Abstract
Background
The anticoagulation and risk factors in atrial fibrillation (ATRIA) score is associated with adverse cardiovascular events. However, its relationship with coronary thrombus burden is unclear. Therefore, we aimed to investigate the relationship between the ATRIA score and thrombus burden in patients with ST-segment elevation myocardial infarction (STEMI) who underwent percutaneous coronary intervention (PCI).
Materials and Methods
The study was designed as a prospective cross-sectional observational study. Our study included 319 patients who were prospectively admitted with STEMI between January 2021 and April 2022. Patients were divided into 2 groups with low thrombus burden (LTB) (grade <3) and high thrombus burden (HTB) (grade ≥3). ATRIA score was calculated and recorded for all patients. ATRIA scores of both groups were compared.
Results
In our study, 58.9% (
Conclusion
In this study, we found that thrombus burden may be associated with ATRIA risk score in patients presenting with STEMI.
Introduction
Coronary artery occlusion due to atherosclerotic plaque rupture and intracoronary thrombus formation are the main pathophysiological mechanisms of ST-segment elevation myocardial infarction (STEMI). 1 High thrombus burden (HTB) in the coronary artery (infarct-related artery [IRA]) responsible for STEMI is associated with distal embolism, stent thrombosis, no-reflow phenomenon, and larger infarct size as well as a significant determinant of long-term cardiovascular mortality and morbidity. The level of intracoronary thrombus burden detected during primary percutaneous coronary intervention (PCI) in STEMI patients has a central role in the follow-up and management of these patients.2–6 Despite improved mechanical and pharmacologic treatment procedures, the clinical management of intracoronary thrombus burden remains complex. Therefore, the prediction of thrombus burden in STEMI patients is critical for optimizing the treatment process and improving clinical outcomes. 7
The anticoagulation and risk factors in atrial fibrillation (ATRIA) risk score is a practical scoring system used to predict the risk of thromboembolism in patients with atrial fibrillation (AF), which is inexpensive and easy to apply. 8 Moreover, the predictive efficiency of the ATRIA score for thromboembolism in AF patients has been shown to be better than traditional scoring systems such as CHADS2 and CHA2DS2-VASc.8–10 Each of the parameters in the ATRIA risk scoring system is generally associated with thromboembolic events. Recent studies have shown that the ATRIA risk score is associated with adverse clinical outcomes in patients with acute coronary syndrome (ACS).11–13
This study aimed to evaluate the effectiveness of the ATRIA score in predicting intracoronary thrombus burden in STEMI patients.
Material and Methods
Study Population and Clinical Evaluations
This study was designed as a prospective, cross-sectional, and observational study. A total of 500 consecutive patients admitted to the emergency department of Düzce University Training and Research Hospital between January 2021 and April 2022 who underwent primary PCI due to STEMI diagnosis were prospectively evaluated. The cardiology clinic where the study was conducted serves as a high-volume tertiary cardiac center in the Western Black Sea Region (>2000 PCI per year).
Exclusion criteria were: previous coronary artery bypass graft (CABG) surgery, stent thrombosis or restenosis, thrombolytic therapy within the last 48 h, presence of active infection including COVID-19, presence of preprocedural AF, recent major surgical procedures or major trauma, presence of known chronic inflammatory disease, history of malignancy, presence of myeloproliferative disease, end-stage liver failure, receiving any antithrombotic therapy and refusal to participate in the study. No upper or lower age limit was used. A total of 500 patients were evaluated as part of the study. One hundred and twenty-seven patients were excluded because they fulfilled at least one of the exclusion criteria. Fifty-four patients were excluded from the study because of inaccurate laboratory results and incorrect or missing data recording. Ultimately, 319 patients were included in the study. The study was approved by the Institutional Ethics Committee.
Patients with STEMI who presented within the first 12 h after the onset of typical chest pain and had diagnostic electrocardiography (ECG) findings were included in the study. Diagnostic ECG findings were as follows: (i) presence of ST-segment elevation in at least 2 consecutive leads, (ii) ST-segment elevation in leads V2–V3 ≥ 2.5 mm in men younger than 40 years, ≥2 mm in men 40 years and older, (iii) ≥1.5 mm in women of all ages, and/or (iv) ≥1 mm in other leads. In addition, (iv) ST-segment elevation >0.5 mm in leads V7–V9 with isolated ST-segment depression when the terminal T wave was positive in leads V1–V3, or (v) the presence of newly developed left bundle branch block.
After a detailed medical history and physical examination, all patients’ age, gender, history of hypertension (HT), diabetes, dyslipidemia, smoking status, history of ischemic or hemorrhagic stroke, previous PCI or CABG, family history of coronary artery disease (CAD), and body mass index (BMI) were noted. Additionally, blood pressure, heart rate, Killip class, and the presence of cardiogenic shock were recorded. Antecubital venous blood samples were taken from all patients included in the study at the time of admission to the emergency department. Cardiac biomarkers, complete lipid profile, high-sensitivity C-reactive protein (hsCRP), complete blood count, urea, creatinine, and hepatic aminotransferases were analyzed. Glomerular filtration rate (GFR) was calculated using the Cockcroft–Gault equation. Periprocedural factors include an IRA, time of onset of chest pain, time of the first presentation to the emergency department, duration of chest pain, time of coronary balloon inflation, stent diameter, length, and characteristics were noted. All patients were evaluated with transthoracic echocardiography at the 48th hour after PCI. Left ventricular ejection fraction (LVEF) was calculated using the modified biplane Simpson's method.
Scores
The ATRIA score was developed from the ATRIA study cohort. It was calculated by adding 1 point for each of the following factors: female sex, diabetes mellitus (DM), congestive heart failure, HT, proteinuria, and renal dysfunction (ie, estimated GFR <45 mL/min/1.73 m2 or end-stage renal disease) and by adding 0 to 9 points depending on the specific score weighting of patients age according to the presence or absence of prior ischemic stroke. 8 All parameters of scoring are summarized in Table 1. The CHA2DS2-VASc risk score was calculated by assigning a score of 1 point for each of the following conditions: congestive heart failure (ejection fraction < 40%), HT, age between 65 and 74 years, DM, vascular disease (myocardial infarction or peripheral arterial disease), and female gender; a score of 2 points for the following conditions: history of stroke or transient ischemic attack (TIA) and age >75 years. 14
ATRIA Score.
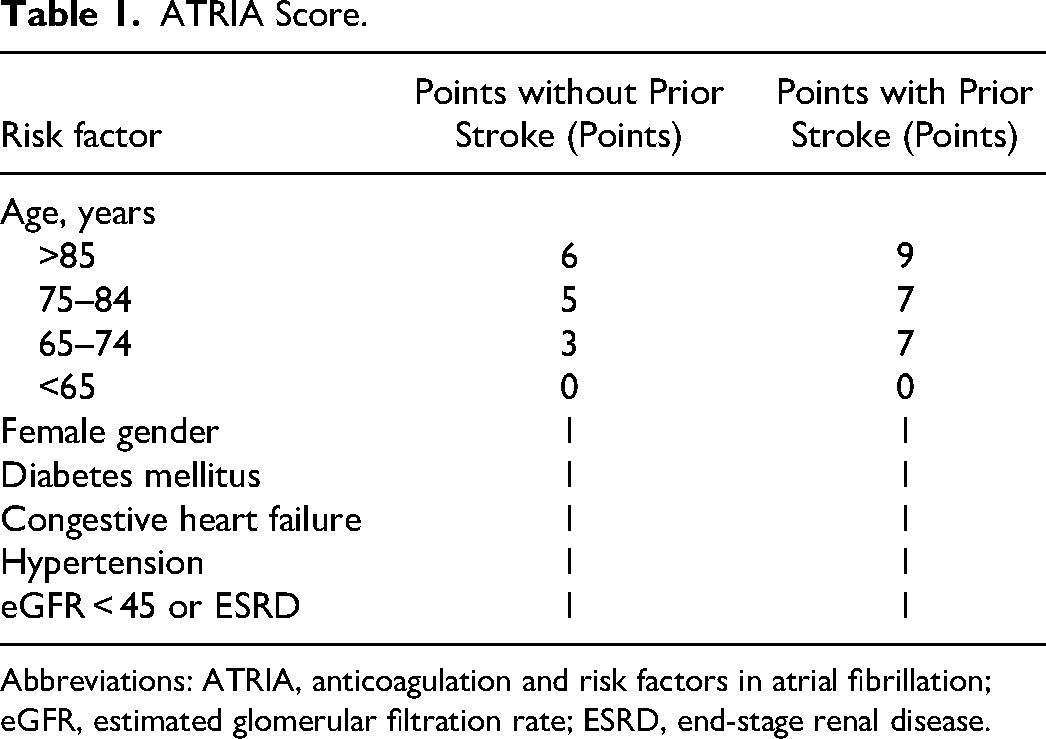
Abbreviations: ATRIA, anticoagulation and risk factors in atrial fibrillation; eGFR, estimated glomerular filtration rate; ESRD, end-stage renal disease.
Angiographic Analyses
Coronary angiography (CAG) via radial or femoral access was performed for each patient. Iodiksanol (Visipaque®) was used as a contrast agent during CAG in all subjects. All patients were given oral aspirin (300 mg) and oral clopidogrel (600 mg) or ticagrelor (180 mg) at the time of admission and intravenous heparin 100 IU/kg just before catheterization. Abciximab was routinely used in our center as a glycoprotein (Gp) IIb/IIIa receptor blocker. Balloon predilatation was not performed in cases suitable for direct stenting. In other cases, stenting was performed after balloon predilatation. Whenever possible, drug-eluting stents were preferred to bare metal stents. Glycoprotein IIb/IIIa receptor blocker use, balloon, and stent selection were left to the operator's discretion. A coronary thrombus aspiration catheter is not used in our center. We did not administer thrombolytic therapy to any patient. Patients referred to our center after thrombolysis were taken directly to the angiography laboratory without waiting time and underwent primary PCI. Patients who had received thrombolytic therapy were not included in the study. Angiography images were evaluated by 2 different cardiologists who had no knowledge of the patients’ clinical data. Thrombus burden in the infarct-related lesion was calculated in five grades based on the classification thrombolysis in myocardial infarction (TIMI) study group. 15 Thrombus burden was classified as: grade 0, no thrombus; grade 1, possible thrombus; grade 2, the greatest dimension of the thrombus is <1/2 vessel diameter; grade 3, largest dimension >1/2 to <2 vessel diameter; grade 4, greatest dimension >2 vessel diameter; grade 5, total vascular occlusion due to thrombus. Grade 5 patients were reclassified after success of flow with guidewire passage or small balloon passage or dilation. 16 Then, final thrombus grades were divided into 2 classes: low thrombus burden (LTB) (grade <3) and HTB (grade ≥3). 17 An additional subgroup analysis of thrombus grade ≥4 or <4 was also performed. Flow in the post-PCI vessel was evaluated with the TIMI flow grade before and after the procedure.18,19 Despite successful dilation and the absence of mechanical complications after completion of the procedure, blood flow in the IRA was considered no-reflow if TIMI ≤2 flow.18,19 The complexity of CAD was evaluated using the Synergy between PCI with Taxus and Cardiac Surgery (SYNTAX) score, which was prospectively calculated using the SYNTAX score algorithm on the baseline diagnostic angiogram. 20 The current online version was used in the calculation of the SYNTAX scores. 21 The Global Registry of Acute Coronary Events (GRACE) score (derived from the GRACE registry) was calculated by using the online calculator during hospital admission. 22 Target vessel diameter and characteristics of the lesion, TIMI frame count, thrombus burden grade, preprocedural TIMI flow rate, postprocedural TIMI flow rate, SYNTAX score, and the procedure used were recorded. Angiography images of the study patients were evaluated by 2 experienced interventional cardiologists who were unaware of the patient's laboratory measurements, clinical status, and ATRIA score. A third cardiologist was consulted when there was inconsistency between the 2 observers.
Statistical Analyses
Mean, standard deviation, median minimum, maximum, frequency, and ratio values were used in the descriptive statistics of the data. The distribution of variables was measured with the Kolmogorov–Smirnov test. Independent sample
Results
The mean age of the patients was 66.2 ± 9.6 years and 234 (73.3%) of them were male. The HTB group consisted of 131 patients (mean age 66.2 ± 12.8 years,
Baseline Characteristics and Laboratory Results of Study Groups.
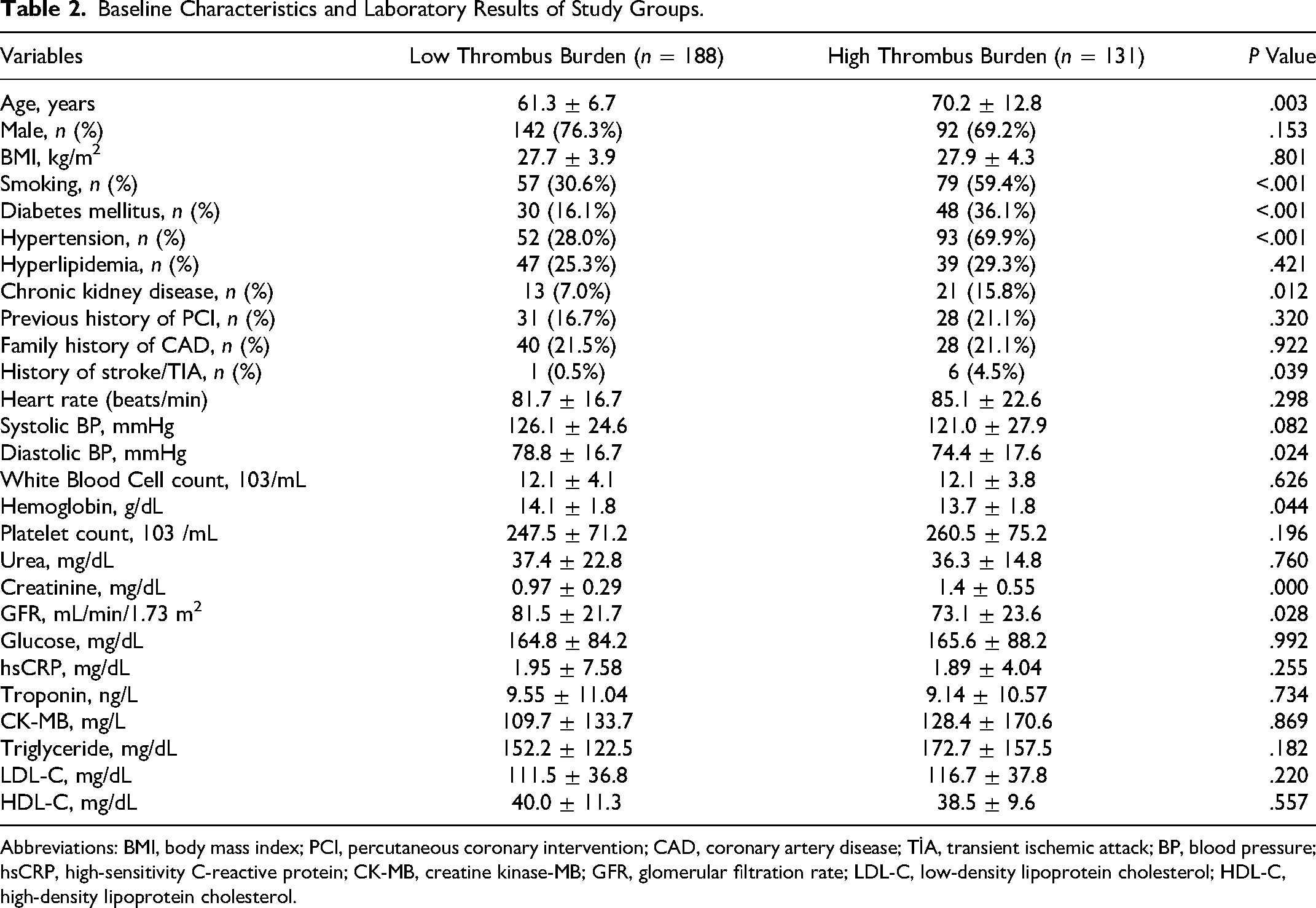
Abbreviations: BMI, body mass index; PCI, percutaneous coronary intervention; CAD, coronary artery disease; TİA, transient ischemic attack; BP, blood pressure; hsCRP, high-sensitivity C-reactive protein; CK-MB, creatine kinase-MB; GFR, glomerular filtration rate; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.
Study groups were similar with regard to age, sex, BMI, dyslipidemia, previous history of PCI, and family history of CAD (
A comparison of the baseline angiographic and procedural characteristics of the two groups is shown in Table 3. There was no significant difference between both groups' infarct-related artery and periprocedural cardiac rhythm. There was no significant difference between both groups' door-balloon time, radial approach, stent diameter, stent length, anterior myocardial infarction, balloon predilatation, TIMI before intervention, and LVEF at 48th hour (
Angiographic and Procedural Characteristics of Study Groups.
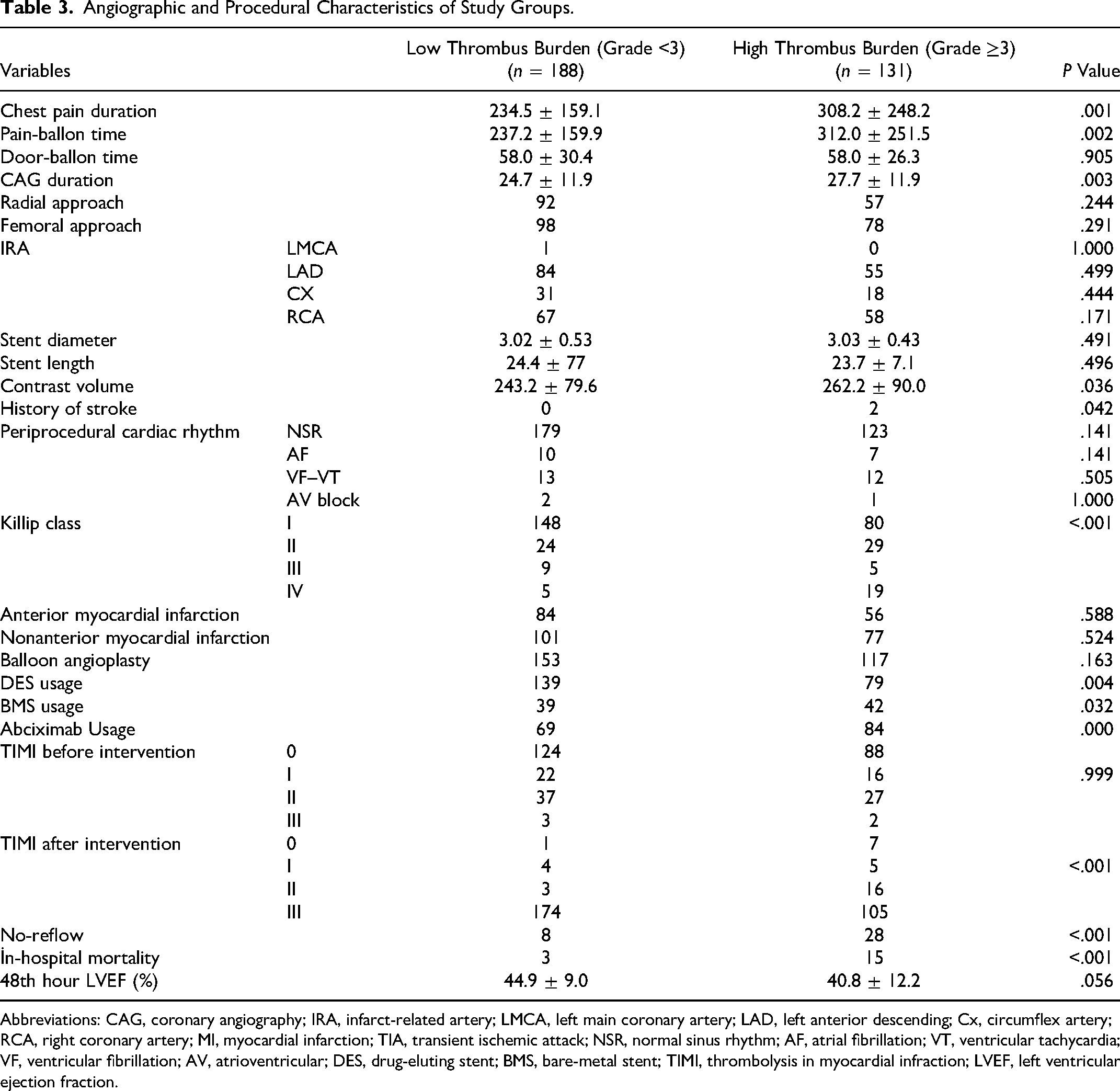
Abbreviations: CAG, coronary angiography; IRA, infarct-related artery; LMCA, left main coronary artery; LAD, left anterior descending; Cx, circumflex artery; RCA, right coronary artery; MI, myocardial infarction; TIA, transient ischemic attack; NSR, normal sinus rhythm; AF, atrial fibrillation; VT, ventricular tachycardia; VF, ventricular fibrillation; AV, atrioventricular; DES, drug-eluting stent; BMS, bare-metal stent; TIMI, thrombolysis in myocardial infraction; LVEF, left ventricular ejection fraction.
Patients with high-thrombus burden had higher smoking (
The ATRIA risk score (
Clinical Scores.

Abbreviations: ATRIA, anticoagulation and risk factors in atrial fibrillation; GRACE, Global Registry of Acute Coronary Events.
In multivariate logistic regression analysis, ATRIA score (odds ratio [OR]: 1.672; 95% confidence interval [CI]: 1.460–1.914;
Univariate and Multivariate Predictors of Coronary Thrombus Burden in Patients with STEMI.
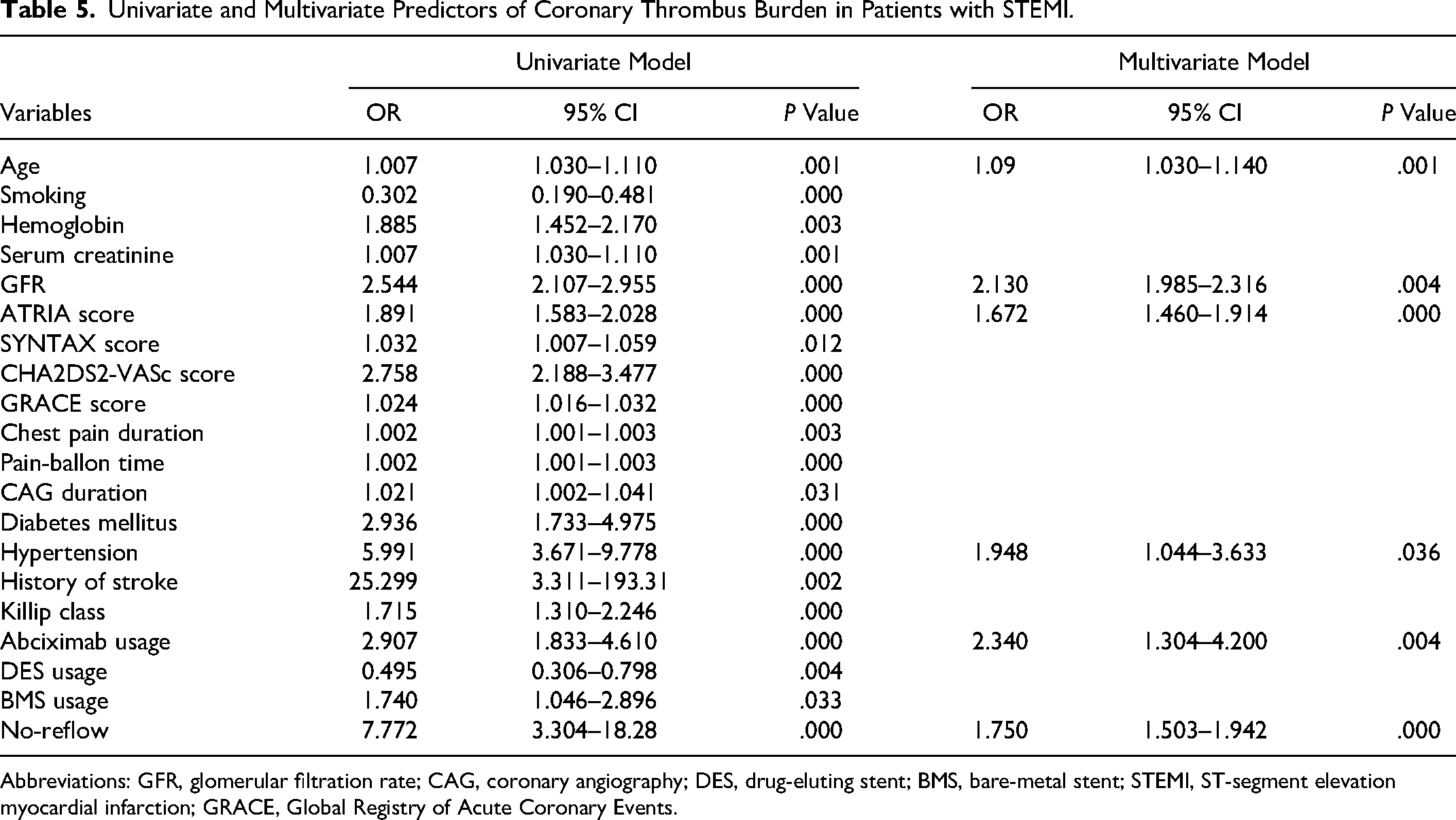
Abbreviations: GFR, glomerular filtration rate; CAG, coronary angiography; DES, drug-eluting stent; BMS, bare-metal stent; STEMI, ST-segment elevation myocardial infarction; GRACE, Global Registry of Acute Coronary Events.
The ability of the ATRIA score to predict HTB was evaluated by ROC curve analysis. The area under curve (AUC) value of this analysis is presented in Figure 1 (AUC = 0.836 (0.788–0.884,
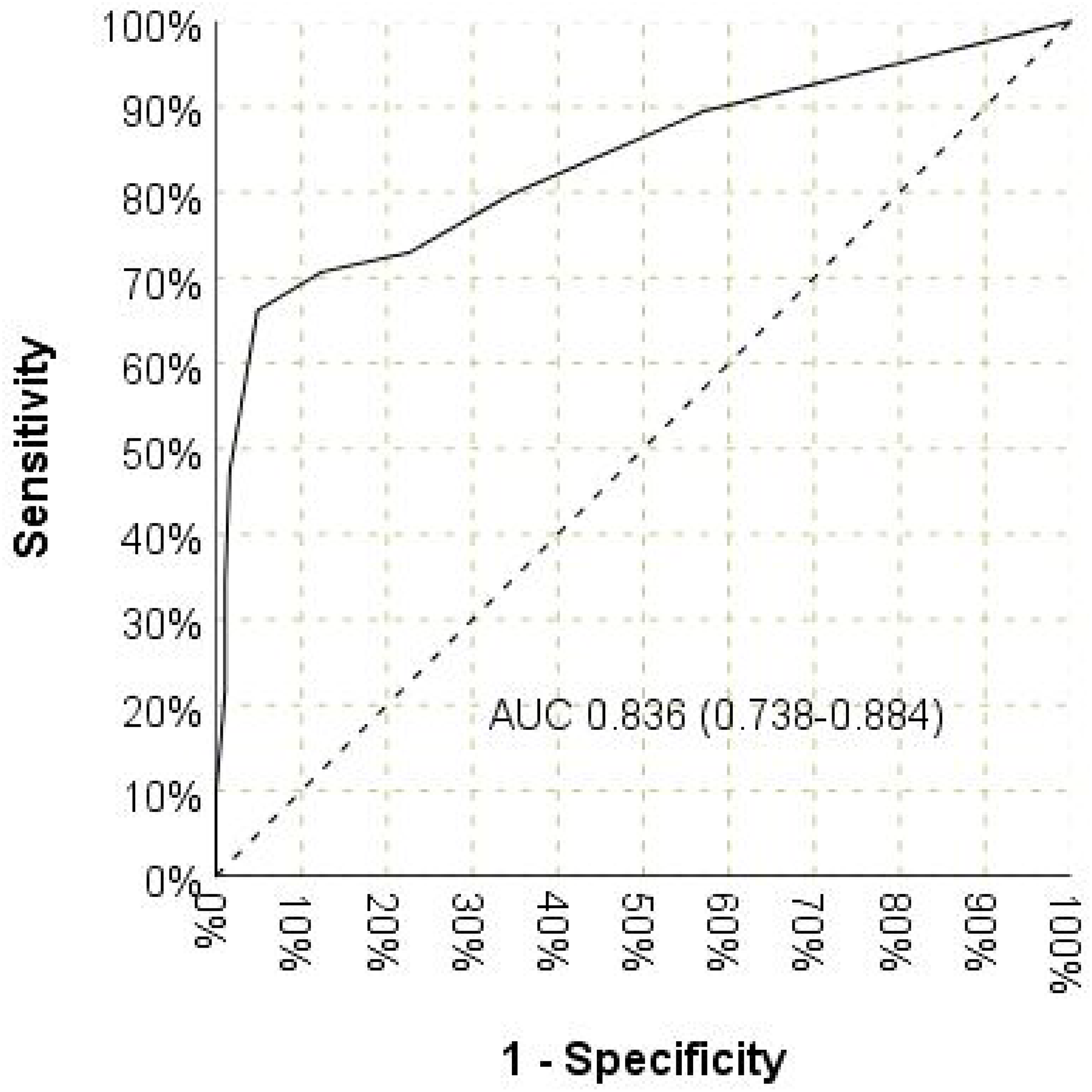
Receiver operating characteristics curves of anticoagulation and risk factors in atrial fibrillation (ATRİA) score in predicting thrombus burden.
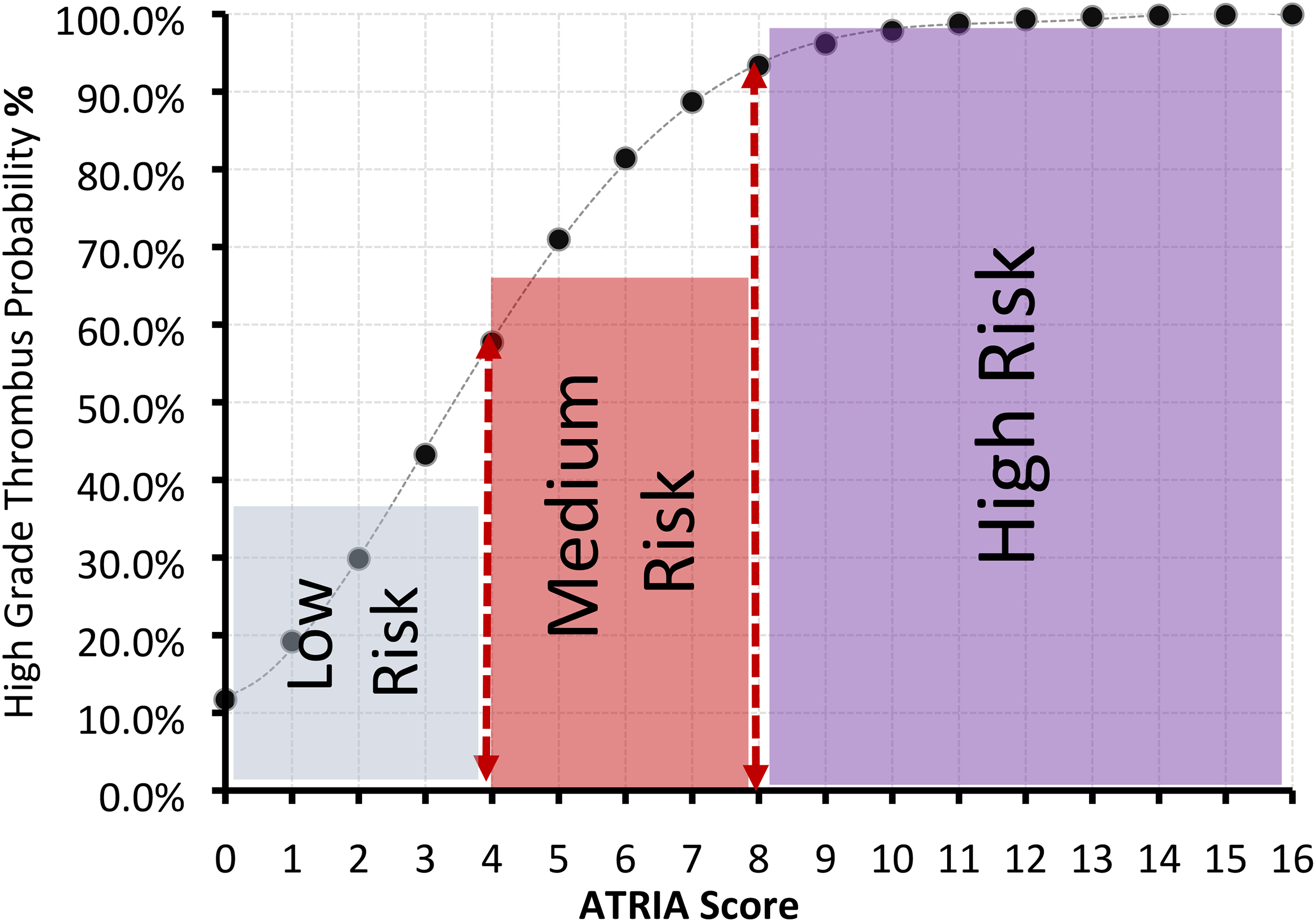
Probability of high thrombus burden according to anticoagulation and risk factors in atrial fibrillation (ATRIA) score.
Discussion
To our knowledge, this is the first study to evaluate the validity of the ATRIA score in predicting HTB in patients with STEMI. The main new finding of the current study was that STEMI patients with an HTB in the IRA had significantly higher ATRIA scores compared to STEMI patients with an LTB. We also demonstrated that as the cut-off value of the ATRIA score increases in these patients, the thrombus burden in the artery associated with infarction might also increase. Moreover, HT, abciximab usage, no-reflow, and low GFR were additional independent predictors of HTB.
Significant advances have been made in the treatment and management of STEMI over the last three decades. These advances have been made possible by demonstrating the critical role of thrombus formed by coronary plaque rupture in coronary occlusion and the resulting myocardial ischemia/infarction. 23 Revascularization based on primary PCI is the central component of the treatment in patients with STEMI. Primary PCI aims to restore patency of the IRA and ultimately myocardial perfusion as early as possible. However, distal embolization of thrombus is common even in primary PCI performed in the most experienced centers. Almost a third of patients have impaired microvascular area perfusion despite TIMI-3 flow in infarct-related coronary arteries. 24 Increasing the thrombus burden further impairs the reperfusion of the microvascular area and increases the risk of complications that may arise. Intracoronary HTB may complicate up to 70% of STEMI cases. 25 In addition, HTB correlates with larger myocardial infarct size and coronary no-reflow associated with distal embolization, increased risk of stent thrombosis, with subsequent increase in mortality and major adverse cardiac events (MACEs).4,6,26–28 In this context, intracoronary thrombus burden appears to be the primary prognostic marker for MACEs in STEMI patients.
Originally, the ATRIA score was used for thromboembolic ischemic stroke risk stratification in patients with AF. The ATRIA score was created to better identify AF patients at high risk of stroke than conventional thromboembolism scores.8,29 The ATRIA score combines renal dysfunction with the CHADS2 score and considers age categories in detail. Age and previous ischemic stroke appear to be the most important factors in ATRIA scoring. These two characteristics account for a large proportion of stroke risk prediction. Advanced age is a dominant risk factor for both cardiovascular and cerebrovascular disease. It is also an independent predictor of poor outcomes in acute myocardial infarction.30–32 The score was validated in the ATRIA–Cardiovascular Research Network cohort comprising about 35 000 patients with AF. It has been revealed that the ATRIA score outperformed the CHADS2 and CHA2DS2-VASc scores regarding c-index and net reclassification improvement for predicting stroke and thromboembolism. 29 These 3 scoring systems were also compared in more than 60 000 AF patients in the UK Clinical Practice Research Datalink cohort. The ATRIA score was better at identifying low thrombosis-risk AF patients who had been classified as high thrombosis-risk by the CHA2DS2-VASc score. 9 All these studies revealed that the ATRIA score performed better than the CHADS2 and CHA2DS2-VASc scores in predicting ischemic stroke in AF patients, especially in the low thromboembolic risk group.8,9,33 Studies in different patient cohorts and a recent meta-analysis also support these results. A subsequent meta-analysis supported these results.10,34
Considering that the parameters in the ATRIA score are directly related to coronary artery diseases, it would be a reasonable hypothesis to think that the ATRIA score can be used to evaluate coronary thrombotic processes. In this context, studies have recently emerged investigating whether the ATRIA score can be used to evaluate early and long-term adverse events caused by CAD. Çetinkal et al 11 suggested that the ATRIA score can predict long-term adverse events in myocardial infarction patients and prognostic assessment can be made with the ATRIA score. Abacıoğlu et al 13 have shown that the ATRIA score may predict no-reflow in patients undergoing primary PCI and may be associated with microvascular dysfunction. Another recent study suggested that the ATRIA score might be useful in predicting contrast-induced nephropathy (CIN) in STEMI patients who underwent primary PCI and that patients at high risk for CIN could be detected by using the ATRIA score in these patients. 35 In the present study, we evaluated the effectiveness of the ATRIA score in predicting thrombus burden during the acute atherothrombotic process in STEMI patients. We found that STEMI patients with HTB had a higher ATRIA score and that thrombus burden increased dramatically as the ATRIA score increased. Furthermore, serum creatinine which is indirectly involved in ATRIA scoring, and HT which is directly involved in ATRIA scoring were significantly higher in patients with HTB even outside the scoring. In this context, the presence of HT and renal dysfunction seems to have a significant effect on the results of the present study. Age was the predominant factor in applying the ATRIA score. Researchers have designed this score keeping in mind the increased risk of ischemic stroke in elderly patients. This situation probably increases the diagnostic accuracy of the ATRIA score in a patient with AF. Especially when the ATRIA score is calculated in patients with a previous stroke, age becomes even more critical. We hypothesize that the age distribution of the patients significantly influenced the results of this study, while a history of previous ischemic stroke had a weak contribution. Another parameter in ATRIA scoring is gender. Since the female gender represents 1 point on the ATRIA score, gender distribution might have impacted this study's results. Previously, it has been shown that female patients have a higher risk for poor outcomes in acute myocardial infarction than male patients. 36 In the present study, the share of the female gender among patients with HTB was 29.7%, while the share of the female gender among patients with LTB was only 24.4%. Although the difference was not statistically significant, this may have contributed to the higher ATRIA score in high-thrombus burden patients.
Thrombus burden has previously been reported to be an important determinant of coronary microembolization. HTB is associated with an increased risk of procedural complications such as no-reflow, stent malapposition, and stent thrombosis. In addition, HTB has been associated with increased infarct size due to impaired postprocedural epicardial and myocardial perfusion and distal embolization in patients with STEMI undergoing primary PCI. 37 To minimize the risk of no-reflow, current guidelines recommend the use of glycoprotein IIb/IIIa inhibitors in patients with an HTB. 7 In our study, the no-reflow rate and Gp IIb-IIIa antagonist use in HTB patients was significantly higher than in LTB patients
In the current study, HT was more common in STEMI patients with HTB. This result suggests that HT may be associated with more severe prothrombotic processes in STEMI patients. In a recent meta-analysis, HT was identified as the only predictor of STEMI, while DM, dyslipidemia, and advanced age were predictors for non-ST-elevation myocardial infarction and unstable angina pectoris. 38 Furthermore, HT is associated with an increased incidence of cardiovascular risk factors and directly contributes to the atherosclerotic process. 39 However, it is also a controversial clinical situation, as HT facilitates the action of other factors causing endothelial dysfunction. 40 It has been suggested that coronary occlusion in patients with preexisting HT might be associated with more extensive myocardial injury, more significant myocardial dysfunction, and consequently, adverse clinical outcomes.41,42 Lots of clinical evidence supports the existence of a prothrombotic process in patients with HT. This situation may be due to hypertensive end-organ damage paradoxically activating some coagulation factors such as fibrinogen. 43 In patients with HT, mean platelet volume and mean platelet mass are higher, and mean platelet granularity is lower. In addition, catecholaminergic discharge and renin-angiotensin-aldosterone system activation trigger platelet aggregation and activation in hypertensive patients. 44 Our data are partially consistent with these previous findings and may at least explain the high incidence of HT in STEMI patients with an HTB.
Our study has some limitations. First, this is a single-center study with a relatively small study population. Second, the thrombus grade was calculated by evaluating only angiographic images. We did not use intravascular imaging techniques such as intravascular ultrasound and optical coherence tomography. Third, since only STEMI patients were included, the results might not be expanded to all ACS patients. Studies with larger samples are required to confirm our results.
Conclusions
In this study, we have shown that an increase in the ATRIA score correlates with an increase in coronary thrombus burden in STEMI patients undergoing primary PCI. Therefore, the identification of STEMI patients with HTB undergoing primary PCI using this simple scoring system may help to select the best treatment strategy and to take measures to reduce the rate of no-reflow. Further studies are needed to determine the association of the ATRIA score with HTB in STEMI patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
