Abstract
Background:
Ticagrelor is an intriguing antiplatelet agent with a potentially beneficial impact on endothelial dysfunction and confers a mortality benefit beyond 1 month after acute coronary syndrome (ACS). However, little data exist on whether ticagrelor improves endothelial dysfunction in stable patients who survive the acute period and receive guideline-directed medical therapy.
Methods and Results:
This study is a prospective, randomized, parallel, open-labeled study that enrolled 30-day survivors of non-ST-segment elevation ACS (NSTE-ACS). Forty patients with NSTE-ACS were randomly assigned to ticagrelor or clopidogrel groups. The primary end point was the change in the percentage brachial artery flow-mediated dilation (baFMD) from baseline. Baseline characteristics were not different between the 2 groups. The median time from the stent implantation to screening was 269 days. After 30 days of study medication administration, the change in the percentage baFMD value was similar between the ticagrelor and clopidogrel groups (−0.08 [1.42] vs 0.30 [1.69],
Conclusions:
This dual time point baFMD study demonstrated that treatment with ticagrelor was not superior to clopidogrel for improving endothelial dysfunction in stabilized patients with NSTE-ACS.
Introduction
Ticagrelor, a direct-acting, reversibly binding P2Y12 antagonist, has significantly reduced all-cause and cardiovascular death in patients with acute coronary syndrome (ACS), and it has unique side effects such as dyspnea and ventricular pause. 1 -4 Several studies revealed that these profiles of ticagrelor may be associated with pleiotropic effects beyond the potent antiplatelet activity. 4 -8 Mechanisms potentially related to the improvement in endothelial function have been proposed. 4,9 While a series of clinical trials investigated the effect of ticagrelor on endothelial function in patients with coronary artery disease (CAD), the results were contradictory. 10 -18 Probable explanations of these inconsistent findings include different clinical presentations, comorbidities, methods to evaluate endothelial function, timing from the index event, and the various statuses of concomitant medications. In the 2 studies showing a beneficial effect, patients with ACS, not stable angina, in the acute period were enrolled, and the prescription rate of concomitant medications was lower or unknown. 12,15 Conversely, the trials showing a lack of effect enrolled stable patients with prior ACS and demonstrated a higher use of concomitant medications. 16 -18 However, this result was measured by peripheral arterial tonometry, which evaluates the microvascular response, whereas brachial artery flow-mediated dilation (baFMD) measures the macrovascular response. Brachial artery flow-mediated dilation is the most established and commonly used method to evaluate endothelial function. 9,19 Accordingly, we designed the present study to assess endothelial function using baFMD. We hypothesized that ticagrelor is continuously working to improve endothelial dysfunction, which contributes to its sustained survival benefits, even in stabilized patients with ACS receiving guideline-directed treatment who are treated with drug-eluting stents and prescribed statins and neurohormonal blockades. Therefore, the objective of the current study was to determine whether ticagrelor improves endothelial dysfunction, as measured by baFMD, in patients with non-ST-segment elevation ACS (NSTE-ACS) who have been stable for more than 30 days after an index event in real-world clinical practice.
Methods
Patients older than 20 years were eligible to participate in this study if they satisfied all of the following inclusion criteria: (1) a documented history of NSTE-ACS and successful treatment with percutaneous coronary intervention (PCI) using drug-eluting stents and (2) stable condition beyond 30 days after the index PCI using aspirin and clopidogrel. Exclusion criteria were as follows: (1) BaFMD percent values higher than 10% at the screening examination. To focus on whether ticagrelor improves endothelial dysfunction, investigators excluded patients with excellent endothelial function under the dual-antiplatelet therapy including clopidogrel. The value of 10% was decided on the basis of the average value of the previous Korean study including stable patients. 20 The percentage baFMD value was defined as (post-FMD − pre-FMD)/pre-FMD × 100; (2) angina-related symptoms; (3) failed invasive treatment; (4) hypersensitivity to ticagrelor or clopidogrel; (5) anticoagulant therapy; (6) decompensated congestive heart failure; (7) intractable arrhythmia; (8) second- or third-degree atrioventricular block; (9) uncontrolled hypertension; (10) high risk of hemorrhage due to coagulation disorders, gastrointestinal bleeding, gross hematuria, intraocular bleeding, hemorrhagic stroke, or intracranial hemorrhage; (11) more than moderate chronic obstructive pulmonary disease and a history of bronchial asthma; (12) renal replacement therapy; (13) moderate to severe hepatic impairment; (14) platelets <100 000/μL; (15) hematocrit <30%; (16) concomitant oral or intravenous strong CYP3A4 inhibitors, CYP3A substrates with narrow therapeutic indices, or strong CYP3A4 inducers; (17) planned elective surgery or invasive procedure; (18) pregnancy, breastfeeding, or not using acceptable birth control; and (19) unsuitability based on the medical judgment of the investigators.
The current study was a prospective, randomized, parallel, open-labeled study. Figure 1 depicts the study flowchart. A total of 49 patients underwent screening for baFMD. After excluding 9 patients, 40 were randomized into 2 groups of 20. The ticagrelor group received ticagrelor 90 mg twice daily for 30 days, and the clopidogrel group received clopidogrel 75 mg once daily for 30 days. Aspirin was given to all patients, and other optimal medications were administered at the physician’s discretion. Both BaFMD and nitroglycerin-mediated dilation (NMD) were performed during the screening and at 30 days after each treatment. An independent examiner who was unaware of the randomization assignments performed the tests. Blood samples were obtained at the time of randomization and follow-up. The safety profiles including adverse events and vital signs were collected using standard questioning at every visit. The investigators also evaluated the patients’ medication compliance. Any remaining pills were considered to be equal to the number of days of missed treatment, and consumption of the medication was confirmed by compliance questionnaires. All procedures in this study were performed in accordance with the 1964 Helsinki Declaration, and this study was approved by the institutional review boards of Catholic Medical Center, The Catholic University of Korea. Written informed consent was obtained from all enrolled patients before the screening. This study has been registered on the ClinicalTrials.gov (NCT02379676).

The study flowchart.
The primary end point was the change in the percentage baFMD between baseline and 30-day follow-up. The secondary end points were the anti-inflammatory effect and platelet reactivity. Other prespecified efficacy variables regarding the endothelial dysfunction included the mean percentage baFMD value and the incidence of patients with a percent baFMD value of less than 7% after 30 days of treatment with ticagrelor or clopidogrel. The secondary efficacy variables were the changes in the high-sensitive C-reactive protein (hs-CRP) and P2Y12 reaction units between baseline and follow-up.
Endothelial function was assessed by measuring the FMD of the brachial artery as described as follows.
21
After an overnight fast and an absence of caffeine ingestion (such as coffee, tea, cola, alcohol, and energy supplements) and cigarette smoking for 48 hours, the participants underwent baFMD and NMD. Owing to circadian variations in the peripheral vascular tone, the test was performed between 9 and 10
The hs-CRP was assayed by a Hitachi 7600-110 autoanalyzer (Hitachi Co, Tokyo, Japan), with a detection limit of 0.1 mg/L; 90% of the normal value was <3.0 mg/L. The effect on platelet aggregation was measured using the VerifyNow P2Y12 assay (Accumetrics, San Diego, California). The P2Y12 reaction units were estimated according to the increase in the light transmittance resulting from the adenosine diphosphate–induced platelet aggregation in the presence of prostaglandin E1.
All baseline characteristics are summarized as the mean (standard deviation) or median (interquartile range [IQR]) for continuous variables and number (percentage) for categorical variables. Differences between groups were analyzed using the Student
Results
Table 1 presents the baseline characteristics of the patients. There was no significant difference between the treatment groups. The median time from PCI to screening was 269.0 days (IQR: 182.0-313.8) in the ticagrelor group and 232.5 days (IQR: 143.5-285.3) in the clopidogrel group (
Baseline Characteristics.
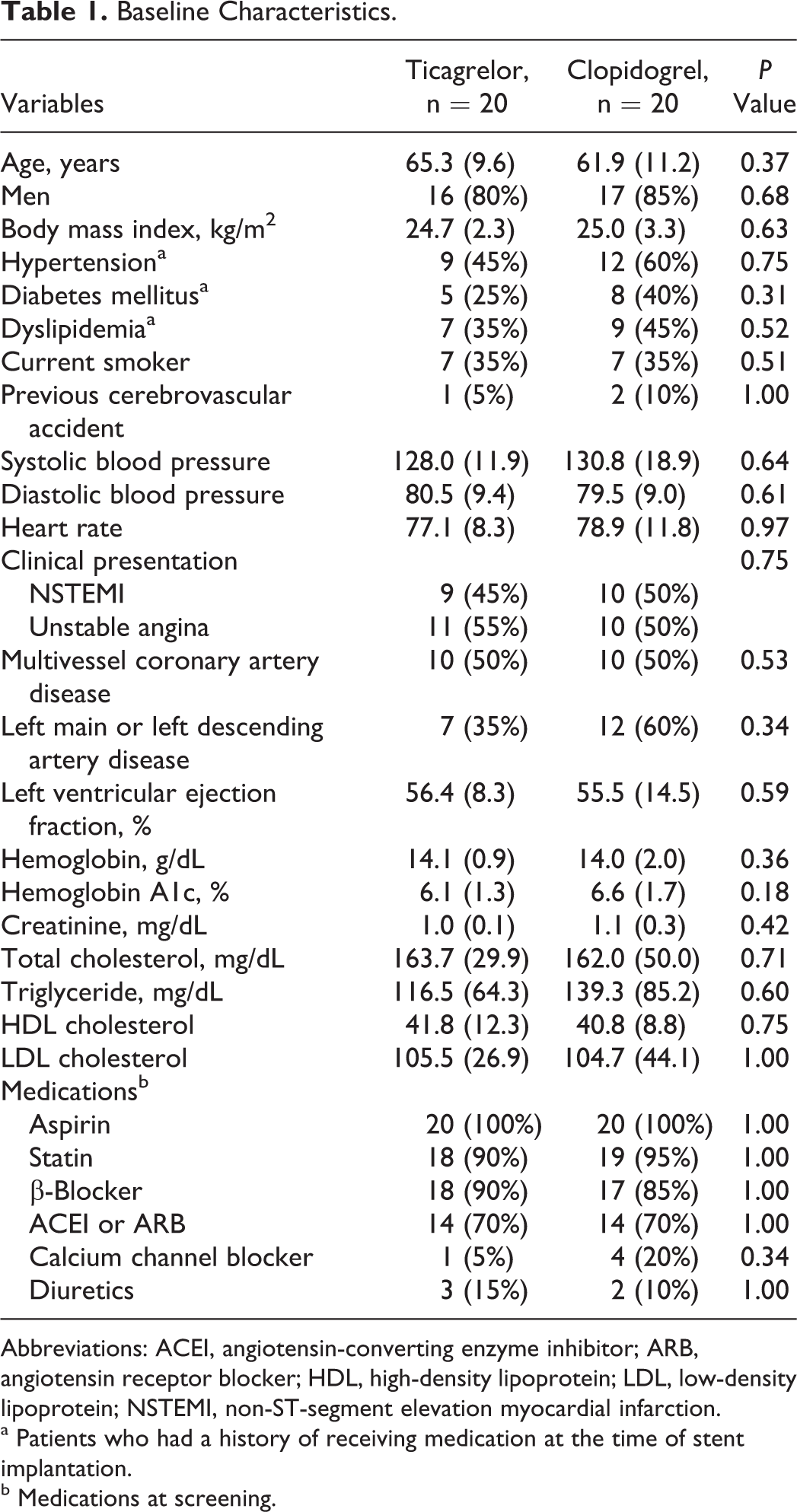
Abbreviations: ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; HDL, high-density lipoprotein; LDL, low-density lipoprotein; NSTEMI, non-ST-segment elevation myocardial infarction.
a Patients who had a history of receiving medication at the time of stent implantation.
b Medications at screening.
After 30 days of treatment, ticagrelor failed to improve the primary end point compared to clopidogrel. The change in the percentage baFMD between baseline and 30-day follow-up was similar in both groups (−0.08 [1.42] vs 0.30 [1.69],
Outcome Measures According to the Treatment Groups.

Abbreviations: FMD, flow-mediated dilation; hs, high-sensitivity; NMD, nitroglycerin-mediated dilation.
a Seventeen patients completed the follow-up tests in the ticagrelor group.
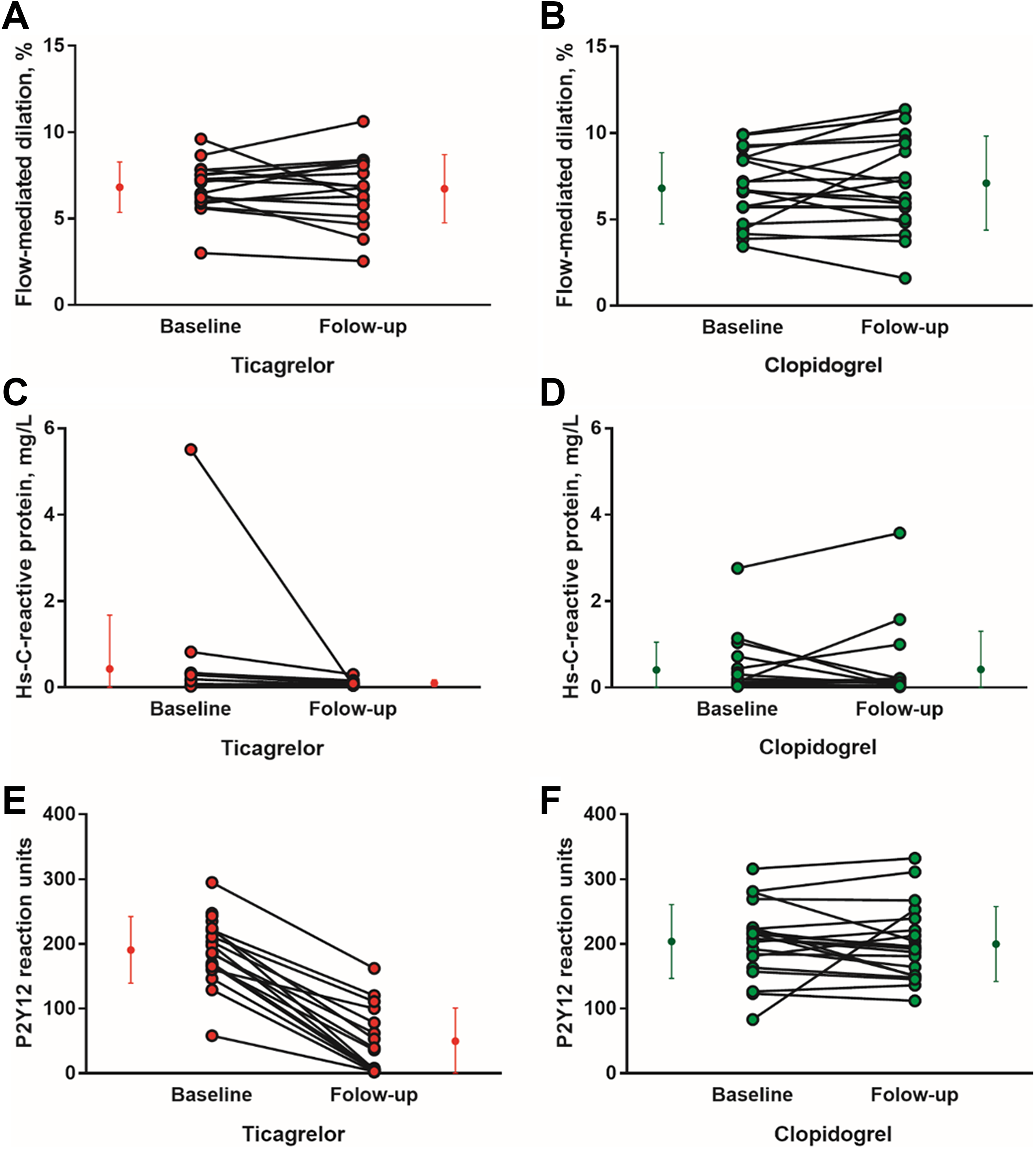
Individual data of the flow-mediated dilation in the (A) ticagrelor and (B) clopidogrel groups, high-sensitivity C-reactive protein levels in the (C) ticagrelor and (D) clopidogrel groups, and P2Y12 reaction units in the (E) ticagrelor and (F) clopidogrel groups.
Neither the initial hs-CRP level (0.50 [1.2] mg/L vs 0.41 [0.64] mg/L,
Discussion
Since the effect of ticagrelor on the vascular endothelium has been proposed as one of the major mechanisms for the steady reduction in the cardiovascular mortality of patients with ACS, we sought to investigate the effect of ticagrelor compared to clopidogrel on endothelial dysfunction using baFMD in stabilized patients who underwent PCI for NSTE-ACS. However, we found that ticagrelor did not improve endothelial dysfunction in the stabilized NSTE-ACS survivors after they had undergone drug-eluting stent implantation and received guideline-directed medical therapy which may have positively affected their endothelial dysfunction.
Ticagrelor is a direct-acting, reversibly binding P2Y12 antagonist, whereas the thienopyridines, such as prasugrel and clopidogrel, are prodrugs whose active metabolites irreversibly bind to the P2Y12 receptor. 2 -4 Ticagrelor, in contrast to prasugrel, has been shown to significantly reduce the incidence of death from any cause including cardiovascular causes. 1,23 The survival curve was shown to diverge even after 30 days from the index event in patients with ACS. Moreover, ticagrelor has unique side effects such as dyspnea and ventricular pauses. These observations have led to the hypothesis that ticagrelor may have mechanisms of action beyond the potent P2Y12 antagonism. 4,5 Ticagrelor inhibits the cellular uptake of adenosine, thereby prolonging the half-life of extracellular adenosine, 8 and induces adenosine triphosphate release from human erythrocytes being degraded into adenosine. The biologic effects of adenosine on ameliorating endothelial dysfunction are well established. 4,9,24 In addition, ticagrelor was found to block the contractile effect of adenosine diphosphate in the vascular smooth muscle that expresses P2Y12 receptors. 25 In humans, a loading dose of ticagrelor 180 mg increased the plasma adenosine concentration in patients with ACS compared to a loading dose of clopidogrel 600 mg. 5 A loading dose of ticagrelor 180 mg also enhanced adenosine-induced coronary blood flow velocity in healthy volunteers 6 and in patients with NSTE-ACS. 7 These pleiotropic effects may contribute to the survival-improving effect of ticagrelor.
Several clinical trials have investigated the effect of ticagrelor on endothelial function in patients with CAD. Whereas some studies showed beneficial effects, 10 -15 others demonstrated that it had a lack of effect on endothelial function. 16 -18 Several probable factors, such as comorbidities, and methods to evaluate endothelial function, will be associated with these inconsistent results. Previous studies examined patients with different comorbidities. Campo et al 14 included patients with stable CAD with chronic obstructive pulmonary disease, and Mangiacapra et al 11 and Jeong et al 15 enrolled diabetic patients. Chronic obstructive pulmonary disease and diabetes are related to endothelial dysfunction and may alter endothelial function. 26,27 Therefore, these various comorbidities may explain differences in the findings. In addition, endothelial function in each study was measured with a different assay: intracoronary agonist infusion with quantitative coronary angiography, brachial artery ultrasound with FMD, vascular tonometry and measurements of vascular stiffness, and the rate of apoptosis in human umbilical vein endothelial cells. 14,28 Because the endothelial function is a very complex phenomenon and each method has its own advantages and disadvantages, different assays may be associated with varying results in the variable clinical settings.
Other probable factors including timing from the index event and the various statuses of concomitant medications also can explain controversial findings among the previous studies. In the studies showing a beneficial effect, 2 randomized trials enrolled patients with ACS at the acute period. 12,15 In addition, the prescription rate of concomitant medications was unknown or significantly lower in the randomized studies with positive results than that in the present study; 3 randomized studies published as a short article did not report the status of concomitant medications, 11 -13 and 1 important clinical trial showed a markedly lower rate of concomitant medication use compared to our study (42% vs 70% for renin–angiotensin system inhibitors and 18% vs 88% for β-blockers). 15 Although only one study showing the positive effect of ticagrelor enrolled stable patients with prior ACS, it was an observational registry-based study. 10 Conversely, 2 recent randomized trials and an observational trial including stable patients beyond 1 month or 1 year after prior ACS revealed the lack of effect of ticagrelor on endothelial function. In these studies, the use of concomitant medications was similar to that in our study (67%-90% for renin–angiotensin system inhibitors and 80%-100% for β-blockers). 16 -18 Since endothelial function is greatly influenced by renin–angiotensin system inhibitors and β-blockers, 29,30 the prescription rate of these medications could be assumed to influence the results of any study testing the association of ticagrelor with improved endothelial dysfunction. Another important consideration is that we recruited patients with stabilized ACS after a median of 269 days from an index event, meaning that all enrolled patients were in a steady state of stabilized ACS under guideline-directed medical therapy. Under these conditions (stable patients with prior ACS and frequent use of neurohormonal blockades), we found that ticagrelor did not improve endothelial dysfunction measured by baFMD.
All of the 3 studies showing the lack of effect of ticagrelor in stable patients with prior ACS used peripheral arterial tonometry to assess endothelial function. 16 -18 Peripheral arterial tonometry and baFMD provide distinct information regarding vascular function, and baFMD is the most established and commonly used method, although baFMD is more technically difficult. 9,19 However, until now, only 2 prior studies have evaluated the effect of ticagrelor on endothelial dysfunction using baFMD. One study evaluated patients with stable CAD but did not enroll patients with prior ACS, 11 while the other enrolled patients with ACS with type 2 diabetes in the acute period. 15 We performed baFMD to assess endothelial function in patients with stabilized ACS and found no beneficial effect of ticagrelor on endothelial dysfunction, in accordance with the results of previous studies using peripheral arterial tonometry. 16 -18
This study has several important limitations. First, the relatively small number of enrolled patients has made this study underpowered for the detection of small differences in the baFMD. As mentioned previously, we calculated the sample size based on the results of a trial with high-dose clopidogrel and baFMD, 22 because there were no available baFMD data on ticagrelor at the time of the study design. Second, we did not measure the serum adenosine level, which is thought to be the main mechanism for the improvement in endothelial function. Third, we did not measure the levels of inflammatory cytokines such as interleukin, interferon, tumor necrosis factor, and epidermal growth factor (EGF), which are potentially associated with endothelial function. A recent study demonstrated that ticagrelor reduced the circulating levels of EGF. 31 A decrease in EGF level correlated with on-treatment platelet reactivity and resulted in the activation of endothelial nitric oxide synthase in the vascular endothelium. Although EGF can negatively affect endothelial function, it has not been established whether the EGF production is related to baFMD. Thus, the results of the present study cannot conclude whether possible mechanisms that are not captured by flow-mediated vasodilation can affect endothelial function after the treatment with ticagrelor.
In conclusion, compared to clopidogrel, ticagrelor did not confer an additional beneficial effect on endothelial dysfunction measured by baFMD beyond its superior antiplatelet effect in stabilized patients with NSTE-ACS. This finding suggests that the beneficial effects of ticagrelor outside of its antiplatelet activity may not be sufficient to make a difference in the long-term clinical outcomes of stable patients with ACS, although potent platelet inhibition may be associated with a better prognosis. Further large, well-designed trials are necessary to elucidate the clinical significance of the pleiotropic effects of ticagrelor.
Footnotes
Authors’ Note
The corresponding author takes responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation. This study was designed and conducted by the investigators. There was no industry involvement in the design, conduct, and analysis of this study.
Acknowledgments
The authors thank Yunhee Yang, RN, MPH, for contributing to our study.
Author Contributions
Sungmin Lim and Eun Ho Choo contributed equally to this study. Sungmin Lim, Eun Ho Choo, and Kiyuk Chang contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. Chan Joon Kim contributed to conception, contributed to acquisition, and drafted manuscript. Ik Jun Choi contributed to design, contributed to interpretation, and drafted manuscript. Kwan Yong Lee contributed to conception and contributed to analysis. Byung-Hee Hwang contributed to design and contributed to analysis. Jong-Min Lee and Wook Sung Chung contributed to conception and contributed to acquisition. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from AstraZeneca.
