Abstract
Introduction:
Clopidogrel is an antiplatelet drug widely used in patients with acute coronary syndromes or stroke. Despite adequate antiplatelet therapy, some patients develop acute ischemic events. This is partly attributed to the fact that they have poor inhibition of platelet reactivity, despite treatment. This study aimed to assess the impact of clinical and demographic variables and of cytochrome P450 2C19 (CYP2C19) loss-of-function polymorphisms on platelet response to clopidogrel evaluated using impedance aggregometry in an East European population.
Methods:
The study included 189 clopidogrel-treated patients with acute coronary syndromes and noncardiogenic ischemic stroke. Platelet aggregation was evaluated by impedance aggregometry. CYP2C19 loss-of-function polymorphisms were detected using the polymerase chain reaction restriction fragment length polymorphism technique. Various clinical and demographic data were also recorded.
Results:
In our data set, 81% of the patients were responders and 19% nonresponders to clopidogrel therapy. The distribution of CYP2C19 polymorphisms was as follows: 61.1% of patients were CYP2C19 wild-type homozygotes, 27.7% of patients were CYP2C19*2 heterozygotes, 1.1% of patients were CYP2C19*3 heterozygotes, and 10% of patients were CYP2C19*2 homozygotes. The highest level of association with clopidogrel response status was found for CYP2C19 polymorphisms, concomitant aspirin treatment, leukocyte and platelet count, history of myocardial infarction, arterial hypertension, and ward where patients were admitted.
Conclusion:
The prevalence of clopidogrel resistance in our East European population was in line with that reported for Western populations. Clopidogrel response was significantly influenced by the presence of CYP2C19 polymorphisms. Interestingly, the concomitant use of aspirin had a significant impact on platelet response to clopidogrel, indicating a synergic interaction between these drugs.
Introduction
Clopidogrel, an irreversible thienopyridine P2Y12 receptor antagonist, is widely used in patients with acute coronary syndrome (ACS) or ischemic stroke. 1 –3 Despite apparently adequate antiplatelet therapy, up to 10% of patients treated with the combination of aspirin and clopidogrel will experience a recurrent acute ischemic event in the first year after an ACS. 4 This is partly attributed to the fact that some patients have poor inhibition of platelet reactivity, despite treatment, and these patients are being considered low responders or nonresponders (“resistant”) to antiplatelet therapy. 5
The prevalence of clopidogrel resistance in various populations appears to be highly variable, ranging from 15.9% to 49.5% in different studies. 6 This large variability between different populations suggests a major impact of genetic and/or environmental factors in clopidogrel resistance. Indeed, in patients treated with clopidogrel, loss-of-function polymorphisms in the gene encoding for cytochrome P450 2C19 (CYP2C19), one of the principal isoenzymes involved in clopidogrel metabolic activation, known as the CYP2C19*2 and CYP2C19*3 allelic variations, have been correlated with higher levels of adenosine diphosphate (ADP)-induced platelet aggregation. 7,8
In addition to genotype, several other factors, often displaying population-specific patterns, have been shown to affect the bioavailability of clopidogrel and to lead to clopidogrel resistance.
To date, the vast majority of the available data are restricted to the North American and West European populations, while Central and East European populations have often been overlooked. Accordingly, we aimed to assess the prevalence of clopidogrel resistance and the most common CYP2C19 loss-of-function polymorphisms (*2 and *3 alleles) in an East European population and to evaluate the impact of these polymorphisms and various clinical and demographic variables on platelet response to clopidogrel using whole blood impedance aggregometry.
Materials and Methods
Participants
The present study included 189 consecutive patients admitted to Cardiology or Neurology Departments between October 2014 and June 2015 either with an ACS unstable angina, non-ST-segment elevation myocardial infarction (NSTEMI), ST-segment elevation myocardial infarction (STEMI), or with noncardiogenic ischemic stroke, respectively.
The study was conducted in accordance with the World Medical Association Declaration of Helsinki and was approved by Ethics Committee of the University of Medicine and Pharmacy Tîrgu Mureş, No.100/06.10.2014. Written informed consent was obtained from all patients.
All patients received clopidogrel according to the current recommendations for ACS and stroke. 2,3,9 Exclusion criteria were use of glycoprotein IIb/IIIa inhibitors, hematological disorders, liver or renal failure, hemoglobin level less than 8 g/dL, a platelet count of less than 100 × 103/µL or more than 450 × 103 /µL, major surgery in the week preceding the enrollment, and admitted noncompliance to antiplatelet therapy.
Blood Sampling
Blood samples were drawn via venipuncture after overnight fasting, 5 days after starting the treatment with 75 mg of clopidogrel, and 24 to 96 hours after performing percutaneous coronary intervention (PCI) in case of patients with ACS treated by this procedure.
For CYP2C19 genotyping, blood was collected into vacutainer tubes containing potassium EDTA as anticoagulant (Becton Dickinson, Franklin Lakes, New Jersey) and preserved at 2°C to 8°C until processing.
In order to reduce procedure-related platelet activation, the second tube was used to evaluate platelet aggregation. Impedance aggregometry was performed between 30 minutes and 2 hours after blood sampling, using whole blood samples collected into commercially available double-wall, hirudin blood tubes (Roche Diagnostics International Ltd, Rotkreuz, Switzerland).
Platelet Function Assessment
The intensity of platelet aggregation was evaluated by impedance aggregometry using the Multiplate analyzer (Roche Diagnostics, Mannheim, Germany) and standard procedures recommended by the manufacturer.
The principle of this test is based on the fact that adhesion and aggregation of activated platelets on the sensors’ surface enhances the impedance between the 2 sensor electrodes. Whole blood of 300 µL was diluted in the test cells with 300 µL NaCl (0.9%) and stirred at 37ºC for 3 minutes. At the end of this incubation period, the platelet agonist was added, and changes in electrical impedance were recorded continuously for 6 minutes.
The agonist used was ADP (6.3 µmol/L) in the presence of prostaglandin E1 (9.4 nmol/L), which was used to suppress the undesirable contribution of P2Y1 receptors, making the assay specific for the P2Y12 receptor pathway (also called ADP high sensitive test). The increase in impedance was transformed into arbitrary aggregation units (AU) plotted against time (AU × min). Platelet aggregation was expressed as the area under the aggregation curve (AUC).
The lowest value of the reference interval provided by the manufacturer (43-100 U) was used as diagnostic cutoff. Patients receiving clopidogrel with results within the reference range of patients not taking the antiplatelet were considered nonresponders to clopidogrel therapy. 10
Genetic Analysis
Genomic DNA (gDNA) was extracted from EDTA-anticoagulated whole blood samples using a PureLink Genomic DNA Mini Kit (Invitrogen), following the manufacturer’s instructions.
CYP2C19 genetic polymorphisms were detected using the polymerase chain reaction (PCR) restriction fragment length polymorphism technique as described previously by Omar. 11
Polymorphisms were analyzed for the nonfunctional alleles *2 and *3 by identifying 2 polymorphic positions, change in the nucleotide guanine for an adenine at position 681 in exon 5 and change in the nucleotide guanine for an adenosine at position 636 in exon 4 of the CYP2C19 gene. Sequences including the polymorphisms were amplified by PCR using a Mastercycler gradient (Eppendorf AG, Hamburg, Germany).
Amplification products were digested using SmaI restriction enzyme for the detection of CYP2C19*2 polymorphism and BamHI for CYP2C19*3 polymorphism (Thermo Scientific FastDigest enzymes; Thermo Scientific).
The following genotypes were examined: CYP2C19*1/*1, CYP2C19*1/*2, CYP2C19*1/*3, CYP2C19*2/*2, CYP2C19*2/*3 and CYP2C19*3/*3. Based on the CYP2C19 genotype results, patients were classified as wild-type homozygote (*1/*1 allele), heterozygote (*1/*2, *1/*3), or variant homozygote (*2/*2, *2/*3, *3/*3), corresponding to extensive (EM), intermediate (IM) and poor metabolizers (PM), respectively. 12
Statistical Analysis
Statistical analysis was performed with IBM SPSS Statistics v22.0 software (Armonk, New York). The results were considered significant at the level α less than .05 (α = .05). Continuous variables were expressed as mean ± standard deviation or median and interquartile range (for nonparametric data), whereas categorical data were represented as counts and percentage. The assumption of normal distribution for the continuous variables was verified with Shapiro-Wilk test and visual inspection of their histograms, normal Q-Q plots, and box plots. Comparisons between groups were performed with the 2-tailed
All independent variables with
Variables able to significantly predict the status of clopidogrel resistance were included in the multivariate regression analysis to identify the independent contribution of each of the factors to the interindividual variability in platelet reactivity. The assumption of independence of residuals was tested with the Durbin-Watson test at a cutoff ≥1. Multicollinearity/singularity of independent variables was excluded using collinearity statistics and a tolerance
Results
Of the 189 patients initially screened, 12 were excluded from the study due to noncompliance to antiplatelet therapy; the remaining 177 patients were included in the analysis. Baseline characteristics of the enrolled patients are summarized in Tables 1 and 2.
Baseline Demographical and Clinical Characteristics of the Study Population.
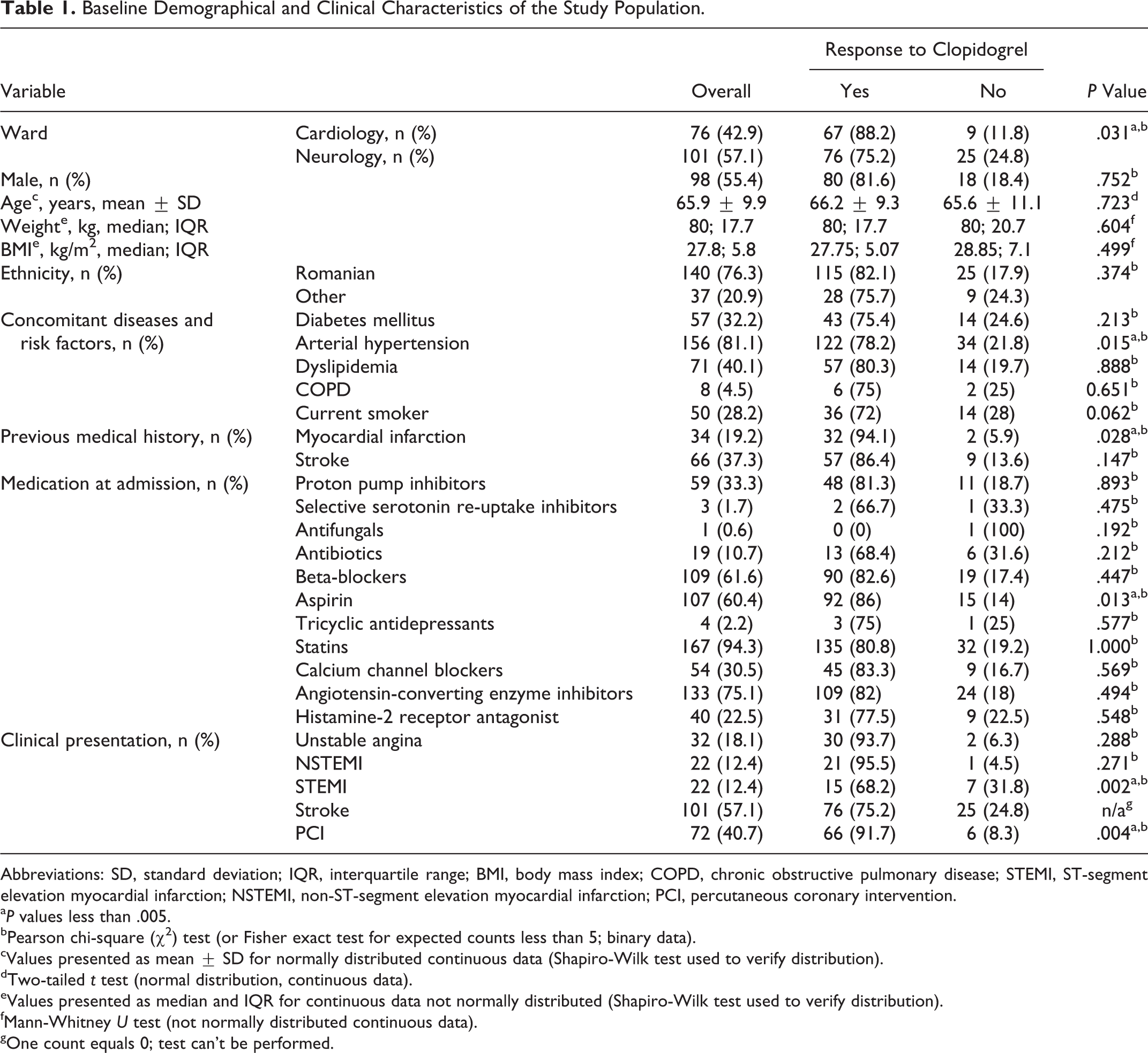
Abbreviations: SD, standard deviation; IQR, interquartile range; BMI, body mass index; COPD, chronic obstructive pulmonary disease; STEMI, ST-segment elevation myocardial infarction; NSTEMI, non-ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention.
a
bPearson chi-square (χ2) test (or Fisher exact test for expected counts less than 5; binary data).
cValues presented as mean ± SD for normally distributed continuous data (Shapiro-Wilk test used to verify distribution).
dTwo-tailed
eValues presented as median and IQR for continuous data not normally distributed (Shapiro-Wilk test used to verify distribution).
fMann-Whitney
gOne count equals 0; test can’t be performed.
Baseline Laboratory Characteristics of the Study Population.
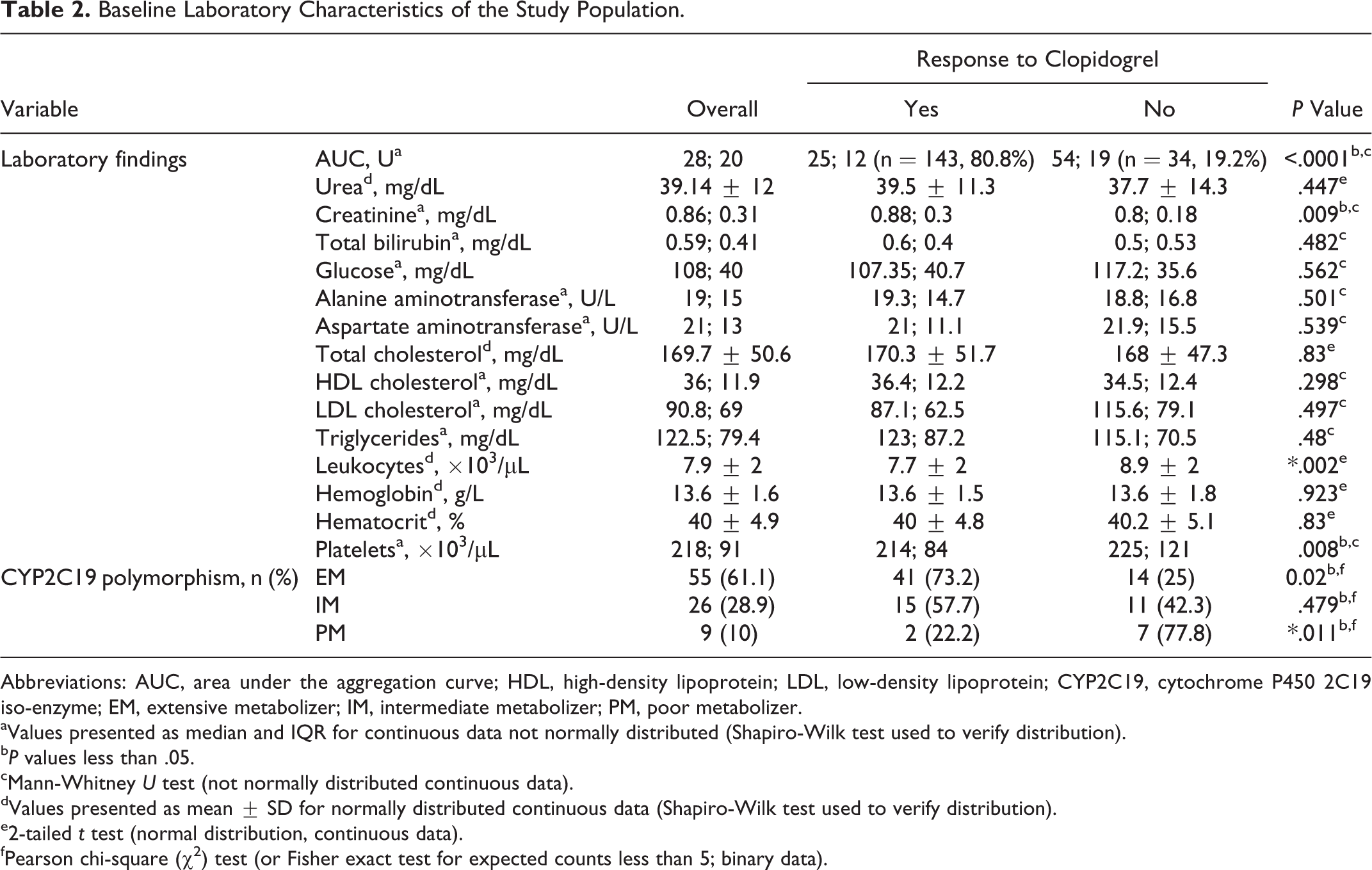
Abbreviations: AUC, area under the aggregation curve; HDL, high-density lipoprotein; LDL, low-density lipoprotein; CYP2C19, cytochrome P450 2C19 iso-enzyme; EM, extensive metabolizer; IM, intermediate metabolizer; PM, poor metabolizer.
aValues presented as median and IQR for continuous data not normally distributed (Shapiro-Wilk test used to verify distribution).
b
cMann-Whitney
dValues presented as mean ± SD for normally distributed continuous data (Shapiro-Wilk test used to verify distribution).
e2-tailed
fPearson chi-square (χ2) test (or Fisher exact test for expected counts less than 5; binary data).
In our data set, 81% (n = 143) of patients were responders with a mean AUC value of 25.15 ± 8.3 U, and 19% (n = 34) were nonresponders to clopidogrel therapy with a mean AUC value of 57.7 ± 12.5 U.
The distribution of CYP2C19 polymorphisms was as follows: 55 (61.1%) patients were CYP2C19 wild-type homozygotes (*1/*1 allele), 25 (27.7%) patients were CYP2C19*2 heterozygotes (*1/*2 allele), 1 (1.1%) patient was CYP2C19*3 heterozygote (*1/*3 allele), and 9 (10%) patients were CYP2C19*2 homozygotes (*2/*2 allele). According to their CYP2C19 genotype, patients were divided into 3 groups (Figure 1): extensive metabolizers (EM; CYP2C19*1/*1)—55 (61.1%) patients; intermediate metabolizers (IM; CYP2C19*1/*2 and CYP2C19*1/*3)—26 (28.9%) patients; and poor metabolizers (PM)CYP2C19*2/*2—9 (10%) patients.
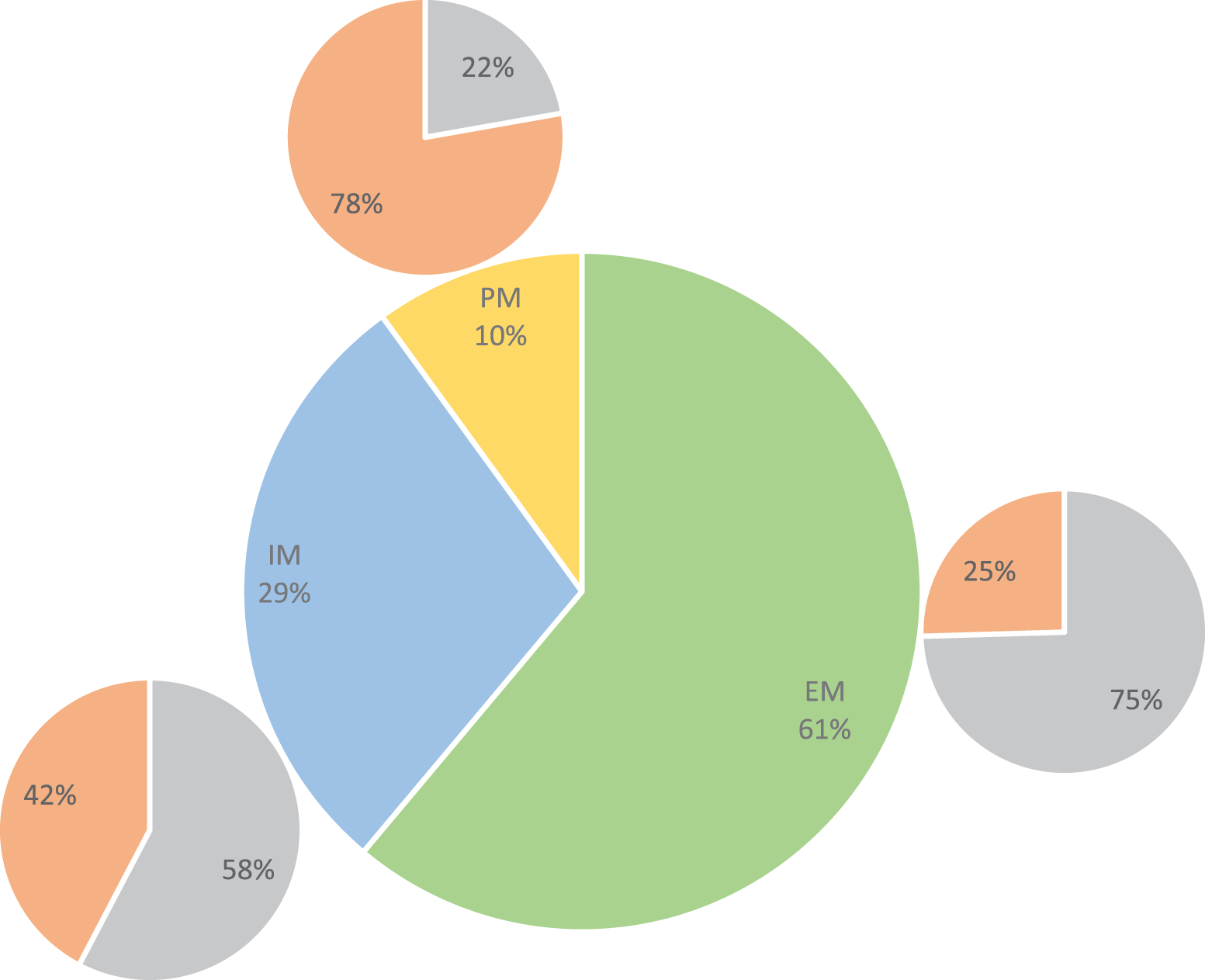
Distribution of patients into poor metabolizers (PM), intermediate metabolizers (IM) and extensive metabolizers (EM) according to the presence of CYP2C19 genetic polymorphisms and the prevalence of nonresponders (nR) to clopidogrel therapy in these groups.
Correlations between AUC values and different clinical and laboratory parameters are presented in Table 3. To identify predictors of clopidogrel resistance, all variables that achieved statistical significance in these correlation analyses were then tested in a univariate regression analysis (Table 4). This latter analysis indicated creatinine, leukocytes, and platelets as significant predictors of AUC values (
Correlation Analysis.
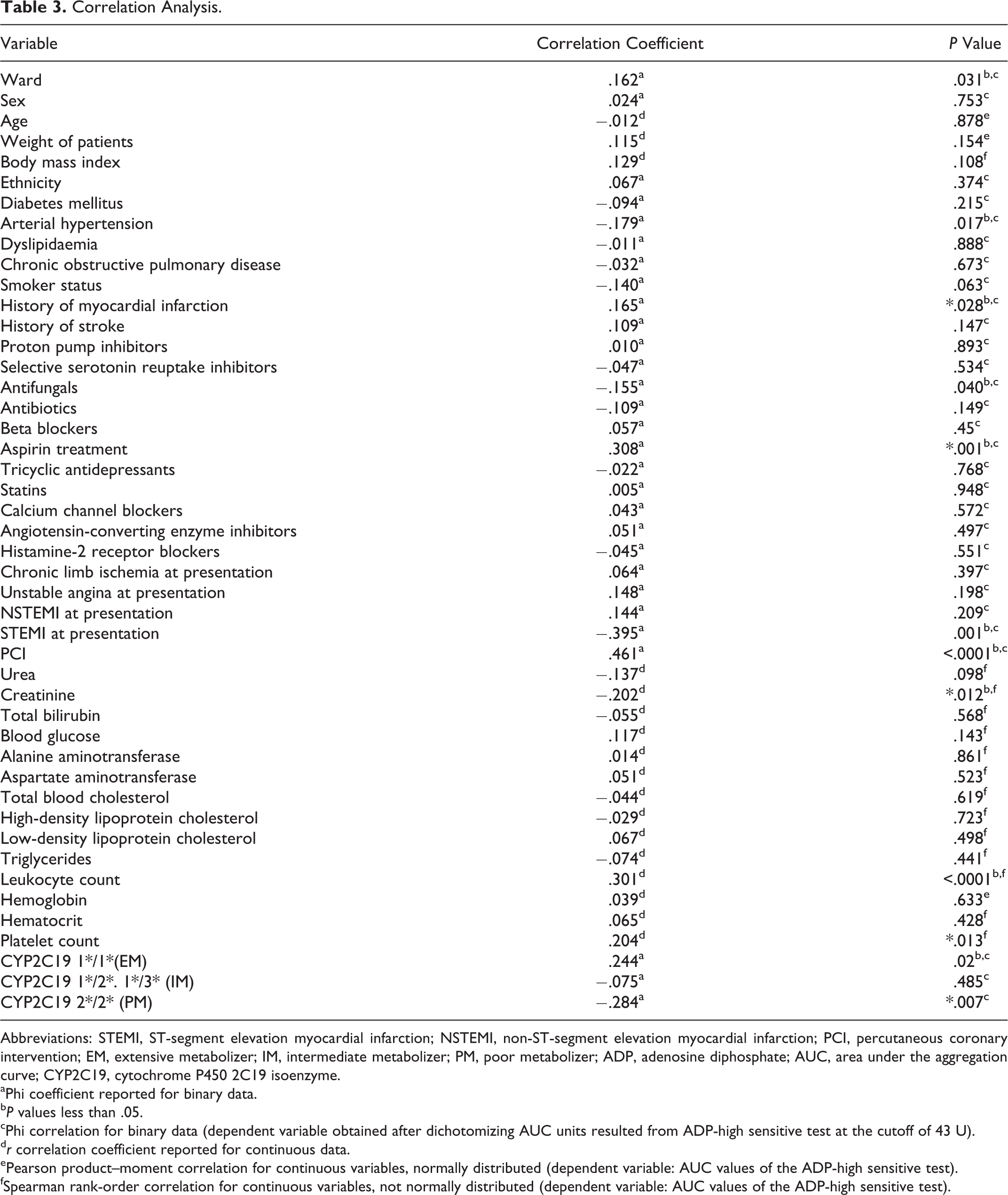
Abbreviations: STEMI, ST-segment elevation myocardial infarction; NSTEMI, non-ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention; EM, extensive metabolizer; IM, intermediate metabolizer; PM, poor metabolizer; ADP, adenosine diphosphate; AUC, area under the aggregation curve; CYP2C19, cytochrome P450 2C19 isoenzyme.
aPhi coefficient reported for binary data.
b
cPhi correlation for binary data (dependent variable obtained after dichotomizing AUC units resulted from ADP-high sensitive test at the cutoff of 43 U).
d
ePearson product–moment correlation for continuous variables, normally distributed (dependent variable: AUC values of the ADP-high sensitive test).
fSpearman rank-order correlation for continuous variables, not normally distributed (dependent variable: AUC values of the ADP-high sensitive test).
Univariate Regression Analysis.a
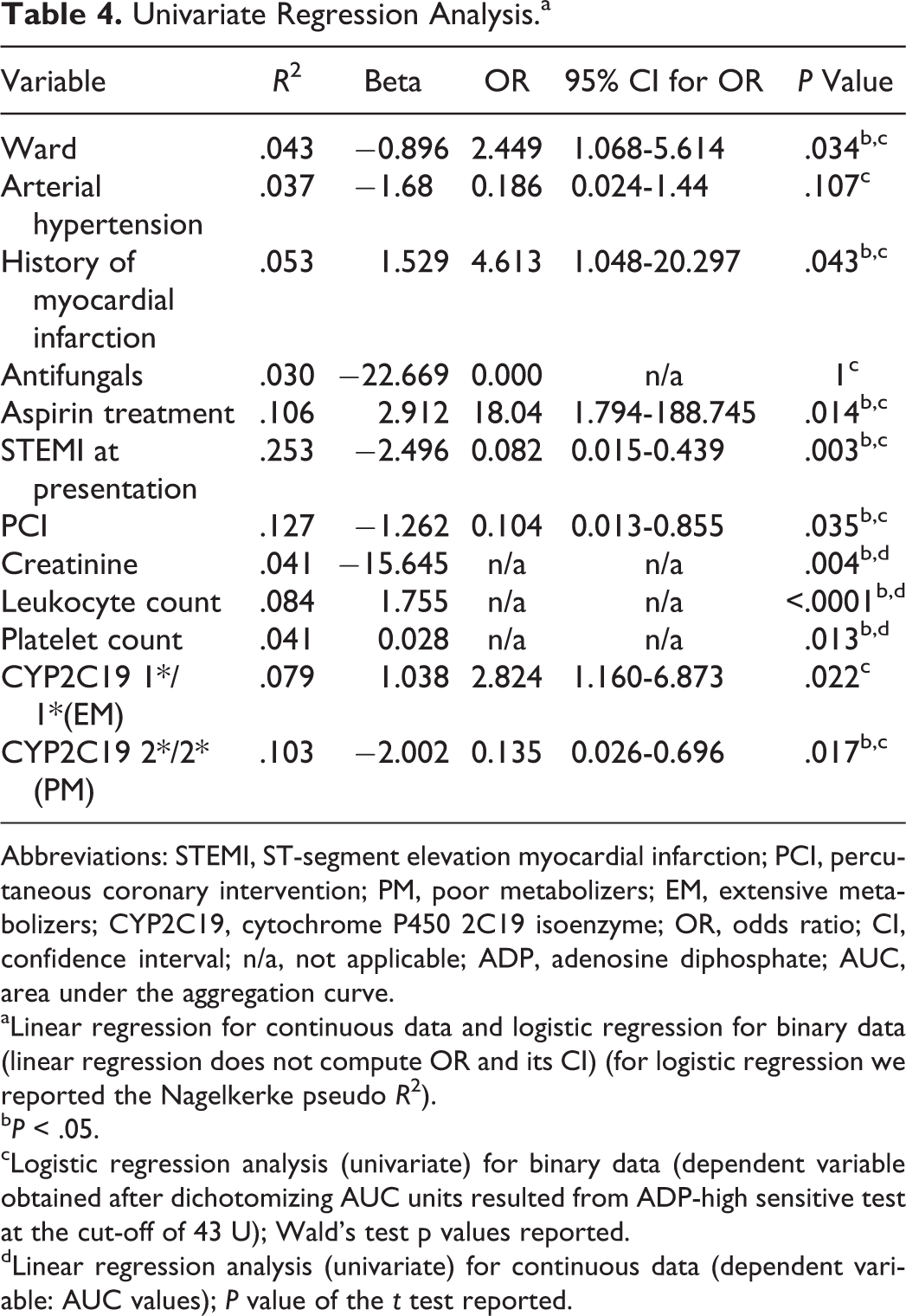
Abbreviations: STEMI, ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention; PM, poor metabolizers; EM, extensive metabolizers; CYP2C19, cytochrome P450 2C19 isoenzyme; OR, odds ratio; CI, confidence interval; n/a, not applicable; ADP, adenosine diphosphate; AUC, area under the aggregation curve.
aLinear regression for continuous data and logistic regression for binary data (linear regression does not compute OR and its CI) (for logistic regression we reported the Nagelkerke pseudo
b
cLogistic regression analysis (univariate) for binary data (dependent variable obtained after dichotomizing AUC units resulted from ADP-high sensitive test at the cut-off of 43 U); Wald’s test p values reported.
dLinear regression analysis (univariate) for continuous data (dependent variable: AUC values);
We also found that patients admitted in the neurology ward were 3.0 times more likely to be responders (and those in the cardiology ward 7.4 times) than nonresponders to clopidogrel therapy. Consequently, patients in the cardiology ward were 1 to almost 6 times more likely to be responders than patients in the neurology ward (odds ratio [OR] 2.45, 95% confidence interval [CI] 1.07-5.61), explaining 4.3% of the variability in response to clopidogrel. A history of myocardial infarction explained 5.3% of the variation in clopidogrel response, with those who have had such an event being 4.6 times more likely to be responders. Patients treated with aspirin were 18.4 times more likely to respond to clopidogrel, explaining 10.6% of variation to treatment, while patients with CYP2C19 2*/2* allele (PMs) were 7.4 times more likely to be nonresponders which accounts for 10.3% of response variation. The presence of the CYP2C19 1*/1* allele increased the chance of responding to clopidogrel treatment 2.8 times, accounting for 7.9% of the variation. Among patients admitted in the cardiology ward with STEMI, the univariate logistic regression predicted that they would be 12.2 times more likely to be nonresponders. Also PCI explained 12.7% of clopidogrel response variability (among patients in the cardiology ward), with those having this intervention being 9.6 times more likely to be nonresponders.
A multivariate logistic regression analysis was then performed to ascertain the independent effects of CYP2C19 polymorphisms (PM and EM), creatinine levels, leukocyte and platelet count, ward where the patients were admitted, history of myocardial infarction, and aspirin treatment on the likelihood that the patients are resistant to clopidogrel therapy.
A test of the full model versus a model with intercept only was statistically significant—χ2(8) = 21.849,
A separate model was created after stratification of patients by ward and included the presence of STEMI at presentation and PCI, in addition to creatinine levels, leukocyte count, platelet count, history of myocardial infarction, and CYP2C19 polymorphisms (PM and EM; for patients in cardiology ward only). A test of the full model versus a model with intercept only was statistically significant—χ2(8) = 20.777,
Discussion
The main findings of this study can be summarized as follows: High on-clopidogrel platelet reactivity was found in 19% of our patients, and 38.9% of the study population had CYP2C19 reduced function genotypes and this influenced response to clopidogrel therapy. Higher leukocyte counts were associated with high platelet reactivity and poor clopidogrel response. History of PCI and STEMI seemed to increase the chances of responding to clopidogrel therapy. Aspirin cotreatment increased the likelihood of responding to clopidogrel therapy. Higher platelet count correlated with high on-clopidogrel platelet reactivity. Arterial hypertension was associated with poorer response to clopidogrel therapy. Statin or angiotensin-converting enzyme inhibitor treatment along with history of diabetes mellitus, hematocrit values, or body mass index didn’t influence clopidogrel response in our data set.
The present study is the first that has investigated the relationship between CYP2C19 loss-of-function polymorphisms together with clinical and demographical variables and clopidogrel resistance in a Romanian population.
Previous studies in different European populations demonstrated a high variability in the prevalence of clopidogrel resistance, from about 5% in a German population to almost 45% in a Spanish population. 13,14 In the present study, clopidogrel resistance was encountered in 19% of patients. The frequency of CYP2C19-reduced function genotypes (IM and PM) shows wide interethnic variation ranging from 20% to 65% in different populations. 15,16 In our population, the frequency of the genotypes mentioned earlier was 38.9%.
It has been demonstrated that there are 3 CYP2C19 genetic variants—*2, *3, and *17—with important influence on the bioavailability of clopidogrel. 17 Poor metabolizers have a very low bioavailability of active clopidogrel, while IMs, which display a reduced bioavailability, are still able to inhibit platelet activity, even if higher doses are usually needed. 18 The majority of the population consists EMs. Rapid (*1/*17) and ultrarapid (*17/*17) metabolizers have also been described, but their prevalence in the general population is much smaller. 18
In our study, the correlation analysis indicated, as expected, that CYP2C19 genetic polymorphisms had a significant impact on clopidogrel resistance. However, in contrast to some previous reports,
7,19
correlation between the status of intermediate metabolizers and clopidogrel resistance did not reach statistical significance (phi = −0.075,
Bhatt et al suggested that the presence of loss-of-function allele (IMs) could be linked to fewer bleeding complications but without a significant increase in the number of ischemic events. 20 Thus, the clinical relevance of the CYP2C19 *1/*2 and *1/*3 mutations remains unclear and deserves further study.
Regarding patients carrying the CYP *2/*2 mutation (poor metabolizers), we found that they are over 7 times more likely to fall into the nonresponder category, demonstrating that the presence of these CYP2C19*2 polymorphisms is associated with higher laboratory clopidogrel resistance. The PM category included the highest percentage of nonresponders (Figure 1). When comparing EMs with PMs, we found that PMs were more likely to be nonresponders than EMs (OR 20.92, 95% CI 9.87-44.60). These findings are in line with most of the existing data, 18,21 suggesting that genetic testing may be helpful in stratifying patients who need alternative newer generation drugs such as ticagrelor or prasugrel or that if no polymorphism is found, the patient may be kept safely on clopidogrel, reducing the risk of bleeding and the financial burden associated with the newer drugs. Previous studies have also suggested that IMs could also benefit from clopidogrel therapy but should have the dose increased. 18
Leukocyte count was also found to be significantly higher in the nonresponder group and was significantly correlated with the status of nonresponder. This variable was able to individually account for 8.4% of clopidogrel response variation. Univariate regression analysis predicted that for every 1000 leukocytes/µL increase, we can expect a rise of 1.75 units in the AUC value. This is in accordance with recent publications. Osmancik et al reported an increased incidence of high on-treatment platelet reactivity for patients with a higher leukocyte count. 22 It seems that the inflammation state, through its mediators (C-reactive protein, P-selectin, platelet soluble CD40 ligand, and interleukin 6), is able to increase platelet reactivity. 22 Moreover, it has been suggested that the controversial aspirin resistance could also be partly related to high leukocyte counts. 23
The same mechanism of acute inflammation commonly associated with PCI procedures
24
and acute STEMI could also explain the increased likelihood of these patients falling into the nonresponder category, with patients who benefited from a PCI being almost 10 times (OR = 9.61, 95% CI 1.17-76.9,
Platelet number was also significantly correlated with AUC values and was able to account for 4.1% of clopidogrel response variation in the univariate regression analysis. From this analysis, we infer that for every 10 000 platelets/µL increase, we should expect a 0.3 rise in AUC units. Our findings are in accordance with the majority of the previously published data. 25,26 To overcome the increased resistance to clopidogrel found in these patients, it has been suggested that doubling the dose would have significant beneficial effect. 27
Interestingly, we found a significant difference in clopidogrel therapy response between the 2 different wards, with patients admitted in cardiology ward being more likely to respond to this therapy. Previous medical history of myocardial infarction was also significantly different between the 2 groups, patients who have had such an ischemic event being 4.6 times more likely to fall into the responder category.
One possible reason for the observed difference could be the higher frequency of concomitant aspirin treatment in patients in the cardiology ward (80.3% vs 45.5%) as well as in all patients having experienced a previous myocardial infarction. This hypothesis is supported by the significant difference that we found regarding the likelihood of a patient also treated with aspirin to fall into the responder category (OR 18.04, 95% CI 1.79-188.75,
Arterial blood pressure differed significantly among the 2 studied groups (responders and nonresponders to clopidogrel therapy). Correlation analysis indicated that hypertension was weakly correlated (phi =−.18,
Baseline characteristics pointed out a slight but significant difference in creatinine levels between the 2 groups. Although the correlation with AUC values was rather weak (
A number of other clinical and laboratory parameters have been related to changes in clopidogrel response in previous studies. While the presence of type 2 diabetes mellitus was associated with reduced effectiveness of antiplatelet therapy, 30,31 lower values of hematocrit have been related to high platelet reactivity. 32
In our data set, none of these factors were significantly different between clopidogrel responders and nonresponders. Similar results have been reported previously. 33,34
Regarding statin concomitant administration, we found no difference between those on or without such treatment in terms of clopidogrel resistance. This is in line with most published data, 35 –37 but detrimental effects of statin treatment (especially those metabolized by CYP3A4) have been suggested. 38,39 Also, a fairly recent article suggested that rosuvastatin (metabolized by CYP2C9 with little involvement of CYP3A4) coadministration results in residual high on-treatment platelet reactivity, but atorvastatin had no such effect. 40 It appears that there is a disagreement between the data obtained from ex vivo studies, suggesting different levels of interaction and the majority of clinical studies which failed to detect any clinically relevant effect. Until a more definite conclusion can be drawn, it is probably safer to assume that statins could decrease clopidogrel response, which could have a significant impact particularly in high-risk patients.
Another class of drugs that are frequently prescribed along with clopidogrel is the angiotensin-converting enzyme (ACE) inhibitors. Again, we couldn’t show any significant interaction between the 2. Concerns arise from the fact that carboxylesterase 1, the enzyme responsible for converting prodrug ACE inhibitors (such as ramipril and perindopril) to their active metabolites, is also involved in the inactivation of clopidogrel by hepatic hydrolysis. It has been suggested that patients receiving cotreatment with clopidogrel and ACE inhibitors may have an increased risk of bleeding. 41 On the other hand, the effect that clopidogrel may have on the ACE inhibitor’s action seems to be of little clinical relevance. 42 Further studies designed to address such matters, using a combination of in vitro and pharmacoepidemiological approaches, may prove useful.
Nardin et al found that high body mass index was related to increased platelet reactivity after PCI and stenting, 43 but this was not the case in our population. Although our multiple regression models were capable of explaining some of the variance in clopidogrel response and showed good correlation between different baseline characteristics and response to clopidogrel therapy, no individual significant predictor was found at the .05 α level (creatinine variable excluded; see explanations earlier). Thus, there seems to be substantial interindividual variability that cannot be fully explained by the variables tested in this study.
The 6-month follow-up showed that 18 (10.2%) patients were readmitted requiring medical assistance: 13 (9.1%) were from responders group, while 5 (14.7%) were from nonresponders to clopidogrel therapy. Regarding all-cause death (one case in responders group), myocardial infarction (one case in responders group), recurrent angina (one case in each group), or stent thrombosis (one case in each group), no definite conclusion can be drawn. Regarding readmissions for ischemic stroke, there seems to be a slightly higher incidence in the nonresponder group (8.8% vs 6.3%), but collected data are not yet sufficient for a robust statistical analysis.
Study Limitations
This study is limited by its cross-sectional design, which only permitted associations to be observed. The lack of clinical outcome data for most of the patients prevented us from creating relevant survival profiles.
The small number of patients may have lowered the study’s statistical power. Plasma levels of clopidogrel and its active metabolite were not detected in the present study; thus, correct treatment administration had to be assumed based on individual interviews.
Conclusion
The prevalence of clopidogrel resistance in our East European population was in line with that reported for Western populations. The highest level of association with the status of clopidogrel response was found to be with CYP2C19 polymorphisms (*2/*2 and *1/*1 alleles), concomitant aspirin treatment, leukocyte and platelet count, history of myocardial infarction, arterial hypertension, and the ward where patients were admitted. Carriers of the variant allele, CYP2C19 *2/*2, appeared to present a higher risk of being nonresponders to clopidogrel therapy (using impedance aggregometry to assess ADP-induced platelet aggregation) than wild-type patients (CYP2C19 *1/*1).
Our findings suggest that genetic testing could be used to stratify patients who need newer antiplatelet therapy (for which the higher risk of bleeding events is justified) or those who may be reverted to clopidogrel therapy, thus limiting the financial burden and the risk of adverse effects. Future research, preferably with larger population sample size, should evaluate additional clinical characteristics and genetic mutations in order to assess and better predict clopidogrel resistance.
Footnotes
Authors’ Note
Alina Mărginean and Claudia Bănescu contributed equally.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sectorial Operational Programme Human Resources Development, financed from the European Social Fund and by the Romanian Government [contract number POSDRU/159/1.5/S/133377]; and the University of Medicine and Pharmacy Tîrgu Mureş [grant number CIGCS 20/11.12.2013].
