Abstract
Intracoronary thrombus burden is associated with some adverse events and poor prognosis in patients with ST-segment elevation myocardial infarction (STEMI). Identifying predictors of the intracoronary thrombus burden may contribute to the management of STEMI. In this study, we evaluated whether monocyte count to high-density lipoprotein cholesterol ratio (MHR) is a predictor of intracoronary thrombus burden in patients with STEMI. The study population consisted of 414 patients with STEMI who underwent primary percutaneous coronary intervention (PCI). Angiographic thrombus burden was classified based on thrombolysis in myocardial infarction (TIMI) thrombus grades. The patients were grouped into 2 categories of low thrombus burden and high thrombus burden. The MHR was significantly higher in the high thrombus burden group compared with the low thrombus group (16.0 [9.2-22.1] vs 25.4 [13.5-44.6]; P < .001). In multivariate logistic regression analysis, MHR was an independent predictor of high thrombus burden (odds ratio: 1.067, 95% CI: 1.031-1.105; P < .001). The area under the receiver–operating characteristic curve of the MHR was 0.688 (0.641-0.733; P < .001) to predict high thrombus burden. In conclusion, MHR was independent predictor of high thrombus burden in patients with STEMI who underwent primary PCI.
Introduction
Intracoronary thrombus formation due to atherosclerotic plaque rupture and the interruption of coronary blood flow constitute the main pathophysiology underlying ST-segment elevation myocardial infarction (STEMI). The quantity of the intracoronary thrombus burden is associated with a poor prognosis in patients with STEMI. 1 –3 Intracoronary thrombus management is still complex, although many pharmacological and invasive treatments have been developed, such as glycoprotein IIb/IIIa antagonists and thrombectomy. Identifying predictors of the intracoronary thrombus burden may contribute to the management of STEMI. Recently, the red cell distribution width (RDW) and bilirubin level were shown to predict the thrombus burden in patients with STEMI. 4,5 However, there are no data on the relationship between the monocyte count to high-density lipoprotein cholesterol (HDL-C) ratio (MHR) and thrombus burden.
Monocytes play a major role in immunity by secreting proinflammatory and prooxidant cytokines. These cells are also one of the most important components of atherosclerotic plaque. Monocytosis is an independent risk marker for coronary artery disease (CAD) and a high monocyte count is associated with plaque progression during the acute phase of acute myocardial infarction (AMI). 6,7 In addition, HDL-C has a protective effect against low-density lipoprotein (LDL) oxidation and monocyte activation. 8 –10 Many studies have described the protective effects of serum HDL-C levels in the atherosclerotic process. 11,12 Recently, MHR was reported to be a new cardiovascular prognostic marker in chronic kidney disease. 13 The MHR is also associated with CAD severity and complexity in stable CAD. 14 To our knowledge, however, no study has evaluated the association of MHR with thrombus burden in patients with STEMI. Therefore, this study investigated the relationship between MHR and thrombus burden and estimated the possible thrombotic adverse events in patients with STEMI.
Materials and Methods
Study Population
Consecutive patients (n = 414) who presented with STEMI within the first 12 hours after the onset of pain were enrolled in the study in Gaziosmanpasa University School of Medicine between January 2012 and December 2014. All patients were treated with primary percutaneous coronary intervention (PCI).
The diagnosis of STEMI was made on the basis of the presence of typical chest pain >30 minutes duration, ST-segment elevation >0.1 mV in at least 2 related electrocardiogram leads, or new-onset left bundle branch block. Exclusion criteria were as follows: thrombolytic treatment within 24 hours, any systemic inflammatory disease, any hematological disease including anemia (<12 g/dL for females, <13 g/dL for male), malignancies, renal and/or hepatic failure, recent infection, and history of blood transfusion in the last 3 months. The study protocol was approved by the local ethics committee and informed consent was obtained from all patients.
Cardiovascular risk factors, previous PCI or coronary artery bypass graft, previous medications, and anthropometric characteristics were recorded. Body mass index (BMI; weight (kg)/height square [m2]) was calculated for all patients. Hypertension was defined as systolic blood pressure ≥140 mm Hg and/or diastolic blood pressure ≥90 mm Hg on repeat measurements, or use of any antihypertensive drug. Diabetes mellitus was defined as a fasting plasma glucose level of >126 or >200 mg/dL at any measurement or use of any antidiabetic drug.
Laboratory Measurements
Peripheral venous blood samples of the patients were obtained upon their admission to the hospital. The levels of blood biochemical parameters including baseline lipid panel, glucose, creatinine, creatine kinase, creatine kinase-MB, troponin, and C-reactive protein (CRP) levels were measured.
Samples for the complete blood count (CBC) analysis were collected in EDTA-anticoagulated Monovettew tubes (Sarstedt Monovette, Nuembrecht, Germany). An automated blood cell counter (Beckman Coulter analyzer; Beckman Coulter, California) was used for measuring the CBC parameters including hemoglobin, white blood cell, neutrophil, lymphocyte, monocyte, RDW, platelet counts, and mean platelet volume (MPV).
Primary PCI Procedure
All patients diagnosed with STEMI received 300 mg aspirin and 600 mg clopidogrel loading dose at the hospital admission, and 70 IU/kg unfractionated heparin during PCI processing. Primary PCI was performed to only culprit artery except for cardiogenic shock. The standard Judkins technique (Expo; Boston Scientific Corporation, Natick, Massachusetts, USA) and Siemens Axiom Sensis XP device (Munich, Germany) were used to perform the baseline coronary angiography. The contrast agent used was iopromide (Ultravist-370 Schering AG, Berlin, Germany).
We assessed the thrombus grade after restoring antegrade flow through guidewire or small balloon dilatation. Angiographic thrombus burden was classified as previously defined by the thrombolysis in myocardial infarction (TIMI) study group. 15
Grade 0: no evidence of thrombus, grade 1: suspected thrombus (low contrast density, haziness, irregular lesion contour, or a smooth convex meniscus at the site of occlusion), grade 2: definite thrombus and the thrombus greatest dimension is ≤1/2 vessel diameter, grade 3: definite thrombus and the thrombus greatest dimension >1/2 to <2 vessel diameters, grade 4: definite thrombus and the thrombus greatest dimension >2 vessel diameters, and grade 5: total thrombotic occlusion. The patients were grouped into 2 categories of low thrombus burden (grades 0-3) and high thrombus burden (grades 4 and 5).
Statistical Analysis
The SPSS 21.0 for Windows was used for statistical analyses (SPSS, Chicago, IL). Data were tested for normality using the Kolmogorov-Smirnov test. Normally distributed continuous data were expressed as mean ± standard deviation; nonnormally distributed continuous variables were presented as median and interquartile range (quartiles 1–3). In statistical analysis for numeric variables, Student’s t test or Mann-Whitney U test was used, and for categorical data chi-square test was used. Correlations between variables were tested by Pearson or Spearman correlation tests where appropriate. Multivariate logistic regression was used to identify the independent predictors of thrombus burden. Receiver–operating characteristic curve was also used to demonstrate the sensitivity and specificity of MHR and the optimal cutoff value for predicting thrombus burden. A value of P < .05 was considered statistically significant.
Results
A total of 414 patients with STEMI were included in the study (mean age 62.6 ± 12.8 years; 76% male). According to the basic clinical and demographic characteristics, both study groups were similar with regard to age, sex, BMI, diabetes mellitus, hypertension, dyslipidemia, previous history of CAD, family history of CAD, and smoking habits (P = .521, P = .132, P = .233, P = .414, P = .299, P = .390, P = .071, P = .819, and P = .688, respectively). Also, no significant differences were observed in systolic or diastolic blood pressures between the 2 groups (P = .112, P = .186, respectively). Previous medications, including aspirin, β-blocker, statin, angiotensin-converting enzyme (ACE) inhibitor or angiotensin receptor blockers (ARB), and calcium channel blocker were similar between groups (P = .711, P = .849, P = .540, P = .911, and P = .732, respectively). Infarct-related arteries were not significantly different between the groups. Postprocedural TIMI flow was low and tirofiban administration was high in the high thrombus burden group as expected (P = .001, P < .001, respectively). The demographical, clinical, and angiographic characteristics of the individuals are shown in Table 1.
Baseline Clinical and Angiographic Characteristics of the Study Population.a
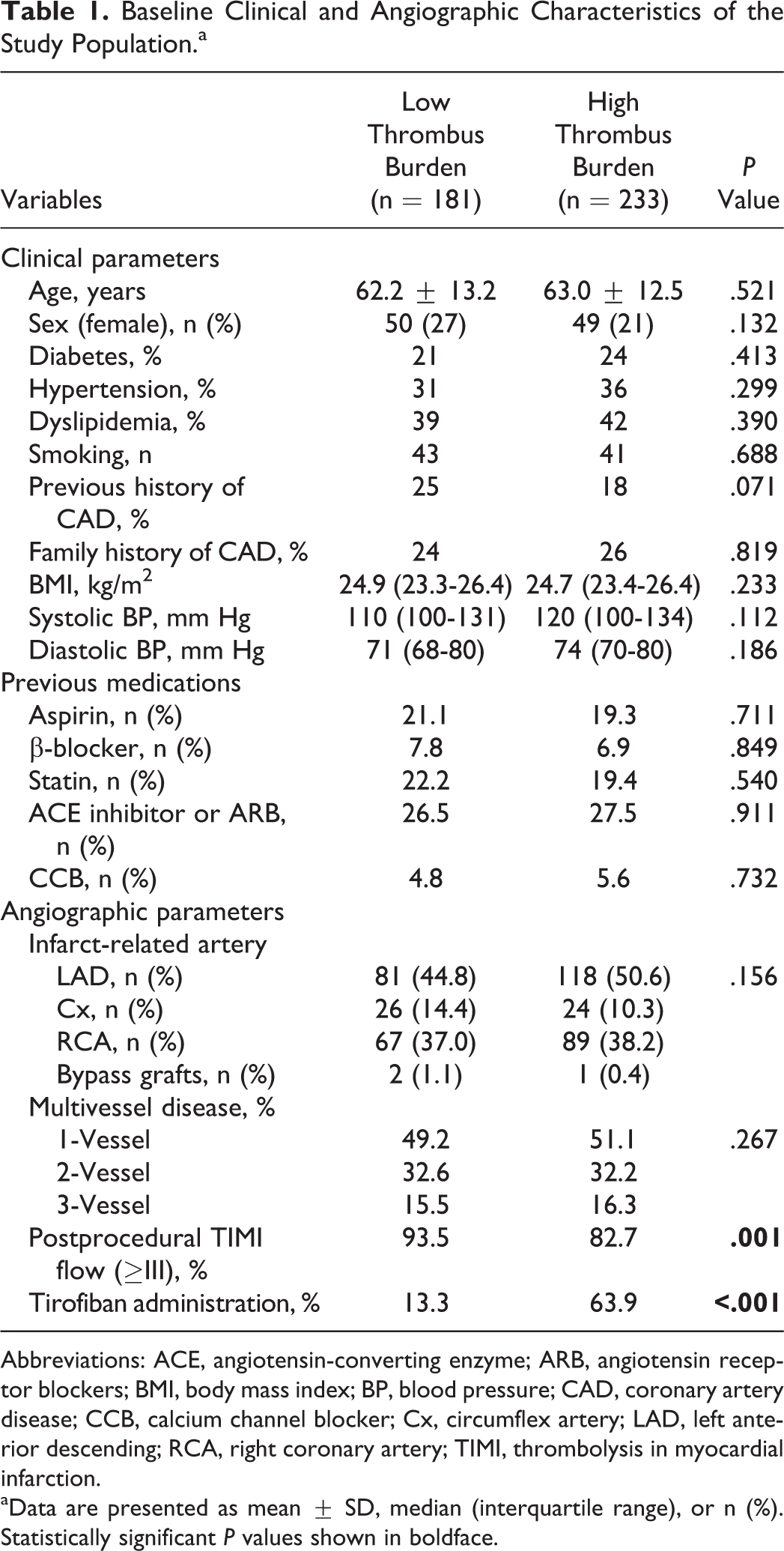
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blockers; BMI, body mass index; BP, blood pressure; CAD, coronary artery disease; CCB, calcium channel blocker; Cx, circumflex artery; LAD, left anterior descending; RCA, right coronary artery; TIMI, thrombolysis in myocardial infarction.
aData are presented as mean ± SD, median (interquartile range), or n (%). Statistically significant P values shown in boldface.
Laboratory characteristics of the individuals are shown in Table 2. Hemoglobin, neutrophil count, lymphocyte count, platelet count, MPV, and neutrophil–lymphocyte ratio were similar between groups (P = .222, P = .054, P = .769, P = .341, P = .058, and P = .364, respectively). White blood cell count (10.7 [8.8-12.9] vs 11.4 [9.6-14.6]; P = .002), monocyte count (0.60 [0.41-0.94] vs 0.93 [0.52-1.40]; P < .001), MHR (16.0 [9.2-22.1] vs 25.4 [13.5-44.6]; P < .001; Figure 1), RDW (15.2 [14.1-16.6] vs 16.0 [15.0-16.8]; P = .004) were significantly higher in the high thrombus burden group when compared with the low thrombus burden group. Total cholesterol, triglyceride, LDL, and creatinine levels were not different between the groups (P = .060, P = .077, P = .069, and P = .315, respectively). Baseline troponin (1.1 [0.2-2.3] vs 1.4 [0.8-4.4]; P < .001) and baseline CK-MB (37 [23-71] vs 39 [31-65]; P = .038) were significantly higher, and HDL-C (40 [33-49] vs 36 [31-43]; P = .004) was significantly lower in the high thrombus burden group when compared with the low thrombus burden group. In correlation analysis, there was a significantly positive correlation between MHR and CRP value (r = .233, P < .001).
Laboratory Characteristics According to Thrombus Burden.a
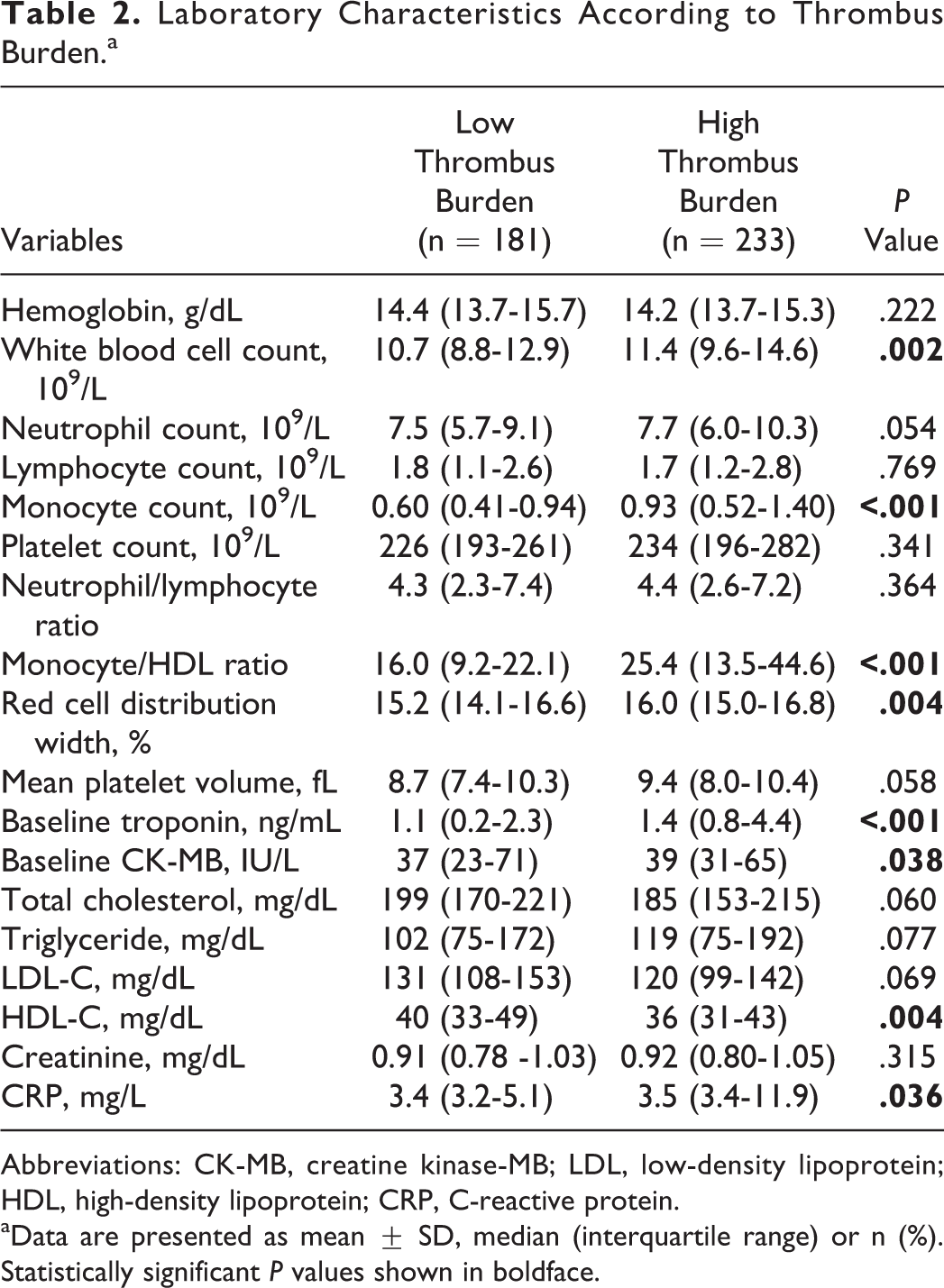
Abbreviations: CK-MB, creatine kinase-MB; LDL, low-density lipoprotein; HDL, high-density lipoprotein; CRP, C-reactive protein.
aData are presented as mean ± SD, median (interquartile range) or n (%). Statistically significant P values shown in boldface.
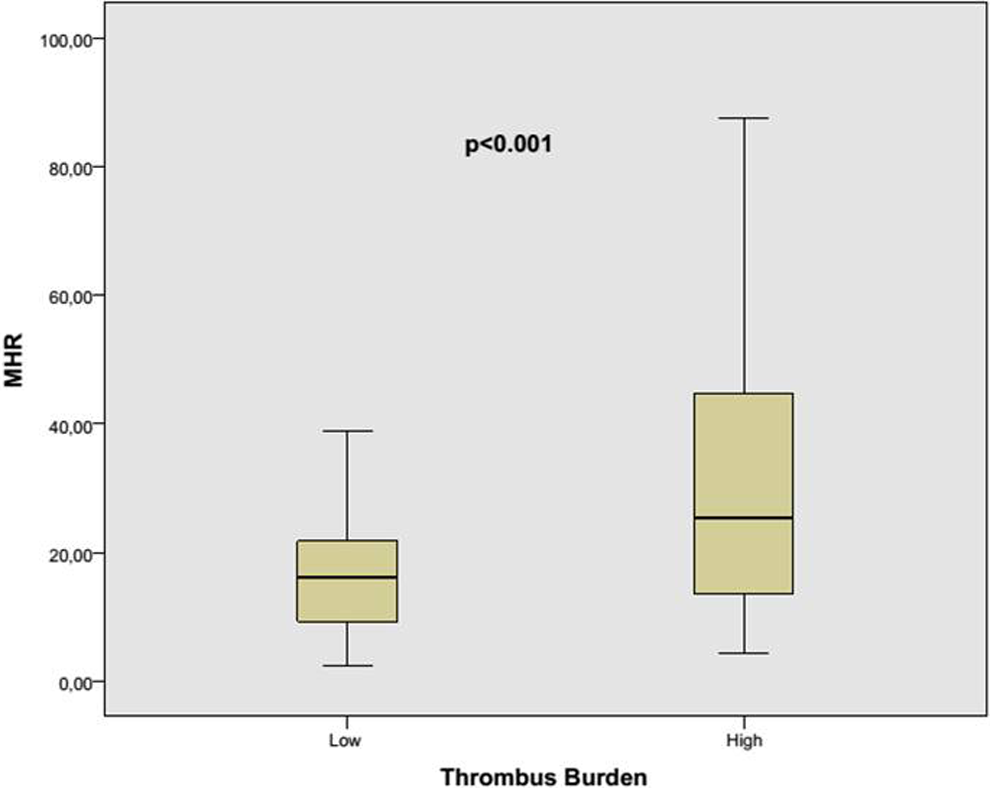
Monocyte count to high-density lipoprotein cholesterol ratio (MHR) values according to thrombolysis in myocardial infarction (TIMI) thrombus burden.
The predictors of high thrombus burden were studied by multivariate logistic regression analysis. The MHR (odds ratio: 1.067, 95% CI: 1.031-1.105; P < .001) and baseline troponin level (odds ratio: 1.179, 95% CI: 1.036-1.342, P = .013) were found as independent predictors of high thrombus burden (Table 3). The cutoff value of MHR was 19.7 with a sensitivity of 60.5% and specificity of 69.6% (AUC, 0.688; 95% CI: 0.641-0.733; P < .001; Figure 2).
Multivariate Logistic Regression Analyses to Detect the Independent Predictors of High TIMI Thrombus Burden.
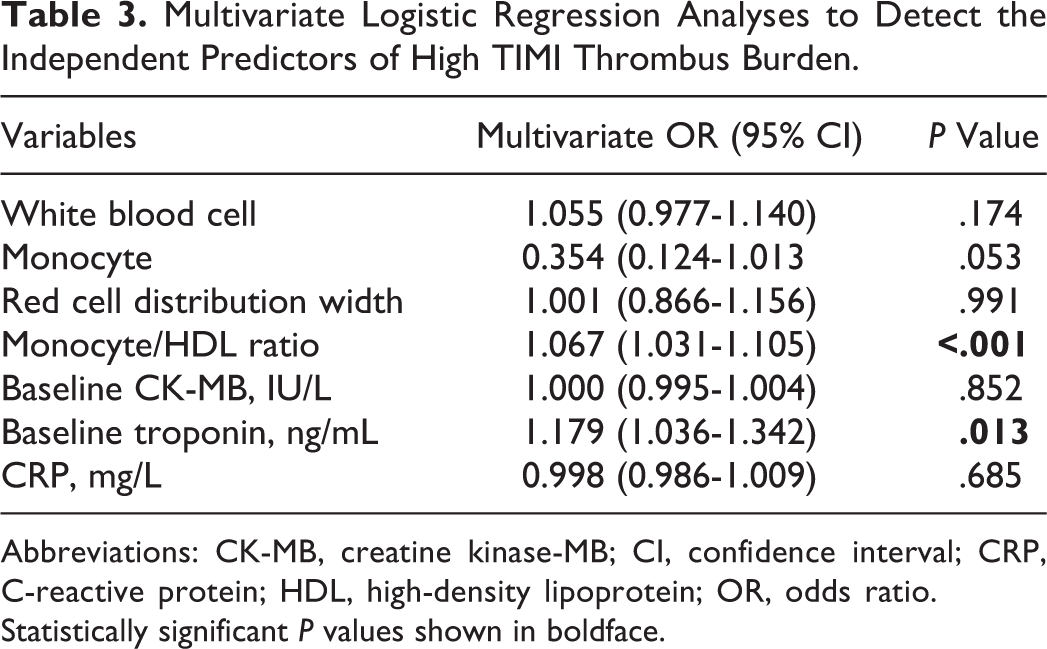
Abbreviations: CK-MB, creatine kinase-MB; CI, confidence interval; CRP, C-reactive protein; HDL, high-density lipoprotein; OR, odds ratio.
Statistically significant P values shown in boldface.
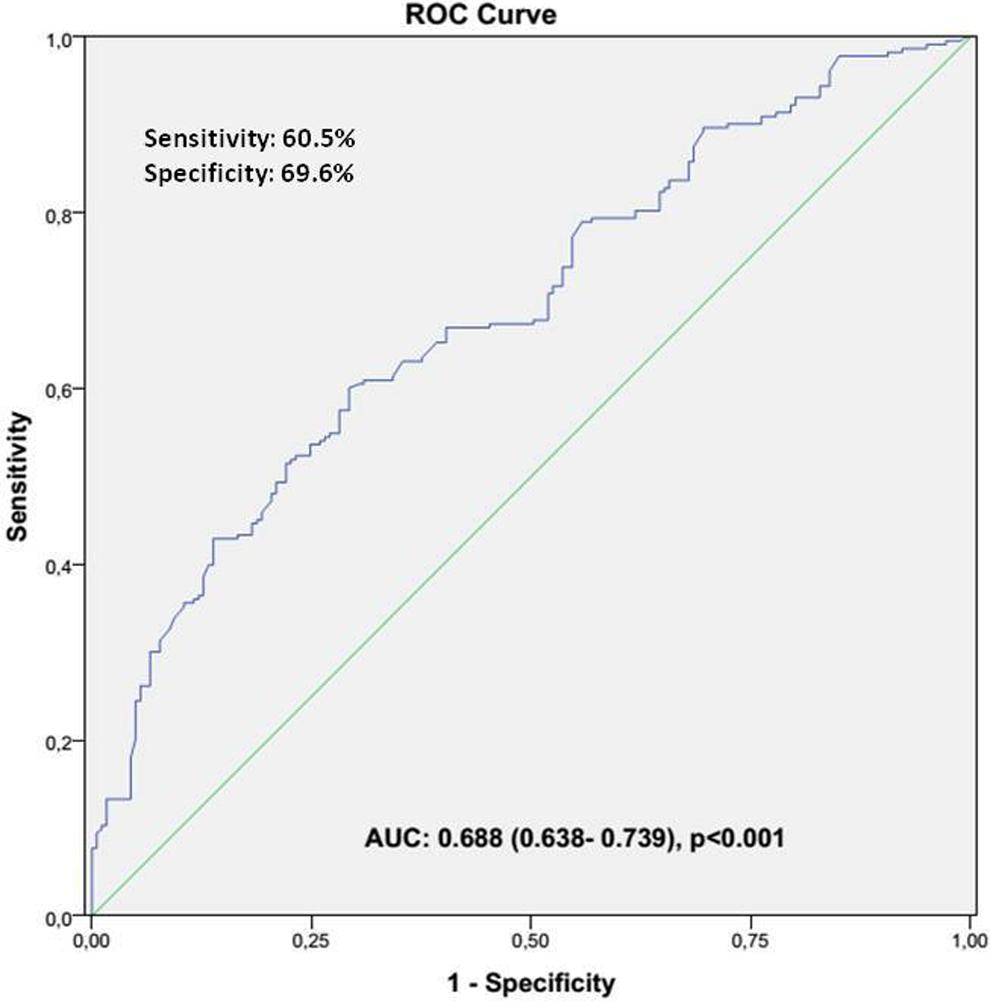
The receiver–operating characteristic (ROC) curve of monocyte count to high-density lipoprotein cholesterol ratio (MHR) for predicting of high thrombolysis in myocardial infarction (TIMI) thrombus burden.
Discussion
Our study revealed that MHR was an independent predictor of a high thrombus burden in patients with STEMI who underwent primary PCI. We also found that a high thrombus burden is associated with low postprocedural TIMI flow and higher baseline troponin and CK-MB levels.
Thrombotic occlusion of a coronary artery due to atherosclerotic plaque rupture is the main pathophysiological cause of STEMI. The size of the intracoronary thrombus is associated with some adverse events, such as no reflow phenomenon, distal embolization, and lower procedural success. 16 A higher thrombus burden is associated with greater 1-year mortality and a higher rate of stent thrombosis. 1 Novel developments such as glycoprotein IIb/IIIa antagonists and thrombectomy reduce the thrombus burden and increase myocardial perfusion. 17 Nevertheless, the management of STEMI with a high thrombus burden is still difficult.
Inflammation and oxidative stress play important roles in atherosclerosis. 18 Monocytes, which play substantial roles in immunity, activation, and cholesterol oxidation, are important milestones in the process. When activated monocytes interact with damaged endothelium, they secrete proinflammatory cytokines and adhesion molecules. Monocytes migrate to the subendothelium and differentiate into macrophages. The macrophage phagocytose oxidized lipoprotein particles and form foamy cells. 19 Therefore, the monocyte count in the circulation may be directly related to the development and severity of atherosclerotic disease. For example, Afiune et al determined that the monocyte count is an independent risk marker for CAD and AMI. 7 Olivares et al found that a high monocyte count was strongly associated with the risk of coronary heart disease in middle-aged men. 20 Nozawa et al reported that a high monocyte count was associated with atherosclerotic plaque progression in patients with STEMI. 6 Similarly, we found that the circulating monocyte count was higher in the high thrombus burden group compared with the low thrombus group in patients with STEMI.
The HDL-C has cardioprotective effects and many studies have shown that HDL-C improves endothelial function via its anti-inflammatory and antioxidative effects. In addition, HDL-C modulates monocyte activation, adhesion, and transmigration. 21,22 Navab et al demonstrated that HDL-C inhibits monocyte transmigration to the subendothelium. 23 Murphy et al found that HDL-C could prevent and reverse monocyte activation. 24 Therefore, the circulating HDL-C level and its relationship with the monocyte count are important for atherosclerotic disease development, progression, and severity. In light of these studies, we hypothesized that the monocyte count and MHR may be related to the intracoronary thrombus burden in STEMI patients.
Kanbay et al identified the MHR as a novel inflammatory and oxidative marker; they found that MHR is an independent predictor of cardiovascular events and is associated with a poor cardiovascular prognosis in patients with chronic kidney disease. 13 Kundi et al reported that MHR is significantly associated with CAD severity and found that the MHR was significantly higher in patients with stable CAD with high Synergy Between Percutaneous Coronary Intervention With TAXUS and Cardiac Surgery (SYNTAX) scores. 14 It was demonstrated that the MHR is significantly related to the presence of isolated coronary artery ectasia and coronary slow flow. 25,26 Çiçek et al demonstrated that the MHR is significantly associated with the short- and long-term mortality in STEMI patients who undergo primary PCI. 27 Recently, Cetin et al reported that the MHR is an independent predictor of stent thrombosis. 28 Similar to the previous studies, we found that the MHR was significantly higher in the high thrombus burden group compared with the low thrombus group in patients with STEMI who underwent primary PCI. We also found that MHR is an independent predictor of a high thrombus burden. These results can be explained by the increased inflammatory cell content in the circulation and low antioxidant effects of HDL-C in these patients. In addition, the correlation between MHR and CRP in our study supports its role in systemic inflammation.
This study has several limitations. Its primary limitations are that it was a single-center study with a small study population. Another limitation is that we analyzed only the admission MHR, and the MHR may change after the acute phase of STEMI. We also assessed the thrombus burden visually, and a quantitative method would have been better.
In conclusion, the MHR, which is a simple, inexpensive marker, can help to predict the intracoronary thrombus burden. Using the MHR, we can detect high-risk patients for a high intracoronary thrombus burden and modify our treatment to deal with possible adverse events related to the high thrombus burden in patients with STEMI. However, the MHR alone is not sufficient for the detection of a high thrombus burden due to its relatively low sensitivity and specificity, but it could complement other predictors, if needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
