Abstract
We compared thrombophilia in 67 cases (59 men and 8 women) with thrombotic events after starting testosterone therapy (TT) versus 111 patient controls having unprovoked venous thrombotic events without TT. In the 67 patients, thrombosis (47 deep venous thrombosis–pulmonary embolism, 16 osteonecrosis, and 4 ocular thrombosis) occurred 6 months (median) after starting TT. Cases differed from controls for factor V Leiden heterozygosity (16 of the 67 [24%] vs 13 [12%] of the 111, P = .038) and for lupus anticoagulant (9 [14%] of the 64 vs 4 [4%] of the 106, P = .019). After a first thrombotic event and continuing TT, 11 cases had a second thrombotic event, despite adequate anticoagulation, 6 of whom, still anticoagulated, had a third thrombosis. Screening for thrombophilia before starting TT should identify men and women at high risk for thrombotic events with an adverse risk–benefit ratio for TT. When TT is given to patients with familial and acquired thrombophilia, thrombosis may occur and recur in thrombophilic men despite anticoagulation.
Keywords
Introduction
In June 2014, based on postmarketing surveillance reports including our studies, 1 –7 both the US Food and Drug Administration (FDA) 8 and Canada Health 9 added a warning regarding the risks of venous thromboembolism (VTE) to the label of all testosterone (T) products. Venous thromboembolism, particularly pulmonary embolism (PE), is associated with significant mortality risk. 10 The FDA has subsequently emphasized the importance of a prospective, blinded, placebo-controlled clinical trial to assess cardiovascular and thrombotic safety of testosterone therapy (TT). 11
Previously, in aggregate, 1 –7 we have described first-event VTE and osteonecrosis (ON) 12 developing after 5 months (median) on TT (TT-VTE) in 59 patients, 57 of whom had measures of thrombophilia–hypofibrinolysis, compared to 22 controls with thrombotic events but not taking TT (VTE-no TT). Of the 57 TT-VTE cases with measures of thrombophilia–hypofibrinolysis, 23% was heterozygous for the factor V Leiden mutation versus 14% of VTE-no TT controls (NS), and 25% had high factor VIII (>150%) versus 50% of controls (P = .06). In the current study, we have expanded the number of TT-VTE cases to 67 and the VTE-no TT controls to 111 to better assess relationships of TT to thrombosis in larger cohorts of cases and controls.
Beyond interacting with familial and acquired thrombophilia, 1 –7 TT is associated with prothrombotic changes including hypertension, 13 elevated hemoglobin, 14 low high-density lipoprotein cholesterol, 13,14 hyperviscosity, polycythemia, 15 and platelet aggregation. 14,16,17 Testosterone therapy also increases circulating estrogens 18 that subsequently play a role in thrombotic events. 6 Given that T is aromatized to estradiol (E2), 19 it may be prothrombotic by the same mechanism as reported in women, where hormone replacement therapy interacts with the factor V Leiden mutation to increase the risk of VTE. 20
Our specific aim was to assess thrombophilia in our total cohort of 67 patients who sustained VTE on TT and to compare these 67 cases to 111 controls who had unprovoked thrombotic events without TT or exogenous estrogens.
Materials and Methods
Patients
The procedures followed were in accordance with the ethical standards of the Institutional Review Board of the Jewish Hospital, Cincinnati, Ohio, which approved the research protocol. The protocol was carried out with the understanding and signed informed consent of each participant.
We excluded patients and controls whose VTE was provoked, associated with cancer, and with polycythemia vera, recent soft tissue trauma, bone fracture, hip–knee–foot surgery, airline flights >8 hours, or extensive immobilization.
The 67 TT-VTE patients (59 men and 8 women) were studied in their serial order of referral over the last 4 years to our thrombosis center by their family physicians–gynecologists–urologists because of thrombotic events. Thrombophilia had been assessed previously in 57 of these patients. 7 Testosterone therapy had been prescribed by the referring physicians, and if not already stopped, it was discontinued by us before our assessment of thrombophilia and hypofibrinolysis. Measurement of thrombophilia–hypofibrinolysis was carried out at least 2 months after the cases’ initial thrombotic event.
At study entry, a detailed history 21 –23 for VTE, deep venous thrombosis (DVT), PE, ON, ocular thrombosis, recurrent miscarriage, preeclampsia, eclampsia, and HELLP syndrome (hemolysis, elevated liver enzymes, low platelet count, pre-eclampsia) was taken for all of the TT-VTE cases and VTE-no TT controls.
Controls
Thrombophilia and hypofibrinolysis were assessed in 111 patient controls in the temporal sequence of their referral, not taking TT (VTE-no TT). These patients had been referred to us for the evaluation of unprovoked thrombotic events, DVT-PE, ON, and ocular thrombosis. Previously, we had assessed thrombophilia in 22 of these 111 VTE-no TT controls. 7
Laboratory Assessment of Thrombophilia and Hypofibrinolysis
Polymerase chain reaction assays
Polymerase chain reaction (PCR) measures of thrombophilia (G1691A factor V Leiden, G20210A prothrombin, and methylene tetrahyrofolate reductase C677T/A198C) and hypofibrinolysis (plasminogen activator inhibitor [PAI-1 SERPINE 1] gene polymorphism 4G/5G) were performed in all cases and controls using previously published methods 24,25 by laboratory staff blinded to the participants’ status (diagnosis and severity of the disease).
Serologic Measures of Thrombophilia and Hypofibrinolysis
Serologic measures of thrombophilia included factors VIII and XI, homocysteine, antigenic proteins antithrombin III, C, S, and free S, as well as the antiphospholipid (APL) syndrome (anticardiolipin antibodies [immunoglobulin (Ig) G, IgM], the lupus anticoagulant, and anti–β2-glycoprotein). Established, previously published methods were used. 25 High homocysteine was identified by levels more than the laboratory 95th percentile. 26 The 3 components of the APL antibody syndrome were measured by enzyme-linked immunosorbent assay using previously published methods. 27 –29 Plasminogen activator inhibitor 1 activity levels, associated with hypofibrinolysis, were not measured.
Statistical Methods
All statistical analyses were done using SAS version 9.4. Fisher exact test was used to compare thrombophilia–hypofibrinolysis in cases versus VTE-no TT controls
Results
Patients and Controls
By selection, none of the 67 TT-VTE cases or the 111 VTE-no TT controls (Tables 1 and 2) had DVT-PE provoked by cancer, elevated hemoglobin, antecedent trauma or surgery, recent airline travel longer than 8 hours, or extended immobilization.
Demographics of 67 Patients With Thrombotic Events After Starting TT and 111 Controls With Thrombotic Events Not Receiving TT.

Abbreviation: DVT, deep venous thrombosis; HCG, human chorionic gonadotropin; PE, pulmonary embolism; SD, standard deviation; TT, testosterone therapy. a The thrombotic event types did not differ between cases and controls, Fisher’s P = .47.
Thrombophilia in 67 Patients With DVT-PE 6 Months (Median) After Starting TT (TT-VTE) Compared to 111 Controls With DVT-PE Not Taking TT (VTE-No TT).
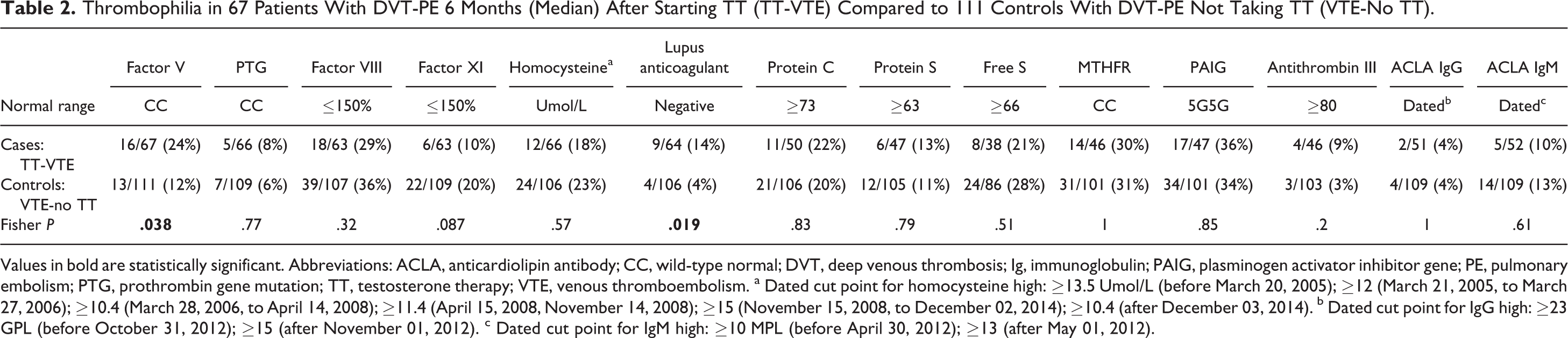
Values in bold are statistically significant. Abbreviations: ACLA, anticardiolipin antibody; CC, wild-type normal; DVT, deep venous thrombosis; Ig, immunoglobulin; PAIG, plasminogen activator inhibitor gene; PE, pulmonary embolism; PTG, prothrombin gene mutation; TT, testosterone therapy; VTE, venous thromboembolism. a Dated cut point for homocysteine high: ≥13.5 Umol/L (before March 20, 2005); ≥12 (March 21, 2005, to March 27, 2006); ≥10.4 (March 28, 2006, to April 14, 2008); ≥11.4 (April 15, 2008, November 14, 2008); ≥15 (November 15, 2008, to December 02, 2014); ≥10.4 (after December 03, 2014). b Dated cut point for IgG high: ≥23 GPL (before October 31, 2012); ≥15 (after November 01, 2012). c Dated cut point for IgM high: ≥10 MPL (before April 30, 2012); ≥13 (after May 01, 2012).
Because we did not initiate TT in the TT-treated patients with VTE, we do not know the etiology of the hypogonadism in the TT-treated group.
Testosterone therapy–VTE cases and VTE-no TT controls differed by age (median, 53 vs 60 years; Table 1). The VTE-no TT controls were more likely to be female than TT-VTE cases (Table 1). Of the TT-VTE cases, 58% used T gels, 31% took intramuscular TT, 4% pellets, 3% patches, 3% human chorionic gonadotropin (HCG), and 1% Clomid (Table 1).
Thrombotic Events and Thrombophilia in TT-VTE Cases and in VTE-no TT Controls
Testosterone therapy–VTE cases did not differ from VTE-no TT controls in regard to the distribution of thrombotic events (P =.47), with most cases and controls having DVT-PE, then ON, and then ocular thrombosis (Table 1).
The median duration of time from starting TT to development of thrombotic events was 6 months, with the 25th to 75th percentile being 3 to 12 months (Table 1). Cases differed from VTE-no TT controls for factor V Leiden heterozygosity (16 [24%] of the 67 vs 13 [12%] of the 111, P = .038) and for lupus anticoagulant (9 [14%] of the 64 vs 4 [4%] of the 106, P = .019; Table 2).
After a first thrombotic event and with TT continued by the patients’ physicians, 11 TT-VTE patients with familial or acquired thrombophilia had a second thrombotic event, despite adequate anticoagulation with warfarin to an international normalized ratio of 2.0 to 3.0 or with apixaban (10 mg twice/day for the first 7 days and then 20 mg once/day) or rivaroxaban (15 mg twice/day for 21 days and then 20 mg once/day). Six of these 11 TT-VTE patients, with TT continued and still anticoagulated, had a third thrombotic event.
In 66 of the 67 TT-VTE cases, the thrombotic event after starting TT was the patients’ first, in 1 participant, unprovoked DVT that had occurred 5 years earlier. In 110 of the 111 VTE-no TT controls, the thrombotic event was the patients’ first, in 1 participant, unprovoked DVT that had occurred 3 years earlier.
Discussion
We speculate that TT is likely to result in DVT, pulmonary embolus, ON, or ocular thrombosis among men or women 2 with underlying familial or acquired thrombophilia. Similarly, in women on hormone replacement therapy (HRT), the presence of the factor V Leiden mutation or high factor VIII increases the risk of DVT 17-fold compared to women on HRT without these thrombophilias. 30 Moreover, the time course in our current study between starting TT and development of a thrombotic event, 6 months (median), is comparable to VTE in women receiving HRT, where VTE rates are highest in the first year of treatment and are much more pronounced in women at higher risk for VTE by virtue of thrombophilia. 31
Our finding that 24% of our TT-VTE patients had factor V Leiden heterozygosity versus 12% of our VTE-no TT controls illustrates the importance of the factor V mutation in VTE, 32,33 and its interaction with TT, promoting thrombotic events. The incidence of factor V Leiden heterozygosity in our VTE control group of 111 patients was 12%, comparable to 12.8% in the large RIETE study of 2744 patients, 32 14.5% in 165 patients studied by Leroyer et al, 34 and 14.1% in 99 patients studied by de Moerloose et al, 35 but lower than 19% in 1668 patients studied by Tick et al. 36 By promoting VTE in patients with underlying thrombophilia, TT and/or HCG or Clomid therapy may enrich TT-VTE cohorts with more cases of thrombophilia than observed in patients with DVT-PE not taking TT, as in the current study.
In men, most E2 comes from aromatization of T. 37 We speculate that when exogenous T is aromatized to E2 and E2-associated thrombophilia is superimposed on familial and acquired thrombophilia, as in our current and previous 6,7 reports, thrombosis occurs. We have reported previously that 45% of men receiving conventional gel TT (50 mg/d) have high serum E2 (≥42.6 pg/mL).3 In our recent study 7 of patients with thrombophilia sustaining thrombotic events after starting TT, of the 9 men having measures of E2 on TT, 7 (78%) had high E2. The high E2 may also reflect overtreatment with TT. In our recent study, 7 of the 10 men having measures of T during TT, 6 (60%) had supranormal serum T values (>800 ng/dL), and 5 of these 6 men also had high E2 or free E2.
The potency of the prothrombotic stimulus of exogenous TT is testified to by the finding that 11 men with thrombophilia sustained a first thrombotic event on TT, and while continuing TT, despite adequate anticoagulation, sustained a second VTE. Moreover, 6 of these 11 men, remaining on TT and still anticoagulated, had a third VTE.
After a thrombotic event, if TT is continued in thrombophilic patients, concomitant and adequate anticoagulation does not appear to prevent recurrent thrombotic events. 6,7
Before starting TT, to facilitate the prevention of VTE, we recommend, at a minimum, PCR measure of the factor V Leiden mutation and measure of the lupus anticoagulant. In a parallel fashion, the issue of pretreatment thrombophilia screening before giving exogenous estrogens has also been raised in women. 30,38 –40
Our findings may have important clinical implications, since VTE risk is an important determinant of the benefit–risk ratio of TT. In women, for example, PE accounts for about one-third of the incidence of potentially fatal VTE events associated with HRT, 41 and HRT increases the risk of VTE by 2- to 3-fold. 31
Only 1 of the 67 TT patients had a thrombotic event preceding TT, so that using history of thrombosis in lieu of measurement of thrombophilia would not be predictive in predicting the risk of thrombosis in patients about to receive TT.
We studied only morbid VTE in the current study, a limitation, since mortal VTE might be associated with a larger percentage of participants with thrombophilia. Because we did not initiate TT in the TT-treated patients with VTE, we do not know the etiology of their hypogonadism.
An optimal study, paralleling the Women’s Health Initiative studies in women, 42,43 would be prospective, placebo-controlled, and double-blind 11 with thrombotic and cardiovascular disease end points and evaluation of the elevated hemoglobin, hypertension, prostate cancer, and osteoporosis. 8,11
Footnotes
Authors’ Note
The study was carried out following a protocol approved by the Jewish Hospital Institutional Review Board, with signed informed consent. The manuscript has been reviewed and approved by all named authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported in part by the Lipoprotein Research Fund of the Jewish Hospital of Cincinnati
