Abstract
Introduction:
This study compared the efficacy of Aryoseven with Novoseven to control bleeding episodes in patients with hemophilia A with inhibitors.
Methods:
Sixty-six patients were randomized into 2 groups, with 4 consecutive block randomization. These groups received Aryoseven and Novoseven dosages of 90 to 120 μg/kg intravenously every 2 hours.
Results:
Median (interquartile range) level of factor VIII (FVIII) inhibitor in groups A and B was 15.0 and 19.0 Bethesda Unit (BU) preadministration. Bleeding onset in group A was 1246 ± 1104 minutes and in group B was 2301 ± 1693 minutes (P = .311). The Kavakli global response scores and treatment success rate was comparable in both the groups. The side effects in groups A (9.7%) and B (2.9%) were comparable.
Conclusion:
Biosimilar recombinant activated FVII is found to be as effective as Novoseven in the treatment of acute joint bleeding in patients with hemophilia with inhibitors. Its usage will decrease the gaps in hemophilia.
Introduction
Hemophilia A represents a commonly observed hereditary coagulation disorders. 1 The blood levels of functional procoagulant factor VIII (FVIII: C) is reduced to less than coagulation level. 2,3 The cardinal signs of hemophilia are bleeding episodes mainly hemarthrosis. 3 –7 In some of the congenital coagulation disorders, there is no relationship between bleeding rate and plasma factor and inhibitor level. Some genetic and thrombophilic factors are involved. 3,4 Factor replacement is the main strategy for treatment. 5 –10 Hemophilia care consists of on-demand treatment for bleeding episodes or prophylaxis for patients with FVIII level less than 1% to maintain factor level at more than 1% for prevention of severe bleeding episodes. 9 –13 Quality of life of patients with hemophilia has improved with current treatment options. However, complications are still important issues. 8,12 –15 Approximately one-third of patients with severe hemophilia A will develop neutralizing alloantibodies called an inhibitor directed against to the product with FVIII. 13,14 Developing an inhibitor is a major cause of morbidity and one of the most serious and costly complications of hemophilia. 14 –16 Inhibitors most often appear during the first year of therapy, most of them on prophylactic treatment, but they can occur at any time. 15 –17 Inhibitors are measured by the Bethesda Unit (BU) assay and clinically are classified as low- and high-responder inhibitors. 16 –18 Hemophilia with inhibitors remains a special challenge. 14 –21 Treating options for patients with inhibitor consist of high-dose clotting factor concentrates, use of bypassing agents (recombinant activated FVII [rFVIIa] and factor eight inhibitor bypass activator), immune tolerance induction (ITI) therapy, and plasmapheresis with immunoadsorption. 13 –24
Additionally autoantibodies (acquired inhibitors) may develop in nonhemophiliacs, resulting in acquired hemophilia A. 25 Acquired inhibitors arise mainly in adults, pregnancy, autoimmune diseases, and hematologic neoplasms (chronic lymphatic leukemia and lymphoma). 25 –33 The treatment goal of patients with inhibitors is to control the bleeding episodes and neutralize the inhibitor by ITI strategy. 34 –39
Treatment of patients with hemophilia with inhibitors rFVIIa (Novoseven) has added a new dimension to hemophilia management. 38 –44 The rFVIIa is successfully administered for patients with inhibitor. 1,2 Generally rFVIIa does not reverse hemophilia but causes thrombin generation. 38 –42 It is recommended as first-line therapy in bleeding episodes of patients with hemophilia with high-responder inhibitor. 36 –44
According to Transplantation and Special Disease Center in Iran and also Iranian Comprehensive Hemophilia Care Center (ICHCC) data registry, 7128 patients were registered with hereditary bleeding disorder. Hemophilia A has been reported in 4304 patients. 45 Studies from Iran reported that 184 (14.4%) of the 1280 patients developed inhibitor. In accordance to unpublished data by ICHCC and Iranian ministry of health, there were 323 patients with hemophilia with inhibitor. 45,46 In March 1999, Novoseven received Food and Drug Administration approval letter to be used for treatment of bleeding episodes in patients with hemophilia A or B with inhibitors to FVIII or factor IX. 40,47
Aryoseven is a biosimilar version of Novoseven. 40 In this study, we compared therapeutic outcome, side effects, and safety of rFVIIa product (Aryoseven) with Novoseven in patients with hemophilia A with inhibitor.
Patients and Methods
Trial Design
This trial was a double-blind, prospective randomized, multicenter study in 8 comprehensive hemophilia care centers around Iran under the direct supervision of Iranian Blood Transfusion Organization (IBTO). Randomization tables were designed based on 4 blocks of consecutive balanced approach between the 2 groups (group A: Aryoseven as the intervention group and group B: Novoseven as the control group). We designed a case report form (CRF) to gather patient’s data including demographic information, medical history as well as physical examination, and laboratory results in each visit by face-to-face interview. Patients with hemophilia A with inhibitors against FVIII are not to be evaluated in terms of treatment efficacy in controlling bleeding by objective criteria. The elevated plasma FVII (FVII: C) after injection of rFVIIa does not mean effectiveness. We evaluated effectiveness of the rFVIIa in treatment of hemarthrosis episodes by subjective criteria, which is named Kavakli Global Response Scoring System. 48 This scoring system is based on patient’s self-assessment. 48 Eligible patients were randomized to receive rFVIIa (Aryoseven or Novoseven) 90 µg/kg as a starting dose intravenously, for bleeding episode followed by administration 2 consecutive 120 µg/kg doses to control the bleeding every 2 hours. 48 Sixty-six patients were enrolled and allocated to the 2 groups by consecutive balanced block randomization tables (fixed block size of 4) per hemophilia center. Group A (31 patients, 53%) received Aryoseven and group B (35 patients, 47%) received Novoseven.
Ethical Consideration
The study was approved by ethics committee of IBTO and the ministry of health in agreement with Declaration of Helsinki and good clinical practice. The trial was registered on the Iranian Registry of Clinical Trials (IRCT) Web site with registration number 201202106302N2.
Global Response Scoring System
The global response scoring system by Kavakli et al worked based on patient’s self-assessment of pain and mobility of joint at 1, 3, 6, and 9 hours after the first injection. 48 The global response to therapy was efficient if the patient had 6 or more positive scores 48 (Table 1).
Global Response Scoring System.
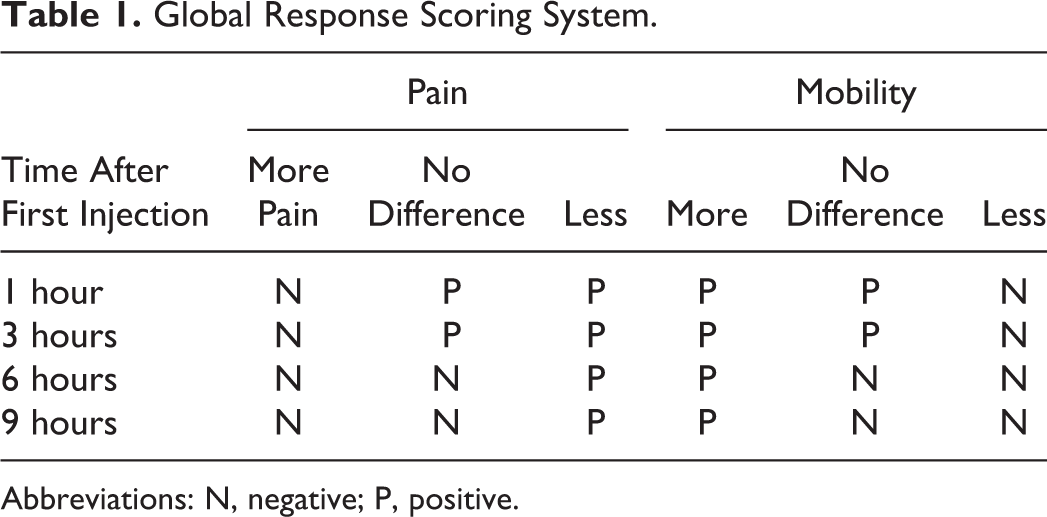
Abbreviations: N, negative; P, positive.
Patient Eligibility
Inclusion criteria were patients with hemophilia A with inhibitor level more than 5 BU having a bleeding episode, age older than 2 years, and severity of bleeding is a guesstimate. Exclusion criteria were any other coagulation disease, ITI therapy during the last month, history of severe atherosclerosis, and platelet count <50 000 per μL.
Written informed consent was signed by all patients or their legal guardians. Consenting patients who met these criteria were randomly assigned to an intervention or a control group. Patients met inclusion criteria by scientific committee of clinical trial team eligible for study. Patients in the intervention group (group A) received the biosimilar rFVIIa (Aryoseven) and patients in the control group (group B) received Novoseven.
Laboratory Methods
Factor VII clotting activity (FVII: C) was measured by 1-stage clotting method using recombinant-tissue factor preparation (STA-VII deficient and STA-Neoplastin; Diagnostica Stago, France). The FVIII inhibitor was measured by classic “Bethesda assay,” and the inhibitor titer was reported in “BU.”
Outcomes and Safety
The primary outcome was reducing pain and improving joint movement on Kavakli global response scoring system. Secondary outcome was increase in plasma FVII: C 20 minutes after the first injection. Treatment safety was analyzed by evaluation of adverse events reported throughout the study period.
Statistical Analysis
Participants’ data were collected in the CRF during the study, and their accuracy was monitored by the study supervisor to ensure their adherence to study protocol. Data were analyzed by SPSS software (version 16.0; SPSS Co, Chicago, Illinois) and was described based on their statistical analysis. Quantitative data were expressed as mean ± standard deviation, and qualitative data were expressed as frequency and proportion (percentage). Factor VII level (FVII: C) and global response scoring system did not exhibit a normal distribution; therefore, these variations were presented as median and interquartile range (IQR). Quantitative data to compare the case and control groups were analyzed utilizing parametric t test for data with normal distribution and parametric Mann-Whitney U test for data with abnormal distribution (FVII levels and global response scoring system). Independent qualitative data were compared using chi-square test and if necessary Fisher exact test, and dependent qualitative data were compared using the McNemar test. The standard level of statistical significance was considered to be P ≤ .05.
Results
During the study, 66 patients were enrolled in 2 arms. All patients were male. The mean age was 20.5 ± 10.9 years (range: 2-53 years). Comparison of baseline data between the 2 groups regarding age, time interval between onset of bleeding to start treatment, and inhibitor levels showed no significant difference. The distributions of these variables were similar between the 2 groups (Table 2). Other factors, such as vital signs (respiratory rate, heart rate, blood pressure, and body temperature), coagulation tests (prothrombin time and partial thromboplastin time), liver and renal function tests, and blood count were not significantly different between the 2 groups. Median (IQR) plasma level of FVII clotting activity (FVII: C) in groups A and B was 103.8 ± 38.4 and 98.6 ± 26.7 IU/dL before injection, respectively. Median (IQR) plasma level of FVIII inhibitor in groups A and B was 15.0 BU (IQR: 8.8-58.0) and 19.0 BU (IQR: 11.0-31.0), respectively. The average time from onset of bleeding to start treatment was 1246 ± 1104 and 2301 ± 1693 minutes (P = .311) in groups A and B, respectively (Table 2). This may be due to differences in patient care protocols at different hospitals.
Comparison of Baseline Characteristics in 2 Treatment Groups.a
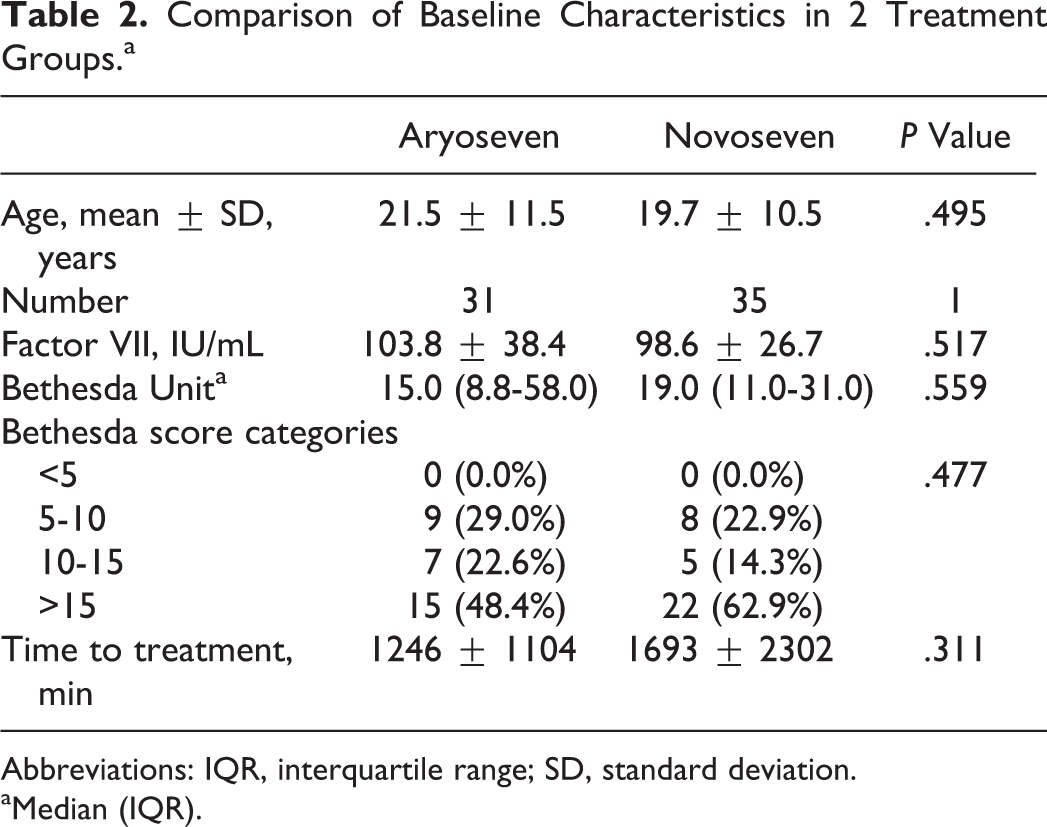
Abbreviations: IQR, interquartile range; SD, standard deviation.
aMedian (IQR).
The results of this study showed that increased levels of FVII were comparable between 2 groups after rFVIIa injection (Table 3). A comparison of the Kavakli global response scores after injection and the treatment success rate in terms of achieving to score 6 or higher showed that both groups were comparable in treatment success rates (Table 4). The global treatment response rate was 96.8% in group A and 91.4% in group B. Administration of either Aryoseven or Novoseven had comparable effect on controlling pain and joint mobility (Tables 5 and 6). Comparison of side effects reported during the treatment between the 2 groups is shown in Table 7. Reported side effects were minor (headache, nausea, and rash) and occurred in similar frequency. The side effects occurred in 3 (9.7%) patients of group A and in 1 (2.9%) patient of group B. Both groups were comparable in terms of number and type of side effects, and there was no significant difference between the 2 groups (Fisher exact test). No withdrawal or discontinuation of treatment was occurred during the study time, and no severe side effects were observed during the study time until 1 year after the injection of Aryoseven.
Comparison of Factor VII Plasma Level (FVII: C) Before and After rFVIIa Injection Between 2 Groups.

Abbreviations: IQR, interquartile range; rFVIIa, recombinant activated factor VII.
aAll units is IU/dL.
bData are shown as median (IQR).
Kavakli Score and the Rate of Treatment Success in the 2 Therapy Groups.
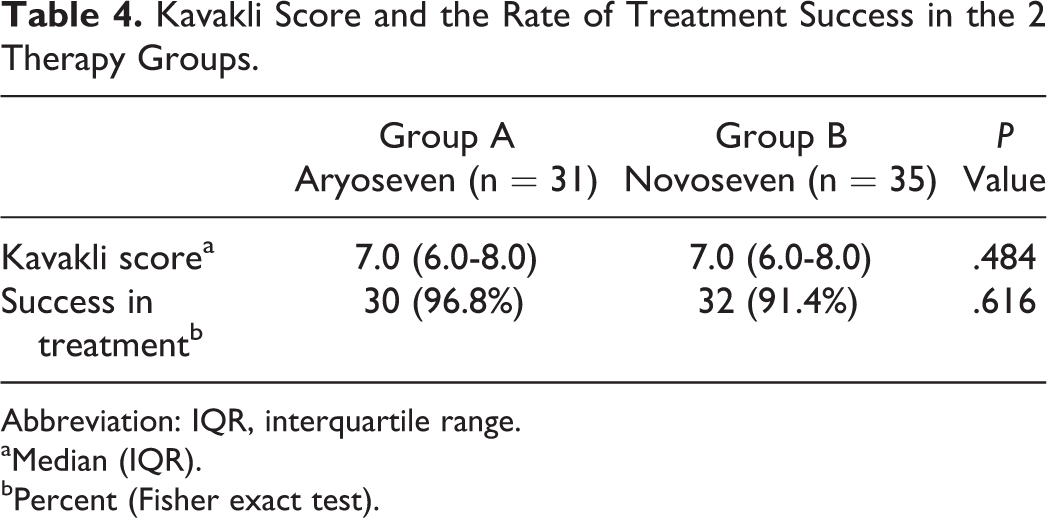
Abbreviation: IQR, interquartile range.
aMedian (IQR).
bPercent (Fisher exact test).
Pain After Treatment in 2 Therapy Groups.
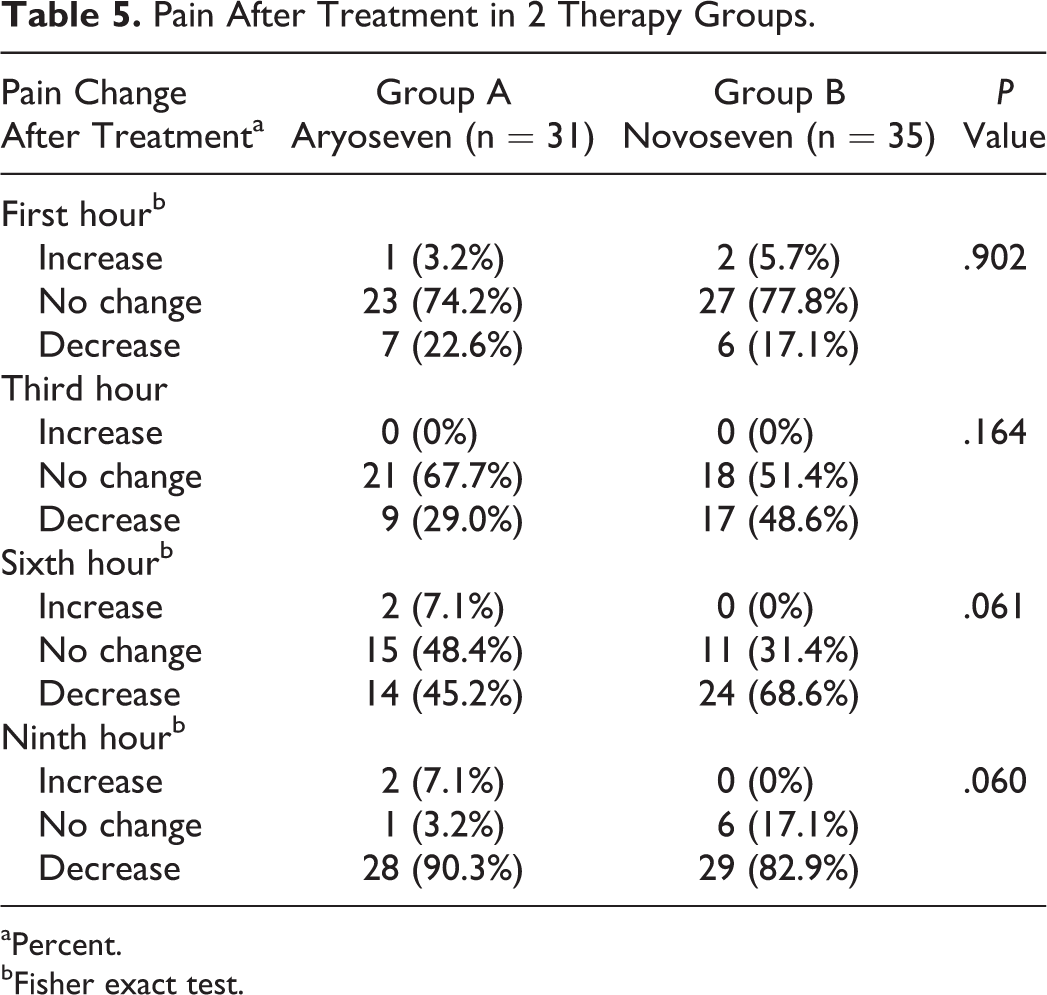
aPercent.
bFisher exact test.
Joint Range of Motion After Treatment in 2 Therapy Groups.
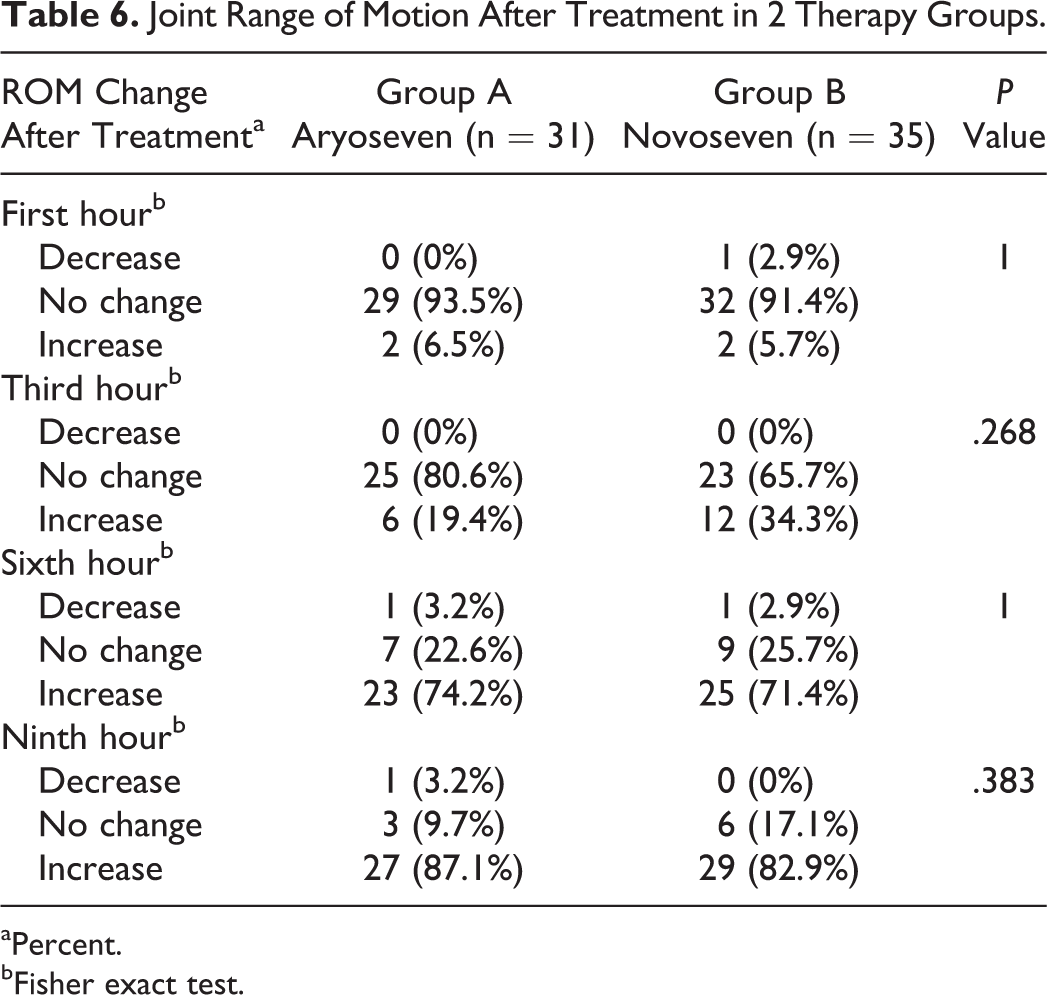
aPercent.
bFisher exact test.
Types of Side Effects Observed During Treatment Period.
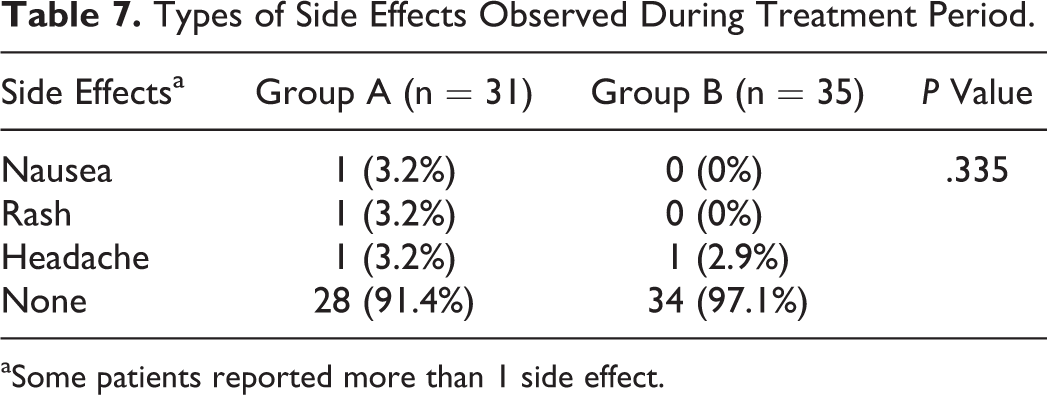
aSome patients reported more than 1 side effect.
Discussion
The development of biosimilar products has met global acceptance and has a major impact on the management of various diseases. 49 –53 The biosimilar FVIIa provides an affordable mean of otherwise costly branded product. 50 –52 The primary reason for production of biosimilar drugs is reduction in the overall health care cost to design better comprehensive management and closing the gap between the poor and developed countries. 52–53
Double-blind, comparative clinical trials were designed for biosimilar rFVIIa (Aryoseven) in terms of clinical efficacy and noninferiority to Novoseven. This study showed increased FVII levels and clinical efficacy in the control of the bleeding episodes in patients with hemophilia A with inhibitor for both the drugs. The definition of efficacy was to measure pain reduction, improve joint range of motion, and bleeding cessation. The first treatment strategy for bleeding episodes is the use of rFVIIa or activated prothrombin complex concentrate (aPCC). 54 –57 Knight et al in systematic review compared the efficacy of rFVIIa and PCC in several studies and showed that the efficacy of rFVIIa was 81% to 91% and aPCC was 64% to 80%. 56 The expert suggests that therapy of bleeding episodes in patients with FVIII inhibitor by rFVIIa has faster response compared to aPCC. 55 –59
In this study, baseline characteristics of patients were comparable to other studies considering the age, bleeding severity and FVII levels before and after intervention, and FVIII inhibitor plasma level. 9,14,56 The number of patients with hemophilia having inhibitor who were treated in most reported trials varied from 15 to 62; in the current study, the sample size was 66 patients. 56 –58 In most of the other main reported trials, double blinding had not been possible due to sample size. 54 –58 Some similarities exist between our findings and the previous clinical trials designed for Novoseven. 54 –56 Different doses of rFVIIa ranged from 75 to270 μg/kg, 2 hours apart, and have been used. We chose 90 to 120 µg/kg dosage, which is similar to other studies. 21,58 –64 Efficacy and reduction in bleeding were similar in both treatment groups. The definition of efficacy of hemostatics agents in most studies was a combination of reduction in bleeding, improvement in range of motion in the joint, and apparent alleviation of pain. 56,63 Evaluation of efficacy was designed by Kavakli et al, Shirahata et al, and Young et al, and we used Kavakli global response scoring system. 48,63,67 In other studies, the treatment success rate using the global response scoring system has been 70% to 86% by different rFVIIa dosage regimen (eg, 3 × 90 and 1 × 270 μg/kg), and in our study, it was above 90% in both treatment groups. 53 –58 In most studies, it was reported more than 90% achieving to cessation of bleeding at 9 hours after first injection of rFVIIa as response time. 53 –56 This was also similar to our study. We checked mean levels of FVII before and after administration of the 2 rFVIIa preparations to prove FVII: C level was comparably increased. 6,38 In the previous studies, the rate of reported adverse events was 1% to 10%, 50 –60 and the most of adverse events had been headache, rashes, hypertension, and rarely thrombosis. We did not encounter any thrombosis in either treatment groups. 28,38 Also, similar to other studies, our study did not show any serious adverse events, and the difference between the frequency of side effects in both treatment groups was not statistically significant. 5,29 Most adverse events in our study were mild and included headache, nausea, and vomiting.
This study showed noninferiority of Aryoseven compared to Novoseven in cessation of bleeding episodes in patients with hemophilia A with inhibitor. There was no evidence of significant difference between the efficacy and safety of 2 drugs.
Health-related quality of life is an important consideration in the management of chronic disease such as hemophilia. The modern treatment regimens in hemophilia with inhibitor help to decrease bleeding episodes, morbidity, and improve quality of life. 55 –64 New therapeutic strategies are relatively expensive, and they are not affordable for underdeveloped and poor countries. 40,64 –67
Biosimilar products are comparable hemostatic agents to the branded product and provide better care and wider affordability. 13,48 –53 Evidently there are no clinically meaningful differences between the biosimilar product and the original product in terms of the safety, purity, and efficacy. 40,48 –52 The World Federation of Hemophilia promotes treatment for all patients by making the clotting factors available at reasonable cost. 15
Conclusion
Aryoseven is similar to Novoseven in clinical efficacy, which was evaluated by Kavakli Global Response Scoring System as well as in postinjection FVII clotting activity (FVII: C). The frequency of side effects was also similar. However, the current study was underpowered to detect differences in rare complications between the 2 drugs. The results of this study warrant additional clinical investigations to demonstrate the similar efficacy and safety profile of Aryoseven in comparison to the branded Novoseven.
Footnotes
Acknowledgments
The authors thank the patients with hemophilia, staff in hospitals (Emam Khomeini, Mofid, Dastgheib, Shafa, seyed al shohada, Taleghani, and Afzalipoor), Aryogen Biopharma Company, Iranian Comprehensive Hemophilia Care centers and Iranian Blood Transfusion Organization for their help in conducting the present study. We are also thankful to Drs Massimo Iacobelli and Nasir Sadeghi, and reviewer of Clinical and Applied Thrombosis/Hemostasis for their review of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was granted by Aryogen Biopharma Company Tehran-Iran.
