevalent, affecting 89% and 91% of patients, respectively. The groups were generally comparable, except for a significantly higher proportion of male patients in the ticagrelor group than in the clopidogrel group (80.5% vs 40.0%, P < .001).
Baseline Characteristics.
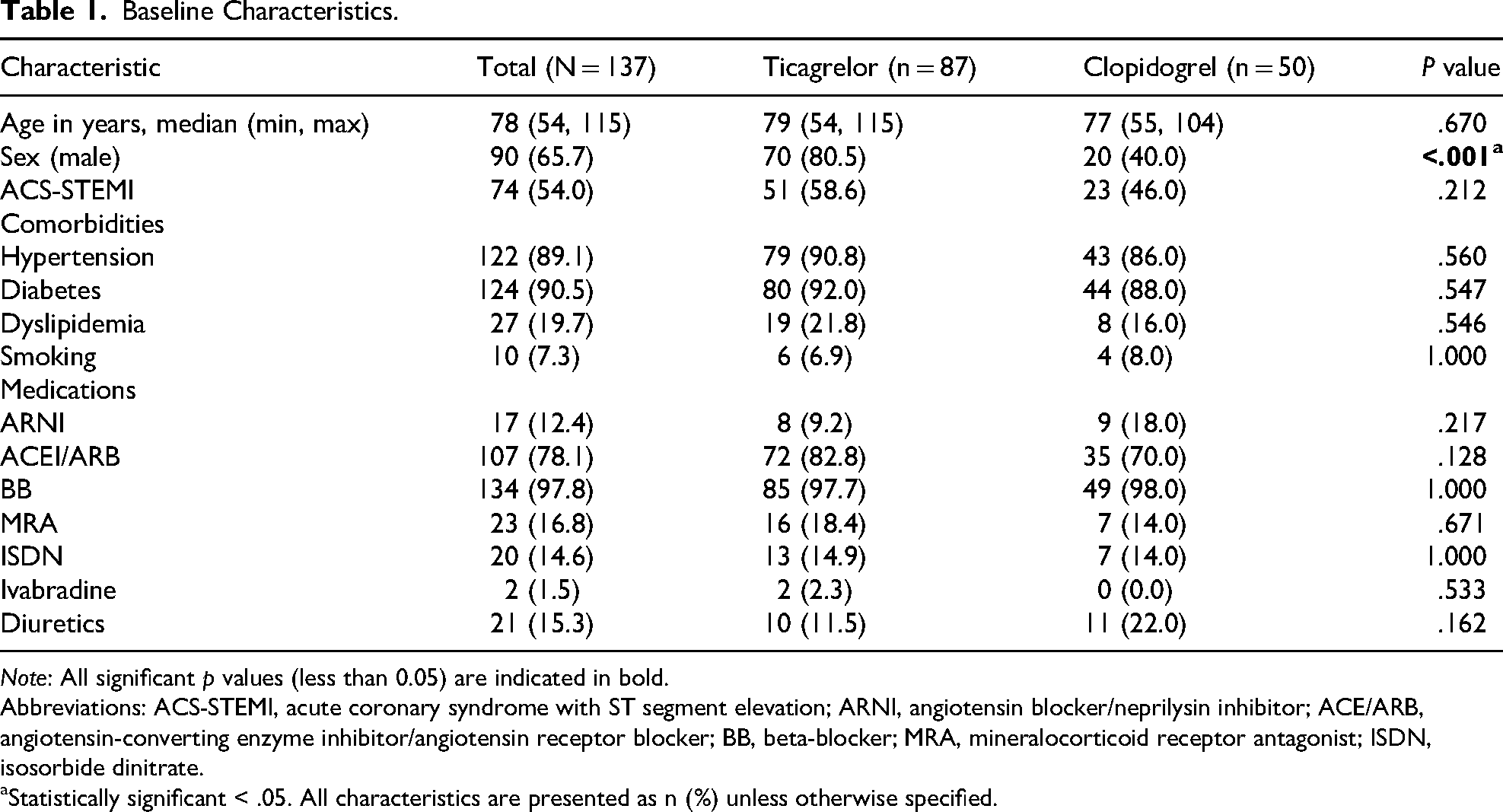
Note: All significant p values (less than 0.05) are indicated in bold.
Abbreviations: ACS-STEMI, acute coronary syndrome with ST segment elevation; ARNI, angiotensin blocker/neprilysin inhibitor; ACE/ARB, angiotensin-converting enzyme inhibitor/angiotensin receptor blocker; BB, beta-blocker; MRA, mineralocorticoid receptor antagonist; ISDN, isosorbide dinitrate.
Statistically significant < .05. All characteristics are presented as n (%) unless otherwise specified.
Cardiac outcomes at baseline and during a one-year follow-up period are detailed in Table 2. Patients in the ticagrelor group exhibited significantly greater improvements in key cardiac parameters. Median reductions in LVEDV and LVESV were more pronounced in the ticagrelor group compared to the clopidogrel group (P = .027 and P = .006, respectively). Similarly, although the median change in LVEF was 0.0% in both groups, the distribution favored the ticagrelor group, which showed significantly higher improvement (P < .001). B-type natriuretic peptide levels declined more substantially in the ticagrelor group (median change −43.0 pg/mL vs −4.5 pg/mL; P = .004).
Cardiac Outcomes at Baseline and Within One-Year Follow up.
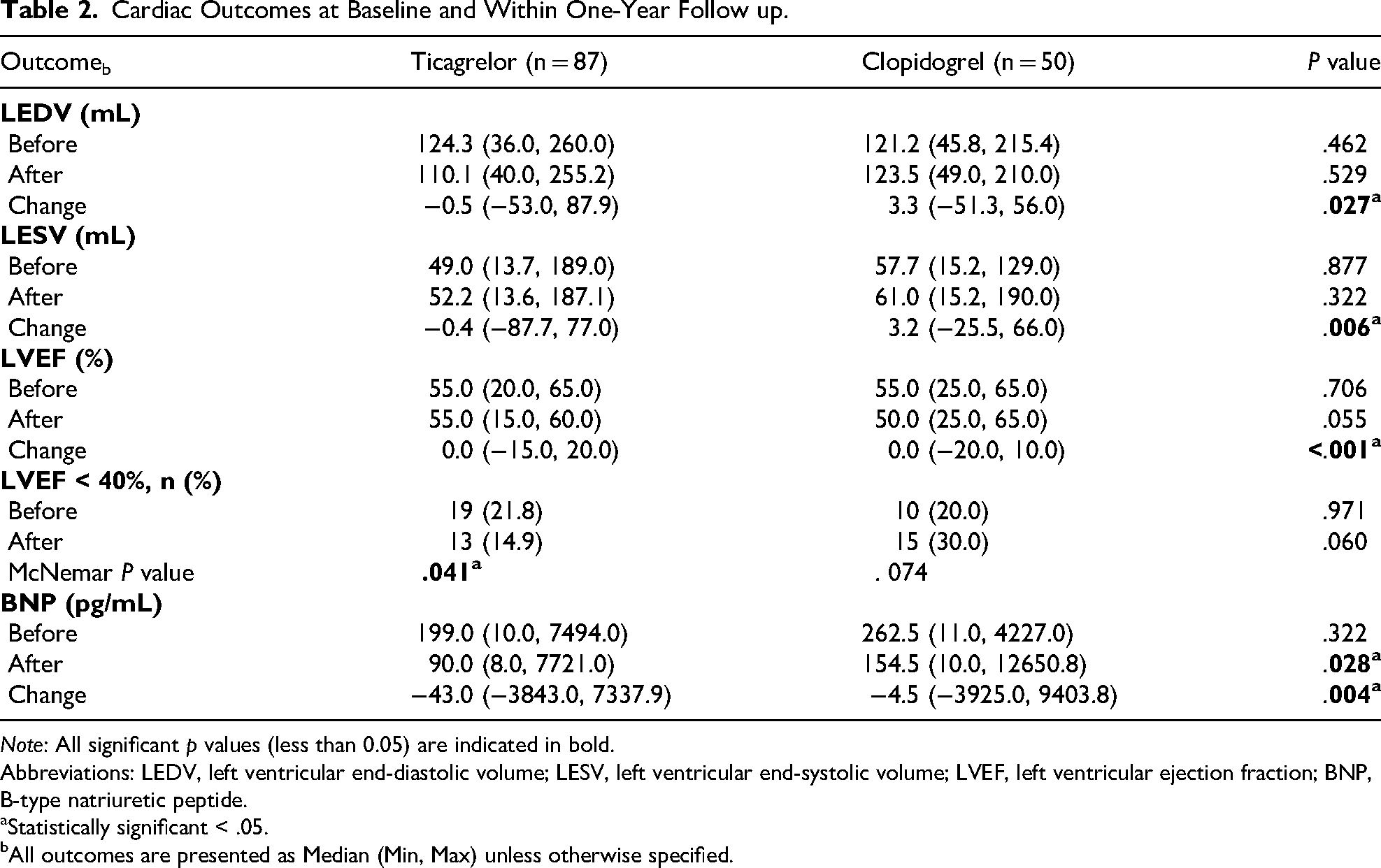
Note: All significant p values (less than 0.05) are indicated in bold.
Abbreviations: LEDV, left ventricular end-diastolic volume; LESV, left ventricular end-systolic volume; LVEF, left ventricular ejection fraction; BNP, B-type natriuretic peptide.
Statistically significant < .05.
All outcomes are presented as Median (Min, Max) unless otherwise specified.
Multivariable robust regression results are presented in Table 3. After adjusting for age, sex, ACS type, comorbidities, and concurrent medications, ticagrelor remained significantly associated with greater reductions in LVEDV (−8.17 mL; 95% CI: −15.84 to −0.50; P = .039), LVESV (−8.09 mL; 95% CI: −13.88 to −2.29; P = .007), and BNP (−73.56 pg/mL; 95% CI: −144.08 to −3.05; P = .043). Ticagrelor was also associated with an adjusted LVEF increase of +4.05% (95% CI: 2.41-5.70; P < .001).
Estimates of the Effect of Ticagrelor Versus Clopidogrel on Cardiac Outcomes Adjusted for Age, Sex, Comorbidities, and Medications by Robust Regression.

Note: All significant p values (less than 0.05) are indicated in bold.
Abbreviations: LEDV, left ventricular end-diastolic volume; LESV, left ventricular end-systolic volume; LVEF, left ventricular ejection fraction; BNP, B-type natriuretic peptide; ACS-STEMI, acute coronary syndrome with ST segment elevation; ARNI, angiotensin blocker/Neprilysin inhibitor; ACE/ARB, angiotensin-converting enzyme inhibitor/Angiotensin receptor blocker; BB, beta-blocker; MRA, mineralocorticoid receptor antagonist; ISDN, isosorbide dinitrate.
Statistically significant < .05.
Subgroup analysis revealed that dyslipidemia was associated with adverse remodeling, including an increase in LVEDV (+12.12 mL; P = .007) and reduction in LVEF (−1.99%; P = .037). In contrast, diabetes mellitus was associated with higher LVEF (+3.71%; P = .005) and lower BNP levels (−141.22 pg/mL; P = .012). The STEMI presentation was also linked to a significant reduction in BNP compared to NSTEMI (−75.12 pg/mL; P = .024). Other covariates such as age, smoking, and use of renin-angiotensin system inhibitors or beta-blockers were not significantly associated with the outcomes.
Discussion
Clinical trials have established that the combination of aspirin and a P2Y₁₂ inhibitor constitutes the primary pharmacotherapeutic strategy for preventing both acute and chronic thromboembolic complications in infarct-related arteries. 1 The Platelet Inhibition and Patient Outcomes (PLATO) study, which involved patients with ACS, demonstrated that ticagrelor was associated with reductions not only in the incidence of MI but also in all-cause and cardiovascular mortality compared to clopidogrel. 10 The underlying mechanism is believed to be related to the adenosine-mediated effects of ticagrelor on vascular and other cellular functions. 3 Moreover, ticagrelor provides greater systemic exposure over a 24-h period due to its active metabolite, in contrast to the shorter plasma half-life of active thienopyridine metabolites of clopidogrel (∼6 h), 11 which may contribute to its more favorable impact on LV remodeling.
Animal studies have corroborated the beneficial effects of ticagrelor on LV remodeling, demonstrating upregulation of cyclooxygenase-2 activity, increased myocardial adenosine concentrations, and phosphorylation of Akt and endothelial nitric oxide synthase—effects that were reversed by adenosine receptor antagonists. 5 Consequently, 4 weeks of ticagrelor therapy led to reduced myocardial fibrosis and inflammation, alongside improvements in LV remodeling. Additionally, ticagrelor-treated animals exhibited higher LVEF compared to those receiving clopidogrel. 12
Consistent with these preclinical findings, our study demonstrated that ticagrelor was associated with significantly reduced LV remodeling, as reflected by reductions in LVEDV and LVESV, and with a significantly higher LVEF compared to clopidogrel. Notably, the HEALING-AMI study also reported that ticagrelor reduced adverse LV remodeling and N-terminal pro-BNP levels; however, it did not demonstrate a significant difference in LVEF between ticagrelor and clopidogrel. 8 Additionally, Liu et al demonstrated in a retrospective study of 80 STEMI patients post-PCI that ticagrelor was associated with significantly faster myocardial perfusion, higher LVEF and cardiac index, and lower BNP and C-reactive protein levels compared to clopidogrel, suggesting that ticagrelor's beneficial effects on ventricular function may be partly mediated through improvements in myocardial microcirculation and attenuation of postinfarction inflammation. 13
While our findings align with the general trend of ticagrelor demonstrating favorable cardiac effects, several aspects distinguish the present study from prior investigations. First, unlike HEALING-AMI 8 and Liu et al, 13 which enrolled exclusively STEMI patients, our cohort included both STEMI and NSTEMI presentations, enhancing the generalizability of findings to the broader spectrum of ACS encountered in routine clinical practice. Second, our one-year follow-up duration extends beyond the 6-month assessment in HEALING-AMI, providing insight into more sustained remodeling changes rather than acute or early subacute effects. The observed improvements in LVEF and reductions in LV volumes, although statistically significant, merit careful interpretation in terms of clinical relevance. Previous studies have shown that even modest improvements in LVEF—such as a 5% to 10% absolute increase—are linked to meaningful reductions in cardiovascular mortality and heart failure hospitalizations among post-MI patients. For example, in the REFINE study, patients with a 1% to 9% increase in LVEF experienced a 47% lower risk of cardiac adverse events compared to those without improvement, with even greater benefit observed in those achieving a ≥10% improvement. 14 Similarly, reductions in LVESV index (LVESVI) have been closely associated with prognosis, and even modest reverse remodeling has been linked to improved survival.15,16 Notably, the adjusted mean reduction in LVESV observed with ticagrelor (−8.09 mL) corresponds to an approximate 13% to 16% decrease from typical baseline volumes in this cohort, a magnitude that aligns with the ≥10% to 15% reduction in LVESVI frequently cited in heart failure and cardiac resynchronization therapy trials as a benchmark for clinically meaningful reverse remodeling. 16
Interestingly, diabetes in our cohort correlated with slightly improved LVEF and lower BNP. This contrasts with the general expectation of worse post-MI outcomes in diabetics. One possible explanation is that diabetic patients derive greater benefit from revascularization and optimized therapy—consistent with reports of larger EF improvement after PCI in diabetics. 17 Nonetheless, given meta-analytic evidence that diabetes promotes adverse remodeling, 18 these findings should be interpreted cautiously and may reflect the efficacy of aggressive post-PCI management in diabetic patients.
Consistent with our results, prior studies have identified dyslipidemia as a driver of adverse remodeling—dyslipidemic MI patients showed greater 6-month LV dilation and EF decline. This reinforces the critical role of intensive lipid-lowering therapy post-PCI. Effective dyslipidemia treatment may improve microvascular recovery and limit remodeling, 19 thereby potentially avoiding the larger end-diastolic volumes and lower EFs observed in patients with uncontrolled lipids.
From a health economic perspective, the choice between ticagrelor and clopidogrel involves balancing clinical efficacy against drug acquisition costs. Multiple cost-effectiveness analyses from diverse healthcare systems have yielded heterogeneous results. In elderly patients (≥70 years) with non-ST-elevation ACS, a Dutch analysis based on the POPular AGE trial demonstrated that clopidogrel was cost-saving (€1485 per patient) with negligible differences in quality-adjusted life years (QALYs) compared to ticagrelor. 20 Conversely, studies from middle-income countries have found ticagrelor to be cost-effective. An Iranian economic evaluation reported an incremental cost-effectiveness ratio (ICER) of US$332 per QALY gained, 21 while a Vietnamese analysis yielded an ICER of approximately US$2010 per QALY, 22 both well within acceptable willingness-to-pay thresholds for their respective healthcare systems.
The present findings on LV remodeling add an important dimension to this economic calculus. Traditional cost-effectiveness analyses of P2Y₁₂ inhibitors have primarily incorporated thrombotic endpoints—recurrent MI, stroke, and cardiovascular death—but have not systematically accounted for the potential impact on heart failure progression, which represents a major driver of long-term healthcare expenditure in post-MI populations. Our observation that ticagrelor is associated with structural cardiac improvements of a magnitude previously linked to reduced heart failure hospitalizations suggests that the clinical benefits of ticagrelor may extend beyond the immediate postdischarge period into the chronic phase of postinfarction care. If these remodeling benefits translate into prevention or delay of heart failure decompensation, the incremental acquisition cost of ticagrelor relative to generic clopidogrel could be partially offset by reductions in downstream resource utilization associated with heart failure management, including hospitalizations, outpatient visits, and intensification of medical therapy.Despite these promising findings, several limitations should be acknowledged. Retrospective cohort studies are inherently limited by the challenge of controlling for multiple confounders. A notable limitation of our study is the significant baseline imbalance in sex distribution, with a higher proportion of male patients in the ticagrelor group. Given the known sex-related differences in post-MI remodeling and outcomes, this imbalance may have influenced our findings. While we applied robust linear regression models to adjust for sex and other potential confounders, the absence of randomization precludes full control for such differences. Another limitation is that LV volumes are influenced by body size, and indexing to Body Surface Area would have improved accuracy; however, the absence of height data prevented such calculations. In addition, the study did not stratify patients according to the infarct-related artery. Evaluating antiplatelet effects in relation to infarct territory would have added further depth to our analysis. Furthermore, our study was not powered to detect subgroup differences, and therefore any observations within subgroups should be interpreted with caution. Given the paucity of data on the impact of antiplatelet therapy on LV remodeling, our study provides additional observational evidence suggesting a potential association between ticagrelor use and improved remodeling outcomes compared to clopidogrel. However, given the inherent limitations of retrospective designs, future prospective, multicenter randomized controlled trials are warranted. These studies should incorporate standardized timing for echocardiographic assessments, longer follow-up durations, and stratification based on key comorbidities such as diabetes and hypertension. A focus on both functional (LVEF) and structural (LV volumes) endpoints, alongside biomarker analysis, would further strengthen the evidence base and guide clinical decision-making in antiplatelet therapy selection post-PCI.
Conclusion
In this retrospective cohort study, ticagrelor was associated with significantly more favorable LV remodeling compared to clopidogrel in ACS patients undergoing PCI, as evidenced by greater improvements in LV volumes, EF, and natriuretic peptide levels over a one-year follow-up period. These findings carry clinical relevance, as the observed structural improvements align with thresholds previously associated with reduced cardiovascular mortality and heart failure hospitalizations in post-MI populations, suggesting that antiplatelet agent selection may influence not only thrombotic outcomes but also the trajectory of myocardial recovery following acute ischemic injury.
Given the inherent limitations of the retrospective design, these findings should be regarded as hypothesis-generating and require confirmation in prospective randomized trials specifically powered to evaluate LV remodeling as a primary endpoint. Future studies incorporating standardized imaging protocols, longer follow-up durations, and adequate sample sizes to assess potential effect modification by infarct territory and baseline ventricular function are needed to definitively establish whether the structural cardiac advantages observed here translate into durable long-term clinical benefit.
Footnotes
Acknowledgments
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Large Research Project under grant number RGP1/59/46.
Ethical Considerations
The study protocol was approved by the institutional review board at King Fahad Medical City (Approval No. 21-155) in accordance with the ethical guidelines of the 1975 Declaration of Helsinki.
Author Contributions
Fawaz H Tawhari contributed to the conceptualization and methodology of the study, supervised the clinical direction, led the writing of the original draft, reviewing and editing the manuscript. Mohammad F Zaitoun contributed to the conceptualization, methodology, data curation, and formal analysis. He participated in the writing of the original draft, reviewing and editing the manuscript. Ehab Elmongui was responsible for formal analysis and validation of the findings and participated in writing, reviewing and editing the manuscript. Arwa Alhuwail contributed to data curation, reviewing and editing of the manuscript. Ali Najmi contributed to data curation, reviewing and editing of the manuscript. Wasayef M Alqahtani contributed to data curation, reviewing and editing of the manuscript. Zeyad H Sabey contributed to data curation, reviewing and editing of the manuscript. Atheer Almohammed contributed to data curation, reviewing and editing of the manuscript. Abdulrahman Al Qahtani contributed to data curation, reviewing and editing of the manuscript. Hani Sultan contributed to data curation, reviewing and editing of the manuscript. Ahmed R. N. Ibrahim contributed to data curation, reviewing and editing of the manuscript. Sultan Alshahrani contributed to data curation, reviewing and editing of the manuscript. Reem Bahmaid served as the research supervisor, supported project administration, ensured compliance with research governance, and contributed to reviewing and editing the manuscript.
Data Availability
Data could be available only upon official request and approval from the institutional review board.
