Abstract
Introduction:
Careful monitoring of the hypercoagulable state is required during pregnancy. However, coagulation and fibrinolysis markers are not fully utilized because there are no reference values reflective of coagulation and fibrinolysis dynamics during pregnancy, which differ from the nonpregnant state.
Methods:
Changes in antithrombin (AT), fibrinogen (Fbg), prothrombin fragment 1+2 (F1+2), thrombin–antithrombin complex (TAT), soluble fibrin (SF), D-dimer (DD), and protein S (PS) were investigated in healthy pregnant women, and reference ranges in the early, mid, late, and end stages of pregnancy were established.
Results:
The AT was essentially constant throughout pregnancy. The Fbg, F1+2, TAT, and DD increased significantly as pregnancy progressed. In contrast, SF did not show a significant increase throughout the entire pregnancy period. Total PS antigen and total PS activity showed a corresponding decrease from early gestation. When test data in 3 cases in which deep vein thrombosis or intrauterine fetal death occurred during pregnancy were compared to the established reference ranges, all of the cases had multiple markers with values that exceeded the reference ranges.
Conclusion:
Establishing reference ranges for each week could potentially make it possible to evaluate abnormalities of the coagulation and fibrinolysis systems during pregnancy. Of note, SF might be a useful marker that reflects thrombus formation during pregnancy. Larger-scale studies will be required to establish reference ranges for every gestational week.
Introduction
Pregnancy brings about a physiological hypercoagulable state in the mother’s body and increases the risk of deep vein thrombosis (DVT) and pulmonary embolism. In some of the serious complications of pregnancy, such as preeclampsia, placental abruption, intrauterine growth restriction (IUGR), or intrauterine fetal death (IUFD), one of the etiologic factors is thought to be maternal–fetal circulatory failure due to intraplacental thrombosis. 1,2 Typically, the hypercoagulable state during pregnancy is controlled by the body’s innate anticlotting mechanisms, with no thrombosis ever occurring within the body, but in pregnant women with congenital thrombophilia or antiphospholipid antibodies, pregnant women with a history of thrombosis, and so forth, it is important to attentively monitor whether thrombosis is developing. Coagulation and fibrinolysis markers are typically useful for such monitoring. 3,4 However, the fact that the coagulation and fibrinolysis dynamics seen during pregnancy are different from the nonpregnant state means that the routinely used reference ranges for the coagulation and fibrinolysis markers cannot be directly applied to pregnant women.
On the other hand, it is well known that the plasma protein S (PS) level decreases during pregnancy. 5 Physiological decreases of PS increase the risk of thrombosis, especially in women with congenital PS deficiency. Since the prevalence of congenital PS deficiency is higher among Japanese than other racial groups, accurate diagnosis of congenital PS deficiency is important when pregnant women develop thrombotic diseases. However, evaluation of PS levels of pregnant women is difficult because of its physiological decrease.
Various coagulation and fibrinolysis system markers have been used clinically in recent years, but these coagulation and fibrinolysis system markers are currently underutilized because there are no reference ranges for determining “how much of an increase (decrease) in a given marker indicates that the level of the physiologically hypercoagulable state from pregnancy has been exceeded.” In the present study, therefore, multiple blood coagulation and fibrinolysis system markers were investigated in healthy pregnant women by gestational week to establish reference ranges for the early stage, midstage, late stage, and end stage of pregnancy. Test results in cases where DVT or IUFD occurred during pregnancy were also compared to these reference ranges, and the usefulness of establishing reference values by gestational week was confirmed.
Participants and Methods
The present study was conducted with the approval of the Kanazawa University medical ethics review committee (approval number: 513-2). Written informed consent was obtained from all participants.
Changes of Coagulation and Fibrinolysis System Markers and PS in Healthy Pregnant Women
The participants were 32 volunteer women (age: 20-39 years, median: 34 years) who had no history of miscarriage, stillbirth, IUGR, or the like, and who delivered normally after an uncomplicated course. Blood was collected when Participants were able to visit during the early stage (10 ± 2 weeks), midstage (20 ± 2 weeks), late stage (30 ± 2 weeks), and end stage (36 ± 2 weeks) of pregnancy, as well as 1 month after birth, yielding a total of 81 samples (Table 1). The number of times that blood could be collected varied from participant to participant, as did the gestational age, and blood was collected from 1 to 5 times from the same participant. Blood was drawn into a 3.2% sodium citrate tube containing a 9:1 ratio of blood to anticoagulant. Specimens were centrifuged for 10 minutes at 2500g to separate the plasma, which was then stored frozen at −80°C; measurements were taken as soon as possible after thawing.
Gestational Ages of the Participants.
The studied items and reagents were as follows—antithrombin (AT), Testzym S ATIII (Sekisui Medical, Tokyo, Japan); fibrinogen (Fbg), Thrombocheck Fib (Sysmex, Kobe, Japan); prothrombin fragment 1+2 (F1+2), Enzygnost F1+2 (monoclonal; Siemens, Munich, Germany); soluble fibrin (SF), Iatoro SF-II (LSI Medience, Tokyo, Japan); thrombin–antithrombin (TAT) complex, HISCL TAT (Sysmex); and D-dimer (DD), Nanopia D-dimer (Sekisui Medical). Total PS antigen (tPSAg) and total PS activity (tPSAc) were measured by recently commercialized reagents (Shino-Test, Kanagawa, Japan). Detailed explanations of the principles underlying the quantification assays for tPSAg and tPSAc have been provided elsewhere. 6 Total PS-specific activity was calculated by dividing the tPSAg by the tPSAc. The devices used included the Coapresta 2000 (Sekisui Medical) to measure AT, Fbg, SF, and DD; the BEP III (Siemens) to measure F1+2; the HISCLE-2000i (Sysmex) to measure TAT; and the H7170S (Hitachi High-Technologies, Tokyo, Japan) to measure tPSAg and tPSAc.
The Kruskal-Wallis test was used to compare each item between gestational ages, and the Pearson product-moment correlation coefficient was used for correlation analyses between items. SPSS version 19 was used as the statistical software, with the level of significance set to P < .05.
Values of Coagulation and Fibrinolysis-Related Markers in Cases Where DVT or IUFD Occurred During Pregnancy
Measurements of coagulation and fibrinolysis-related markers taken in immediate proximity to onset were extracted from the electronic medical records of 2 cases that developed DVT and 1 case that experienced IUFD during pregnancy follow-up at the Department of Hematology, Kanazawa University Hospital, and these were compared to the measurement values obtained at the closest blood collection point for the healthy pregnant women. The cases are presented below.
Case 1
A 36-year-old primigravida was referred to our hospital because she developed left lower limb DVT in gestational week 12. A search for congenital thrombophilia or antiphospholipid antibodies failed to identify any thrombotic predisposition. The pregnancy was managed with anticoagulation therapy by subcutaneous heparin injection (10 000 units twice daily). Later, though no new thrombus was confirmed, she had IUGR; at gestational week 24, she experienced a disruption of umbilical cord blood flow, and the fetus was expelled by emergency cesarean section. After expulsion, no thrombus or infarction was noted in the placenta. Blood was collected at gestational week 14 when she was undergoing anticoagulant therapy with heparin.
Case 2
A 32-year-old primigravida with congenital AT deficiency developed left lower limb DVT in gestational week 6. Because she had a family history of subarachnoid hemorrhage (2 blood relatives had died of subarachnoid hemorrhage), a decision was made to postpone plans for anticoagulant therapy with heparin at that time, and she was observed while taking oral aspirin; 1 week later, her DVT worsened rapidly. Her pregnancy was then managed with subcutaneous heparin injection (10 000 units twice daily) and intravenous AT formulation (1500 U/week) replenishment; delivery was at 38 weeks, with no new thrombus. Blood was collected at gestational week 7 when she was only taking aspirin and was aware of the left leg pain.
Case 3
A 30-year-old woman had a history of systemic lupus erythematosus, antiphospholipid antibody syndrome, cerebral infarction, and 2 miscarriages (gestational weeks 7 and 20). She had been receiving subcutaneous heparin injections (5000 units twice daily) since the early stages of this pregnancy, but she had persistent hypertension and experienced IUFD at gestational week 15. Findings of slight bleeding were observed on placental pathology, but there was no indication of a thrombus or infarction. Blood was collected at gestational week 14 (immediately prior to IUFD); the patient herself had no subjective symptoms, and lower limb ultrasound had ruled out DVT.
Results
Changes in Coagulation and Fibrinolysis-Related Markers
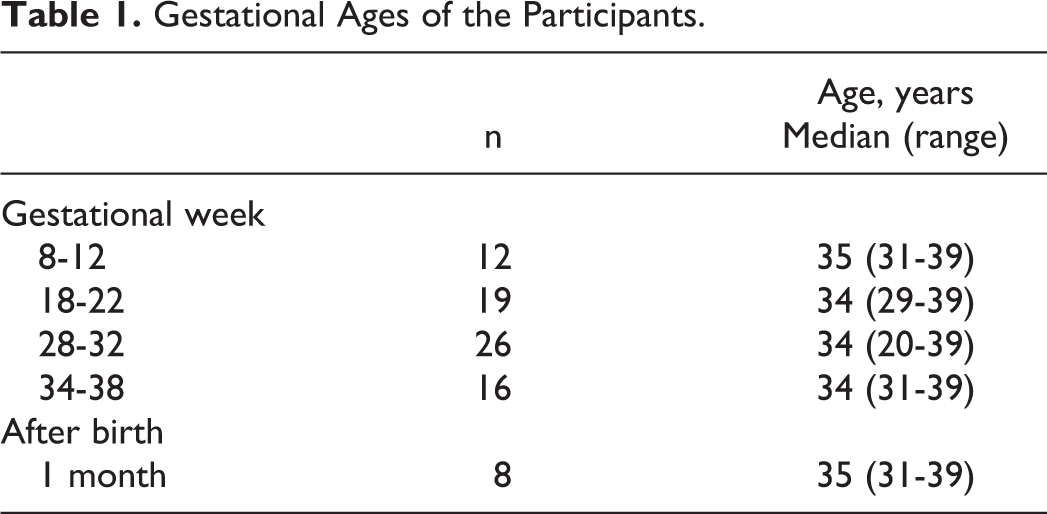
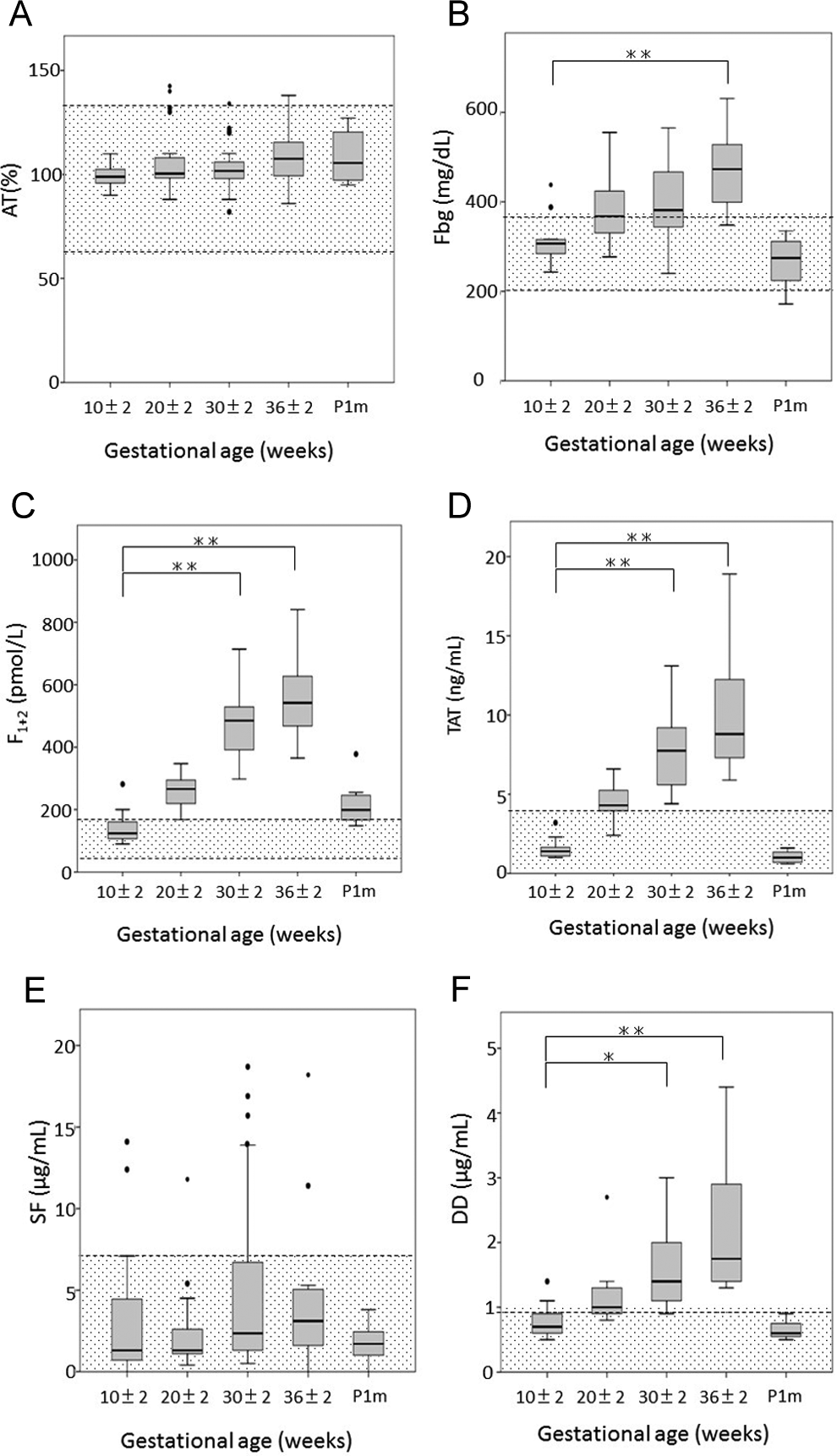
Figure 1 shows the changes in each of the coagulation and fibrinolysis-related markers. Normal reference ranges of our institution (indicated by shadows) were used to evaluate the values of each item. The AT (reference range: 50%-130%) was essentially constant throughout the observation period and remained largely within the reference range (Figure 1A). The Fbg (reference range: 183-381 mg/dL) increased as pregnancy progressed; the median value exceeded the normal range from the midstage to the end stage of pregnancy. The median value in the end stage (473 mg/dL) was about 1.5-fold that in the early stage of pregnancy (307 mg/dL; P < .001; Figure 1B). Of the coagulation activation markers, F1+2 (reference range: 47-171 pmol/L) and TAT (reference range: <4.0 ng/mL) increased markedly from the midstage to the late stage of pregnancy; in the late stage, they were 3.9-fold (485 vs 124 pmol/L; P < .001) and 5.5-fold (7.8 vs 1.4 ng/mL; P < .001), what they were in the early stage, respectively, and in the end stage, they were 4.4-fold (542 vs 124 pmol/L; P < .001) and 6.3-fold (8.8 vs 1.4 ng/mL; P < .001), respectively (Figure 1C and D). However, SF (reference range: <7.0 μg/mL), also a coagulation activation marker, increased less as pregnancy progressed; in the late stage and end stages, it was 1.8-fold (2.4 vs 1.3 μg/mL) and 2.4-fold (3.1 vs 1.3 μg/mL) than in the early stage, respectively; neither was significantly different from the early stage. Nonetheless, SF had a broad distribution of measurement values in each week, and more so than was the case with other items, it was common for specimens to exhibit higher values than other specimens from the same week (Figure 1E). When the SF and DD time series were checked, in 4 participants, SF at 30 ± 2 weeks was higher than the 75th percentile for the same week (6.7 μg/mL), 2 of whom had also been measured at 36 ± 2 weeks, and in both, the high SF values had persisted (cases A and B). One of them also had a high DD value at 36 ± 2 weeks (Table 2). The DD (reference range: <1.0 μg/mL), an activation marker of both coagulation and fibrinolysis, increased significantly to 2-fold (1.4 vs 0.7 μg/mL; P < .005) and 2.6-fold (1.8 vs 0.7 μg/mL; P < .001), what it was in the early stage during the late and end stages of pregnancy, respectively (Figure 1F).
Changes associated with pregnancy of coagulation and fibrinolysis-related markers. AT indicates antithrombin; DD, D-dimer; Fbg, fibrinogen; F1+2, prothrombin fragment 1+2; P1 m, 1 month after delivery; SF, soluble fibrin; TAT, thrombin–antithrombin complex. Boxes indicate median and upper/lower quartiles, whiskers indicate largest/smallest values excluding outliers, and dots indicate outliers. Significant differences (vs 10 ± 2 weeks) are demonstrated with *(P < .005) and **(P < .001). Shadows indicate reference ranges at Kanazawa University Hospital, which are as follows—AT = 50% to 130%; Fbg = 183 to 381 mg/dL; F1+2 = 47 to 171 pmol/L; SF < 7.0 μg/mL; TAT < 4.0 ng/mL; DD < 1.0 μg/mL.
Time Series Changes of SF and DD in Cases With High SF Values During the Late Stage of Pregnancy.
Abbreviations: DD, D-dimer; ND, not done; SF, soluble fibrin.
aValues that are larger than the upper quartile of healthy pregnant women at identical gestational weeks.
Changes in Plasma PS Levels
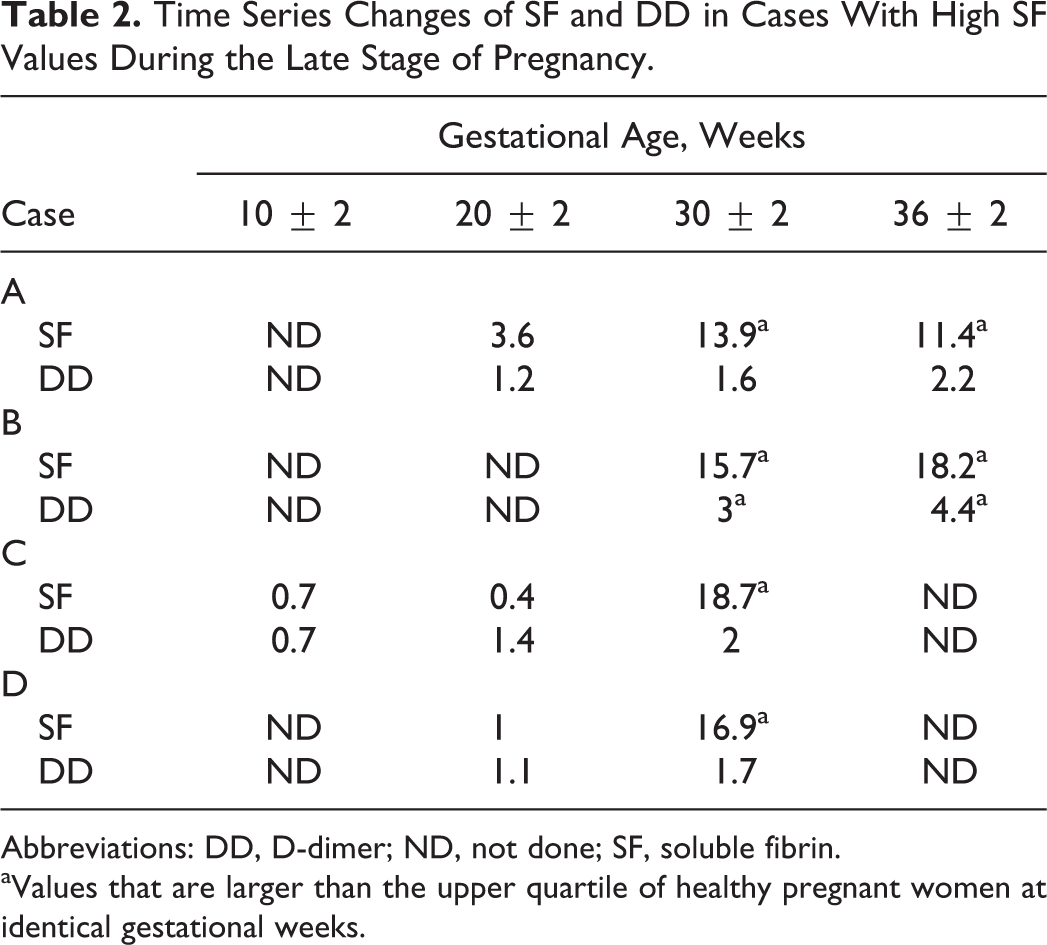
The changes in tPSAg, tPSAc, and PS-specific activity are shown in Figure 2. Normal reference ranges in healthy nonpregnant women reported by Tsuda et al 6 (indicated by shadows) were used to evaluate the values of each item. The level of tPSAg (reference range: 16.0-28.8 μg/mL) decreased from early gestation and remained approximately 70% of that in healthy nonpregnant women (Figure 2A). The decrease in tPSAc (reference range: 15.1-28.7 μg/mL PS-equivalent) was similar to that of tPSAg (Figure 2B); therefore, PS-specific activity (reference range: 0.76-1.20) remained relatively constant at all time points (Figure 2C).
Changes associated with pregnancy of plasma protein S (PS) levels. Shadows indicate reference ranges described in the study by Tsuda et al, 6 which are as follows—Total PS antigen (tPSAg) = 16.0 to 28.8 μg/mL; total PS activity (tPSAc) = 15.1 to 28.7 μg/mL PS-equivalent; PS-specific activity = 0.76-1.20.
Correlations Between Coagulation Activation Markers (TAT, F1+2, and SF) and the Fibrinolysis Activation Marker (DD)
Investigating the correlations among the 3 coagulation activation markers (TAT, F1+2, and SF) and DD showed that TAT and F1+2 had very highly positive correlation coefficients, 0.746 and 0.724, respectively, whereas SF had a correlation coefficient of 0.422, somewhat weaker than TAT and F1+2 (Figure 3).
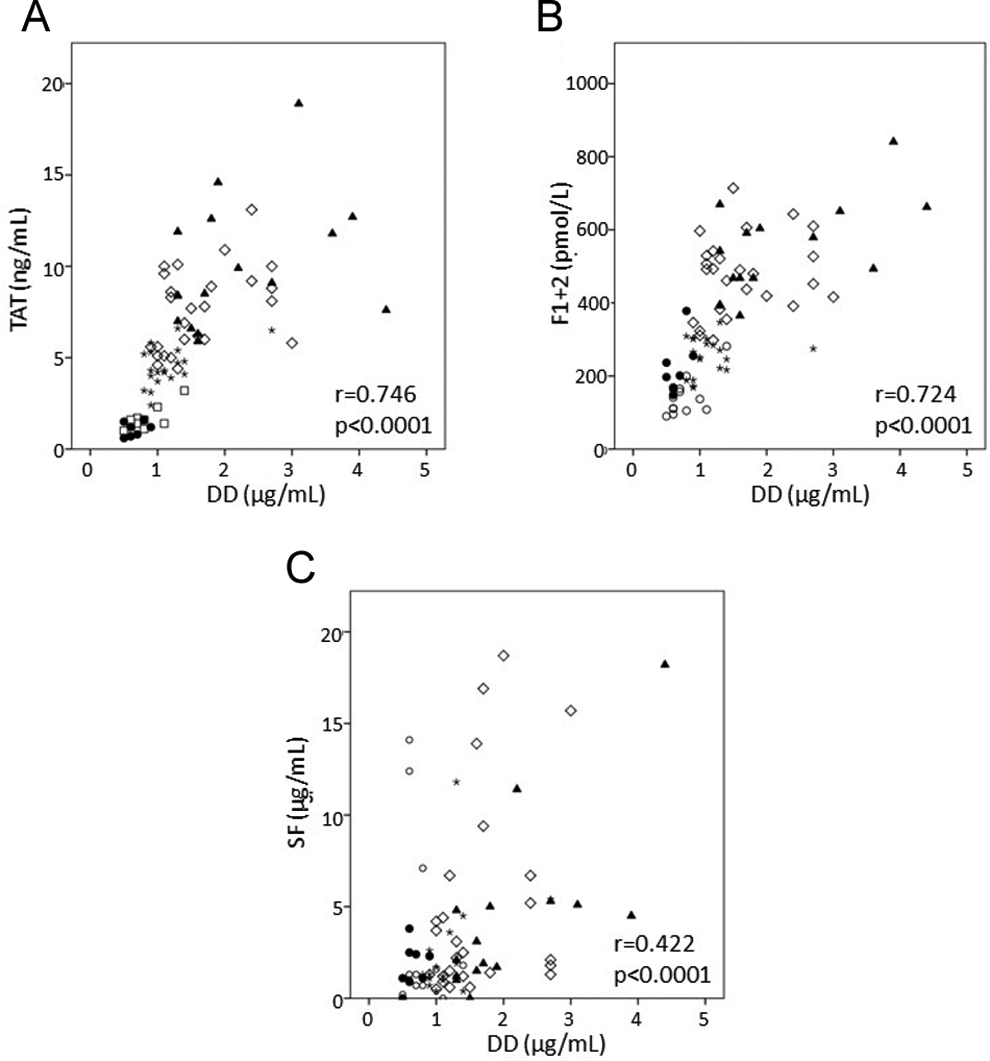
Correlations of thrombin–antithrombin (TAT) complex, fragment 1+2 (F1+2), and soluble fibrin (SF) with D-dimer (DD). ○ : 8 ± 2 weeks; ★ : 20 ± 2 weeks; ⋄ : 30 ± 2 weeks; ▴ : 36 ± 2 weeks of gestation; and • : 1 month after delivery.
Values of Coagulation and Fibrinolysis-Related Markers in Cases Where DVT or IUFD Occurred During Pregnancy
The blood collection points in cases 1, 2, and 3 were gestational weeks 14, 7, and 14, respectively, making it impossible to compare them to values for the exact same weeks in healthy pregnant women; therefore, data for 10 ± 2 weeks, which was the closest time period, were compared. The results showed that the 75th percentile of the healthy pregnant women was exceeded by Fbg, F1+2, and DD in case 1, by Fbg, TAT, and SF in case 2, and by F1+2, TAT, and SF in case 3 (Table 3).
Values of Coagulation and Fibrinolysis-Related Markers in Cases With DVT or IUFD During Pregnancy.

Abbreviations: APS, antiphospholipid antibody syndrome; AT, antithrombin; DD, D-dimer; DVT, deep vein thrombosis; Fbg, fibrinogen; F1+2, fragment 1+2; IUFF, intrauterine fetal death; IUGR, intrauterine growth restriction; ND, not done; SF, soluble fibrin; SLE, systemic lupus erythematosus; TAT, thrombin–antithrombin complex.
aValues that are larger than the upper quartile of healthy pregnant women at 10 ± 2 weeks of gestation.
bValues that are smaller than the lower quartile of healthy pregnant women at 10 ± 2 weeks of gestation.
Discussion
It has long been known that, during pregnancy, the mother’s body is in a hypercoagulable state, and fibrinolysis is inhibited to prepare for bleeding during labor. 7 However, coagulation data from peripheral blood in the present study showed that the coagulation activation markers TAT and F1+2 increased along with DD, a marker of activation of both coagulation and fibrinolysis, indicating a state where coagulation and fibrinolysis are both enhanced. The reason for this may be that the products of the coagulation and fibrinolysis activation constantly occurring in the uteroplacental circulatory system were circulating through the peripheral blood. In the placenta, the fibrinoid layer, acting as the glue that binds the placental tissue and the uterine tissue, is derived from Fbg and is a place where thrombus formation and dissolution are incessantly repeated. 8,9 In their comparison of coagulation and fibrinolysis-related markers in the peripheral venous blood and uterine venous blood of healthy pregnant women who underwent a cesarean section at the end of pregnancy, Komatsu et al showed that coagulation activation factors such as TAT and F1+2 and fibrinolysis activation markers such as plasmin inhibitor complex (PIC) and ibrin degradation products (FDP) were more highly concentrated in the uterine venous blood than in the peripheral venous blood. 8 This implies that the values of thrombosis-related markers in the peripheral blood during pregnancy are more strongly affected by coagulation changes in the uteroplacental circulatory system than the dynamics of thrombus formation in the peripheral blood vessels. For this reason, it has been thought difficult to infer the presence of thrombus formation from coagulation data of peripheral blood during pregnancy. To solve the problem, an attempt was made to establish reference ranges for the coagulation and fibrinolysis system markers in each stage of pregnancy, as well as to find a marker that is not affected by the hypercoagulability of the uteroplacental circulatory system. The established reference ranges of the coagulation and fibrinolysis system markers were applied to 2 cases of DVT and 1 case of IUFD that actually occurred during pregnancy. The results showed that all cases had multiple markers exceeding the distribution ranges of the healthy pregnant women. This demonstrates that, though coagulation data that are clearly different from outside pregnancy are seen during pregnancy, establishing reference ranges for each week could potentially make it possible to evaluate abnormalities of the coagulation and fibrinolysis systems during pregnancy. Case 3, in particular, showed high levels of F1+2, TAT, and SF at a stage where the patient herself had no symptoms a week before succumbing to IUFD, and it may have been extremely useful to have established reference ranges for every gestational week. Changes in some coagulation and fibrinolysis system markers during pregnancy have been studied in the past, 10,11 but this report is the first to apply them to clinical cases that developed a thrombotic pathology.
Of the 3 coagulation activation markers investigated in this study, TAT and F1+2 had changes showing similar trends as pregnancy progressed, and they also showed a high correlation to DD, whereas SF did not increase markedly during pregnancy and was more weakly correlated to DD than TAT and F1+2. This implies that TAT, F1+2, and DD are strongly reflective of the hypercoagulable and hyperfibrinolytic state of the uteroplacental circulatory system, whereas SF is less affected. The TAT, F1+2, and DD are products that result from coagulation and/or fibrinolysis activation; after they are produced, they circulate through the peripheral blood, whereas SF is an intermediate product from after Fbg is cleaved by thrombin until when it is cross-linked by activated factor XIII. A thrombus grows continuously involving Fbg and SF, which are supplied from the blood flow. 12,13 Therefore, though the uteroplacental circulatory system may be in a hypercoagulable state, Fbg remains in the placenta as fibrin via SF, and it may less readily flow out into the maternal circulating blood. Thus, SF may not be affected by the coagulation activation products originating from the uteroplacental circulatory system, and it may potentially be a useful marker making it possible to identify thrombus formation in the mother’s body.
After being cleaved by thrombin, Fbg passes through intermediate stages such as fibrin monomer (FM), fibrin monomer complex (FMC), and SF before becoming its final form, stabilized fibrin. 14,15 When fibrinolytic activation occurs in the presence of FM, FMC, and SF, the FM degradation products (FMDPs) with various fractions derived from fibrin are produced. 16 As such, FM, FMC, and SF are markers of the initial stages of thrombosis or of a prothrombotic state, and FMDPs are markers that also reflect fibrinolysis activation. 16,17 Currently, there are multiple FM, FMC, and SF measurement reagents on the market that use monoclonal antibodies that recognize different epitopes. 17 –19 In their study on changes in FMC during pregnancy, Onishi et al reported that FMC concentrations were unchanged during early or midpregnancy and slightly elevated during late pregnancy. An antibody (F405) that recognizes all of FM, FMC, SF, and the FMDPs was used in their study. 10 In contrast, the antibody used in the present study (IF-43) specifically recognizes SF and does not react with FMDPs; therefore, it is characterized by increasing in the early stages of coagulation activation, but then rapidly disappearing when fibrinolysis is started. 18,20 Thus, theoretically, IF-43 seems to be more specific for thrombus formation than F405. However, we cannot make a definite conclusion because no direct comparison between those 2 items was made. Comparison in a large-scale study is needed.
In this study, there were 2 participants in whom elevated SF was continuously confirmed in the midstage and late stage of pregnancy. Of these, 1 also had elevated DD. A definitive judgment cannot be made because thrombosis was not confirmed by imaging tests, but the participants were inferred either to have experienced thrombosis asymptomatically or to have been in a prothrombotic state. We have often seen falsely high values for SF due to poor blood collection technique in routine operations. 21 If high SF values are found in a pregnant patient, first checking whether blood was properly collected, retesting if necessary, and checking for thrombosis through imaging tests or the like may be effective for early discovery of thrombosis during pregnancy.
The AT is already known to not be affected by pregnancy. As can also be seen from the fact that TAT is measured in ng/mL, whereas AT is measured in mg/dL, AT is present in overly abundant amounts compared to thrombin, and therefore, AT activities are believed not to fall in a hypercoagulable state to the extent where TAT increases by several fold. The AT increases or decreases depending on hepatic synthesis and renal loss; therefore, these factors must also be considered if a decrease in AT is observed during pregnancy. In pregnant women with gestational thrombocytopenia and/or gestational hypertension syndrome, it has been reported that AT activity decreases significantly more than in pregnant women in a control group, and that measuring AT in the late stage of pregnancy is effective for early discovery of acute fatty liver of pregnancy. 22,23 However, considerable amounts of thrombin are believed to be generated if AT has decreased, despite the absence of a suspected decrease in hepatic synthesis or increase in renal loss.
The Fbg is also known to increase, and this depends on a balance between the increase in hepatic synthesis during pregnancy and consumption owing to the hypercoagulable state. Thus, even though Fbg levels may be normal during pregnancy, it may be necessary to consider the possibility of Fbg consumption if an increase corresponding to the gestational age is not observed.
Both tPSAg and tPSAc decreased during pregnancy. It was noted that PS-specific activity was relatively constant in pregnant women throughout their entire pregnancy. Previously, PS activity relative to antigen levels was expressed as the free PS activity/free PS antigen ratio since measurement of tPSAc had not been established. However, the conventional determination of free PS activity by the clotting time method may be unsuitable since clotting time tends to shorten during pregnancy because of elevated levels of clotting factors. Measuring tPSAc reduces assay interference because of the increased sample dilution, and the chromogenic assay-based quantification improves test accuracy. 6 The constant value of PS-specific activity during pregnancy may enable successful screening for type II PS deficiency in which PS-specific activity is reduced to <0.69. 6 This may be beneficial, given that the inherited type II PS deficiency associated with the genetic mutation K196E (PS Tokushima) is relatively common (1.8%) in the Japanese population. Further studies in which PS K196E-positive pregnant women are enrolled are needed to confirm the utility of PS-specific activity values.
To date, any report where multiple coagulation-related markers were studied by gestational week, including this report, has been a small scale. 10,11 Furthermore, the 3 cases of DVT and IUFD that were presented do not provide firm evidence to support the utilities of the reference ranges. If a future, large-scale study were to establish reference ranges for every gestational week, this could potentially enable assessment when a state of physiological coagulation activation at the same number of weeks has been assumed in the prediction of thrombus formation during pregnancy or analysis of underlying pathology of recurrent miscarriage. With patients referred from other facilities or patients visiting the emergency room, it may also be useful for interpreting coagulation data where there is no time series.
In conclusion, the potential utilities of early diagnosis of thrombus formation based on reference ranges in each stage of pregnancy were presented. Establishment of reference ranges would enable more effective use of existing coagulation and fibrinolysis-related markers. Finally, SF may become a useful marker for early detection of thrombosis. Regarding thrombosis during pregnancy and other pregnancy complications, more coagulation data for every gestational week should be collected for different diseases to better understand the respective clinical significance of these multiple coagulation markers.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by grant-in-aid from the ministry of Health, Labor and Welfare of Japan [grant number 26070201]; Japan Society for the Promotion of Science [grant numbers 15K19176, 25462818, 15K08643]; and the Hokkoku Cancer Fund.
