Abstract
Background:
We aimed to investigate the association between electrocardiographic (ECG) grade III ischemia and angiographic thrombus burden in patients with acute ST-segment elevation myocardial infarction (STEMI) who underwent primary percutaneous coronary intervention (pPCI).
Methods:
The study population consisted of 307 patients with STEMI. Baseline ECGs of the patients were analyzed for grade III ischemia; angiographic thrombus burden was assessed by thrombolysis in myocardial infarction thrombus classification.
Results:
A total of 108 (35%) patients had low thrombus burden whereas 199 (65%) patients had high thrombus burden. Grade III ischemia was more prevalent in patients with high thrombus burden (25.1% vs 11.1%, P = .004). Only grade III ischemia (odds ratio: 2.59, 95% confidence interval 1.24-5.39, P = .011) and history of coronary artery disease (CAD) were found to be the independent predictors of high thrombus burden.
Conclusion:
Grade III ischemia on ECG and previous history of CAD were independent predictors of coronary thrombus burden in patients with STEMI who underwent pPCI.
Introduction
Electrocardiography (ECG) is known to be a very important tool in both diagnostic and prognostic evaluations of ST-segment elevation myocardial infarction (STEMI). Grade III ischemia, defined as distortion of the terminal portion of the QRS complex, was shown to be associated with poor myocardial and epicardial perfusion. 1 –3 Grade III ischemia may not only provide substantial information about postprocedural epicardial and myocardial flow in patients both receiving thrombolytic treatment and undergoing primary percutaneous coronary intervention (pPCI) but also may provide significant data regarding long-term adverse cardiovascular events. 4,5 However, the reason for the negative impact of grade III ischemia on short- and long-term cardiovascular events is not exactly known. Grade III ischemia has also been noted to have an association with diffuse myonecrosis shown by elevated biomarker release, 2,4 more diffuse microvascular obstruction, 6 lower thrombolysis in myocardial infarction (TIMI) flow, and similarly low postprocedural TIMI flow and myocardial blush. 7,8 Intracoronary thrombus burden is a well-known risk factor for long-term adverse cardiovascular events, distal embolization, and stent thrombosis. 9–12 To establish the predictors of intracoronary thrombus burden and associated clinical and angiographic conditions, therefore, could provide improvement in therapeutic management. We hypothesized that the association of grade III ischemia with short- and long-term adverse events may be due to its relationship with angiographic thrombus burden. To address this issue, we aimed to investigate the relation of grade III ischemia with angiographic thrombus burden in patients with STEMI who underwent pPCI.
Methods
Study Population
We retrospectively reviewed 307 patients who presented to our hospital with STEMI who underwent PCI between August 2011 and August 2012. The inclusion criteria were (1) presentation within the first 12 hours after the onset of pain (18 hours for cardiogenic shock) and no history of thrombolytic therapy (2) and presence of an ST-segment elevation at least 1 mm (2 mm for V1-V3) in 2 or more contiguous leads in ECG. 13 Patients with left bundle branch block, intraventricular conduction delay, and ventricular rhythm were excluded. Informed consent was obtained from all the patients during hospitalization, and the study protocol was approved by our institutional review board.
For all patients, clinical risk factors, age, sex, history of hypertension, diabetes and hyperlipidemia; smoking status; history of previous PCI or coronary artery bypass grafting (CABG); family history; and anthropometric characteristics such as height and weight were recorded. Besides, blood pressure, heart rate, Killip class, presence of cardiogenic shock, previous medications, preinfarctional angina, pain-balloon time and door-balloon time, serum creatinine, and lipid panel were measured for all the patients during hospital admission. Hemoglobin levels were measured as part of the automated complete blood count using a Coulter LH 780 Hematology Analyzer (Beckman Coulter Ireland Inc, Mervue, Galway, Ireland).
Clinical and demographic properties of the patients were recorded. Baseline hemogram, lipid parameters, urea, creatinine, glucose, creatine kinase, creatine kinase-MB isoform (CK-MB), and troponin I levels were obtained on admission. Blood samples were repeated for creatine kinase, CK-MB, and troponin I every 6 hours until peak levels were reached and repeated daily thereafter. An estimated glomerular filtration rate was calculated from serum creatinine using the Modification of Diet in Renal Disease Study equation. The ECG was performed on admission, 90 minutes after the procedure, and daily thereafter. All patients received chewable aspirin 300 mg, loading dose of clopidogrel 600 mg on admission, and intravenous standard heparin 10 000 U before the procedure. The use of glycoprotein IIb/IIIa receptor blockers (tirofiban) with 10 μg/kg bolus and 0, 15 μg/kg per min intravenous infusion was left to the primary operator’s discretion.
All pPCI procedures were performed by experienced interventional cardiologists through a femoral approach using a 7-F guiding catheter. The lesions were passed by 0.014 in guide wires and predilation, if needed, was performed. After stent implementation, postdilatation was performed according to the operators’ discretion. Postprocedural final TIMI flow grade and TIMI myocardial perfusion grade (TMPG) were assessed. Postprocedural TIMI flow, corrected TIMI frame count (cTFC), and TMPG were noted. After the procedures, all patients were followed up in the coronary intensive care unit until clinical stabilization was established.
Clinical Definitions
Hypertension was defined as systolic pressure >140 mm Hg and/or a diastolic pressure >90 mm Hg at least 2 times or if the individual was taking antihypertensive medications. The diagnosis of diabetes mellitus (DM) was based on previous history of DM treated with or without drug therapies. Current smoker was defined as smoking for some period during the past year. Previous history of coronary artery disease (CAD) was defined as diameter lumen stenosis of >50% in at least 1 major coronary artery, previous history of PCI, or CABG. The ST-segment resolution (STR) in ECG was assessed based on ST-segment regression percentage on 90th minute ECG and 70% and above ST resolution was concluded as successful reperfusion. While calculating the STR, the sum of ST elevations (from all the leads) on admission and 90-minute ECGs were used.
Grade III Ischemia
Distortion of the terminal portion of the QRS was defined by Birnbaum et al into 2 groups grade 3 ischemia is characterized by absence of S waves in at least 2 spatially adjacent leads (leads V1 through V3) and/or a takeoff of the ST segment at >50% of the R wave in at least 2 spatially adjacent other leads. ST elevation that does not meet these criteria is classified as grade 2 ischemia 4 (Figure 1). The ECGs were read by 2 independent cardiologists blinded to the patient data (A.B.A. and N.S.).
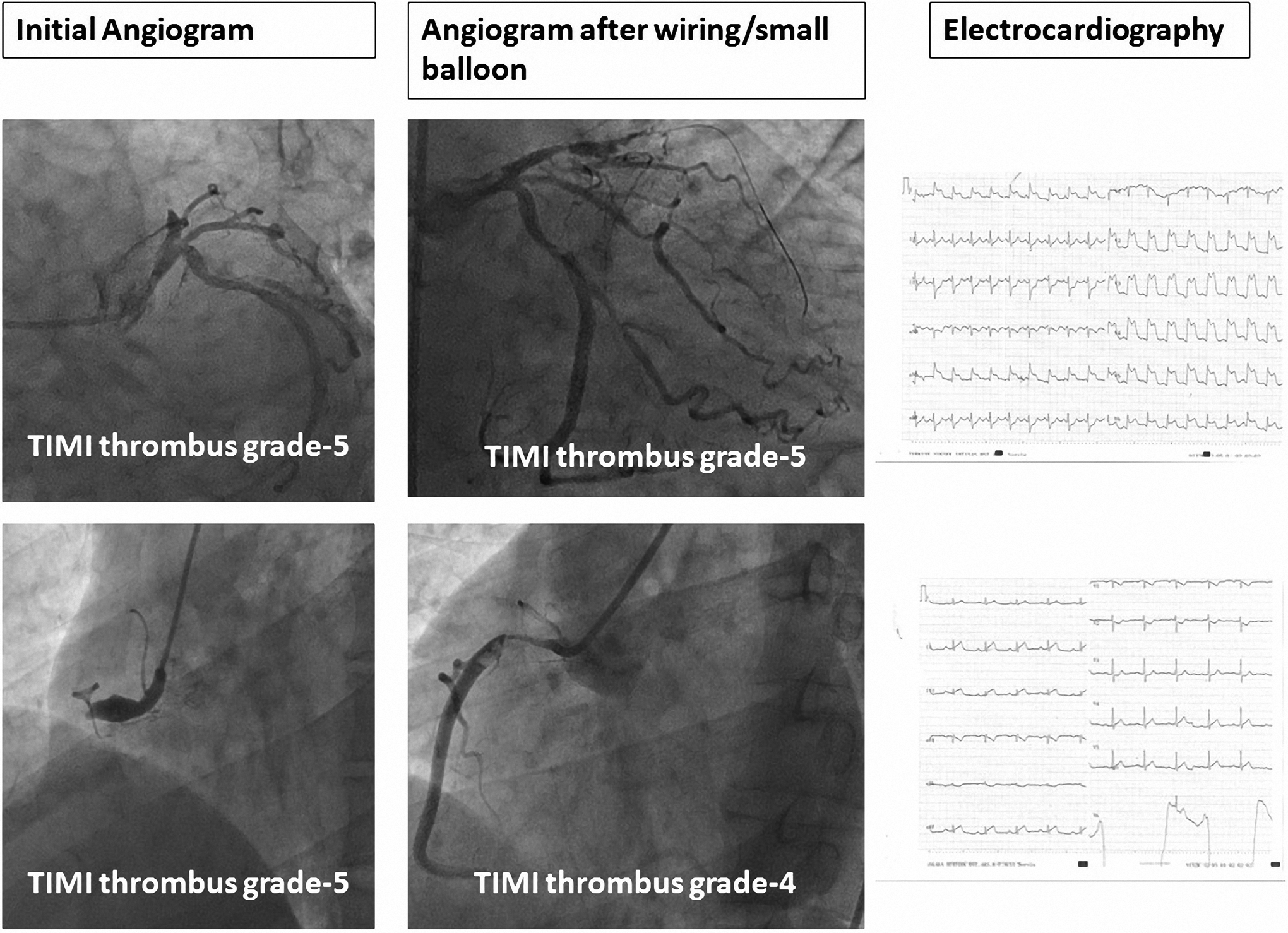
Angiograms in which thrombolysis in myocardial infarction (TIMI) thrombus scores calculated in our study population, and electrocardiographic (ECG) samples.
Angiographic Definitions
Angiographic coronary thrombus burden was scored based on 5 grades, as previously described. 10 Grade 0 defined as no angiographic evidence of thrombus, grade 1 defined as angiographic features suggestive of thrombus (decreased contrast density, haziness of contrast, irregular lesion contour, a smooth convex meniscus at the site of a total occlusion, suggestive, but not firmly diagnostic of thrombus), grade 2 defined as definite thrombus present in multiple angiographic projections (marked irregular lesion contour with a significant filling defect—the thrombus’ greatest dimension is <1/2 vessel diameter), grade 3 defined as definite thrombus appears in multiple angiographic views (greatest dimension from >1/2 to <2 vessel diameters), grade 4 defined as definite large size thrombus present (greatest dimension >2 vessel diameters), and grade 5 defined as definite complete thrombotic occlusion of a vessel (a convex margin that stains with contrast, persisting for several cardiac cycles). We primarily calculated TIMI thrombus grade based on the initial diagnostic angiogram, then, after restoring antegrade flow through guide wire or small balloon dilatation in patients with TIMI thrombus grade 5; coronary angiogram enabled restratification of the underlying residual thrombus (final TIMI thrombus grade).
Finally, we stratified final TIMI thrombus grades as low thrombus burden or high thrombus burden based on scores 1 to 3 or 4 to 5, respectively. In the final diagnostic angiogram, TIMI flow grades, cTFC, and TMPG measurements were assessed as previously described. 14 –16 The TMPG was analyzed by categorizing into 2 groups (TMPG 0, 1 vs TMPG 2, 3). TIMI 0, I, and II were described as angiographic no-reflow.
Statistics
Continuous variables are expressed as mean ± standard deviation or median (interquartile range), whereas categorical variables are expressed as percentage. Comparisons between groups were made by using the Student t test, Mann-Whitney U test, or chi-square tests, as appropriate. Multiple logistic regression analysis was performed to identify the independent predictors of high thrombus burden using variables. Two-tailed P values <.05 were considered to indicate statistical significance. Statistical analyses were performed using SPSS version 17.0 for Windows.
Results
The study population consisted of 307 patients with STEMI (mean age 55 ± 12.7 and 71% male). Eventually, final TIMI thrombus grades of the study population were low thrombusburden in 108 (35%) patients and high thrombus burden in 199 (65%) patients. Similarly, grade III ischemia was present in 20.2% (n = 62) of the whole population. Grade III ischemia is significantly higher in patients with high thrombus burden (25.1% vs 11.1%, P = .004; Figure 2). The comparisons of basic clinical and laboratory findings between thrombus burden groups were presented in Table 1. Patients with high thrombus burden had more family history of CAD (17% vs 3.7%, P = .001), longer pain-balloon time, higher baseline CK-MB and baseline troponin, higher peak CK-MB, and peak troponin values.
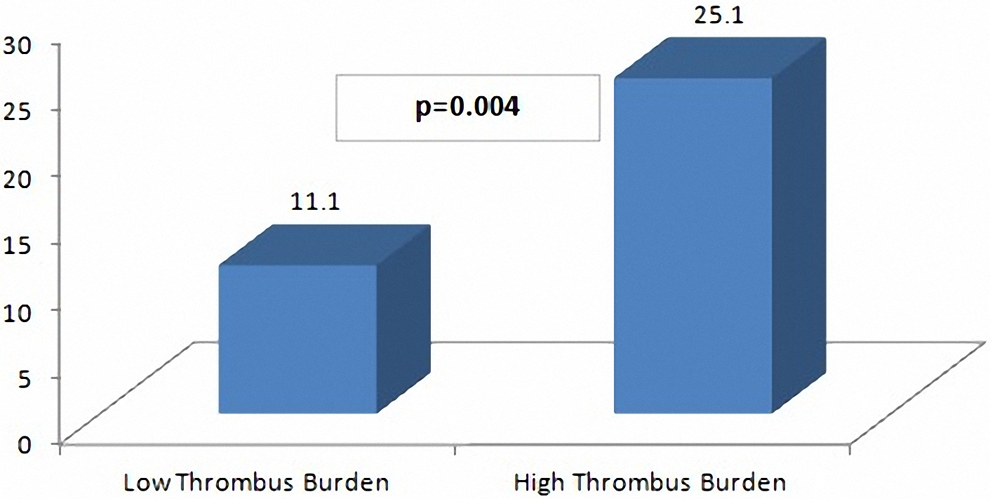
Grade III ischemia according to thrombolysis in myocardial infarction (TIMI) thrombus burden groups.
Baseline Clinical and Laboratory Characteristics According to Thrombus Burden.
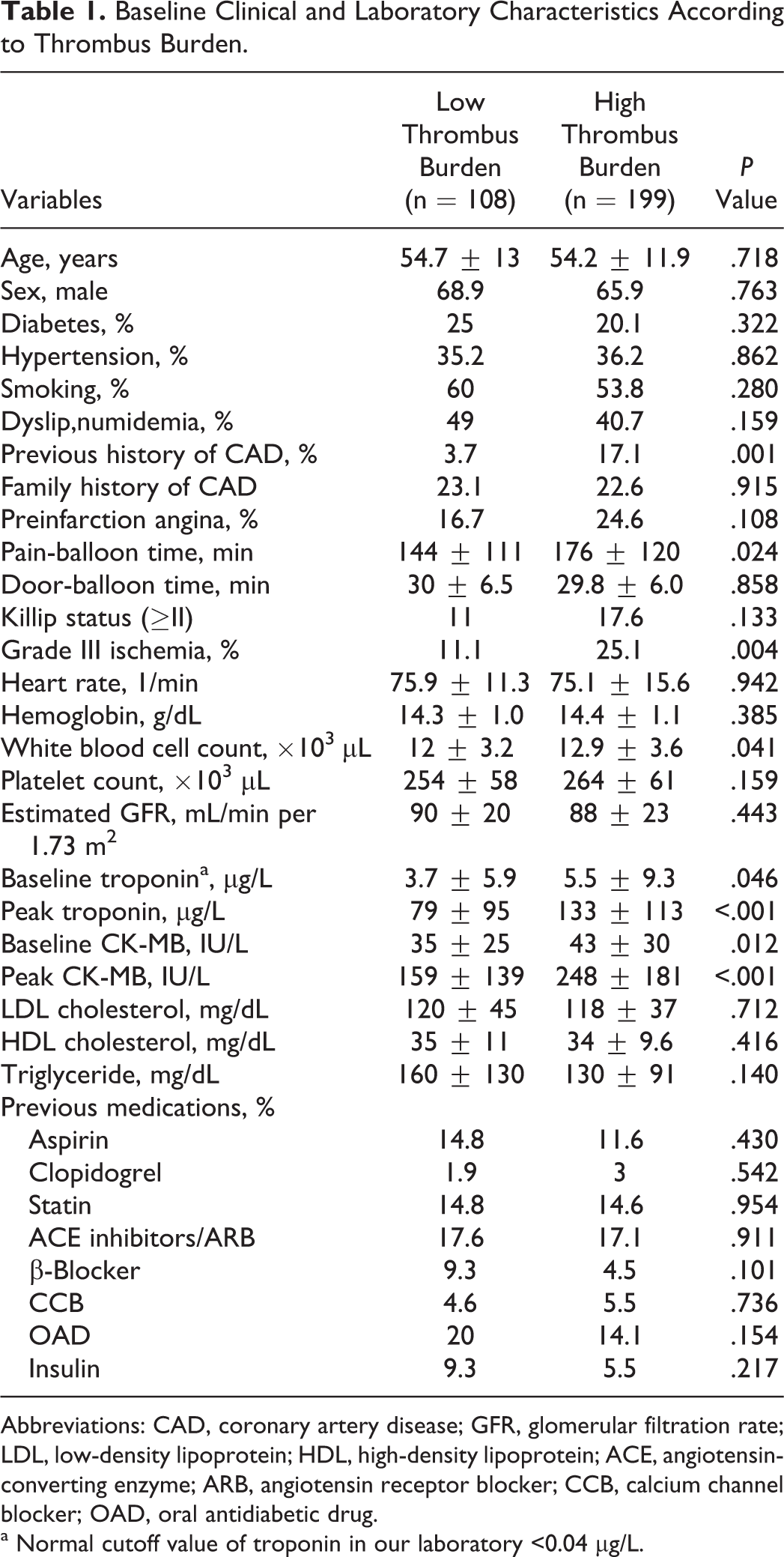
Abbreviations: CAD, coronary artery disease; GFR, glomerular filtration rate; LDL, low-density lipoprotein; HDL, high-density lipoprotein; ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CCB, calcium channel blocker; OAD, oral antidiabetic drug.
a Normal cutoff value of troponin in our laboratory <0.04 µg/L.
Comparison of the baseline angiographic characteristics and postprocedural findings of the groups based on thrombus burden were shown in Table 2. Longer stents were chosen (median 20 mm vs 18 mm, P < .001), and less frequent direct stenting (9.5% vs 37.9%, P < .001), lower epicardial (81.9% vs 95% for TIMI flow III, P = .002 and 26 ± 15.6 vs 19.9 ± 7.1 for cTFC, P < .001) and myocardial perfusion (45.2% vs 62%, P = .005), less initial infarct-related artery patency (TIMI II-III; 6.5% vs 85.2%, P < .001), more frequent ECG (49.8% vs 27.9%, P < .001), and angiographic no-reflow (18.1% vs 5%, P < .001) were observed in patients with high thrombus burden. Besides, comparison of the baseline angiographic characteristics and postprocedural findings of the groups based on grade II and grade III ischemia were shown in Tables 3 and 4.
Baseline Angiographic and Postprocedural Characteristics According to Thrombus Burden.
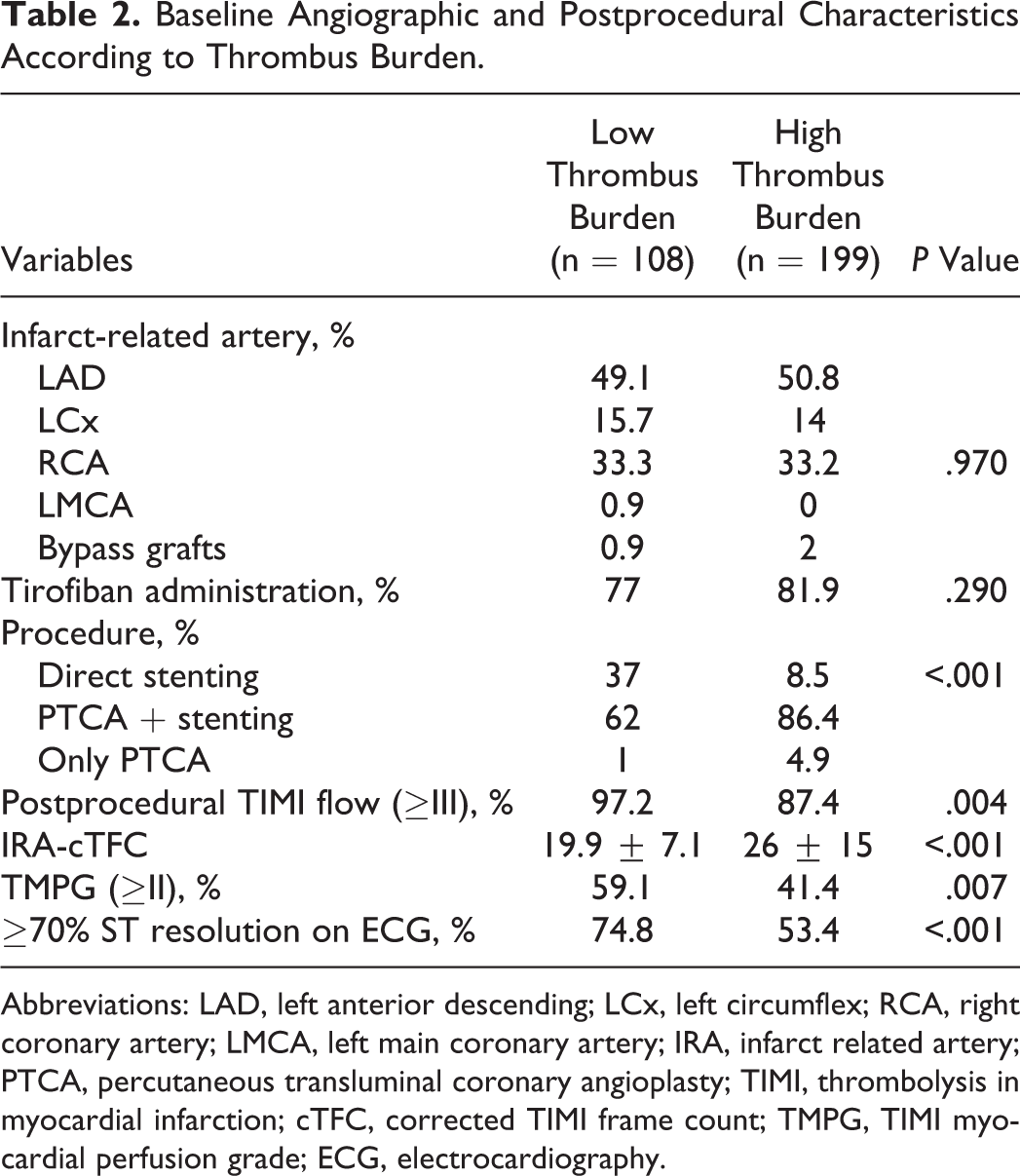
Abbreviations: LAD, left anterior descending; LCx, left circumflex; RCA, right coronary artery; LMCA, left main coronary artery; IRA, infarct related artery; PTCA, percutaneous transluminal coronary angioplasty; TIMI, thrombolysis in myocardial infarction; cTFC, corrected TIMI frame count; TMPG, TIMI myocardial perfusion grade; ECG, electrocardiography.
Baseline Clinical and Laboratory Characteristics According to Grades of Ischemia.
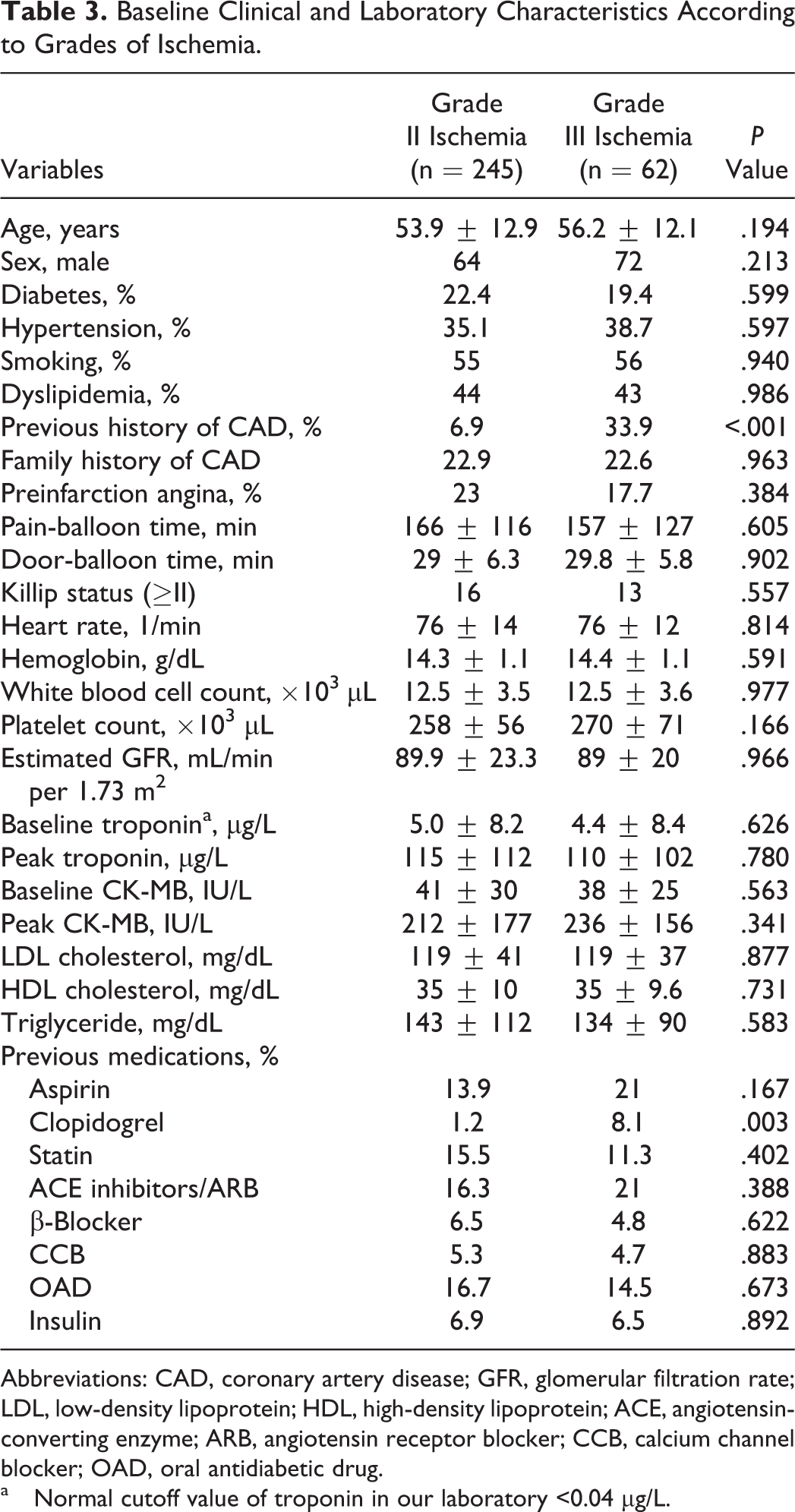
Abbreviations: CAD, coronary artery disease; GFR, glomerular filtration rate; LDL, low-density lipoprotein; HDL, high-density lipoprotein; ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CCB, calcium channel blocker; OAD, oral antidiabetic drug.
a Normal cutoff value of troponin in our laboratory <0.04 µg/L.
Baseline Angiographic and Postprocedural Characteristics According to Grades of Ischemia.
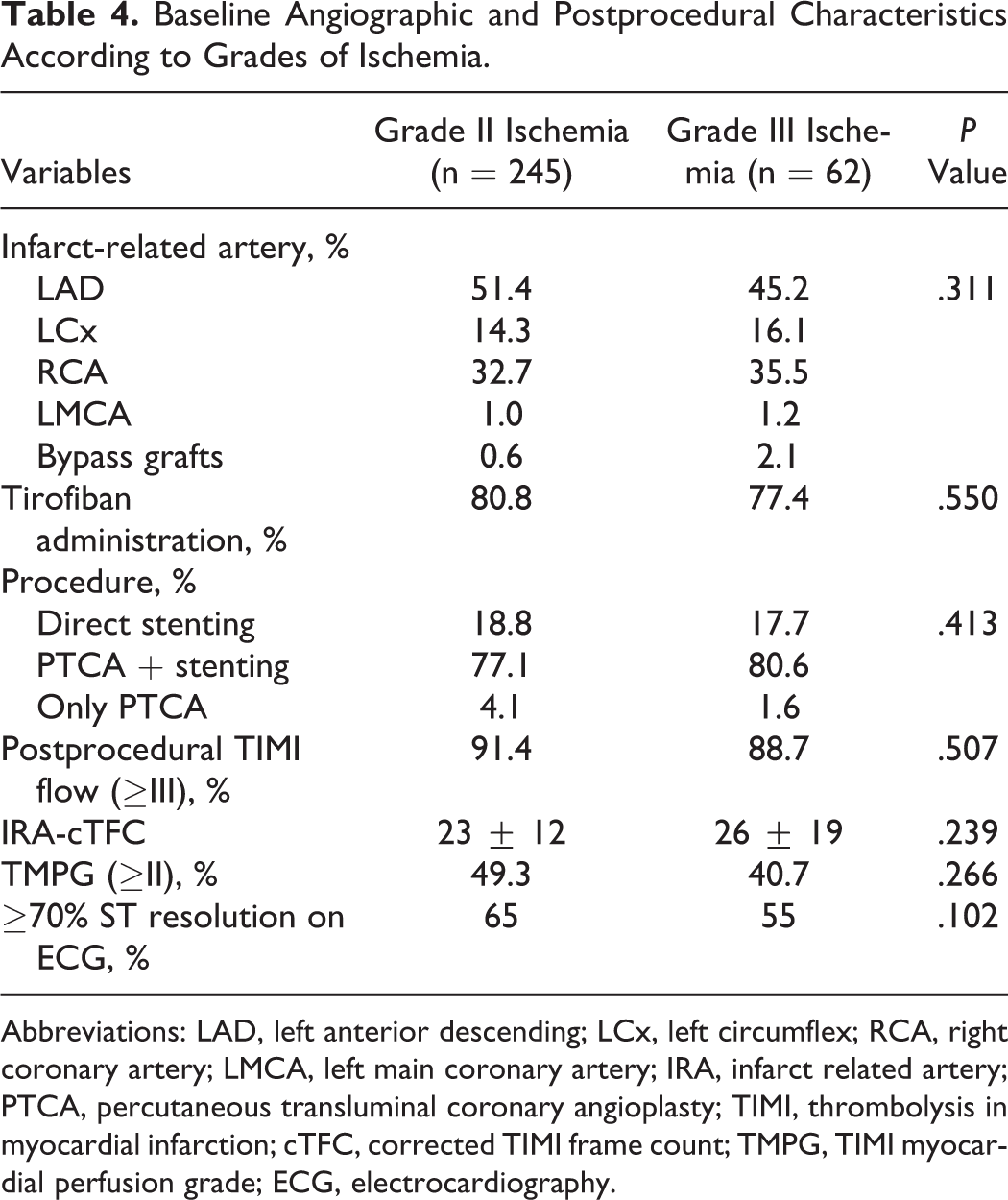
Abbreviations: LAD, left anterior descending; LCx, left circumflex; RCA, right coronary artery; LMCA, left main coronary artery; IRA, infarct related artery; PTCA, percutaneous transluminal coronary angioplasty; TIMI, thrombolysis in myocardial infarction; cTFC, corrected TIMI frame count; TMPG, TIMI myocardial perfusion grade; ECG, electrocardiography.
Multivariate logistic regression analysis was performed in order to determine the independent predictors of high thrombus burden. Only grade III ischemia (odds ratio: 2.59, 95% confidence interval: 1.24-5.39, P = .011) and history of CAD were determined as independent predictors in the analysis performed for the variables found as significant in univariate analysis (history of CAD, previous history of acetylsalicylic acid use, white blood cell, baseline CK-MB and baseline troponin, pain-balloon time, and grade III ischemia; Table 5). Agreement analysis revealed the value of weighted κ for interobserver and intraobserver agreement was 0.77 and 0.86, respectively.
Multivariate Logistic Regression Analysis to Detect the Independent Predictors of High TIMI Thrombus Burden.
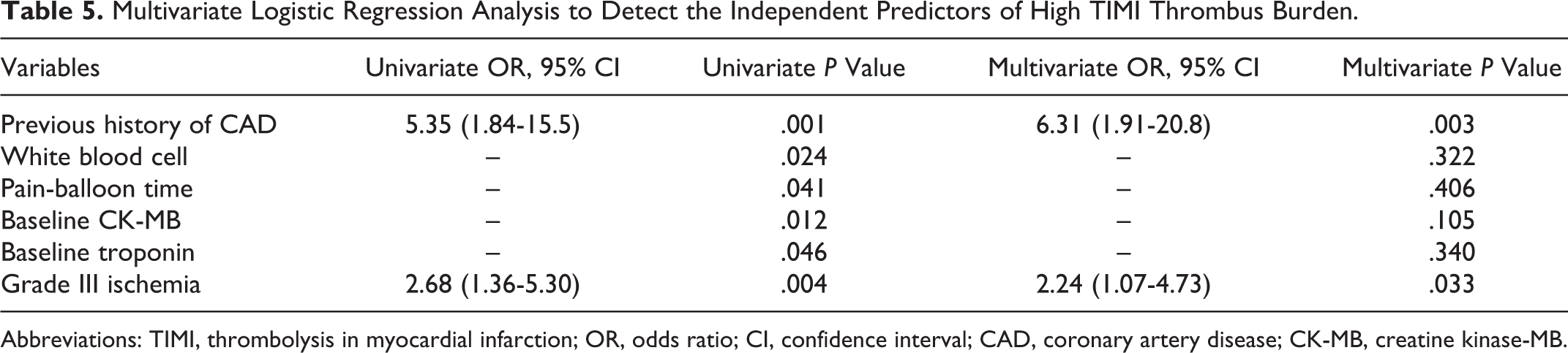
Abbreviations: TIMI, thrombolysis in myocardial infarction; OR, odds ratio; CI, confidence interval; CAD, coronary artery disease; CK-MB, creatine kinase-MB.
Discussion
Our study results indicate that ECG grade III ischemia and previous history of CAD were independent predictors of high thrombus burden in patients with STEMI who underwent pPCI. Besides, we found that high thrombus burden was associated with more impaired epicardial and myocardial perfusion and higher percentage of no-reflow.
Intracoronary thrombosis developed after plaque rupture is the basic pathophysiologic event in patients with STEMI. 17 The presence of intracoronary thrombus and quantity of the thrombus burden are important determinants of prognosis in STEMI. 9,10,18 Intracoronary thrombus impairs epicardial blood flow depending on partial or complete occlusion. In addition, distal embolization coming from the thrombus mass impairs myocardial perfusion. Therefore, treatment modalities (glycoprotein IIb/IIIa inhibitors) or procedures (thrombectomy), which are reducing thrombus burden, improve both epicardial and myocardial perfusion. In the present study, grade III ischemia and previous history of CAD were found as an independent predictor of high thrombus burden. Grade III ischemia is associated with adverse clinical outcomes. 8 Weaver et al revealed that grade III ischemia was more frequently associated with intramyocardial hemorrhages and microvascular obstruction shown by cardiac magnetic resonance imaging. 6 Grade III ischemia is also associated with higher cardiac mortality and no-reflow rates in patients who underwent pPCI. Furthermore, grade III ischemia is associated with angiographic and ECG no-reflow too. 1,4,7,8 Mechanism of this is not exactly known. However, the present study results revealed that grade III ischemia is an independent predictor of angiographic thrombus burden. Angiographic thrombus burden is one of the fundamental determinants of both no-reflow and impaired epicardial and myocardial perfusion after pPCI. 9–11 In conclusion, we propose that grade III ischemia predicts angiographic no-reflow and impaired epicardial and myocardial perfusion. Wolak et al found a higher rate of visible thrombus among patients with grade III ischemia which was supportive to our result. 1
Prediction of thrombus burden in patients with STEMI undergoing pPCI before the procedure may contribute to high thrombus burden management through enhancing utilization of pharmacological agents such as glycoprotein IIb/IIIa antagonists or interventional strategies such as mechanical thrombectomy.
Limitation
Since our study was designed as retrospective, it has common limitations of retrospective studies because of the bias depending on nonmeasurable factors. Similarly, since classification of thrombus burden was based on visual evaluation, an agreement analysis was performed but agreement analyses also have some limitations. 19 Thrombectomy devices were not actively used in our country during the time of our study; therefore, we could not have given information about this subject.
Conclusion
Present study results demonstrated that high thrombus burden in patients with STEMI were associated with impaired postprocedural epicardial and myocardial perfusion; and grade III ischemia on ECG and previous history of CAD were independent predictors of coronary thrombus burden.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
