Abstract
Background:
Patients with sickle cell disease (SCD) are at high risk of renal dysfunction and cardiovascular morbidity. The association between cystatin C and renal function is well known, however, cystatin C has recently emerged as a strong predictor of cardiovascular events and adverse outcomes in patients with and without kidney disease, mostly related to both inflammation and atherosclerosis.
Aim:
To determine cystatin C levels in 53 children and adolescents with SCD compared to 40 age- and sex-matched healthy controls and assess its relation to markers of hemolysis, iron overload, sickle vasculopathy, and carotid intima–media thickness (CIMT).
Methods:
Patients with SCD in steady state were studied, focusing on hydroxyurea therapy, hematological profile, serum ferritin, high-sensitivity C-reactive protein (hs-CRP), urinary albumin–creatinine ratio (UACR), and serum cystatin C. Echocardiography and CIMT were assessed using high-resolution ultrasound. Heart disease was defined by systolic left ventricle dysfunction (shortening fraction <30% or ejection fraction <55%).
Results:
Carotid IMT was significantly higher in patients with SCD compared to controls (P < .001). Patients with SCD having nephropathy, heart disease, or history of frequent sickling crisis (≥3 attacks/y) had significantly higher cystatin C levels than those without (P < .05). Patients with SCD treated with hydroxyurea had lower cystatin C levels than untreated patients (P = .039). High-sensitivity C-reactive protein, UACR, ejection fraction, and CIMT were independently related to cystatin C in multiple regression analysis. The cutoff values of cystatin C for detection of renal or cardiovascular complications were determined.
Conclusion:
Cystatin C may be considered a biological marker for vascular dysfunction and subclinical atherosclerosis in SCD.
Keywords
Introduction
Sickle cell disease (SCD) is one of the most common monogenetic disorders in the world, affecting nearly 1 in 600 African Americans 1 and an estimated 1% to 4% of babies born in sub-Saharan Africa. 2 Patients with SCD are at risk for a progressive vasculopathy characterized by systemic and pulmonary hypertension, endothelial dysfunction, and proliferative changes in the intima and smooth muscle of blood vessels. 1,3 With increasing age, the incidence of chronic end-organ complications increases. 4 The type and severity of complications vary significantly between individuals from mild symptoms to hospitalization with serious life-threatening complications. 5
Chronic anemia in SCD results in cardiac chamber dilation and a compensatory increase in left ventricular mass. This is often accompanied by left ventricular diastolic dysfunction that has also been a strong independent predictor of mortality in patients with SCD. Both pulmonary hypertension and diastolic dysfunction are associated with marked abnormalities in exercise capacity in these patients. 6
Patients with SCD may have renal dysfunction from early childhood. 7 Renal failure is the consequence of a series of events related to nephron loss due to ischemia–reperfusion injury, glomerular hypertension, proteinuria, and renal scarring. 8 Serum creatinine and creatinine clearance are still the most accessible measurements in clinical practice and currently are used to assess renal function in patients with SCD, however, because of the increased creatinine secretion by renal tubules, serum creatinine is generally low and is not a reliable test to detect early signs of decreasing kidney function. 9,10
Cystatin C or cystatin 3, a protein encoded by the CST3 gene, is mainly used as a biomarker of kidney function. Recently, it has been studied for its role in predicting new-onset or deteriorating cardiovascular disease (CVD). 11 –13 Cystatin C is a nonglycosylated low-molecular-weight (13 kD) basic protein that inhibits cysteine proteases and correlates closely to glomerular filtration rate (GFR) in children. 14 Cystatin C crosses the glomerular membrane, and it is reabsorbed and metabolized in the renal tubules and not returned to the bloodstream. Unlike creatinine, cystatin C is not secreted by the tubules, even in cases of reduced GFR, 15 and is not affected by muscle mass, protein intake, metabolic factors, drugs, and inflammatory stimuli. 16
All of these characteristics make it ideal for study in SCD because patients have ongoing hemolysis and inflammation secondary to the sickling phenomenon. It has been reported that serum cystatin C correlates with the level of albuminuria and may be a reliable method to measure renal function in SCD. 17,18 Serum cystatin C levels perform better than other markers of renal function as indicator of cardiovascular outcome both in population-based studies and in patients with non-ST-elevation acute coronary syndrome (ACS). 12
Carotid intima–media thickness (CIMT) is a surrogate marker of atherosclerosis and provides a noninvasive method for the risk assessment of CVD. 19 It is a strong predictor of future cardiovascular events and is associated with conventional markers of cardiovascular risk such as age, diabetes, and serum cholesterol. 20 Cystatin C was more strongly associated with carotid thickening and plaque than other measures of kidney function. 21 Therefore, we sought to measure serum cystatin C levels in children and adolescents with SCD and assess its relation to markers of hemolysis, iron overload, and CIMT as well as its potential value as an early determinant of sickle cell vasculopathy.
Materials and Methods
This cross-sectional study included 53 patients with SCD (39 males and 14 females) recruited from the regular attendants of the Pediatric Hematology Clinic, Pediatric Hospital, Ain Shams University. Forty age- and sex-matched healthy participants were enrolled as a control group (26 males and 14 females). The median age of patients with SCD was 9.5 years, whereas that of controls was 9 years. An informed consent was obtained from the guardian of each patient or control before participation. The procedures applied in this study are approved by the ethical committee of Human Experimentation of Ain Shams University and are in accordance with the Helsinki Declaration of 1975.
All patients were diagnosed with SCD based on complete blood picture, reticulocyte count, and markers of hemolysis as well as hemoglobin analysis using high-performance liquid chromatography (HPLC) and confirmed by genotyping based on identification of β-globin gene mutations by polymerase chain reaction and subsequent reverse hybridization to immobilized allele-specific biotinylated oligonucleotides probes covering the most common Mediterranean mutations. 22,23 Patients with SCD included 32 patients with sickle cell anemia (SCA) and 21 patients with sickle β-thalassemia.
Patients with infection, chronic inflammatory condition other than SCD, renal disease unrelated to SCD, symptomatic heart disease, rheumatoid arthritis or other autoimmune diseases, hypothyroidism, diabetes mellitus, or steroid therapy were excluded. All the studied patients were in a steady state at the time of sample collection, and those who had sickling crisis were excluded.
Patients with SCD were subjected to the following—detailed medical history and thorough clinical examination with special emphasis on disease duration, history of sickling crisis, acute chest syndrome, stroke, priapism, bone manifestations, evidence of pulmonary or cardiac disease, spleen status (for patients with sickle β-thalassemia). A painful crisis was defined as the occurrence of pain in the extremities, back, abdomen, chest, or head that lasted at least 2 hours, led to a clinic visit, and could not be explained except by SCD, 24 while steady state was defined as a period without pain or painful crisis for at least 4 weeks. 25 The frequency of sickling crisis in the previous year was divided into mild (defined as 2 or less episodes requiring medical visits) or severe (defined as 3 or more episodes requiring medical visits). 26 The diagnosis of acute chest syndrome was defined as a new pulmonary infiltrate on chest X-ray and ≥2 of the following—chest, upper abdominal, or rib pain; dyspnea; fever; tachypnea; grunting; nasal flaring; or retractions. 27
All of the patients were transfused. The transfusion received was calculated as the transfusion index—volume of transfused packed red cells in milliliter per kilogram body weight per year (expressed as the mean value of the last 3 years). Thirty-nine (73.6%) patients with SCD received hydroxyurea (Bristol-Meyers-Squibb, New York) as an oral daily dose ranging from 10 to 25 mg/kg/d. Patients with SCD who needed chelation received deferoxamine (DFO) therapy (Desferal; Novartis Pharma AG, Basel, Switzerland), given subcutaneously in a dose that ranged from 30 to 40 mg/kg/d 5 d/wk).
Sample Collection
Peripheral blood samples were collected on potassium–ethylenediaminetetraacetic acid (1.2 mg/mL) as an anticoagulant for complete blood count (CBC) and hemoglobin analysis. For chemical analysis and enzyme-linked immunosorbent assay (ELISA), clotted samples were obtained and serum was separated by centrifugation for 15 minutes at 1000g then stored at −20
Laboratory Assessment
Laboratory investigations included CBC using Sysmex XT-1800i (Sysmex, Kobe, Japan), examination of Leishman-stained smears for differential white blood cell (WBC) count, hemoglobin analysis by HPLC using D-10 (Bio-Rad, Marnes La Coquette, France), liver and kidney function tests, markers of hemolysis (lactate dehydrogenase [LDH] and indirect bilirubin), and high-sensitivity C-reactive protein (hs-CRP) as well as serum ferritin on Cobas Integra 800 (Roche Diagnostics, Mannheim, Germany). Patients with any clinical evidence of infection or hs-CRP >10 mg/L were excluded. Serum ferritin level was measured at the start of the study with calculation of the mean value of the last year prior to the study in order to know the ferritin trend.
Urinary albumin excretion (as an indicator of nephropathy) was measured in an early morning urine sample as albumin to creatinine ratio (UACR) using Cobas Integra 800 (Roche Diagnostics). Nephropathy was defined as micro- or macro-albuminuria assessed by urinary albumin excretion in at least 2 of the 3 consecutive urine samples, 2 months apart. Microalbuminuria or macroalbuminuria is present if UACR = 30 to 299 mg/g creatinine or ≥300 mg/g creatinine, respectively. 28 Determination of serum levels of cystatin C was done by ELISA using Quantikine Human Cystatin C Immunoassay (R&D systems, Minneapolis, Minnesota).
Echocardiography
All studied patients were clinically asymptomatic for pulmonary hypertension and cardiovascular abnormalities. Screening for pulmonary hypertension and cardiovascular abnormalities was performed by the noninvasive echocardiography with different modalities (Doppler, 2D imaging, and M-mode) using Vivid E9 (GE Healthcare, Oslo, Norway) to evaluate left ventricular function, pulmonary artery pressure, and tricuspid regurgitant jet velocity (TRV). A TRV ≥2.5 m/s was used as a proxy for patients at risk for pulmonary hypertension. 29,30 Heart disease was defined by a systolic left ventricle (LV) dysfunction (LV shortening fraction <30% or LV ejection fraction [EF] <55%). 31
Measurement of CIMT
All of the carotid scans were done using carotid Doppler ultrasound scanner (Toshiba Ultrasonography machine [Xario], Tokyo, Japan) with a 10.0-MHz linear array transducer following a predetermined standardized scanning protocol. 32 The left and right common carotid arteries were imaged in a standardized magnification (2 × 2 cm) with images of the posterior wall of the distal 10 mm of the common carotid artery, just proximal to the carotid bulb. To ensure the images were taken perpendicular to the vessel, images were captured when both the anterior and posterior wall margins were clearly seen. A minimum of 4 images of each of the common carotid arteries were taken. All images were taken at end diastole, incident with the R wave on a continuously recorded electrocardiogram and then digitally stored for later analysis. The 3 best quality images for each of the carotid arteries were selected and analyzed. Best quality was defined with those images that produced the most number of points for analysis. For each image, the greatest distance between the lumen–intima interface and media–adventitia interface (IMT) was measured at a minimum of 100 points. The mean and maximum IMT of each image were then averaged to give the final result for each participant. All the ultrasonographic examinations were analyzed by a single experienced radiologist blinded to the group to which the participants belonged.
Statistical Analysis
Analysis of data was done using Statistical Program for Social Science version 15 (SPSS Inc, Chicago, Illinois). Quantitative variables were described in the form of mean and standard deviation or median and interquartile range (75th and 25th percentiles). Qualitative variables were described as number and percentage. In order to compare parametric quantitative variables between 2 groups, Student t test was applied. For comparison of nonparametric quantitative variables between 2 groups, Mann-Whitney test was used. Qualitative variables were compared using χ2 test or Fischer exact test when frequencies were below 5. Pearson correlation coefficient and stepwise multiple linear regression analysis were used to assess the relation between cystatin C and clinical as well as laboratory variables. Receiver–operating characteristic curve was used to determine the best cutoff value of cystatin C, with the highest balanced sensitivity and specificity. The area under the curve (AUC) was calculated for each plot. A P value <.05 was considered significant in all analyses.
Results
The median disease duration was 8 years and the mean transfusion index was 158.5 ± 24.6 (range: 19.2-265) mL/kg/y. Serum creatinine was within normal range but median UACR was 34.9 (range: 6.7-545) mg/g creatinine (Table 1) and nephropathy was found in 50.9% of patients. In this study, 17% patients with SCD had heart disease and 26.4% had pulmonary hypertension risk. None of the patients had acute chest syndrome (Table 2).
Laboratory Data of the Studied Patients With SCD.

Abbreviations: Hb, hemoglobin; hs-CRP, high-sensitivity C-reactive protein; IQR, interquartile range; SCD, sickle cell disease; SD, standard deviation; UACR, urinary albumin creatinine ratio; WBC, white blood cell.
Serum Cystatin C Levels in Relation to Clinical Characteristics of Patients With Sickle Cell Disease.
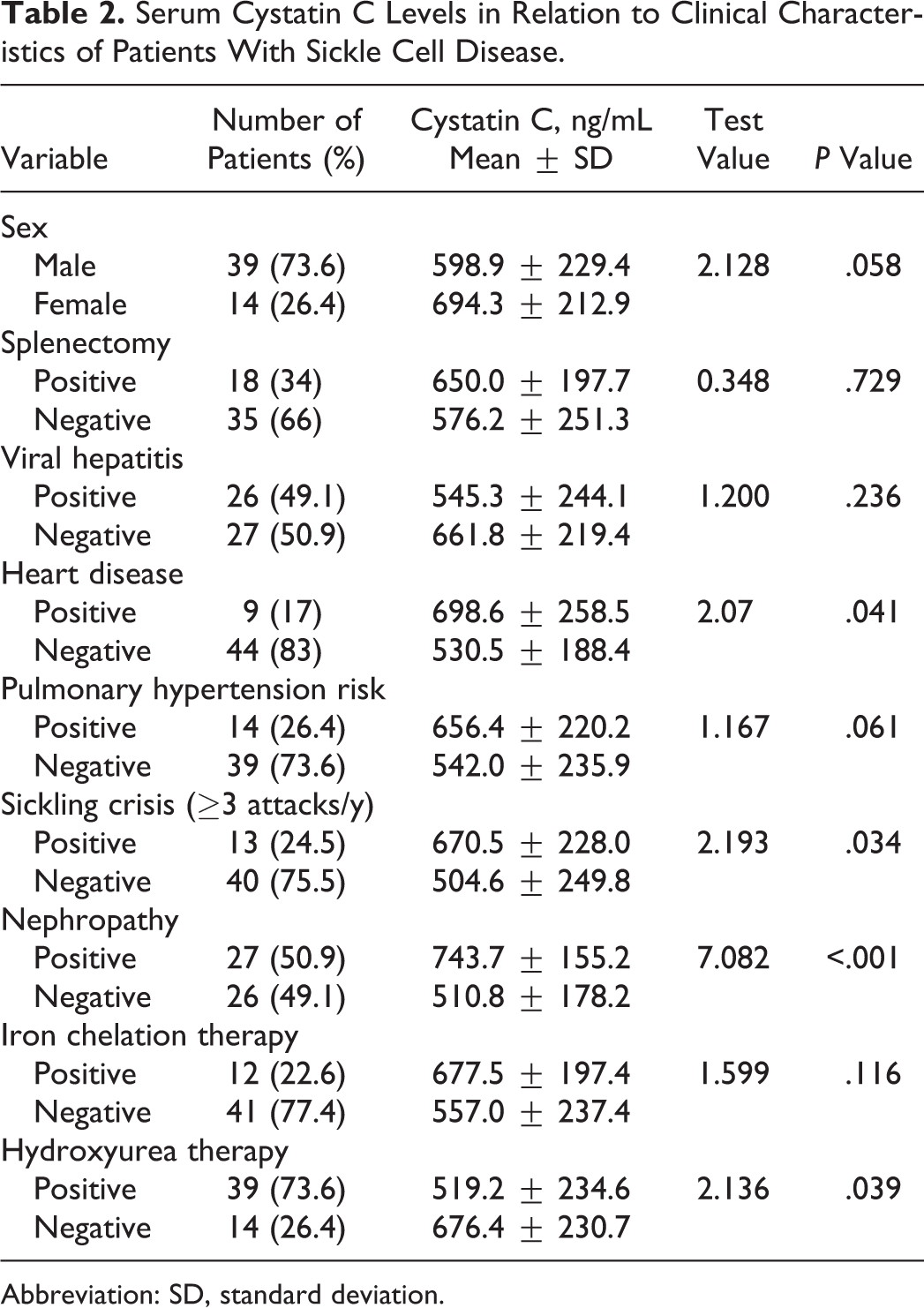
Abbreviation: SD, standard deviation.
Mean serum cystatin C levels in patients with SCD were 624.3 ± 232.8 ng/mL compared to 533.4 ± 35.6 ng/mL in controls (P = .024). Carotid IMT was significantly higher in patients with SCD compared to controls (0.64 ± 0.12 mm vs 0.42 ± 0.02 mm; P < .001; Figure 1).
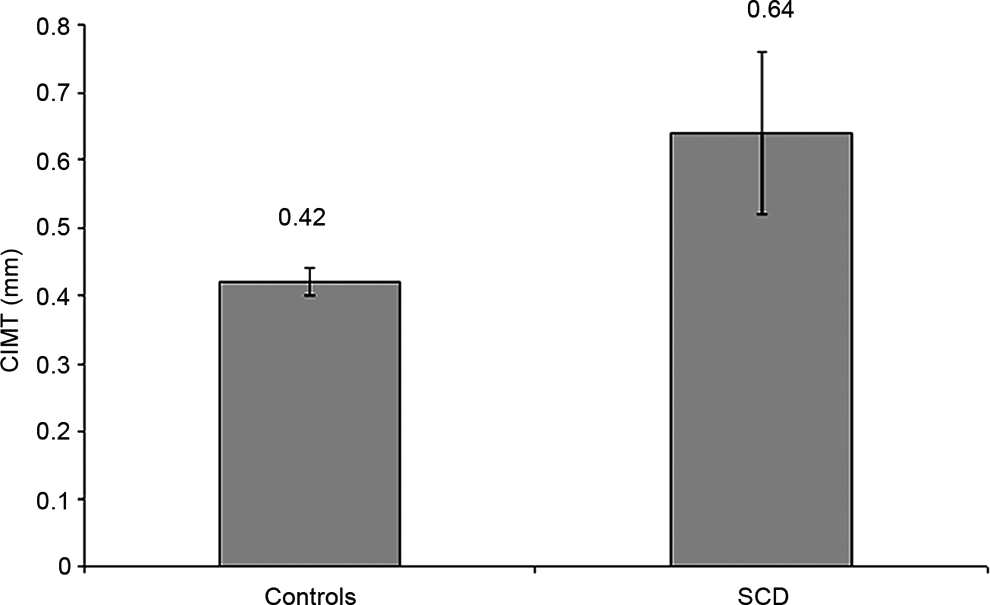
Carotid intima–media thickness in patients with sickle cell disease compared to healthy controls.
As shown in Table 2, patients with nephropathy (P < .001), heart disease (P = .041), or history of frequent sickling crisis (≥3 attacks/y; P = .034) had significantly higher cystatin C levels than those without. Although levels were higher in patients with pulmonary hypertension risk compared to their counterparts, the difference did not reach a significant level (P = .061). Patients with SCD treated with hydroxyurea had lower cystatin C levels than untreated patients (P = .039).
Receiver–operating characteristic curve analysis revealed that the cutoff value of cystatin C at 580 ng/mL could detect nephropathy with 87.8% sensitivity, 84.6% specificity, and AUC: 0.908, P < 0.001, whereas the cutoff value 615 ng/mL could differentiate patients with and without heart disease with 83.2% sensitivity, 81.7% specificity, and AUC: 0.815, P < .001.
Cystatin C levels were positively correlated with WBC count (r = .428; P = .016), hs-CRP (r = .591; P = .006), UACR (r = .677; P < .001), TRV (r = .496; P = .009), and CIMT (r = .688; P < .001; Figure 2), whereas negatively correlated with EF (r = −.614; P < .001) among patients with SCD. No significant relations between cystatin C and other studied variables including LDH or serum ferritin. In multiple linear regression analysis, hs-CRP, UACR, EF, and CIMT were independently related to cystatin C (Table 3).
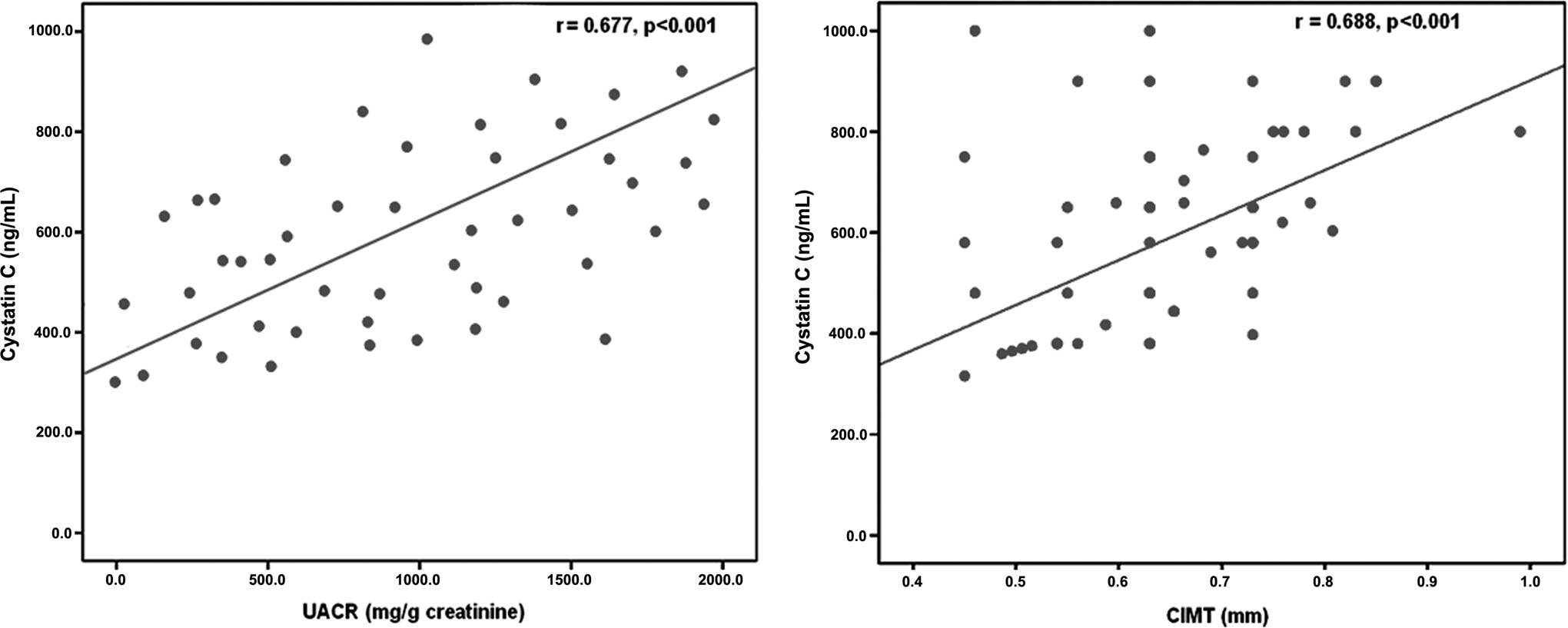
Significant positive correlation between cystatin C levels and urinary albumin creatinine ratio (UACR) and carotid intima–media thickness (CIMT) among patients with sickle cell disease.
Significant Variables Related to Increased Cystatin C Levels in Patients With Sickle Cell Disease Shown by Stepwise Multiple Regression Analysis.
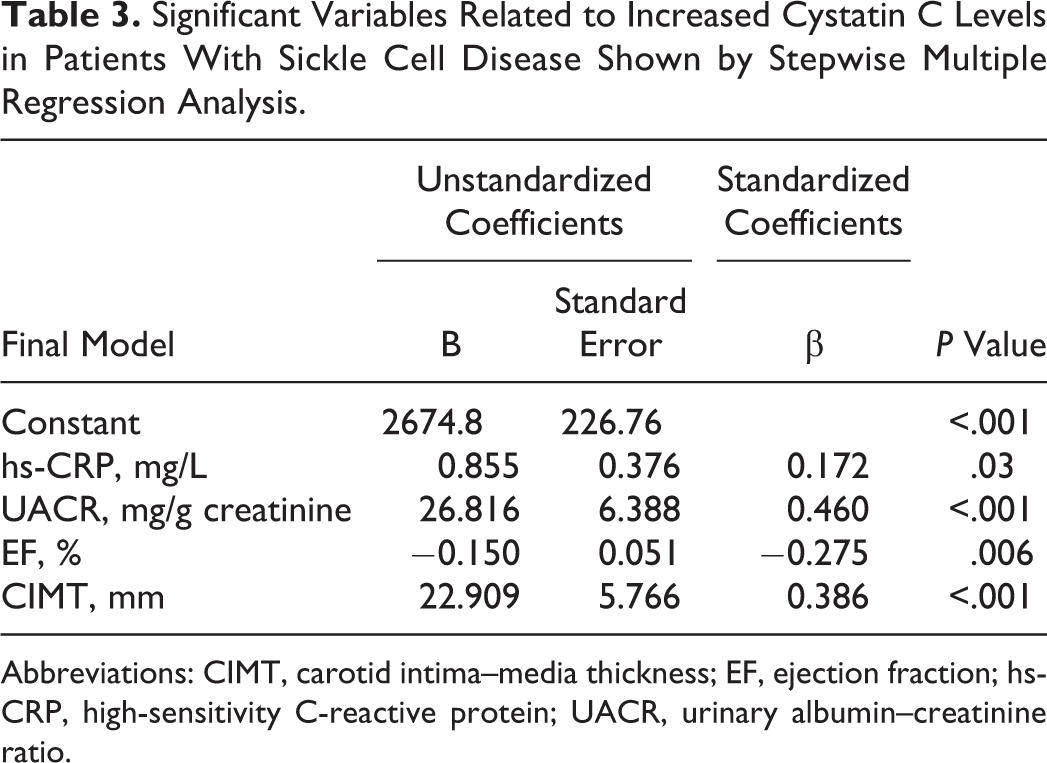
Abbreviations: CIMT, carotid intima–media thickness; EF, ejection fraction; hs-CRP, high-sensitivity C-reactive protein; UACR, urinary albumin–creatinine ratio.
Discussion
Sickle cell disease is as much a disease of endothelial dysfunction as it is a hemoglobinopathy that triggers erythrocyte polymerization. 33 Oxidative stress, chronic endothelial damage, and hemolysis initiate a cascade of events that result in episodic vaso-occlusion, subsequent ischemia–reperfusion injury, and inflammation. 34 Vascular dysfunction is the end result, due to complex and multifactorial interactions that ultimately manifest as the clinical phenotypes of SCD. 35
As renal dysfunction is a leading cause of morbidity in SCD, it is important that clinicians have accurate means of assessing its risk and severity. 18 It is well known that creatinine clearance is generally increased and serum creatinine is low in the sickle cell population due to creatinine secretion by the renal tubules. 36 The early development of glomerular hypertrophy enhances creatinine excretion and gives false normal results. Therefore, the renal dysfunction becomes evident rather late. 37 In this work, serum creatinine was within reference range. Therefore, we assessed UACR, a sensitive and early clinical marker of glomerulopathy, and the evidence of nephropathy was found in 50.9% of patients with SCD that is in line with other studies. 38,39
We found significantly higher serum cystatin C in patients with SCD compared to healthy controls. In particular, patients with nephropathy had higher cystatin levels than those without, and a significant positive correlation was found with UACR. The diagnostic performance of cystatin C in comparison to serum creatinine was analyzed in a meta-analysis of 46 studies, including children and adults. The data compared correlation coefficients between GFR and the reciprocals of serum creatinine and cystatin C in 3703 participants and showed significantly better correlations for cystatin C, suggesting that cystatin is superior to serum creatinine for the detection of impaired GFR in cross-sectional studies. 40 The cystatin C levels of serum and urine increased with increasing degree of albuminuria, reaching higher levels in patients with macroalbuminuria documenting that could be useful markers for renal dysfunction in type 2 diabetic patients with normoalbuminuria. 41
In agreement with our results, Alvarez et al 17 examined the value of serum cystatin C as a marker for GFR in small cohort of 20 children with SCD with and without albuminuria, compared to serum creatinine and creatinine clearance. Mean serum cystatin C was significantly different among the children with macroalbuminuria (n = 4), microalbuminuria (n = 5), and without albuminuria (n = 11; 1.25, 0.84, and 0.78 mg/L, respectively). The mean GFR derived from serum cystatin was significantly different among these subgroups, becoming abnormal in the proteinuric cohort (63 mL/min per 1.73 m2), in contrast to 94 for the microalbuminuric and 103 for the normal subgroups. Serum creatinine or creatinine clearance did not change significantly with the level of albuminuria. The authors concluded that serum cystatin C is higher than serum creatinine in SCD, and this probably relates to the fact that serum cystatin C is not secreted by the kidney, as creatinine. Serum cystatin C correlates with the level of albuminuria and may be a reliable method to measure renal function in SCD. 17 Moreover, Asnani and Reid 18 determined the utility of cystatin C in 98 adults with SCA. Cystatin C was significantly correlated with measured GFR, hemoglobin, serum creatinine, UACR, and systolic blood pressure.
These findings as well as ours suggest that monitoring of serum cystatin C in addition to urine screening for albuminuria may help in the diagnosis of early renal impairment in the patients with SCD before a significant rise in serum creatinine is observed. Receiver–operating characteristic curve analysis revealed that the cutoff value of cystatin C at 580 ng/mL could differentiate patients having SCD with and without nephropathy with 87.8% sensitivity and 84.6% specificity. To the best of our knowledge, no previous studies assessed serum cystatin C cutoff in children with SCD and therefore, further prospective studies are needed to validate this threshold.
On the other hand, Cho et al 42 evaluated the significance of serum cystatin C levels in pediatric patients with chronic kidney disease diagnosed by renal biopsy and who showed normal serum creatinine levels. They found that 95% of the patients showed only slightly increased cystatin C levels from the upper normal limit of the reference range and suggested that mildly increased cystatin C without increased creatinine might not have clinical significance. However, as explained before, serum creatinine is not an accurate measure for renal function in the SCD 36 and their findings may not be applied to our studied patients.
In this study, patients with SCD having heart disease had significantly higher levels than those without and there was a trend toward higher levels among patients with pulmonary hypertension with significant correlations between cystatin C and EF as well as TRV. The relation between serum cystatin C and heart disease among patients with SCD has not been previously explored. However, Ge et al 11 investigated plasma cystatin C in a total of 126 consecutive patients with coronary artery disease (CAD). They found elevated plasma cystatin C in CAD, and it was positively and significantly correlated with age, hs-CRP, WBCs, creatinine, and uric acid, whereas a significantly negative correlation was observed with high-density lipoprotein (HDL) cholesterol. During a 6-month follow-up, 26 patients were found with adverse cardiovascular events and had significantly higher cystatin C levels than the 22 control patients at admission. Thus, cystatin C plays an important role in the development of CAD and is a strong predictor of risk of cardiovascular events. 11
A possible explanation for the predictive value of cystatin C for further cardiovascular events is that, independently from renal dysfunction, elevated serum concentrations of this marker would reflect a defensive response against the augmented secretion of lysosomal cathepsin associated with inflammatory processes related to the atherosclerotic disease. According to this hypothesis, the cystatin C levels should have a pattern similar to that already described for other inflammatory markers such as CRP and interleukin 6 (IL-6) during ACS, that is, increase during acute phase and then return to baseline levels during the stable chronic phase. 12
Whether the relation between elevated serum cystatin C levels and risk of a worse cardiovascular prognosis reflects a more precise measurement of renal function or an association with nonrenal factors such as inflammation is a matter still poorly defined. 12 In a prospective, multicenter study, De Servi et al 43 evaluated the time course of serum cystatin C levels in 222 patients over the first 6 weeks after an episode of non-ST-elevation ACS and a successful percutaneous revascularization. Cystatin C levels slightly but significantly increased from the admission to 6-week samples, contrary to hs-CRP, N-terminal portion of the pro brain natriuretic peptide (NT-proBNP), and IL-6 levels that significantly decreased in the same period. Cystatin C levels were not different in patients with or without elevated troponin (cardiac troponin T [c-TnT]), whereas the other inflammatory and biomechanical markers showed higher values in patients with increased c-TnT versus those without increased c-TnT. Cystatin C was highly correlated with estimated GFR both in ACS and in 6-week samples. Their data seem to contradict the hypothesis that inflammation is a determinant of the cystatin C levels because they did not show the typical pattern of an acute-phase reactant and secondly, they were independent of myocardial necrosis diagnosed by c-TnT levels. They showed that cystatin C remains a reliable marker of renal function also during ACS because it is not influenced by myocardial necrosis or acute left ventricular dysfunction, as detected by increased NT-proBNP values.
In contrast to the findings of De Servi et al, 43 the significant correlation between cystatin C levels and WBCs and hs-CRP among our patients with SCD highlights the role of inflammation in the pathogenesis of vasculopathy in SCD. We also found that patients with SCD who experienced history of frequent sickling crisis (≥3 attacks/y) had higher cystatin C levels than those without. The 2 main pathophysiological processes in the crisis state are ischemia and inflammation. 44 Cystatin C and its fragments may affect the phagocytic and chemotactic ability of neutrophil, participate in the inflammatory process, and regulate inflammatory responses. Inflammation plays an important role in the development of atherosclerosis. 11 Dandana et al 13 showed that cystatin C was correlated with the CAD severity score and associated with biochemical atherosclerosis markers such as hs-CRP and homocysteine.
Furthermore, we found that patients with SCD treated with hydroxyurea had lower cystatin C levels than untreated patients, possibly due to the role of hydroxyurea in decreasing inflammation. The multicenter study of hydroxyurea 45 reported that the decrease in neutrophils, monocytes, reticulocytes, and platelet counts with hydroxyurea therapy were directly associated with decreases in 3-month crisis rates. This further enforces the relation between cystatin C and inflammation in SCD.
However, we could not find a significant association between elevated cystatin C levels and pulmonary hypertension risk among the studied patients with SCD, and cystatin C was weakly correlated with TRV. The pathophysiology of pulmonary hypertension is multifactorial including hemolysis, oxidant stress, inflammatory stress, chronic thromboembolism, in situ thrombosis, parenchymal and vascular injury, iron overload, and asplenia. 46
The oxidative damage caused by hemolysis and iron overload may be the main pathogenic mechanism of alteration of arterial elastic tissue in chronic hemolytic conditions such as SCD. 47 In our study, CIMT was significantly higher in patients with SCD compared to control group and positively correlated with cystatin C. Because there are some common pathophysiological features between atheroma and SCD vasculopathy, Belizna et al 48 assessed carotid arterial stiffness, IMT, and transcranial Doppler ultrasonography in adult patients with SCD and found increased carotid arterial stiffness and transcranial Doppler velocities in patients with SCD having stroke. Hadeed et al 49 assessed the vascular properties of carotid and brachial arteries in children with SCD compared to a control group without cardiovascular risk factors. All patients were symptom free, and endothelial dysfunction risk factors such as hypertension, diabetes, obesity, and hypercholesterolemia were excluded. They reported that children with SCD have no marked endothelial dysfunction or change in arterial stiffness, and these manifestations may be related to disease severity and duration.
The relation between cystatin C and CIMT has been shown in other diseases than SCD. Zhu et al 21 surveyed 927 participants without clinical CVD and chronic kidney disease, and 56.63% of the participants were found to have carotid thickening. Cystatin C was the only measure of kidney function that was significantly associated with carotid thickening and plaque in the adjusted analysis that was performed to select risk factors from sex, age, body mass index, hypertension, diabetes, smoking, total cholesterol, triglyceride, HDL, low-density lipoprotein, apolipoprotein A, apolipoprotein B, cystatin C, creatinine, blood urea nitrogen, blood uric acid, and estimated GFR. Furthermore, in patients with metabolic syndrome and normal estimated GFR, higher cystatin C quartiles were more likely to present with increased CIMT, suggesting the importance of early detection of subclinical atherosclerosis in those patients. The optimal cystatin C cutoff point for detecting elevated CIMT was defined. 50
Limitations of the Study
The relatively small number of patients included is an important limiting factor. However, the results obtained suggest the importance of further larger studies to verify the practical utility of cystatin C measurement in SCD and its potential to predict the clinical severity and disease outcome.
In conclusion, cystatin C may be considered a biological marker for vascular dysfunction and subclinical atherosclerosis in patients with SCD as reflected by positive correlation with CIMT. Serum cystatin C correlates with the level of albuminuria and may be a reliable method to measure renal function in SCD. Elevated cystatin C levels would help to identify patients at risk of cardiac complications. The positive correlation between cystatin C and WBC count strengthens the role of inflammation in the pathogenesis of SCD and the development of atherosclerosis. Low cystatin C levels in patients with hydroxyurea therapy may suggest a potentially valuable tool for monitoring the response to therapy in patients with SCD. Further longitudinal studies are needed to verify whether cystatin C and CIMT should be included in the screening of cardiovascular risk in patients with SCD and validate cystatin C cutoff values for detection of cardiac and renal complications before their incorporation into patients’ risk stratification to guide current therapeutic regimens. The relation between cystatin C and inflammation in SCD represents an interesting area for future research.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
