Abstract
We compared thrombophilia and hypofibrinolysis in 6 men with Klinefelter syndrome (KS), without previously known familial thrombophilia, who had sustained deep venous thrombosis (DVT)–pulmonary emboli (PE) or mesenteric artery thrombosis on testosterone replacement therapy (TRT). After the diagnosis of KS, TRT had been started in the 6 men at ages 11, 12, 13, 13, 19, and 48 years. After starting TRT, DVT-PE or mesenteric artery thrombosis was developed in 6 months, 1, 11, 11, 12, and 49 years. Of the 6 men, 4 had high (>150%) factor VIII (177%, 192%, 263%, and 293%), 3 had high (>150%) factor XI (165%, 181%, and 193%), 1 was heterozygous for the factor V Leiden mutation, and 1 was heterozygous for the G20210A prothrombin gene mutation. None of the 6 men had a precipitating event before their DVT-PE. We speculate that the previously known increased rate of DVT-PE and other thrombi in KS reflects an interaction between prothrombotic, long-term TRT with previously undiagnosed familial thrombophilia. Thrombophilia screening in men with KS before starting TRT would identify a cohort at increased risk for subsequent DVT-PE, providing an optimally informed estimate of the risk/benefit ratio of TRT.
Keywords
Introduction
Klinefelter syndrome (KS) is the most common cause of primary testicular failure, resulting in impairment of both spermatogenesis and testosterone production. 1,2 It is a chromosomal disorder characterized by small, firm testes, azoospermia, gynecomastia, varying degrees of eunuchoidism, and testosterone deficiency with elevated gonadotropin plasma levels. 2 Venous thromboembolism (VTE), deep venous thrombosis (DVT), pulmonary embolism (PE), and other thrombotic events appear to be 5 to 20 times more common in men with KS than in normal men at comparable ages. 3 There have been scattered single-case reports of thrombophilia as a putative cause of VTE in KS, with suggestions that thrombi were related to hypofibrinolysis and thrombophilia, 4 –9 but testosterone replacement therapy (TRT) has not, to date, been implicated as a contributor to DVT-PE.
Although it has been reported that thrombosis may occur when TRT is given to patients with familial and acquired thrombophilia, 10 –13 this interaction has not been evaluated in men with KS who develop VTE. Men with KS are unique in that they are usually given TRT beginning in adolescence, 14 hence, they have a long and usually continuous exposure to TRT, in comparison to the more common initiation of TRT in middle-aged to older men. 11 In 6 men who developed VTE on TRT, our specific aim was to assess whether VTE reflected the superposition of TRT on previously undiagnosed familial or acquired thrombophilia.
Materials and Methods
Patients
Six men with well-documented KS 1,2 were referred to us after having sustained DVT-PE while taking TRT. They were evaluated as a consecutive case series in the order of their referral.
Controls
One hundred ten healthy normal participants (48 men and 62 women), previously evaluated by us, 13 without history of VTE, who had never taken TRT, were used as a normal control group. One hundred ten patients (53 men and 57 women), previously evaluated by us, 11 who had sustained VTE without exogenous testosterone and without external precipitating events were utilized as thrombotic controls (VTE controls).
Laboratory Assessment of Thrombophilia and Hypofibrinolysis
Polymerase chain reaction assays
Polymerase chain reaction measures of the factor V Leiden, prothrombin G20210A, Methylene tetrahydrofolate reductase (MTHFR) C677 T and A1298C, and plasminogen activator inhibitor (PAI) mutations (SERPINE1) gene 11,15 –17 were carried out using established methods. 18 –20
Plasmatic measures of thrombophilia and hypofibrinolysis
Plasmatic measures of activated protein C resistance, 21,22 antigenic antithrombin III, protein C, total protein S, free protein S, homocysteine, factors VIII and XI, 23,24 the lupus anticoagulant, and anticardiolipin antibodies were carried out using established methods. 18 –20 Serologic studies of PAI activity were not done. Coagulation measures were done in patients with KS after TRT had been stopped.
Statistical Methods
All statistical analyses were done using SAS version 9.4. Power and sample size calculations were carried out based on previous comparisons of cases sustaining VTE on TRT versus those sustaining VTE without TRT. 11 With factor V Leiden heterozygosity in 24% of cases versus 12% of VTE controls, 11 ≥160 participants would be needed in each group to detect group differences at a significance level of .05 with power 80%. For the lupus anticoagulant, present in 14% of cases versus 4% of VTE controls, 11 n ≥ 128 would be needed.
To compare cases sustaining VTE on TRT versus normal controls, for factor V Leiden heterozygosity, 31% versus 2%, 13 there would need to be ≥25 participants in each group to detect differences at a significance level of .05 with power 80%. For factor VIII, 25% in cases versus 7% in normal controls, ≥64 participants would be needed for each group; for factor XI 19% versus 3%, ≥59 participants would be needed.
Results
Thrombophilia in Patients With KS, Normal Controls, and Controls With VTE Not Taking TRT
Six patients were assessed in the order of their referral. None had elevated hemoglobin or hematocrit, antecedent trauma or surgery, cancer, extended immobilization, or recent airplane travel longer than 8 hours, all risk factors for VTE. 10 All 6 were documented to have the KS genotype (47,XXY). 25
As displayed in Table 1, the diagnosis of KS was made in early adolescence in 5 men, whereas the sixth (case #2) did not have chromosomal documentation 1,2 of KS until age 48. The TRT therapy had been continuous from adolescence in 5 men and from age 48 in case 2 who had first been treated with TRT and developed mesenteric artery thrombosis 1 year later (Table 1). Subsequently, he was treated with Clomid, 75 mg/d, and had a DVT-PE after 3 years on Clomid (Table 1).
Current Age, Age at Diagnosis, TRT Dose, Time Interval Between Starting TRT and Development of DVT-PE, and Serum Total and Free Testosterone and Estradiol Levels on TRT.
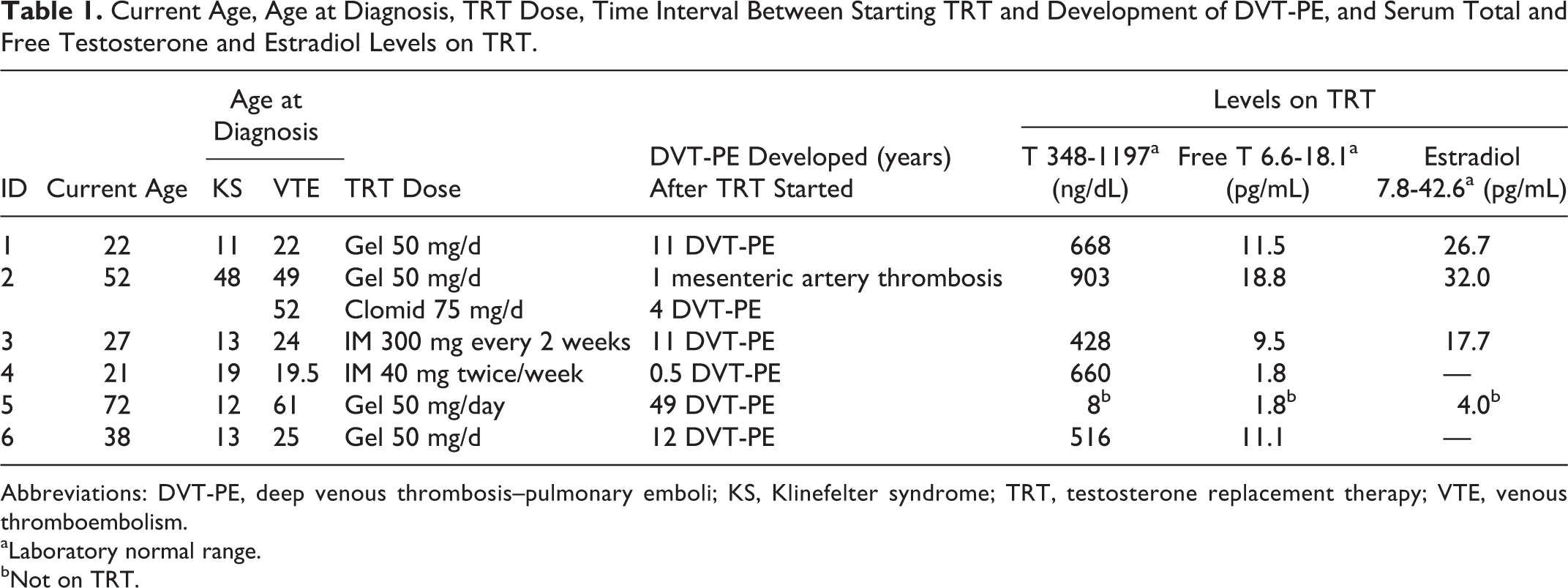
Abbreviations: DVT-PE, deep venous thrombosis–pulmonary emboli; KS, Klinefelter syndrome; TRT, testosterone replacement therapy; VTE, venous thromboembolism.
aLaboratory normal range.
bNot on TRT.
The KS was first diagnosed at ages 11, 12, 13, 13, 19, and 48 years (Table 1). The DVT-PE or mesenteric artery thrombosis was developed in 0.5, 1, 11, 11, 12, and 49 years after TRT had been started at ages 19.5, 22, 24, 25, 49, and 61 years (Table 1).
In the 5 men with measurements of serum testosterone, free testosterone, and estradiol (E2) while on TRT, the total serum testosterone levels were in the normal range, as were E2 levels (Table 1). In case 5, with measures taken off TRT, total and free T were very low, as was E2 (Table 1).
Thrombophilia
As displayed in Table 2, all 6 patients with KS had 1 or more thrombophilia found, with 1 having factor V Leiden heterozygosity, 1 G20210A prothrombin gene heterozygosity, 4 high factor VIII, and 3 high factor 11. The percentage of cases with thrombophilia in the 6 men with KS did not differ (P > .1) from the VTE controls, which may reflect the fact that the numbers of men with KS did not provide the power to discern a significant difference from VTE controls (Table 3).
Coagulation Measures in 6 Patients With Klinefelter Syndrome.
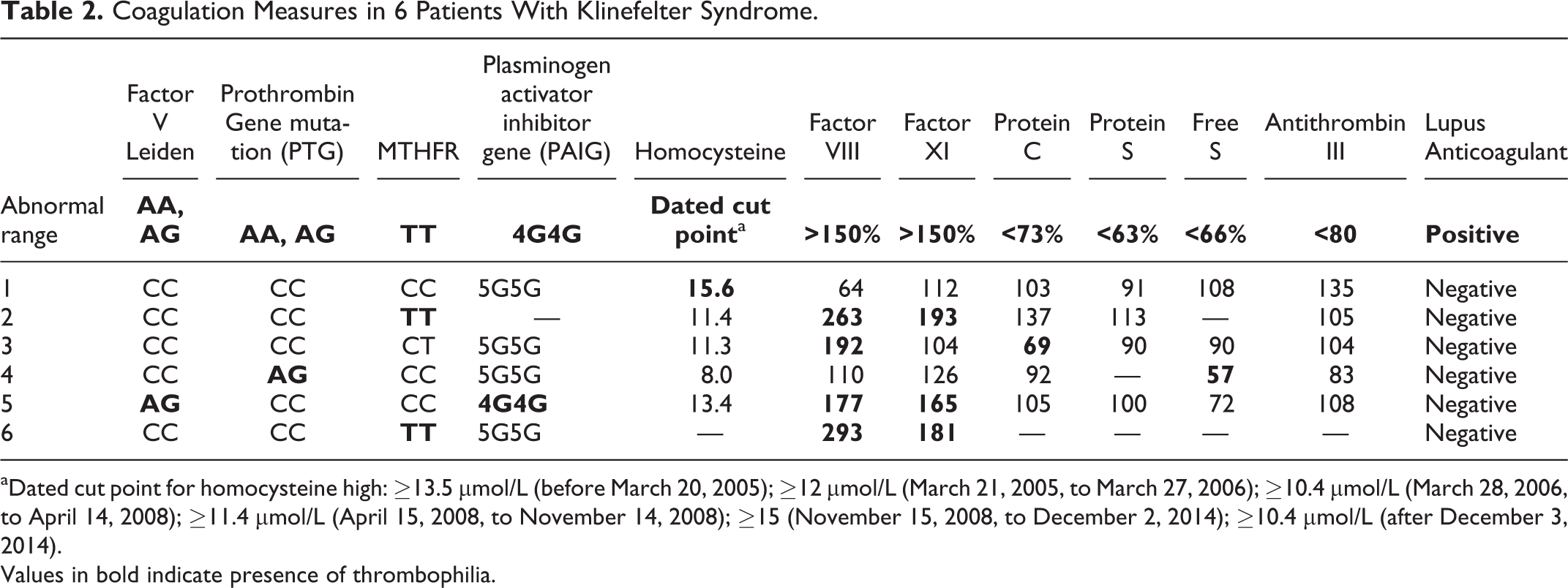
aDated cut point for homocysteine high: ≥13.5 μmol/L (before March 20, 2005); ≥12 μmol/L (March 21, 2005, to March 27, 2006); ≥10.4 μmol/L (March 28, 2006, to April 14, 2008); ≥11.4 μmol/L (April 15, 2008, to November 14, 2008); ≥15 (November 15, 2008, to December 2, 2014); ≥10.4 μmol/L (after December 3, 2014).
Values in bold indicate presence of thrombophilia.
Coagulation Disorders in 6 Patients With KS, Compared With 110 VTE Controls, and With 110 Healthy Normal Controls.
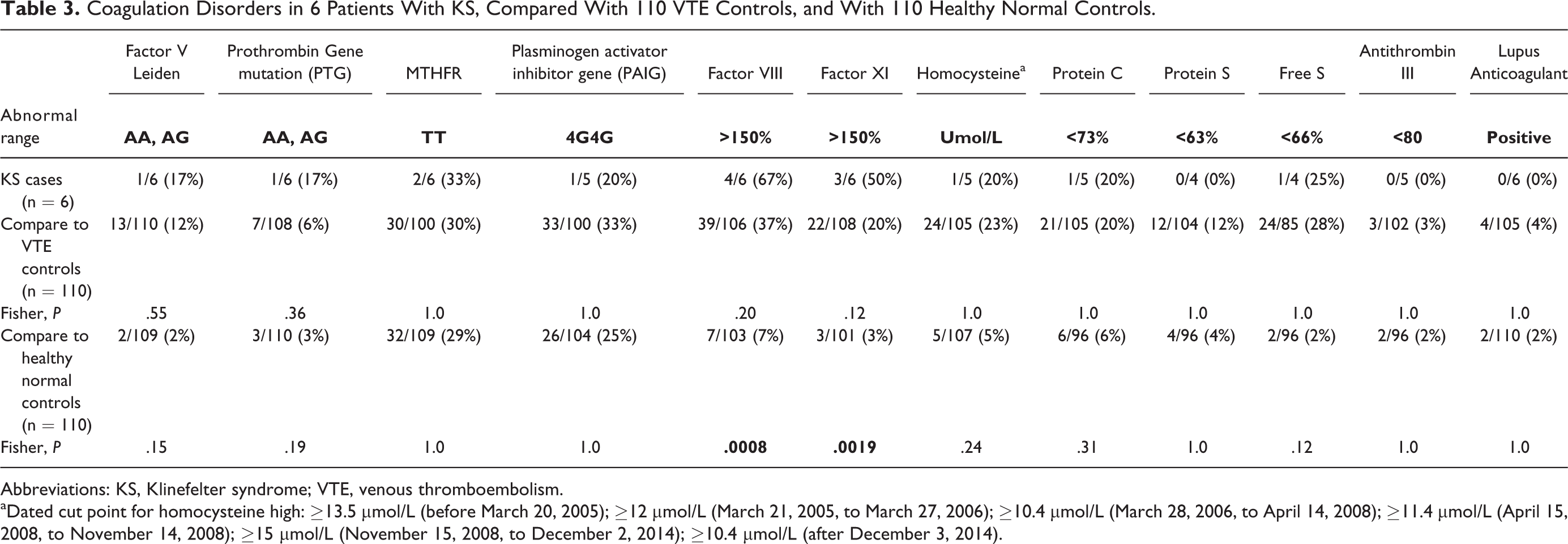
Abbreviations: KS, Klinefelter syndrome; VTE, venous thromboembolism.
aDated cut point for homocysteine high: ≥13.5 μmol/L (before March 20, 2005); ≥12 μmol/L (March 21, 2005, to March 27, 2006); ≥10.4 μmol/L (March 28, 2006, to April 14, 2008); ≥11.4 μmol/L (April 15, 2008, to November 14, 2008); ≥15 μmol/L (November 15, 2008, to December 2, 2014); ≥10.4 μmol/L (after December 3, 2014).
Cases were differed from normal controls by virtue of more frequent high factors VIII (67% vs 7%; P = .0008) and XI (50% vs 3%; P = .0019; Table 3). As displayed in Figure 1, either factor V Leiden or prothrombin gene heterozygosity was present in 2 (33%) of the 6 KS cases versus 17% of the 110 VTE controls versus 5% of the normal controls (P = .04). Either factor VIII or XI or both were high in 66% of the KS cases versus 51% of the VTE controls versus 8% of the normal controls (P = .001; Figure 1).
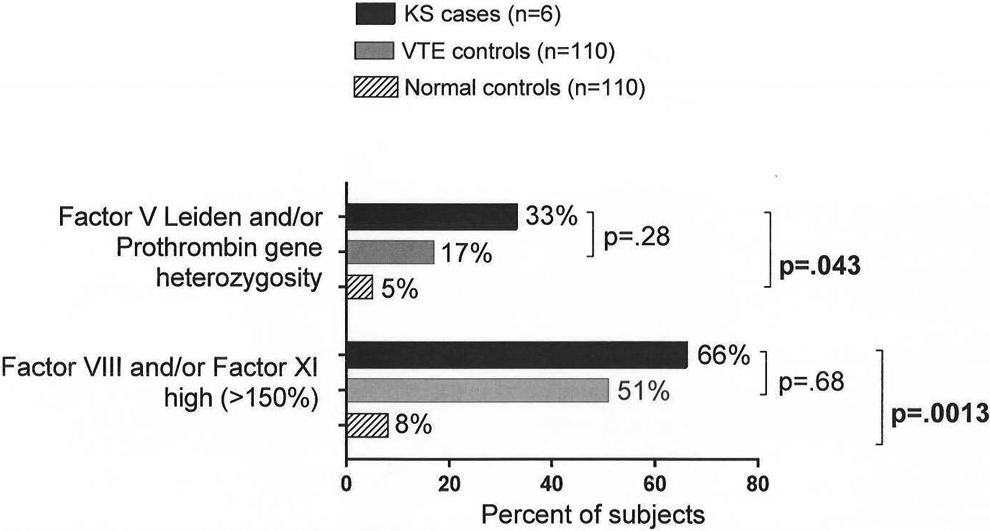
Factor V Leiden and/or G20210A PTG heterozygosity, elevated factor VIII and/or factor XI, in 6 patients with Klinefelter syndrome in 110 controls with VTE without concurrent TRT and in 110 healthy normal controls without TRT. PTG indicates prothrombin gene; TRT, testosterone replacement therapy; VTE, venous thromboembolism.
Discussion
Campbell and Price reported that patients with KS have a ∼20 times greater risk of thromboembolic disease when compared to normal males. 3 Patients with KS also have an increased rate of mesenteric vein thrombosis (present in 1 of our 6 men) and arterial ischemia. 8,26 –30 Additionally, patients with KS have a high prevalence of recurrent venous ulcers, 31 which are thought to be due to postthrombotic syndrome. These findings have been reinforced by large registry-based studies. 26,27,30,32
Despite the high rate of thromboembolic disease in KS, no one has systematically explored the etiology of this phenomenon, and much of our current knowledge is limited and based on single cases or studies with small sample sizes. 4 –9 Currently, there are 8 hypotheses describing the pathoetiology of thromboembolism in KS: (1) vascular abnormalities, 33 –36 (2) hypofibrinolysis with increased activity of PAI-1, 37 –41 (3) high factor VIII, 42,43 (4) platelet hyperaggregability, 44,45 (5) protein C and S deficiency, 9,46 –52 (6) high homocysteine, 8 (7) antithrombin III deficiency, 53 and (8) Factor V Leiden heterozygosity. 4,7,54 We speculate that the development of VTE in KS is influenced by the duration and consistency of TRT beginning, for the most part, during adolescence, in contrast to less consistent and much shorter duration of therapy in adult hypogonadal men, typically begun in mid to late adulthood. 11 –13,55 In parallel with the studies of VTE when exogenous estrogens were given to women heterozygous for the factor V Leiden mutation, 56,57 increasing the likelihood of VTE significantly, we believe that the thrombophilias identified in the current study significantly increase the risk of VTE when TRT is given.
In patients with KS, 58,59 testosterone production is inhibited by virtue of primary testicular failure; this leads to the hypogonadism and infertility, although some patients may have low normal or even normal levels of testosterone. 60,61 Due to the androgen deficiency usually diagnosed (as in the current study) in adolescence, 14 it is common practice to treat patients with KS with exogenous testosterone early in life to ensure proper male development. Nonrandomized trials have documented positive effects of TRT in 77% of 30 men followed for 3.6 years. 62 In patients with KS, TRT has been associated with improvements in endurance, strength, sleep, concentration, learning ability, 62 sexual function, libido, decreased fatigue, 63 increased lean body mass, reduced fat mass, and improved bone mineral density. 64
The benefits of TRT, particularly in men with KS, are tempered by common development of DVT-PE in patients with previously unknown familial and acquired thrombophilia. 11,13,65,66 In our recent study of 67 patients who developed DVT-PE after starting TRT, there were 47 DVT-PE events within 6 months of starting exogenous testosterone. This population was found to have increased rates of factor V Leiden (16 of 67 [24%] vs 13 of 111 [12%] VTE controls not taking TRT; P = .038) and lupus anticoagulant (9 of 64 [14%] vs 4 of 106 [4%] VTE controls; P = .019). 11 Additionally, testosterone therapy was found to be associated with recurrent thromboembolism despite appropriate anticoagulation, 11,13 underscoring its role in the development of DVT-PE. Endogenous testosterone, including in high concentrations, is not associated with thrombotic events 67 ; the association is observed with exogenous testosterone. 11
In addition to increased risk of DVT-PE, there seems to be an increased risk of mesenteric artery thrombosis in men with KS receiving TRT. One case report by Ozbek et al documented an acute case of mesenteric arterial thrombosis in a patient with KS during TRT. 8 Our cohort had 1 patient with mesenteric artery thrombosis 1 year after initiation of TRT, and DVT-PE 3 years later on treatment with Clomid. There are also 2 cases in the literature reporting mesenteric venous thrombosis in patients with KS. 28,29 Taken together, these cases indicate that TRT, given for KS and superimposed with thrombophilia, can cause acute thrombosis in the arterial system, in addition to the venous, low-velocity, low-flow system.
Although the number of participants with KS in the current case series was larger than most published studies of thrombosis and thrombophilia in KS, the small number of men with KS limited our ability to discern significant differences in thrombophilia when compared to VTE or normal controls. Given the small number of cases with KS in the current study, we were not powered to discern a significant difference between cases and VTE controls not taking TRT or to optimally discern significant differences between cases and normal controls. In the future, an optimal study design, based on our power and sample size calculations, would include ≥160 men with KS who had VTE on TRT compared to ≥160 with VTE not on TRT.
The documentation of familial or acquired thrombophilia in a patient with KS who has sustained DVT-PE on TRT raises difficult therapeutic risk/benefit issues. The benefits of TRT in KS are well documented, 62 –64 but DVT-PE is both a morbid and mortal threat. Screening for thrombophilia before starting TRT (for life) in adolescents with KS should identify patients at high risk for DVT-PE. On the other hand, in men with KS with negative thrombophilia screening, TRT should, speculatively, convey little risk for DVT-PE. Prudent use of TRT would allow men with KS the hope of developing normally without putting them at risk of the potentially fatal consequence of mixing TRT and thrombophilia.
Given the link between TRT, thrombophilia, and VTE, we hypothesize that the duration and consistency of TRT, given for most of a lifetime beginning in adolescence, 14 could account for the increased rate of thromboembolism in those men with KS who have familial or acquired thrombophilia. We speculate that the reported 20-fold increase in VTE in KS 3 does not reflect coinheritance of thrombophilia but is associated with very long-term TRT beginning in adolescence. The TRT increases the likelihood of thrombotic events in patients with previously unknown thrombophilia. 11,13 The rate of thromboembolic disease is greater in patients with KS than in a normal population. 3 Since patients with KS are chronically on TRT for decades, we speculate that TRT, in the same fashion as in adult men, 11 when superimposed on preexisting familial or acquired thrombophilia, increases the risk of DVT-PE in KS. In our cohort, 5 of the 6 patients with KS had an inherited thrombophilia, with subsequent increased risk of DVT-PE 11 when given TRT.
Our article is limited by the small number of KS cases with VTE during TRT therapy, and we recognize that documentation of a major gene thrombophilia like the factor V Leiden or prothrombin gene mutation in KS raises the difficult question of balancing the risk of future VTE against the known benefits of TRT in KS.
Conclusion
Prior to starting TRT in KS, we suggest thrombophilia screening to prospectively identify patients with KS at increased risk of DVT-PE. Thrombophilia screening in men with KS before starting TRT would identify a cohort at increased risk for subsequent DVT-PE, providing an optimally informed estimate of the risk/benefit ratio of TRT. Many more patients with KS who have sustained VTE on TRT need to be studied to advance our understanding beyond the preliminary findings of the current article.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
