Abstract
Several small studies have reported an elevated risk of venous thrombosis (VT) with thrombophilia and oral contraceptive (OCP) use. We aimed to summarize the risk of VT among women with thrombophilia and OCP use and to assess the interaction between the 2 factors. We selected 15 studies that assessed the prevalence of OCP use and thrombophilia among reproductive-aged women. Odds ratios (ORs) were calculated for each study and pooled using the random effects model. We found an increased risk of VT among women with OCP use (pooled OR 3.0, 95% confidence interval [CI] 1.9-4.5) and with thrombophilia (pooled OR 4.5, CI 3.4-5.9), respectively. Heterogeneity was significant (I 2 >80%). Women with both thrombophilia and OCP use had a 14-fold risk of VT compared to healthy OCP nonusers (pooled OR 14.25, CI 6.2-32.8). Oral contraceptive use and thrombophilia similarly increase VT risk. Our study confirms an interaction between OCP use and thrombophilia.
Introduction
Oral contraceptives (OCPs) were first introduced in the late 1950s and are used by 8% of reproductive aged women worldwide. 1 They are associated with an increased risk of deep vein thrombosis (DVT) and pulmonary embolism (PE). Compared with age-matched nonusers, the risk of DVT and PE among OCP users is increased by 3- to 5-fold,2–4 and is more pronounced with third-generation compared with second-generation OCPs.5,6 Furthermore, thrombophilic disorders, and in particular the gene mutation of Factor V Leiden (FVL), have been reported to amplify the effect of OCP use on the risk of DVT and PE by at least 30-fold, though estimates vary widely.7,8 A similar synergistic effect for OCP and thrombophilia has been reported for atypical site venous thrombosis (VT), such as cerebral vein thrombosis (CVT). 9 However, there remains uncertainty on how to counsel women with thrombophilia contemplating OCP use. This is in part due to the wide variation in published estimates of association between thrombophilia and OCP use that are largely derived from small observational studies with heterogeneous study populations. As a result, the aim of our meta-analysis was to provide an updated summary estimate of the association between VT including atypical site thrombosis, OCP use, and thrombophilia and to evaluate the combined effect of OCP and thrombophilia.
Methods
Study Identification
We reviewed all language articles containing original data on thrombophilia, OCP use, and venous thromboses of typical and atypical sites. We searched the databases Medline, Embase, CINAHL, and Cochrane Review for articles published between 1950 and 2008 using the following Mesh terms for thrombophilia: thrombophilia, activated protein C resistance, FVL gene mutation, prothrombin (PT) mutation G20210A, antithrombin III deficiency, protein C deficiency, protein S deficiency, elevated factor VIII levels, antiphospholipid antibodies, lupus anticoagulant, anticardiolipin antibodies, and hyperhomocysteinemia; the following Mesh terms for oral contraceptives: oral contraceptive, ethinyl estradiol, desogestrel, levonorgestrel, combined contraceptives, synthetic contraceptives; and the following Mesh terms for venous thromboembolism: pulmonary embolism, thromboembolism, thrombophlebitis. The terms were further exploded to include thrombosis in all areas, including DVT, PE, CVT, and upper-extremity DVT (UEDVT). We limited our search to “female” and “adolescent 13 to 18 years” or “adult 19 to 44 years.” We excluded studies published prior to 1950 because the association between OCP and thrombosis was not known prior to 1950. We supplemented our search by manually reviewing the reference list of retrieved articles.
Study Selection
Two independent reviewers (N.D. and V.T.) selected the studies for review. Studies were included if all 6 of the following criteria were met: (1) case−control or cohort design, (2) objectively documented VT, including DVT, PE, CVT, and UEDVT, (3) control groups for comparison, (4) thrombophilia measured by validated assay, (5) population included reproductive aged women (13-44), and (6) OCP use recorded in all women. We excluded case reports and series, and when multiple articles for a single study were published, we used the earliest publication.5,10–12 The κ statistic was used to assess agreement between reviewers for study selection. 13 κ values of at least .6 represent substantial agreement, while values .8 or greater represent near perfect agreement.
Study Validity Assessment
Two reviewers (N.D. and V.T.) independently assessed validity of the selected studies. Because qualitative validity scales are lacking in observational studies, we applied the Meta-analysis of Observational Studies in Epidemiology (MOOSE) criteria 14 put forth by the consensus statement of meta-analysis reporting of observational studies. If at least 2 of the following criteria were met, the study was considered “high quality”: (1) sufficient follow-up, (2) potential confounders addressed, (3) blinded assessment, (4) the appropriate analysis not conducted in a study population subgroup, and (5) similar selection criteria for cases and controls.
Data Extraction
Two reviewers (N.D. and V.T.) independently extracted data from studies meeting inclusion criteria. Any disagreement was resolved by discussion. We collected data on the number of reproductive aged women using OCP (including definition of “current use” and type of OCP if specified) with and without thrombosis, and the number of women with thrombophilia (FVL mutation, PT mutation, hyperhomocysteinemia, elevated factor VIII levels, protein C deficiency, protein S deficiency, antithrombin III deficiency, antiphospholipid antibodies, lupus anticoagulant, or anticardiolipin antibodies) with and without thrombosis. Baseline characteristics included number of participants, mean or median age of participants, family or personal history of thrombosis, and smoking status. Attempts were made to contact authors if relevant data were not available in the published report.
Statistical Analyses
The outcomes included DVT, UEDVT, PE, and CVT. Each study is summarized according to its unadjusted odds ratio (OR) and stratified by OCP use and thrombophilia type. Where appropriate, meta-analyses were carried out to calculate the pooled ORs with 95% confidence intervals (CIs) using a random-effects model with DerSimonian-Laird weighting. 15 Heterogeneity was assessed using the I squared (I 2) statistic with corresponding 95% CIs and meta-regression to determine whether specific clinical or study characteristics were significantly associated with variation of the log OR. Interpretation of I 2 values was based on suggested guidelines in the Cochrane Handbook. 16 Meta-regression models were constructed with the log OR as the outcome and with appropriate covariates such as thrombophilia type and study type (case−control vs cohort design). Funnel plots were completed to assess possible publication bias. Analyses were conducted using MIX software, version 1.7.17–19
Ethics
The study was exempt from Institutional Review Board approval since our study was a meta-analysis that included nonidentifying patient data.
Results
Our search strategy identified 415 studies (Figure 1). There were 39 studies identified more than once. We excluded 323 studies based on title and abstract screening, and 53 studies were retrieved for more detailed evaluation. This subset included studies retrieved by manual review of references. The k value for interobserver agreement for study selection was 0.77, representing substantial agreement. Of the 53 studies, 38 were subsequently excluded for the following reasons: 32 studies did not meet inclusion criteria4,20–50; 4 studies contained duplicate data5,8,13,51; 1 study presented data as proportions rather than absolute counts 52 ; and 1 study lacked data on thrombophilia and OCP use for some study participants. 53 Therefore, 15 studies were included in our meta-analysis.7,9,11,54–65
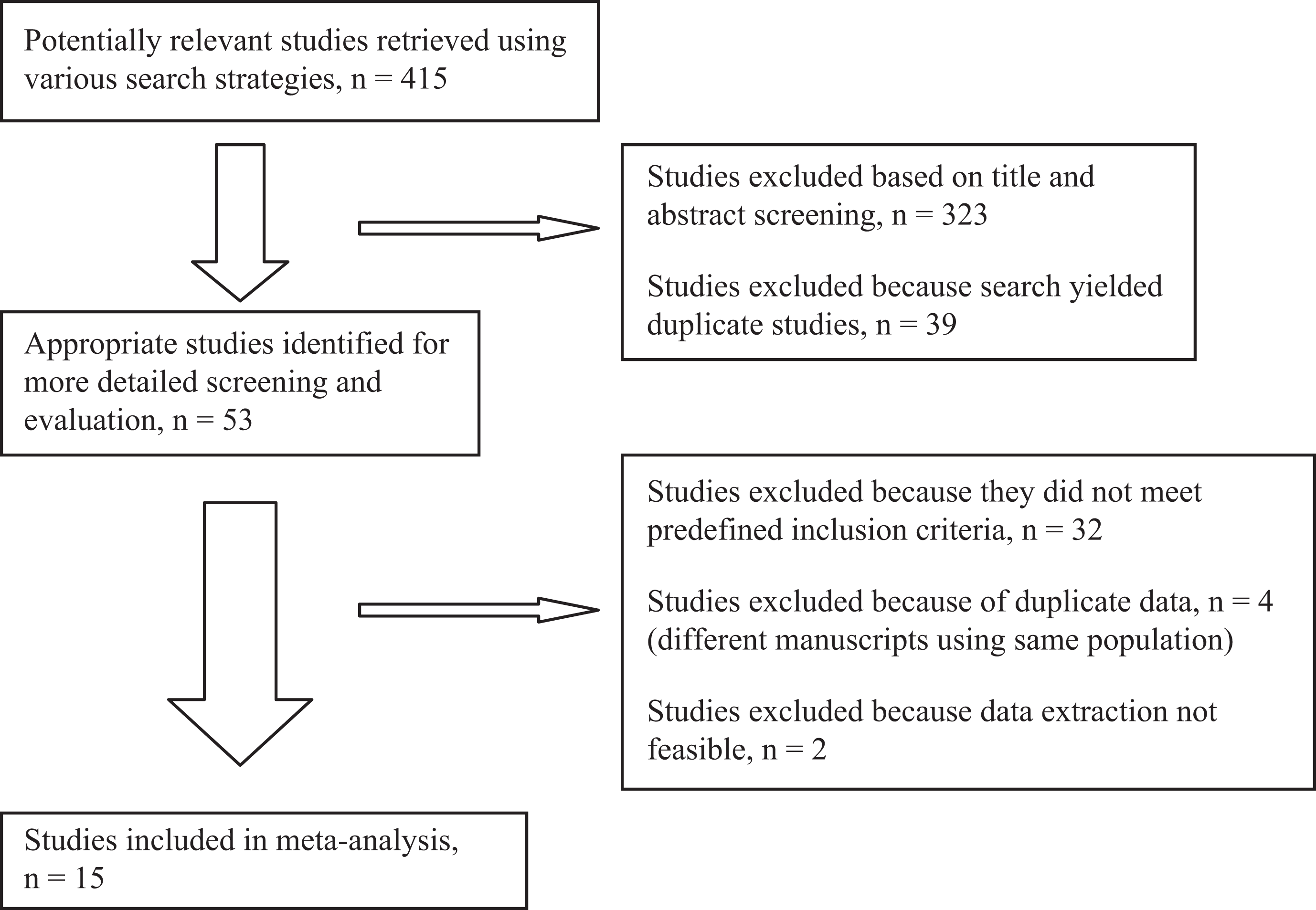
Study selection process flow diagram.
Study Characteristics
Table 1 summarizes the baseline study characteristics. Two were cohort studies57,58 and 13 were case−control studies. One study evaluated the interaction between elevated factor VIII levels and OCP use on the risk of DVT 56 ; 4 studies evaluated the effect of FVL and OCP use on the risk of DVT and PE7,11,60,62; 2 studies evaluated the effect of various thrombophiliass and OCP use on the risk of UEDVT55,59; 6 studies evaluated the effect of thrombophilia and OCP use on the risk of CVT9,54,61,63–65; 1 study analyzed families with inherited thrombophilia to determine thrombosis risk with thrombophilia compounded by OCP use 57 ; and 1 study followed women at high risk of thrombsis to determine thrombosis risk with thrombophilia compounded by OCP use. 58
Baseline Characteristics of Included Studies, N = 15 Studies
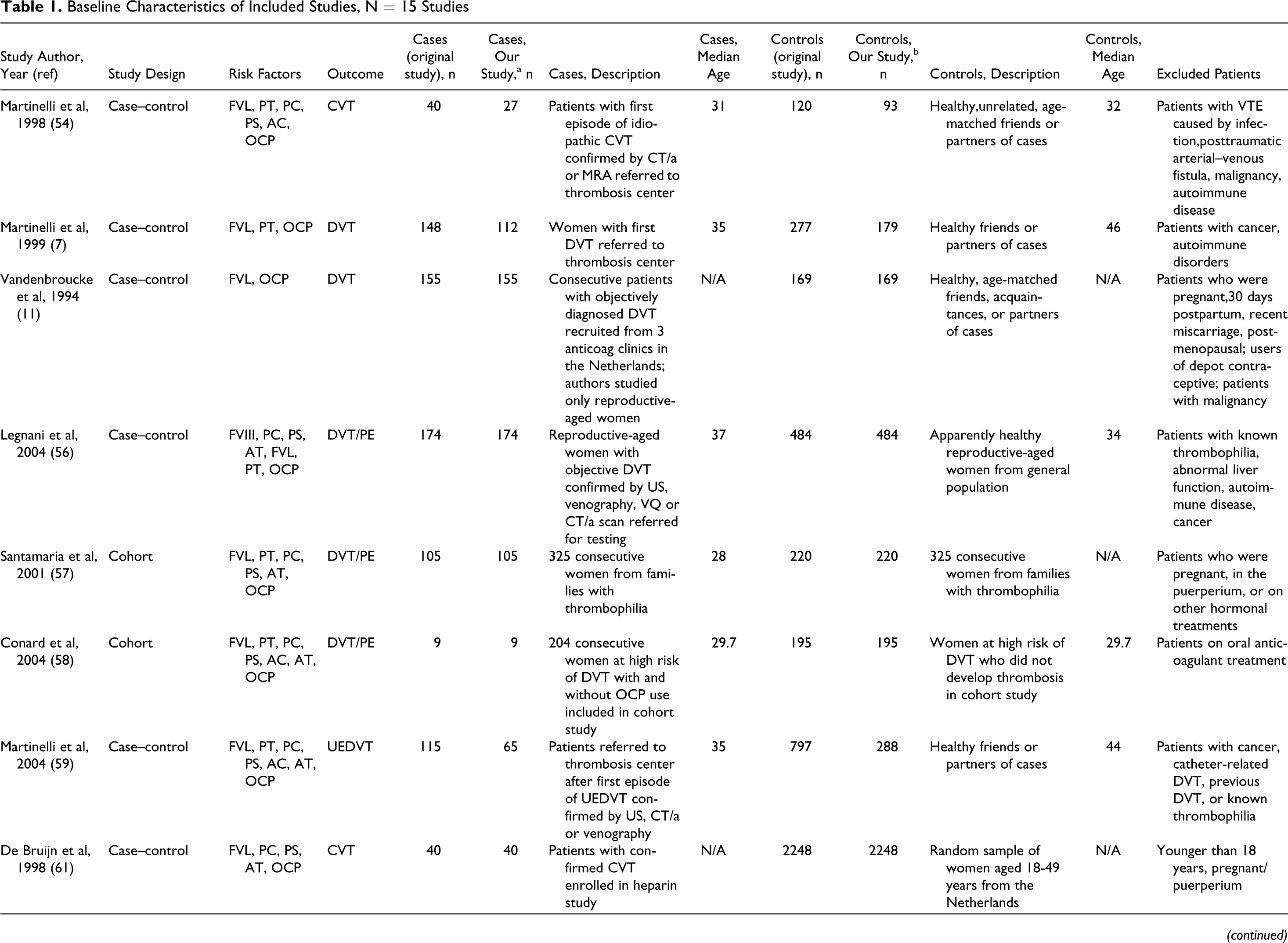
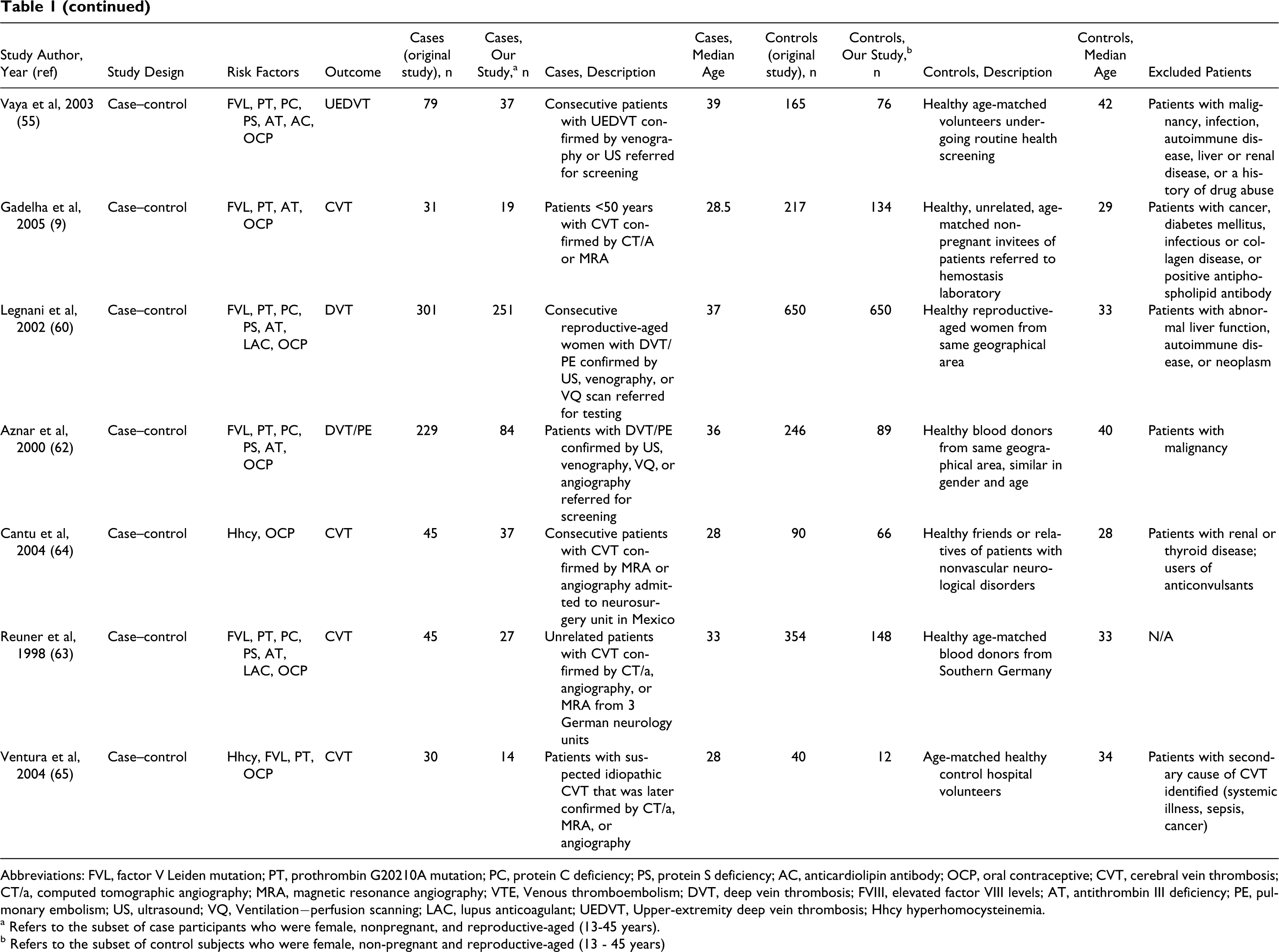
Abbreviations: FVL, factor V Leiden mutation; PT, prothrombin G20210A mutation; PC, protein C deficiency; PS, protein S deficiency; AC, anticardiolipin antibody; OCP, oral contraceptive; CVT, cerebral vein thrombosis; CT/a, computed tomographic angiography; MRA, magnetic resonance angiography; VTE, Venous thromboembolism; DVT, deep vein thrombosis; FVIII, elevated factor VIII levels; AT, antithrombin III deficiency; PE, pulmonary embolism; US, ultrasound; VQ, Ventilation−perfusion scanning; LAC, lupus anticoagulant; UEDVT, Upper-extremity deep vein thrombosis; Hhcy hyperhomocysteinemia.
a Refers to the subset of case participants who were female, nonpregnant, and reproductive-aged (13-45 years).
b Refers to the subset of control subjects who were female, non-pregnant and reproductive-aged (13 - 45 years)
Study Quality
The quality of included studies was high. In 7 studies, control participants were matched to cases on age.9,11,54,55,57,58,63
Effect of OCP Use on VT Risk
The effect of OCP on the risk of thrombosis was assessed by pooling data from the 15 studies (Figure 2). A funnel plot of OR versus standard error exhibited symmetry about the mean, suggesting lack of publication bias. The pooled analysis contained 1016 women with VT (including DVT, PE, UEDVT, CVT) and 5057 controls. Of the 15 studies, 12 reported OR estimates greater than 1, suggesting an increased risk of thrombosis with OCP use.7,9,11,54–57,59–63 We observed a 3-fold increased risk of thrombosis with OCP use compared to non-OCP use (pooled OR 3.01 95% CI 2.00-4.54; Figure 2). Heterogeneity between the studies was significant (I 2 = 82%, 95% CI 71%-89%) and was maintained after exclusion of studies with cohort design, which we hypothesized may have contributed to the observed heterogeneity. Moreover, a meta-regression analysis confirmed that study type did not significantly explain the variability in the log OR for thrombosis and OCP use (P = .11). As a result, differences among study populations in characteristics such as smoking and family history of VT likely explain the heterogeneity observed. Finally, we performed a subgroup analysis limited to studies reporting only on DVT or PE9,54,61,63–65 and observed a pooled OR of 2.28 (95% CI 1.55-3.35) and heterogeneity between studies remained important (I 2 = 79%, 95% CI 60%-89%).
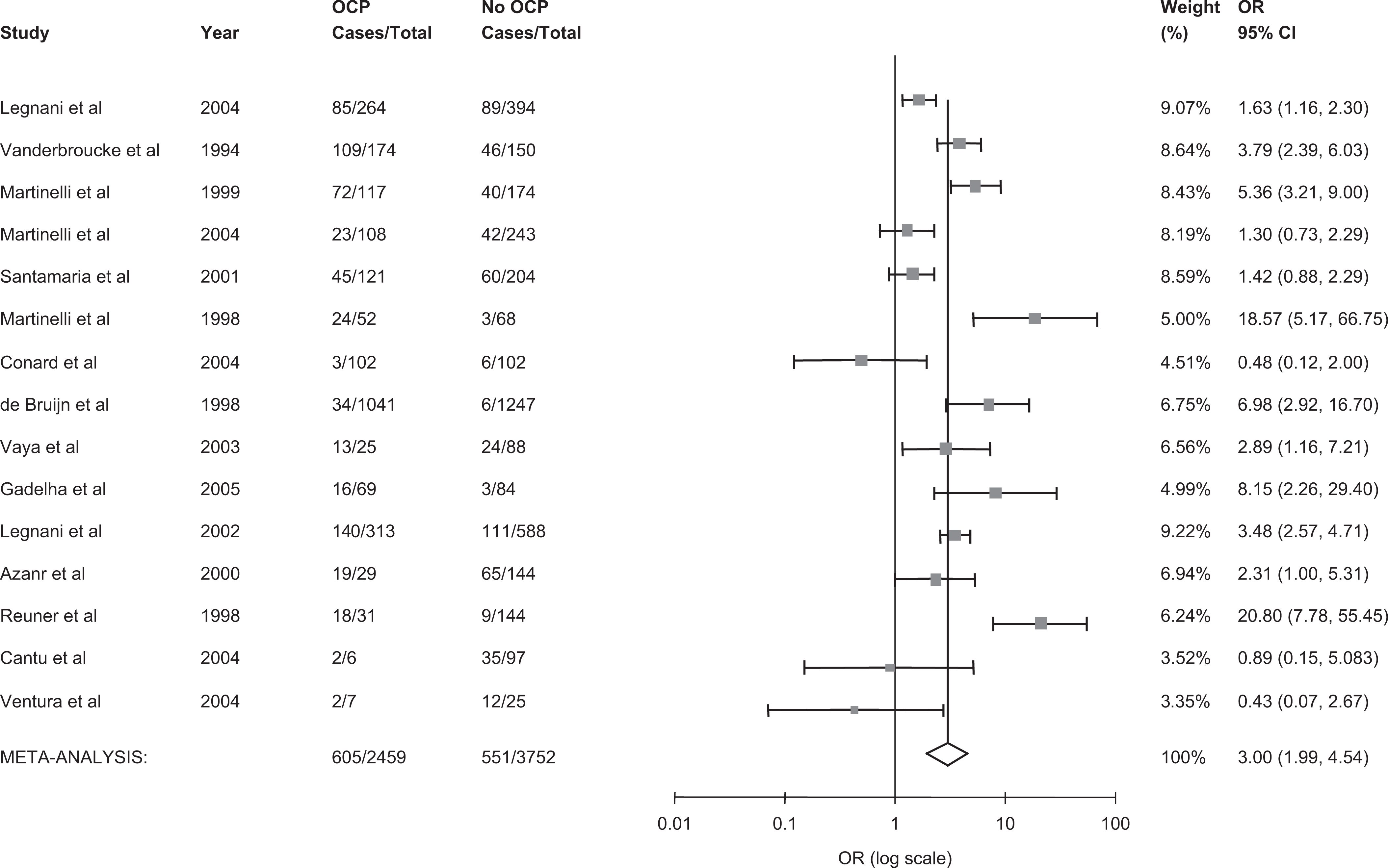
The pooled odds ratio for venous thrombosis in oral contraceptive users vs nonusers. OCP indicates oral contraceptive use; CI, confidence interval; OR, odds ratio; cases, number with venous thrombosis.
Effect of Thrombophilia on VT Risk
We included 7 studies in our evaluation of the effect of thrombophilia on the risk of thrombosis (Figure 3).7,11,54,56–59 Eight studies were excluded from this analysis because of missing thrombophilia information for reproductive-aged women in 6 studies,9,55,60,63–65 for the control group in 1 study, 61 and for non-OCP users in 1 study. 62 In Martinelli’s study that measured both FVL and PT20210A gene mutations in the same patients, 54 we report only their analysis of FVL to avoid duplicating the results. As a result, we pooled together 647 women with VT and 1628 controls from 7 studies. A funnel plot revealed symmetry around the mean, suggesting lack of publication bias. Overall, compared to women without thrombosis, thrombophilia carriers had 4-fold the odds of having a thrombotic event as compared to noncarriers (OR= 4.52, 95% CI 3.46-5.89). Heterogeneity between the studies was not important, (I 2 = 0%, 95% CI 0%-71%), and a meta-regression analysis revealed that study design (case−control vs cohort design) and thrombophilia type (FVL mutation vs other thrombopilia) were not statistically significant in explaining variability in the OR (P = .21 and P = .80, respectively). In all, 4 studies7,11,54,57 for a total of 399 case and 661 control participants reported positive associations between FVL and thrombosis. A pooled odds ratio of 4.77 (95% CI 3.05-7.47) was observed with no between-study heterogeneity (0%, 95% CI 0%-85%; Figure 4). Overall, 3 studies7,54,57 for a total of 244 case and 492 control participants reported positive associations between PT G20210A mutation and thrombosis, though there was high variability among the reported estimates. A pooled OR of 3.28 (95% CI 0.81-13.13) was observed and there was significant heterogeneity (83%, 95% CI 48-95%; Figure 5.
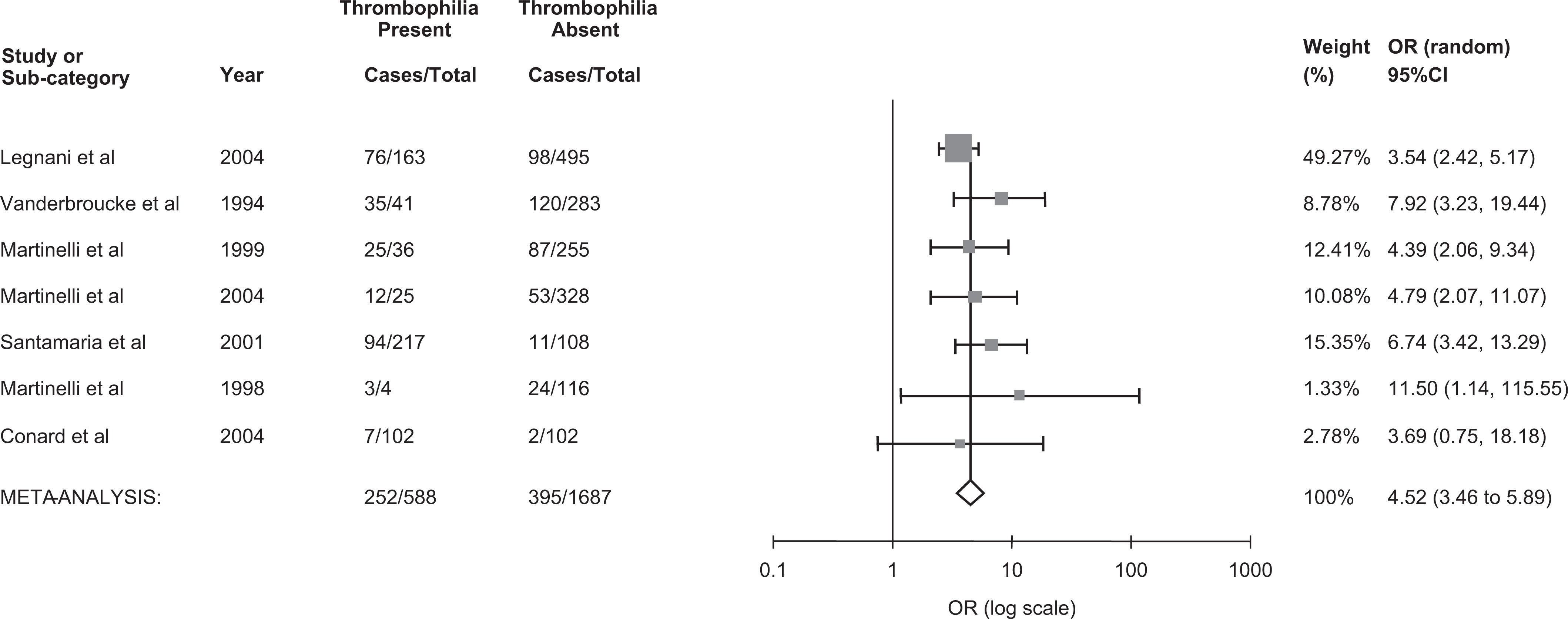
The pooled odds ratio for venous thrombosis in thrombophilia carriers. OR indicates odds ratio; CI, Confidence interval.
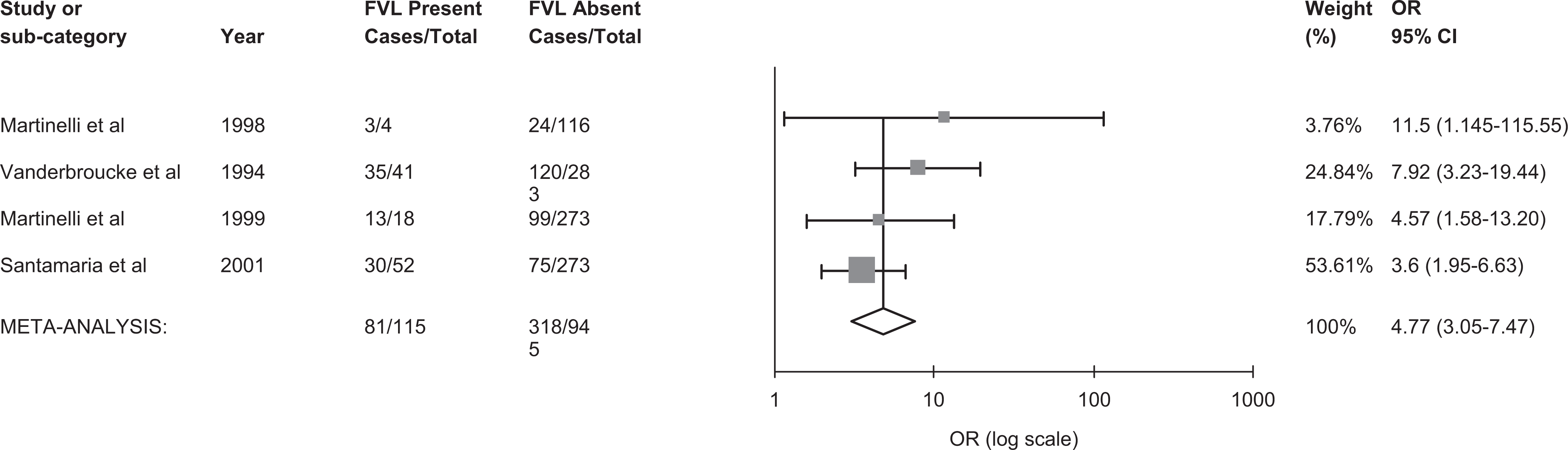
The pooled odds ratio for venous thrombosis in FVL carriers. FVL indicates factor V Leiden; CI, confidence interval; OR, odds ratio; cases, number with venous thrombosis.
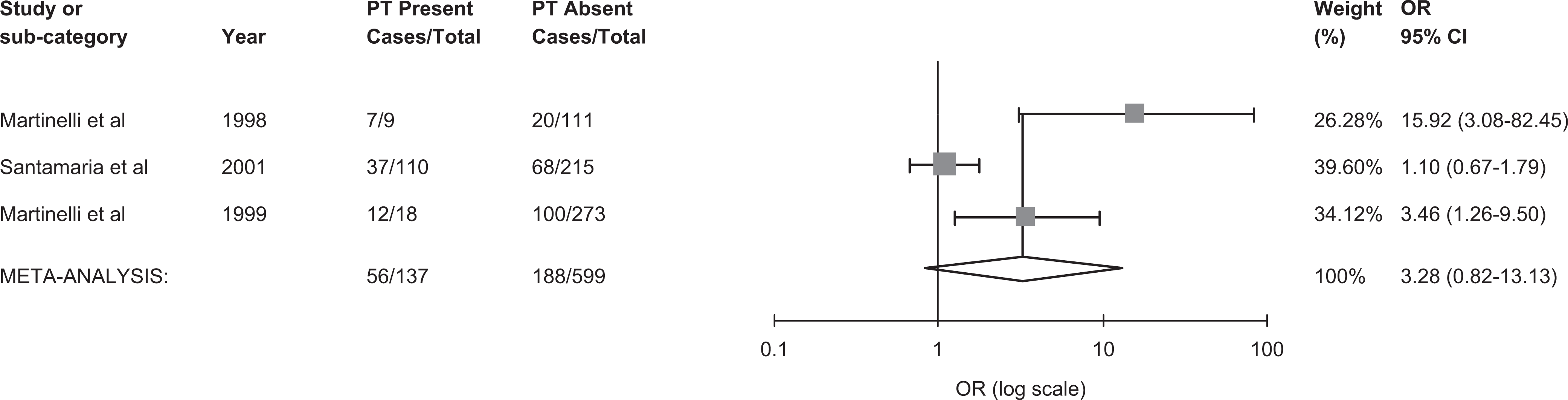
The pooled odds ratio for venous thrombosis in prothrombin 20210A carriers. PT indicates prothrombin 20210A mutation; OR, odds ratio; cases, number with venous thrombosis.
Combined Effect of OCP Use and Thrombophilia on VT Risk
Seven studies that included a total 647 women with thrombosis and 1528 controls were included to assess the interaction between OCP and any thrombophilia on the risk of thrombosis (Figure 6).7,11,54,56–59 Compared to women without thrombophilia and non-OCP use, women with any thrombophilia and OCP use had a 14-fold increased risk of VT (pooled OR 14.11, 95% CI 6.27-31.72). Moderate heterogeneity was observed (I 2 = 57%, 95% CI 0%-81%). A meta-regression revealed that thrombophilia type (FVL vs no FVL) was a statistically significant covariate (P = .024) and therefore explained variability in the log OR, while study type (case−control vs cohort design) was not significant (P = .897). Among 4 studies7,11,54,57 examining the effect of FVL mutation in combination with OCP on the risk of VT (Figure 7), the individual OR estimates ranged widely between 5.86 and 129 and were associated with large confidence intervals. The pooled analysis showed a 17-fold increased risk of thrombosis among FVL carriers with OCP use compared to women without FVL and without OCP use (pooled OR 17.84, 95% CI 5.61-56.78; Figure 7). Heterogeneity was moderate and was not explained by study design in a meta-regression. It is likely that differences among the studies in the selection of the populations analyzed explain the heterogeneity. Among 3 studies7,54,57 examining the effect of PT G20210A mutation in combination with OCP on the risk of VT (Figure 8), the individual OR estimates ranged widely between 1.45 and 149 and were associated with large confidence intervals. A pooled OR estimated of 12.4 with a wide confidence interval that crossed unity (95% CI, 0.9-165.8) was observed, and as a result it was difficult to draw any conclusions based on the pooled result.
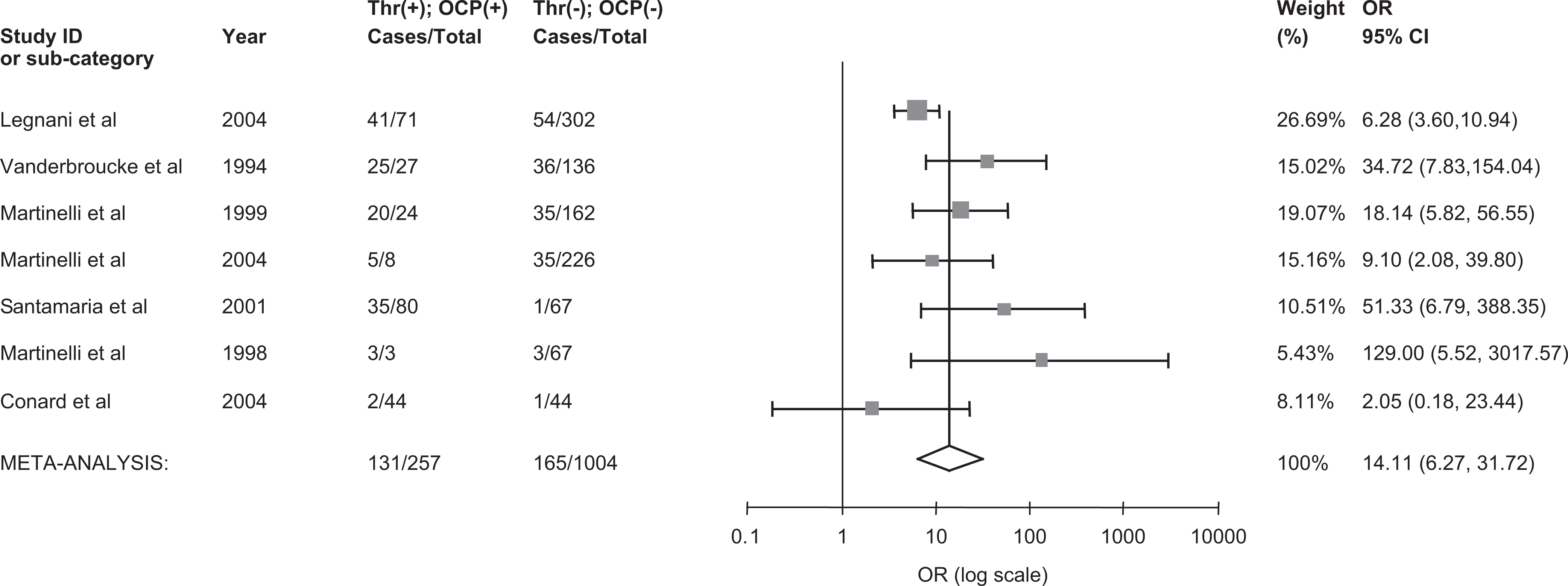
The pooled odds ratio for venous thrombosis in oral contraceptive users with thrombophilia. Thr (+) indicates thrombophilia present; Thr (−), thrombophilia absent; OCP (+), oral contraceptive use; OCP (−), oral contraceptive nonuse; OR, odds ratio; CI, confidence interval; cases, number with venous thrombosis.
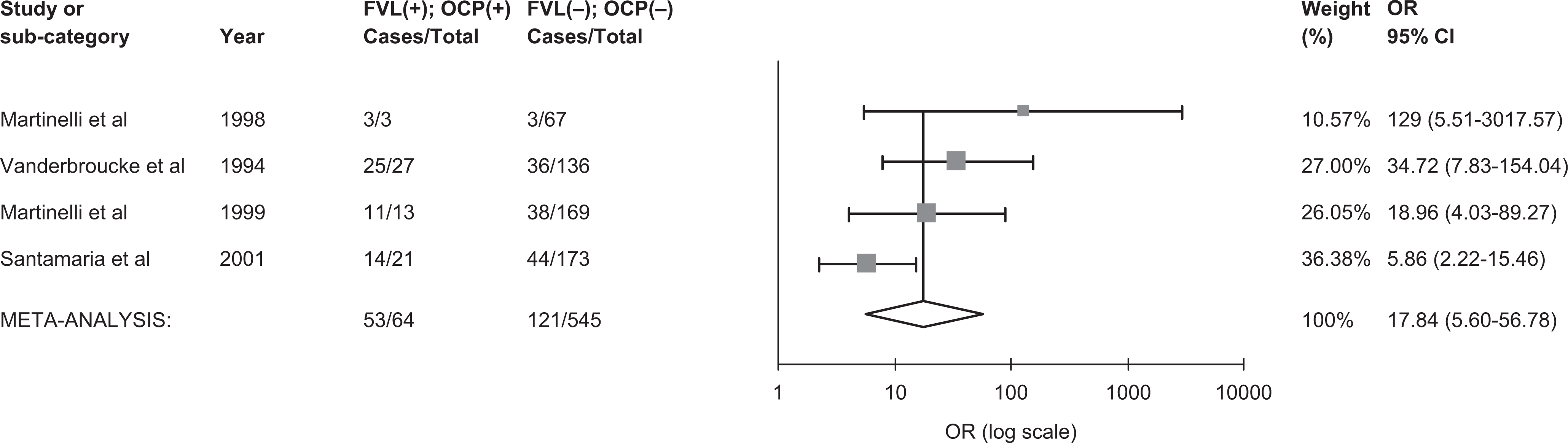
The pooled odds ratio for venous thrombosis in oral contraceptive users with FVL. FVL (+), factor V Leiden present; FVL (−), factor V Leiden absent; OCP (+), oral contraceptive use; OCP (−), oral contraceptive nonuse; OR, odds ratio; CI, confidence interval; cases, number with venous thrombosis.
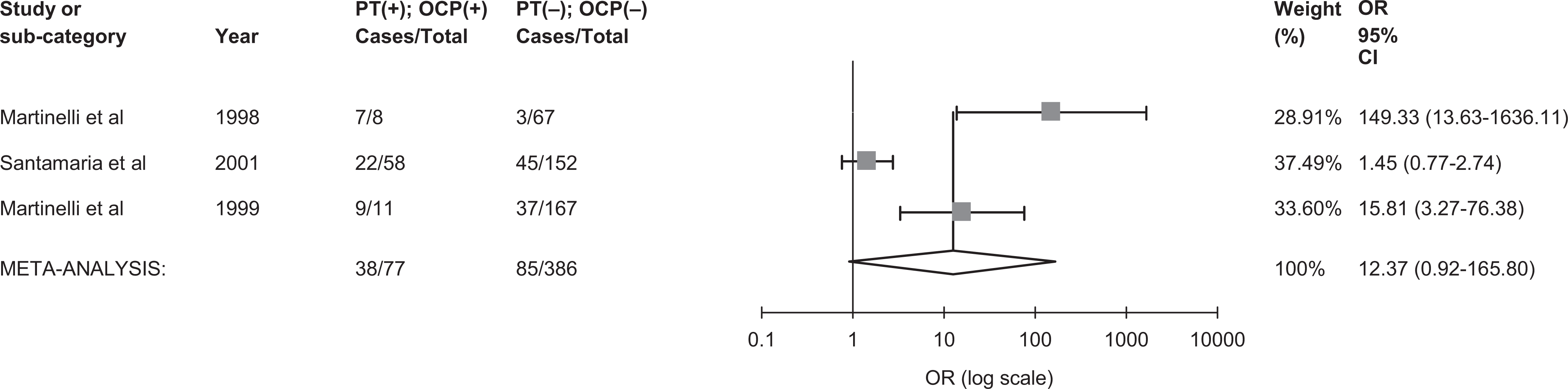
The pooled odds ratio for venous thrombosis in carriers of prothrombin 20210A mutation with oral contraceptive use. PT (+) indicates prothrombin present; PT (−), prothrombin absent; OCP (+), oral contraceptive use; OCP (−), oral contraceptive nonuse; OR, odds ratio; CI, confidence interval; cases, number with venous thrombosis.
Comment
In this latest and most up-to-date meta-analysis of 15 observational studies that included reproductive aged women with and without OCP use and with and without thrombophila, we determined that OCP users were approximately 3 times more likely to develop any thrombosis compared to non-OCP users. This is in agreement with Wu et al who in their meta-analysis of 7 studies addressing the effect of OCP on the risk of DVT and PE reported an OR of 3.10 (95% CI 2.17-4.42), 66 and slightly lower than that reported by Dentali et al in their meta-analysis evaluating the effect of OCP on the risk of CVT alone (OR of 5.59; 95% CI 3.95-7.91). 67 Moreover, having any thrombophilia conferred a 4.5-fold increased risk of thrombosis, which is comparable to previously reported estimates.6,11,67,68 In particular, the association between thrombosis and FVL mutation was equally strong and robust (add OR and CI here) with a relatively narrow confidence interval, though that of PT mutation and thrombosis was less robust and as a result difficult to interpret. We evaluated the combined effect of thrombophilia and OCP on the risk of thrombosis. A supra-additive effect was seen. Specifically, the risk of thrombosis in women with thrombophilia and OCP use was 14 times that of non-OCP users and without thrombophilia. Moderate heterogeneity was found and thrombophilia type was statistically important in explaining some of the variability among the studies. As a result, among women with OCP use and FVL only, a 17-fold increased risk of thrombosis compared to women without OCP use and without FVL mutation was observed. This estimate is consistent with that reported by Wu and colleagues in their meta-analysis of the effect of FVL with OCP use on thrombosis risk (OR = 15.62, 95% CI 8.66-28.15) 66 but greater than that reported by Emmerich et al in their review of 3 studies that reported on the combined effect of FVL and OCP use on thrombosis risk (OR = 10.25; 95% CI 5.69-18.45). 68 Variabilty in study inclusion criteria likely explains the range of reported summary risk estimates. Nonetheless, there is agreement of a supra-additive effect for FVL and OCP on thrombosis risk. A synergistic effect is suggested for PT mutation and OCP use though the estimate of association is difficult to interpret given the wide variability in the individual studies and wide CI associated with the pooled estimate.
Our study has limitations. First, meta-analysis of observational studies is controversial because of inherent biases in the included studies. To minimize potential for bias, we selected only case−control and cohort studies with objective confirmation of thrombotic events and standard measurement methods for thrombophilic defects. Furthermore, we employed all of MOOSE criteria. 14 Nonetheless, selection bias remains a strong possibility. For example, over one third of included studies recruited controls from relatives of people with known thrombophilia. This may have led to an overrepresentation of control women with thrombophilia who may have been counseled to avoid OCP use, resulting in an underestimation of the association between OCP and thrombosis. Second, studies included in our meta-analysis had different population inclusion and exclusion criteria. For example, some studies included men and women of all ages, and we subsequently extracted data on nonpregnant reproductive-aged females, while other studies only recruited reproductive-aged women. However, when including observational studies in a meta-analysis, it is suggested that an initial search strategy using broad inclusion criteria be used, and that subsequent analyses should relate study design features to outcome, as in our attempt to explain heterogeneity by exclusion of cohort studies from our analysis. 14 Third, heterogeneity remained substantial for some analyses, even after considering potential sources of heterogeneity such as study design, and the results should therefore be interpreted with caution.Moreover, the small number of thrombosis events in some analyses and paucity or lack of information on patient-related characteristics, such as prior history of VTE, smoking, and family history of VTE, limited our ability to adequately assess heterogeneity. Finally, we were not able to evaluate the impact of age and OCP type on the risk of thrombosis because few studies provided separate analyses according to these factors. We would expect that risk of thrombosis with OCP use with and without thrombophilia would be more pronounced among older women, and quantifying this risk according to age group would be clinically relevant. As well, the specific preparations of OCPs have changed since their inception and thus likely impart different degrees of risk on thrombosis. For example, third-generation OCPs which contain desogestrel have been shown to be more thrombogenic than second-generation OCPs which do not contain desogestrel. 69 Only 1 of our included studies reported data on the type of OCP used, and hence it was not possible to perform a pooled analysis.
In summary, our meta-analysis suggests important associations between OCP use and thrombophilia on the risk of VT. A particularly robust association was observed between FVL mutation and thrombosis, confirming what has previously been proposed from observational studies. Our study also confirmed results of previous studies supporting a supra-additive effect of OCP combined with thrombophilia (FVL in particular) on thrombosis risk. However, considering the significant heterogeneity in our meta-analyses, our findings should be interpreted with caution. Furthermore, we were unfortunately unable to determine the absolute risk of thrombosis in women with OCP use and thrombophilia, which we recognize to be critical information for physicians advising women with thrombophilia who are contemplating OCP use. As a result, large prospective cohort studies are needed to more precisely define the effect of OCP use on the risk of thrombosis among women with and without thrombophilia, who are carefully selected from the general population. This would allow for determination of absolute risk of thrombosis with OCP use in women with thrombophilia compared to women without thrombophilia. Moreover, such a study would provide a more accurate assessment of the interaction between OCP and thrombophilia type, while addressing potential modifying effects of known risk factors for thrombosis with OCP use such as age and smoking.
Footnotes
This study was presented on July 16, 2009, at the XXII Congress of The International Society on Thrombosis and Haemostasis, July 11-16, 2009, Boston, Massachusetts USA.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article
