Abstract
Recombinant factor VIIa (rFVIIa) is used in the management of bleeding in patients with hemophilia. A generic biosimilar version of NovoSeven is also developed (AryoSeven). To compare the activation profile of NovoSeven and AryoSeven, 2 commercially available protein complex concentrates (PCCs) were used. Profilnine activated by RecombiPlasTin 2G resulted in conversions of prothrombin to prethrombin and thrombin at 5 to 30 minutes. However, addition of rFVIIa at final concentration range of 0.25 to 0.5 µg/mL to the same mixture resulted in total conversion of prothrombin to thrombin with a doublet at 36 kDa. Recombinant factor VIIa alone did not generate thrombin in native Beriplex, and the addition of rFVIIa to Beriplex failed to generate thrombin. Beriplex activated by RecombiPlasTin 2G resulted in complete conversion of prothrombin to thrombin. Both NovoSeven and AryoSeven exhibited similar activation profiles. These studies indicate that the activation of PCCs by both rFVIIa preparations results in comparable generation of thrombin.
Introduction
Commercially available recombinant coagulation factors are widely used in the management of bleeding disorders. A commercially available recombinant factor VIIa (rFVIIa) namely NovoSeven is approved for the management of bleeding with inhibitors in patients having hemophilia A . Factor VIIa, when complexed with tissue factor, can activate FX to FXa and FIX to FIXa. Following complexation with other factors, FXa converts prothrombin to thrombin, which eventually converts fibrinogen to fibrin. The effect of FVIIa in patients with or without hemophilia has been evaluated in different in vitro experimental models. In 1 such model, supplementation of FVIIa at a concentration as low as 10 nmol/L with tissue factor increased both the rate and the level of thrombin generation in blood drawn from healthy volunteers and in patients with hemophilia A. 1 In another model, escalating dosage of FVIIa (NovoSeven) demonstrated a dose-dependent increase in thrombin generation in plasma from patients with hemophilia A. 1
Fresh frozen plasma is a treatment option for FVII deficiency. 2,3 Prothrombin complex concentrates have also been used. 4 –6 However, these products are not standardized for their FVII content, so multiple doses of protein complex concentrates (PCCs) may be utilized, which increased the risk of thrombotic event. 2 Several studies have reported that utilizing NovoSeven did not cause an uncontrolled thrombin generation and fibrin deposition in an experimental setting. 7,8 Addition of rFVIIa to FVIII- and FIX-deficient plasma did not induce free thrombin generation. 9 However, addition of activated protein complex concentrates (aPCC) and PCC enhanced the thrombin generation in the same experimental model. 10
In some clinical conditions, the concomitant use of aPCC/FVIII and rFVIIa resulted in cerebrovascular events or disseminated intravascular coagulation (DIC). 11 In contrast, some patients with DIC or severe bleeding have been treated with NovoSeven. 12 For rapid warfarin, reversal in the setting of intracranial hemorrhage combination of PCCs and rFVIIa was reportedly used. 13 Variable efficacy results may due to the compositional differences in various PCCs. 14 Protein complex concentrates are purified plasma-derived concentrates with hemostatic activity containing 4 vitamin K–dependent coagulation factors II, VII, IX, and X. 15 These agents were originally developed as a source of factor IX for the treatment of hemophilia B. 16 The use of PCCs has been suggested in several liver diseases and in preoperative and trauma-related bleeding. 17,18 Four-factor concentrates such as Beriplex contain adequate amounts of vitamin K–dependent factors, whereas 3-factor concentrate, such as Profilnine SD, contains significantly lower amount of factor VII. 19,20 Furthermore, differences were also noted between the determined and the label values for various clotting factors. Besides these coagulation factors, PCCs may contain small quantities of anticoagulants like protein C, protein S, and protein Z. Heparin and antithrombin are also added to reduce thrombogenicity and prevent activation of coagulation factors by endogenous processes. 21 –23
AryoSeven (AryoGen, Tehran, Iran) is a biosimilar product comparable to NovoSeven and is reported to have a similar efficacy and safety profile. 24,25 This product is clinically being used in Iran for the management of bleeding in patients with hemophilia. 26 There are reported studies on the effect of biosimilar version of rFVIIa on PCCs. Despite the availability of some studies on the effect of NovoSeven and PCCs, variable results have been reported. The current study was designed to compare 2 recombinant VIIa preparations in the generation of thrombin in an in vitro experimental studies utilizing 2 different PCCs. These comparative studies included sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) analysis and immunoblotting studies utilizing a polyclonal (rabbit) antihuman factor VIIa antibody.
Materials and Methods
Profilnine SD and Beriplex were purchased from Grifols Biologicals, Inc (Los Angeles, California) and CSL Behring Canada, Inc (Ottawa, Ontario), respectively. Tissue factor, RecombiPlasTin 2G (RCP) was obtained from Instrumentation Laboratory Company (Bedford, Massachusetts). Two batches of NovoSeven were purchased from Novo Nordisk (Copenhagen, Denmark). Seven batches of AryoSeven were obtained from AryoGen (Tehran, Iran). As per the product insert, all AryoSeven products contained 1.2 mg of rFVIIa. The batches of NovoSeven contained 2.4 or 1 mg of rFVIIa. All batches were initially diluted in sterile water to obtain concentrations of 1 mg/mL solution. Further dilutions were made in saline.
Preparation of Mixtures
Controls were prepared by mixing 25 µL of 10 U/mL Profilnine SD or 10 U/mL Beriplex with 75 µL saline and 25 µL of 10 U/mL Profilnine SD or 10 U/mL Beriplex with 50 µL of saline and 25 µL RCP. Sample mixtures were prepared by mixing 25 µL of 10 U/mL Profilnine SD or 10 U/mL Beriplex with 25 µL saline, 25 µL RCP, and 25 µL of various batches of rFVIIa with different working concentration of 0.5, 1.25, and 2.5 µg/mL. Samples were incubated for 5, 15, and 30 minutes and then 25 µL of EDTA (2 mg/mL) was added to stop reaction.
Sodium Dodecyl Sulfate Polyacrylamide Gel Analysis
Five microliters of Laemmli sample buffer (Boston BioProducts, Ashland, Massachusetts) was added to 20 µL of PCC mixtures. The samples were then denatured and subjected to electrophoresis under reducing condition through 4% to 20% gradient Tris-N-2-hydroxyethylpiperazine-N-2-ethanesulfonic acid–sodium dodecyl sulfate (SDS) polyacrylamide mini gels (Pierce Biotechnology, Rockford, Illinois). Gel was then stained with Coomassie blue (Bio-Rad Laboratories, Hercules, California) for band visualization.
Immunoblotting Studies Using Rabbit Antihuman Recombinant Thrombin Antibody
The immunoblotting studies were carried out utilizing immunoglobulin G (IgG) isolated from antisera generated against recombinant human thrombin in rabbits. A commercial lot of Recothrom (ZAC0712A) was obtained from ZymoGenetics Inc (Seattle, Washington). This product was used for immunization of the rabbits and as a reference thrombin for immunoblotting studies.
To generate specific antisera in rabbits, Recothrom was provided to Lampire Biologics (Pipersville, Pennsylvania). Individual groups (n = 3) of rabbits were challenged repeatedly with human recombinant thrombin, over a 9-month period. To generate specific antisera in rabbits, Recothrom was conjugated with keyhole lymphocyte hemocyanin as a carrier and was administered intravenously at a dosage of 100 mg using standard immunologic methods. A total of 10 injections were administered during this time, with a 4-week interval between each injection. Preimmune blood and antiserum were collected from each rabbit. The antiserum from each rabbit and the pooled antisera obtained from 3 individual rabbits in each treatment group were stored at −80°C prior to IgG isolation.
HiTrap Protein G HP 1-mL columns (GE Healthcare Bio-Science Crop, Piscataway, New Jersey) were used to isolate IgG from rabbit anti-Recothrom antisera. The eluted IgG fractions were carefully collected, and the concentration of harvested IgG was adjusted with saline to 1 mg/mL based on the protein concentration determined using a modified Lowry assay. All isolated IgG samples were aliquoted and stored at −80°C.
In order to determine the immunoreactive component in the Profilnine mixture preparations, immunoblotting studies were carried out using anti-Recothrom IgG isolated from antisera generated against recombinant human thrombin in rabbits.
Various mixtures of the Profilnine samples were electrophoresed through 4% to 20% gradient gel and then were electrotransferred onto nitrocellulose membrane overnight at 4°C. Precision Dual Color Plus protein standards (Bio-Rad Laboratories, Hercules, California) were included in each gel. After electrotransfer, the nitrocellulose membranes were blocked with 5% milk/TBST (Tris-buffer saline, pH 7.6, 0.1% Tween-20) for 1 hour with gentle shaking, then briefly washed in TBST, and incubated for 24 hours at 4°C with anti-Recothrom IgG (5 µg/mL in 5% milk/TBST). Blot was then washed and incubated for 1 hour at room temperature with 1:1 00 000 dilutions in 5% milk/TBST of horseradish peroxidase-conjugated donkey antirabbit IgG (H + L; Thermo Scientific, Rockford, Illinois). The blots were then extensively washed, and immunoreactive bands were detected with SuperSignal West Pico Chemiluminescent Substrate (Pierce Biotechnology) followed by film exposure for about 10 seconds. For each run besides the molecular-weight standards, each of the PCCs alone, PCCs with rFVIIa, and PCCs with recombiplastin served as internal control.
Results
These studies were designed to evaluate thrombin generation in Profilnine and Beriplex upon activation with recombiplastin in the presence of various rFVIIa preparations. In this study, 2 sets of analysis were performed. In the first set of experiment, activated PCCs with tissues factor were supplemented with different working concentration of rFVIIa (1.25 and 2.5 µg/mL) and incubated for 30 minutes. Second set of experiment was performed with fixed working concentration of rFVIIa (0.5 µg/mL) and different incubation time points, 5 and 15 minutes, in order to the activities to various batches of rFVIIa in the generation of thrombin in different concentrations and time points.
Sodium Dodecyl Sulfate Polyacrylamide Gel Analysis
The SDS-PAGE studies were performed to evaluate the protein composition in Profilnine. Figure 1A shows SDS-PAGE analysis of various Profilnine mixtures contained rFVIIa (Final concentration [FC]: 0.25 µg/mL) incubated for 30 minutes loaded in gel as follows: molecular-weight markers (lane 1), Profilnine (lane 2), Profilnine/NovoSeven JV60316 (lane 3), Profilnine/RCP (lane 4), Profilnine/AryoSeven 1020/RCP (lane 5), Profilnine/AryoSeven 1021/RCP (lane 6), Profilnine/AryoSeven 1022/RCP (lane 7), Profilnine/AryoSeven 1023/RCP (lane 8), Profilnine/NovoSeven JV60316/RCP (lane 9), and control recombinant thrombin (lane 10). Coomassie blue staining revealed that native Profilnine SD exhibited an intense 75-kDa band, which may indicate prothrombin and 2 unidentified faint bands at 250 and 140 kDa (lane 2). Mixture of rFVIIa and Profilnine showed similar profile to native Profilnine (lane 3). Activation of Profilnine with RCP resulted in the generation of new bands at 50 and 36 kDa (lane 4). Mixture of Profilnine–RCP–rFVIIa exhibited different protein profile with intense bands at 36 kDa (lanes 5-9). Generation of thrombin was confirmed, which was comparable to the control thrombin (lane 10). Figure 1B depicts similar Profilnine mixtures with 1 exception, which is the supplementation of 2.5 µg/mL of working concentration of rFVIIa (FC: 0.5 µg/mL) to the mixture. There was no difference in protein profile with higher concentration of rFVIIa.
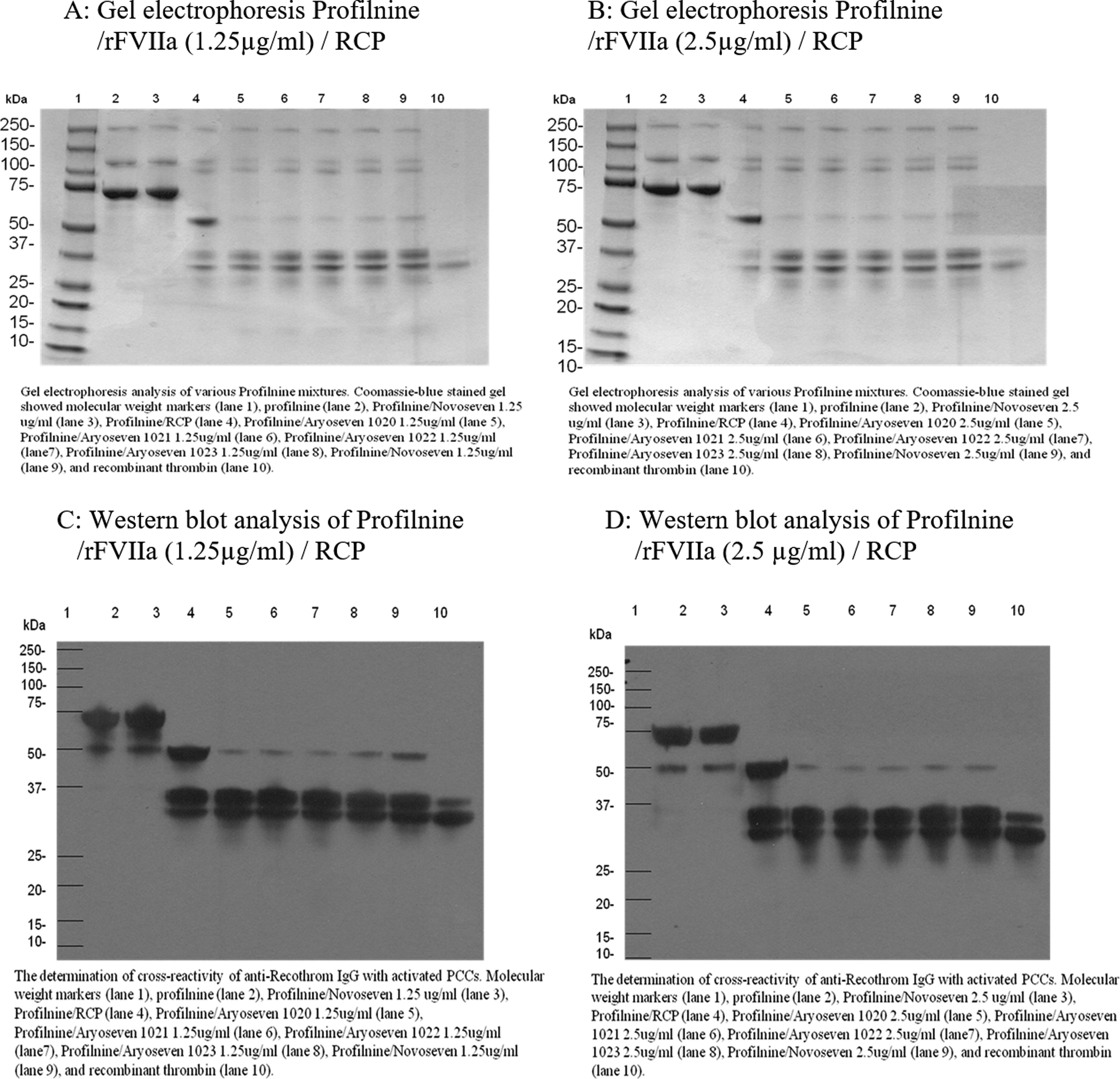
Gel electrophoretic profile and Western blot analysis of Profilnine/rFVIIa/ RCP, 30-minute incubation time. RCP indicates recombiplastin; rFVIIa, recombinant factor VIIa.
Similar experiment was performed on 4-factor PCC, Beriplex. Figure 2A exhibits SDS-PAGE analysis of various Beriplex mixtures containing rFVIIa (FC: 0.25 µg/mL) incubated for 30 minutes loaded in gel as follows: molecular-weight markers (lane 1), Beriplex (lane 2), Beriplex/NovoSeven JV60316 (lane 3), Beriplex/RCP (lane 4), Beriplex/AryoSeven 1020/RCP (lane 5), Beriplex/AryoSeven 1021/RCP (lane 6), Beriplex/AryoSeven 1022/RCP (lane 7), Beriplex/AryoSeven 1023/RCP (lane 8), Beriplex/NovoSeven JV60316/RCP (lane 9), and control recombinant thrombin (lane 10). Coomassie blue staining revealed that native Beriplex exhibited an intense 75-kDa band, which may indicate prothrombin and other intense band around 66 kDa that may represent albumin (lane 2). Mixture of rFVIIa and Beriplex showed similar profile to native Beriplex (lane 3). Activation of Beriplex with RCP resulted in generation of new bands at 36 kDa (lane 4). Mixture of Beriplex–RCP–rFVIIa exhibited similar protein profile with bands at 36 kDa (lane 5-9). Generation of thrombin was confirmed, which was comparable to the control thrombin (lane 10). Similar protein profile was observed when 2.5 µg/mL of rFVIIa (FC: 0.5 µg/mL) was supplemented in Beriplex mixtures as shown in Figure 2B.
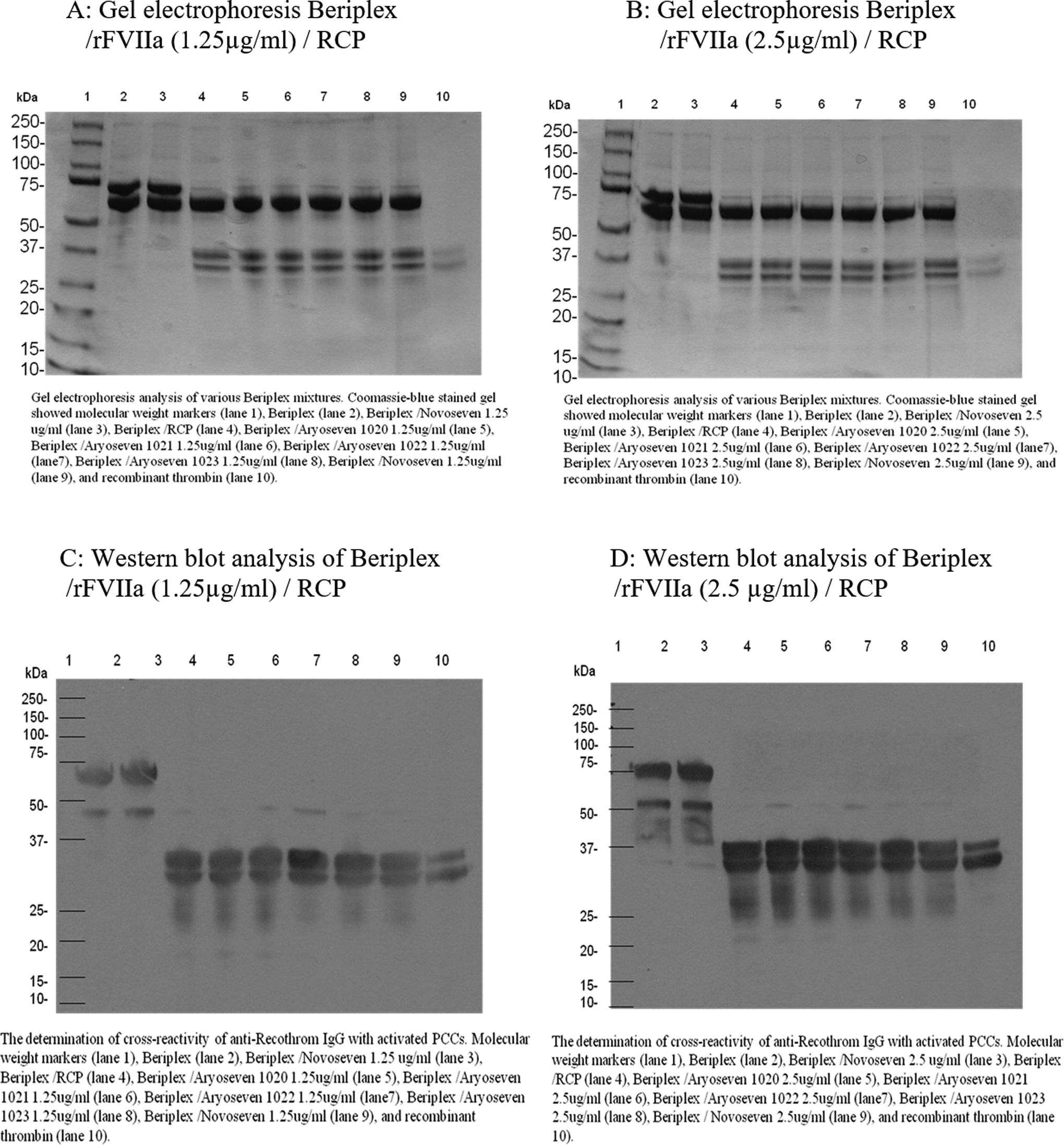
Gel electrophoretic profile and Western blot analysis of Beriplex/rFVIIa/RCP, 30-minute incubation time. RCP indicates recombiplastin; rFVIIa, recombinant factor VIIa.
After utilizing different concentration of rFVIIa for the generation of thrombin in 3- and 4-factor PCCs, similar experiments were performed with fixed concentration of rFVIIa (FC: 0.1 µg/mL) and various incubation time points. This was done to understand the time necessary for complete generation of the thrombin and potency of different rFVIIa in the generation of thrombin. In addition, different batches of branded and biosimilar rFVIIa were utilized to compare their effect on generation of thrombin upon activation of PCCs.
Figure 3A shows gel electrophoretic analysis of various Profilnine mixtures containing rFVIIa (FC: 0.1 µg/mL) incubated for 5 minutes loaded in gel as follows: molecular-weight markers (lane 1), Profilnine (lane 2), Profilnine/RCP (lane 3), Profilnine/AryoSeven 1026/RCP (lane 4), Profilnine/AryoSeven 1027/RCP (lane 5), Profilnine/AryoSeven 1028/RCP (lane 6), Profilnine/NovoSeven CS6C150/RCP (lane 7), and control recombinant thrombin (lane 8). Coomassie blue staining revealed that native Profilnine SD exhibited an intense 75-kDa band, which may indicate prothrombin and 2 unidentified faint bands at 250 and 140 kDa (lane 2). Activation of Profilnine with RCP resulted in the generation of new bands at 50 and 36 kDa (lane 3). Mixture of Profilnine–RCP–rFVIIa exhibited similar protein profile with intense bands at 50 kDa and faint bands at 36 kDa (lanes 4-7). Generation of thrombin was confirmed, which was comparable to the control thrombin (lane 8). Figure 3B depicts similar Profilnine mixtures with 1 exception, which is incubation time of 15 minutes. The 50-kDa bands were almost disappear as the incubation time increased from 5 to 15 minutes.
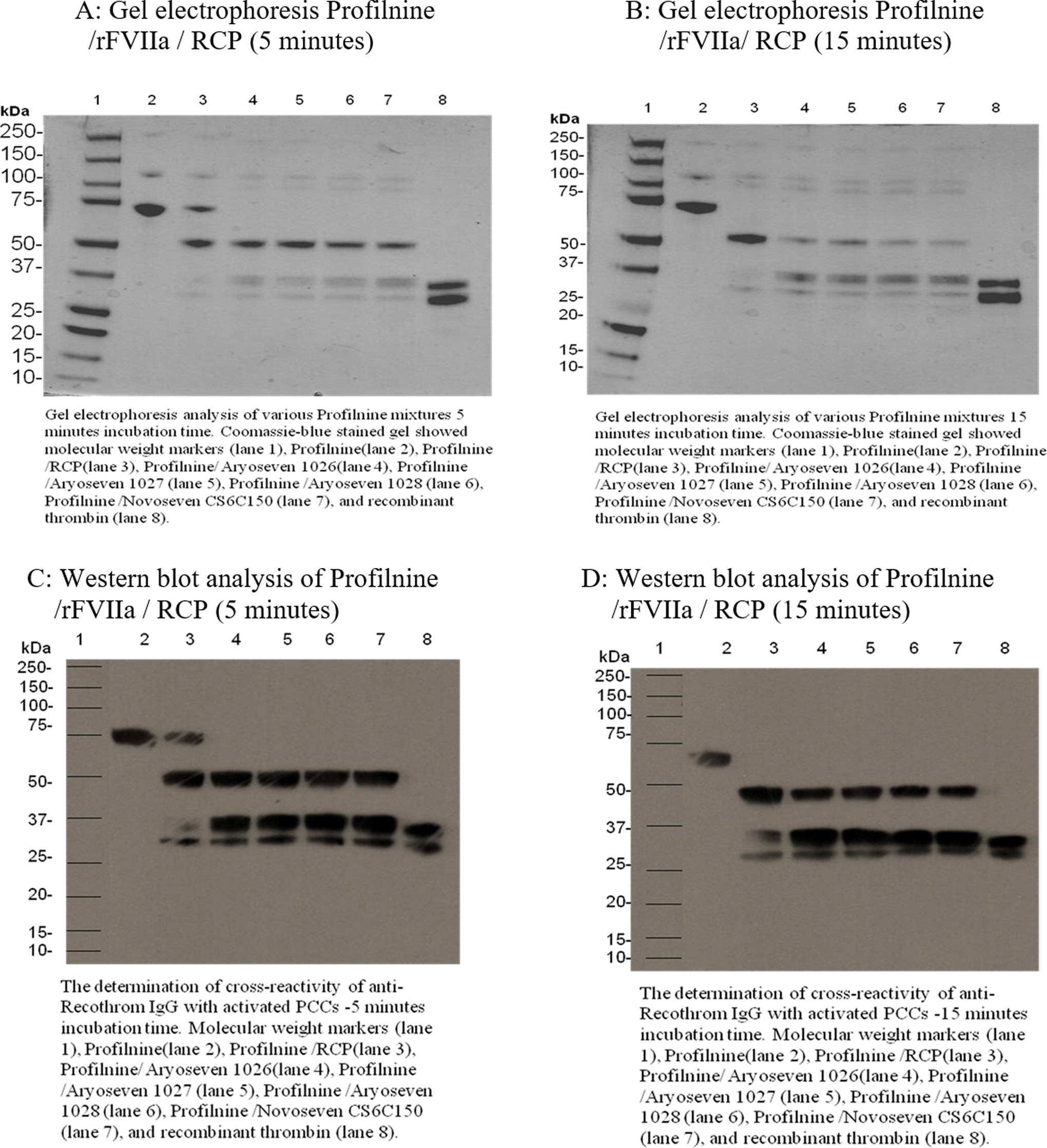
Gel electrophoretic profile and Western blot analysis of Profilnine/rFVIIa 0.5 µg/mL/RCP, 5- and 15-minute incubation time. RCP indicates recombiplastin; rFVIIa, recombinant factor VIIa.
Figure 4A shows gel electrophoretic analysis of various Beriplex mixtures containing rFVIIa (FC: 0.1 µg/mL) incubated for 5 minutes loaded in gel as follows: molecular-weight markers (lane 1), Beriplex (lane 2), Beriplex /RCP (lane 3), Beriplex/AryoSeven 1026/RCP (lane 4), Beriplex/AryoSeven 1027/RCP (lane 5), Beriplex/AryoSeven 1028/RCP (lane 6), Beriplex/NovoSeven CS6C150/RCP (lane 7), and control recombinant thrombin (lane 8). Coomassie blue staining revealed that native Beriplex exhibited an intense 75-kDa band, which may indicate prothrombin and other intense band around 66 kDa that may represent albumin (lane 2). Activation of Beriplex with RCP resulted in the generation of new band at 36 kDa (lane 3). Mixture of Beriplex–RCP–rFVIIa exhibited similar protein profile with intense bands at 36 kDa (lanes 4-7). Generation of thrombin was confirmed, which was comparable to the control thrombin (lane 8). Figure 4B depicts similar Beriplex mixtures with 1 exception, which is incubation time of 15 minutes. There was no difference in protein profile with different incubation time.
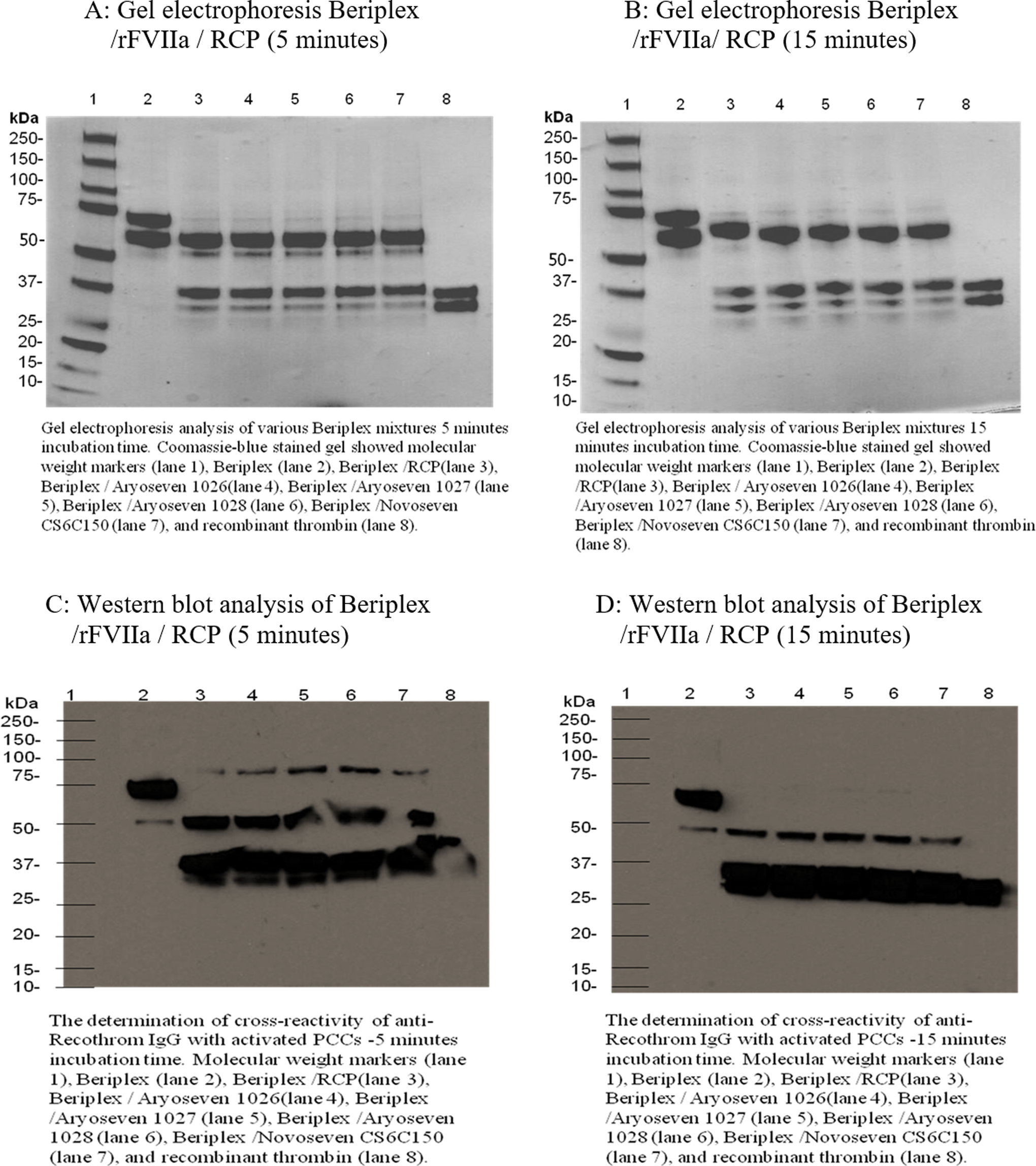
Gel electrophoretic profile and Western blot analysis of Beriplex/rFVIIa 0.5 µg/mL/RCP, 5- and 15-minute incubation time. RCP indicates recombiplastin; rFVIIa, recombinant factor VIIa.
Immunoblotting Studies
The immunoblotting studies were performed to confirm the generation of thrombin in both native and activated Profilnine and Beriplex with different concentrations of various batches of rFVIIa and incubation times. Figure 1C and D shows Western blot analysis of Profilnine in the presence of rFVIIa with different concentrations while incubated for 30 minutes: the molecular-weight markers (lane 1), Profilnine (lane 2), Profilnine/NovoSeven JV60316 (lane 3), Profilnine/RCP (lane 4), Profilnine/AryoSeven 1020/RCP (lane 5), Profilnine/AryoSeven 1021/RCP (lane 6), Profilnine/AryoSeven 1022/RCP (lane 7), Profilnine/AryoSeven 1023/RCP (lane 8), Profilnine/NovoSeven JV60316/RCP (lane 9), and control recombinant thrombin (lane 10). Activation of Profilnine with RCP generates 2 bands of 50 and 36 kDa (lane 4). Addition of various rFVIIa with different concentrations diminishes the amount of prethrombin and increases thrombin (lanes 5-9).
Similar experiment was performed on 4-factor PCC, Beriplex. Figure 2C and D exhibits Western blot analysis of various Beriplex mixtures containing 2 different concentrations of rFVIIa incubated for 30 minutes: molecular weight markers (lane 1), Beriplex (lane 2), Beriplex/NovoSeven JV60316 (lane 3), Beriplex/RCP (lane 4), Beriplex/AryoSeven 1020/RCP (lane 5), Beriplex/AryoSeven 1021/RCP (lane 6), Beriplex/AryoSeven 1022/RCP (lane 7), Beriplex/AryoSeven 1023/RCP (lane 8), Beriplex/NovoSeven JV60316/RCP (lane 9), and control recombinant thrombin (lane 10). Western blot analysis revealed that native Beriplex exhibited an intense 75-kDa band, which may indicate prothrombin and other intense band around 66 kDa that may represent albumin (lane 2). Mixture of rFVIIa in both concentrations and Beriplex showed similar profile to native Beriplex (lane 3). Activation of Beriplex with RCP resulted in the generation of new bands at 36 kDa (lane 4). Mixture of Beriplex–RCP–rFVIIa exhibited similar protein profile with bands at 36 kDa (lane 5-9). Generation of thrombin was confirmed, which was comparable to the control thrombin (lane 10).
Figure 3C and D shows Western blot analysis of various Profilnine mixtures containing rFVIIa (FC: 0.1 µg/mL) incubated for 5 and 15 minutes, respectively: molecular-weight markers (lane 1), Profilnine (lane 2), Profilnine/RCP (lane 3), Profilnine/AryoSeven 1026/RCP (lane 4), Profilnine/AryoSeven 1027/RCP (lane 5), Profilnine/AryoSeven 1028/RCP (lane 6), Profilnine/NovoSeven CS6C150/RCP (lane 7), and control recombinant thrombin (lane 8). Western blot analysis revealed that native Profilnine SD exhibited an intense 75-kDa band, which may indicate prothrombin and 2 unidentified faint bands at 250 and 140 kDa (lane 2). Activation of Profilnine with RCP resulted in the generation of new bands at 50 and 36 kDa (lane 3) in 5- and 15-minute incubation time. The only difference was disappearance of 75-kDa band in 15-minute incubation time. Mixture of Profilnine–RCP–rFVIIa exhibited similar protein profile with intense bands at 50 kDa and faint bands at 36 kDa (lane 4-7). Generation of thrombin was confirmed, which was comparable to the control thrombin (lane 8).
Figure 4C and D shows Western blot analysis of various Beriplex mixtures containing rFVIIa (FC: 0.1 µg/mL) incubated for 5 and 15 minutes: molecular-weight markers (lane 1), Beriplex (lane 2), Beriplex/RCP (lane 3), Beriplex/AryoSeven1026/RCP (lane 4), Beriplex/AryoSeven 1027/RCP (lane 5), Beriplex/AryoSeven 1028/RCP (lane 6), Beriplex/NovoSeven CS6C150/RCP (lane 7), and control recombinant thrombin (lane 8). Western blot analysis revealed that native Beriplex exhibited an intense 75-kDa band, which may indicate prothrombin and other band around 50 kDa that may represent prethrombin (lane 2). Activation of Beriplex with RCP resulted in the generation of new band at 36 kDa (lane 3). Mixture of Beriplex–RCP–rFVIIa exhibited similar protein profile with intense bands at 36 kDa (lanes 4-7). Generation of thrombin was confirmed, which was comparable to the control thrombin (lane 8). At 5-minute incubation time, both 75- and 50-kDa bands were present, while at 15-minute incubation time, 75-kDa bands completely disappeared and more prethrombin bands converted to thrombin bands.
Discussion
The comparative studies carried out on the 3 batches of AryoSeven clearly indicate that the individual batches of AryoSeven are comparable. The studies carried out on the 3 batches of AryoSeven and 1 batches of NovoSeven to determine thrombin generation in an activated PCCs also revealed comparable results.
A comparison of the thrombin generation in activated Profilnine in the presence of rFVIIa with both final concentrations of 0.25 and 0.5 µg/mL in 30-minute incubation time with different batches of AryoSeven and NovoSeven in the SDS-PAGE analysis under reduced condition revealed a distinct doublet at 36-kDa bands in all batches of AryoSeven and NovoSeven. Several faint bands were visible in the molecular-weight range of 50 to 250 kDa. The distinct 50-kDa band in activated Profilnine with RCP almost disappeared by addition of rVIIa at both concentrations. The relative intensity of these bands was also comparable. Although rFVIIa improved the generation of thrombin in activated Profilnine, addition of rFVIIa to native Profilnine did not generate any thrombin. On the other hand, activation of Beriplex, 4-factor PCC, with RCP resulted in complete generation of thrombin in 30-minute incubation time even with rFVIIa at both concentrations of 0.25 and 0.5 µg/mL. The relative intensity of these bands was also comparable and no differences between 2 concentrations.
The effect of different concentrations of rFVIIa at 30-minute incubation times was similar between biosimilar and brand rFVIIa in the generation of thrombin in both PCCs. To reveal an effect of incubation time on the generation of thrombin with a fixed final concentration of rFVIIa (0.1 µg/mL), SDS-PAGE was carried out. This study revealed 2 distinct bands at 50 and 36 kDa, which were comparable in all products upon activation of Profilnine. A faint band at 75 kDa at 5-minute incubation time in the presence of RCP disappeared upon addition of rFVIIa. Shorter incubation time prevented complete conversion of prothrombin to thrombin as evidenced by prethrombin band at 50 kDa in the presence of rFVIIa upon supplementation in activated Profilnine. Intensity of prethrombin bands was comparable in both rFVIIa preparations and higher in 5-minute incubation time. At 5-minute incubation time, activated Beriplex exhibited faint band of prothrombin at 75 kDa even in the presence rFVIIa. Also, 3 distinct bands at 66, 50, and 36 kDa were observed, which represent albumin, prethrombin, and thrombin bands, respectively. At 15-minute incubation time, a faint band at 50 kDa and high-intensity bands at 66 and 36 kDa were seen. Generation and band intensity of prethrombin and thrombin were comparable between biosimilar and brand rFVIIa preparations. The 36-kDa band in the presence of generation of thrombin from prothrombin has been previously reported in a study to represent thrombin on the immunoblotting human and bovine thrombin. 27
In order to further investigate the effect of AryoSeven and NovoSeven preparations with final concentration of 0.25 and 0.5 µg/mL in 30-minute incubation time in generation of thrombin upon supplementation in activated Profilnine, immunoblotting studies were carried out. This study revealed a strong band at 36 kDa and a very faint band at 50 kDa, which were comparable in all products. Similar results under same condition were observed when rVIIa supplemented in activated Beriplex. At 5- and 15-minute incubation time with rFVIIa (FC: 0.1 µg/mL), 2 distinct bands at 50 and 36 kDa were seen, which were comparable in both rFVIIa products upon supplementation in activated Profilnine. At 5-minute incubation time, trace prothrombin bands were seen in all sample mixtures in addition to distinct bands at 50 and 36 kDa. The prethrombin band at 75 kDa completely disappeared after supplementation of RCP or RCP/rFVIIa to Beriplex in 15-minute incubation time. The intensity of prethrombin band decreased when incubation time increased from 5 to 15 minutes. The thrombin generation and intensity of bands were similar in both rFVIIa preparations.
The results reported in this study utilized different rFVIIa concentrations and incubation time to compare the effect of biosimilar and brand rFVIIa in the generation of thrombin in activated 3-factor and 4-factor PCCs. In all of the studies carried out, the 2 products exhibited similar profile. Thus, the biosimilar version of NovoSeven named the AryoSeven appears chemically and biologically equivalent to NovoSeven.
Although the clinical data on the comparison of NovoSeven and AryoSeven are somewhat limited, 2 recent studies have reported on the safety and efficacy of the 2 products in parallel investigations. 24,25 In these studies, the investigators have reported similar safety and efficacy of the 2 products in patients having hemophilia with inhibitors and in patients with congenital FVII deficiency. The results confirmed the differential activation of PCC similar to the previous study. 14 However, both NovoSeven and AryoSeven exhibited similar activation profiles. Taken together, the reported study further validates the biosimilarity of the 2 rFVIIa products. Our studies warrant further investigations and postmarketing surveillance data to collect additional information on the similarity of the 2 products.
Footnotes
Acknowledgments
The authors acknowledge the help of technical assistance of the staff of the Hemostasis and Thrombosis Research Laboratories of the Department of Pathology in facilitating these studies. The authors are also grateful to Dr John Fanikos for his advice and support to these studies. Finally, the authors wish to thank Dr Eva Wojcik, Chairman of Department of Pathology at the Loyola University Medical Center, for her support and providing resources to carry out these studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
