Abstract
We evaluated the effectiveness and safety of prothrombin complex concentrate (PCC) in urgent warfarin reversal in Iran. This is a nonrandomized, pre–post intervention study. Thirty-seven high-risk patients with prolonged international normalized ratio ([INR] >8) or with bleeding symptoms were enrolled in this study. Prothrombin complex concentrate was infused with a dose of 22.4 ± 11.2 IU/kg. The INR was measured 30 minutes postinfusion. Mean age of the participants was 63.9 ± 14.5 (22-89 years). After 30 minutes of PCC injection, INR reversed to <3.5 in 31 patients (84%) in whom 12 patients achieved INR 1.5 to 2.5, and 19 patients achieved INR 2.5 to 3.5. Bleeding symptoms were subsided during 0.5 hour after PCC infusion in patients with hemorrhagic symptoms. Not any adverse reaction was observed in patients during 3-month follow-up. Our experience in use of single injection of PCC in high-risk patients with prolonged INR or with bleeding symptoms was successful without any complication.
Introduction
Warfarin was licensed as the first anticoagulant medication in the United States, and it has been remained as the most commonly used oral anticoagulant to date.1,2 Warfarin acts as a vitamin K antagonist and prevents the production of vitamin K-dependent procoagulant factors including factors (F) II, VII, IX, and X. There are some mechanisms causing warfarin overdose including high doses of administration, impaired protein binding, decreased vitamin K intake, reduced production or increased clearance of vitamin K-dependent coagulation factors, as well as the concurrent use of other drugs competing with warfarin for protein binding. 3 The most serious complication of anticoagulant therapy is intracranial hemorrhage (ICH). 4 Traditional method for reversal of warfarin therapy consisted of fresh frozen plasma (FFP) and administration of vitamin K. This method of therapy is not an idealistic method, due to infection risks, requirement for large amount of fluid and no rapid response particularly in case of high international normalized ratios (INRs). 5
Prothrombin complex concentrates (PCCs) produce a rapid action and is an effective method for replacement of the deficient coagulation factors and correcting INR. 5 Compared with FFP, the PCCs have no any risk of infection transmission, allergic reaction, transfusion-related lung injury, or volume overload, also, it can reverse markedly prolonged INR values more rapidly. 2 There are many evidences regarding safety and effectiveness of PCC in patients with hemophilia A who were complicated by acute episodes of bleeding. Only rare and mild adverse reactions including dizziness, nausea, hives, flushing, and headache were reported in using PCC in these patients.6–9
The successful use of PCCs to control bleeding caused by overanticoagulation therapy was first reported by Taberner et al. 10 Fortunately in the recent years, several prospective and retrospective studies have been designed to evaluate the efficacy and safety of PCC in urgent reversal of warfarin-induced bleeding. 1 Chong et al 11 and Imberti et al 4 reported their successful experiences on using PCC in patients complicated by ICH caused by oral anticoagulation therapy. Leissinger et al 5 conducted a review discussing the effect of PCC in warfarin reversal including 14 published studies with promising successful results. The reason for warfarin reversal in all articles in this review except one was mentioned as major bleeding or the need of rapid reversal in surgical setting. Considering the results of this review, single injection of PCC can reverse hemostatic effect of warfarin completely and immediately.
In Iran, we did not have any experience using PCC in reversing warfarin. So we decided to design a study evaluating the safety and effectiveness of PCC in high-risk patients who were on warfarin therapy and came with bleeding symptoms or prolonged INR (>8) for the first time in Iran.
Materials and Methods
This is a nonrandomized, pre–post intervention study. Thirty-seven patients with warfarin ongoing treatment were enrolled in this study. Patients were selected from high-risk patients whom referred with prolonged INR (>8) to the warfarin clinic as well as patients with bleeding symptoms who referred to emergency wards of hospitals affiliated to Shiraz University of Medical Sciences, after giving written informed consent. Inclusion criteria were having a positive history of ischemic heart disease (IHD), arrhythmia, congestive heart failure, valvular heart disease, or cerebrovascular accident. All patients had positive history of previous bleeding and all of them had come from far distance and they did not have access to an equipped tertiary clinical and diagnostic center in their residence.
Based on our clinical experience, we observed the occurrence of severe bleeding in some of these high-risk patients under warfarin therapy who had INR >8 and because all patients were living in places without developed diagnostic and therapeutic facilities, we considered 2 groups of these patients for evaluating the effect of PCC infusion including asymptomatic patients with INR >8 as well as patients with bleeding symptoms.
Exclusion criteria were disseminated intravascular coagulation, sepsis, pregnancy, breast feeding, and mental retardation. 4 Selection and monitoring of the patients were performed by hematologists based on the first assessment of the patients including medical history, physical examination, and determination of vital signs. The study protocol was approved by the Ethics Committee of Shiraz University of Medical Sciences.
Patients were infused 10 to 50 IU/kg of PCC (3 factors: FII, FIX, and FX Uman complex, Kedrion Company, Italy). The PCC dose was individualized based on clinical status, baseline INR, and weight of the patients. Dosage of PCC was 10 to 25 IU/kg in patients without bleeding symptoms and 25 to 50 IU/kg in patients with bleeding symptoms.
Concomitant therapy with whole blood, plasma, or plasma fractions was not allowed within the first 30 minutes after the PCC infusion, unless urgently required as judged by the attending clinician. Patients were assessed by clinical response and INR using Fisher Scientific Company kit. An INR control was performed 30 minutes after the end of PCCs infusion. Because all patients were at high risk, the primary end point of the study was determined as the proportion of patients who achieved normalized INR after 30 minutes from the first infusion. If target INR was not obtained at that time, further evaluation was done at 24 and 48 hours, and further management with PCC, FFP, or vitamin K was performed in case of bleeding based on the requirement for reversing anticoagulation and according to the attending physician’s clinical judgment. All patients were observed for at least 24 hours at the hospital after normalizing INR.
Patients were followed for 3 months for the occurrence of any adverse events mainly consisted of allergic reactions, thromboembolic complications, death, and recurrences of the hemorrhage. 4 Blood sampling was done for the determination of INR before infusion and at intervals of 0.5, 24, 48 hours afterward based on the requirement. Prothrombin time (PT), hemoglobin, and platelet count were checked also at baseline. In patients with bleeding symptoms, hemoglobin level was rechecked 24 hours after stop bleeding.
Data were analyzed by descriptive statistics for safety and efficacy using SPSS v. 17. Descriptive data were presented as mean ± standard deviation (SD) and median. Response rate was evaluated based on the proportion of patients who achieved target INR in 30 minutes of PCC infusion. Baseline PT and INR of the patients were compared with the values 30 minutes after infusion by paired samples t test. P value less than .05 was considered statistically significant.
Results
Mean age of the patients was 63.9 ± 14.5 (22-89 years) including 18 men and 19 women. Clinical diagnosis, underlying disease or associated risk factors, and frequency of bleeding sites are presented in Table 1. Fifteen patients (40%) had no any bleeding symptoms (INR > 8, mean ± SD: 9.6 ± 1.5, range: 8-15.3). In other group of patients (n = 22) who were complicated by hemorrhagic symptoms, mean baseline INR was 10.4 ± 4, and range was 4.6 to 16.9. In this group, all patients had mild to moderate bleeding except 2 patients who referred with Gastrointestinal Bleeding (melena).
Frequency of the Patients With Warfarin Toxicity With Regard to Diagnosis, Associated Risk Factors and Underlying Disease, and Bleeding Symptoms.
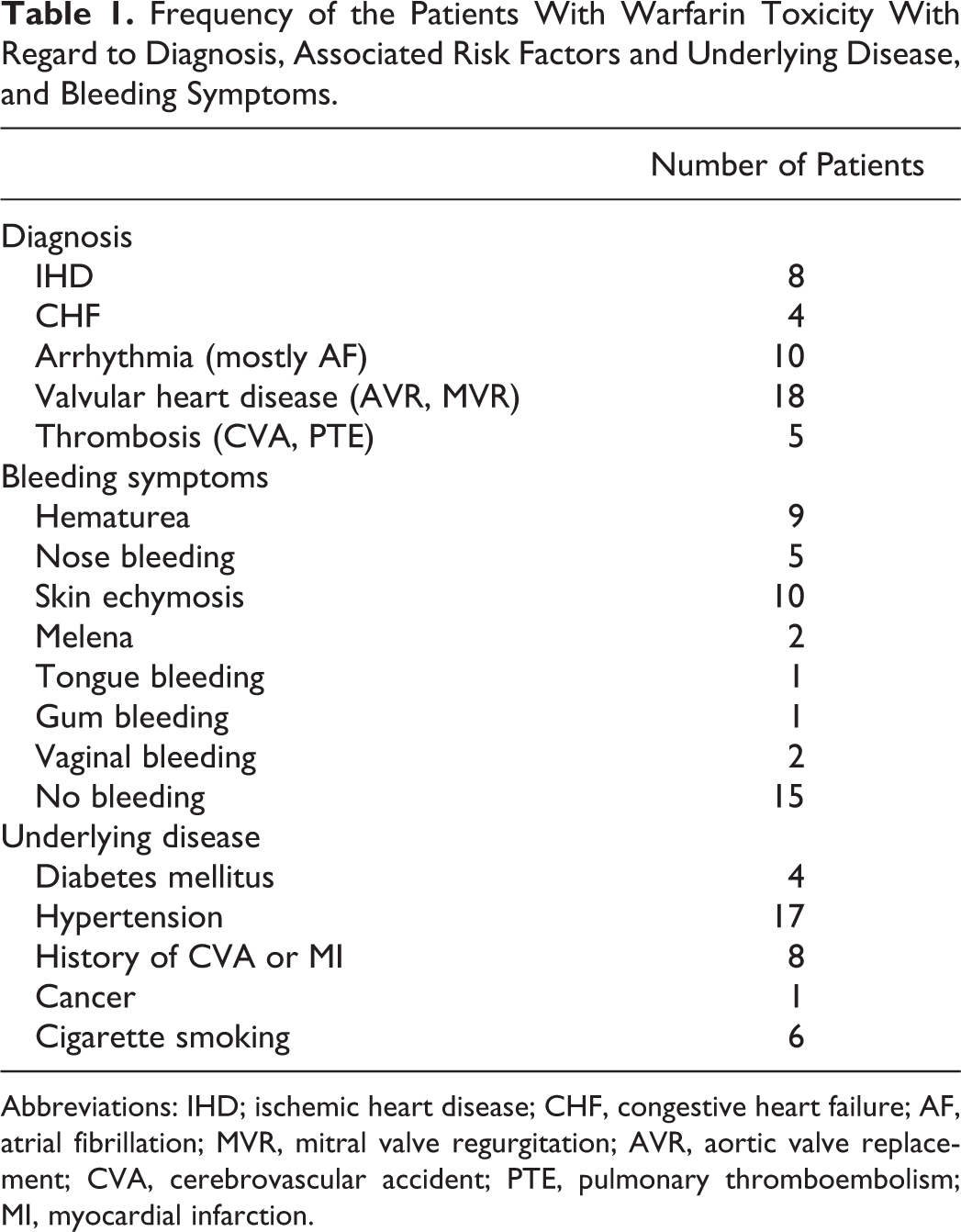
Abbreviations: IHD; ischemic heart disease; CHF, congestive heart failure; AF, atrial fibrillation; MVR, mitral valve regurgitation; AVR, aortic valve replacement; CVA, cerebrovascular accident; PTE, pulmonary thromboembolism; MI, myocardial infarction.
The PCCs were infused by mean ± SD of 22.4 ± 11.2 and range of 10 to 50 IU/kg (Table 2). After 30 minutes of PCC injection, INR reversed to <3.5 in 31 patients (84%): 12 patients achieved INR 1.5 to 2.5 and 19 patients achieved INR 2.5 to 3.5.
Laboratory Data and Treatment Characteristics of the Patients Referring With Warfarin Toxicity.a
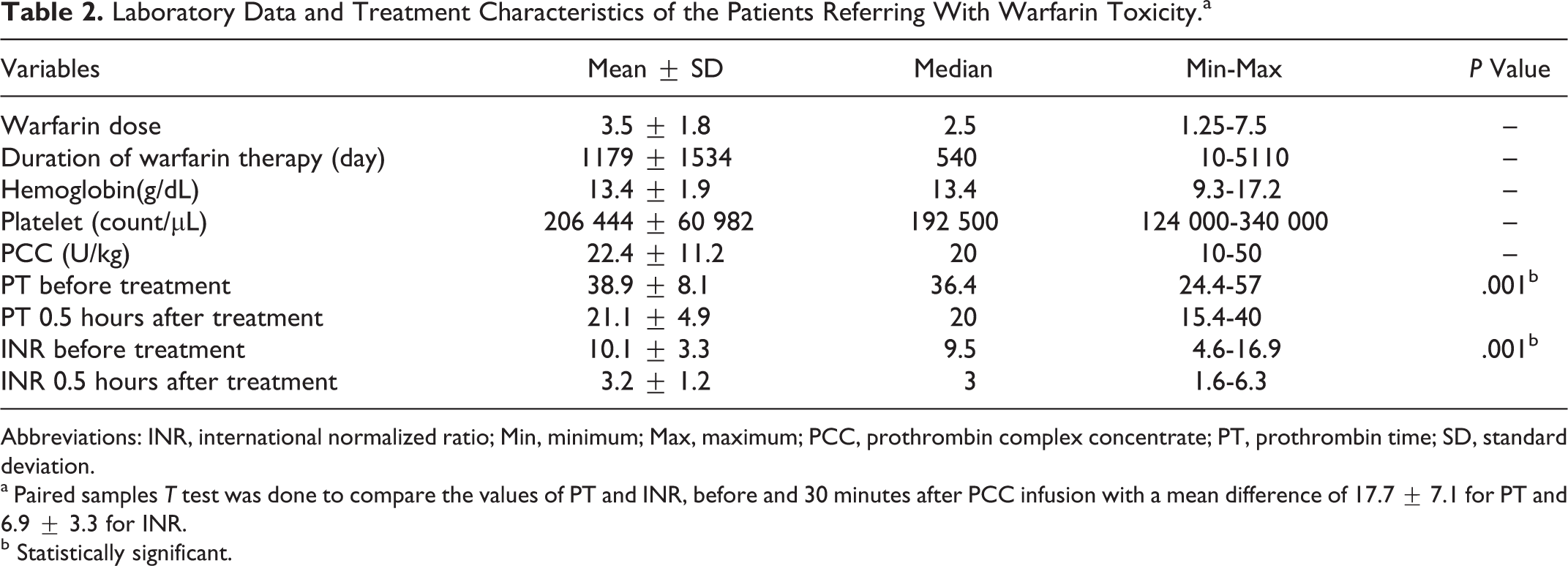
Abbreviations: INR, international normalized ratio; Min, minimum; Max, maximum; PCC, prothrombin complex concentrate; PT, prothrombin time; SD, standard deviation.
a Paired samples T test was done to compare the values of PT and INR, before and 30 minutes after PCC infusion with a mean difference of 17.7 ± 7.1 for PT and 6.9 ± 3.3 for INR.
b Statistically significant.
The mean INR values reversed to 3.2 ± 0.75 in the group of patients with no bleeding symptoms and to 3.2 ± 1.5 in patients with hemorrhagic symptoms after 30 minutes of PCC injection. Bleeding symptoms were subsided during 30 minutes after PCC infusion in this group. From the 6 patients with 30-minute post-INR values >3.5, 3 were normalized after 24 hours without the need for additional dose of PCC infusion. In other patients, management was individualized according to INR and clinical status of the patients as follow.
Patient 1
A 89-year-old man referred with atrial fibrillation (AF) and cardiomyopathy, without any bleeding symptoms, only headache with normal brain computed tomography scan, INR decreased from 14.8 to 6.3 and 5.6 after 30 minutes and 24 hours of the first PCC infusion, respectively, so warfarin was discontinued and 5 mg of vitamin K was administered but additional dose of PCC was not used due to AF. After 5 days, his INR value was 1.7.
Patient 2
A 80-year-old woman referred with IHD and skin bleeding. Thirty minutes after a single PCC injection (17 U/kg), INR decreased from 16 to 5.98. After 48 hours, INR increased to 7.77. Warfarin was discontinued and vitamin k was used (5 mg). After 3 days, his INR was 2.
Patient 3
A 73-year-old woman referred with IHD and skin bleeding. She was admitted in hospital and INR decreased from 8 to 7 after taking FFP and vitamin K; this patient was first excluded from the study protocol by mistake. But after that due to uncorrected INR and skin bleeding, she received a single dose of PCC injection (28 IU/kg) with a decrease in INR to 1.5 after 30 minutes.
Decrease in PT and INR of the patients after 30 minutes of PCC injection was statistically significant (P = .001 for PT and P = .008 for INR). In group of patients with bleeding symptoms, decrease in hemoglobin level in each patient was less than 0.5 g/dL 24 hours after the bleeding stopped.
No thrombotic complication was seen in any of these patients.
Discussion
Increasing use of long-term oral anticoagulant therapy has been resulted in higher occurrence of complications. Bleeding is the most important and frequent side effects of oral anticoagulant therapy. More caution should be considered especially in elderly patients to avoid overdosing complications. 12 We evaluated the safety and effectiveness of PCC in warfarin reversal in 37 high-risk patients with INR >8 or who were complicated by hemorrhage in southern Iran. This is the first experience using PCC for reversing warfarin in Iran. Good responses to single PCC injection (22.4 ± 11.2, range: 10-50 IU/kg) were achieved after 30 minutes in 84% and after 24 hours in 8% of patients without any adverse events. Our results are in agreement with the previous reports documenting the successful result of using PCC in correction of anticoagulation.13 –17 Similar to our results, Tran et al 18 investigated the safety and efficacy of PCC in patients who needed urgent warfarin reversal. They divided their patients to 2 groups: partial and complete reversal arms based on initial INR and patient weight. They showed 91% success rate by achieving 30-minute post-INR <1.4 in complete reversal arm and 93% success rate by achieving 30-minute post-INR 1.4 to 2 in partial reversal arm. Consistent with our results, no adverse events were observed in this group of patients. Imberti et al 4 showed successful result of PCC in INR correction without any adverse effects in patients with ICH (median initial and post-INRs were 3.3 and 1.4, respectively). Chiu et al 19 reported a successful and safe result of using PCC in patients who were undergoing vascular surgery or angiography. In all patients, baseline INR (2-3.9) was successfully reversed to INR 1 to 1.3. Also they did not report any side effects. Yasaka et al 20 investigated the effectiveness of PCC (10-60 minutes after infusion) in 42 patients with hemorrhagic complications or required interventional procedures. Based on their results they concluded that PCC with a dose of 500 IU could be effective for warfarin reversal in patients with INR level <5, but in INR levels >5 higher dose of PCC and/or vitamin K is needed to correct INR and keep the lower INR value for 12 to 24 hours. Pabinger et al 21 similarly conducted a study to evaluate the efficacy and safety of PCC (25-50 IU/kg) in a setting of emergency surgical or major bleeding. The INR level reversed to ≤1.3 after 30 minutes in 93% of patients. Lankiewicz et al 15 evaluated safety and efficacy of PCC (25-50 IU/kg) in urgent warfarin reversal in 58 patients from whom 62% presented with ICH. Immediately after PCC infusion, INR decreased to <1.5 in 76% and <2 in 96.5% of patients.
The difference between our study and previous studies is that our trial included high-risk patients with prolonged INR (>8) without any bleeding in addition to the patients with bleeding symptoms. Also, the mean baseline level of INRs in our patients was higher than that reported in previous studies. So the primary end point in our study was considered in a higher level (1.5-3.5) compared with the desired target INR in those studies (<1.5). Most of the related studies have been carried out in a setting of major bleeding with successful result or the need of rapid reversal in surgical setting without any major side effects. Based on the previous and our results, it seems that single injection of PCC can reverse hemostatic effect of warfarin completely and immediately. It remains only a question about the minimum effective and maximum safe dose of PCC in this regard. 5
Our study was limited due to low number of study participants and the lack of control group to compare the INR correction time between PCC and FFP or vitamin k. But our study, showed using PCC on warfarin reversal in high-risk patients is effective in short period of 30 minutes and safe with dose of 10 to 50 IU/kg without any complication.
Footnotes
Acknowledgments
We would like to thank Shiraz University of Medical Sciences for approval support and Kedrion Company for its support to do this research. We also thank Shirin Parand at the Hematology Research Center for help with manuscript preparation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: project No. 2399.
