Abstract
Introduction:
Improved life expectancy of persons with hemophilia (PWHs) has led to a greater interest in the role of age-related chronic diseases, such as hypertension, in this cohort. Several observational studies have reported an increased prevalence of hypertension in PWHs; however, this has not been assessed using a large, national database in the United States.
Aims:
We hypothesized the prevalence of hypertension is increased in PWHs and compared the prevalence of hypertension and associated risk factors among patients with and without hemophilia.
Methods:
A cross-sectional analysis was performed using discharge data among adult males from the National Inpatient Sample over the 3-year period, 2009 to 2011. Hypertension was compared across groups using Rao-Scott χ2 test. Multivariable logistic regression was used to estimate the odds of hypertension in patients with hemophilia after adjustment for hypertension-associated risk factors.
Results:
The prevalence of hypertension in patients with hemophilia was less than the prevalence of hypertension in patients without hemophilia (39.5% vs 56.3%, P < .001). Hemophilia was associated with a decreased odds of hypertension after adjusting for associated risk factors (odds ratio: 0.87; 95% confidence interval: 0.81-0.94).
Conclusion:
In contrast to the findings of several other recent studies, we report a decreased prevalence of hypertension in PWHs. The discrepancy among the reported prevalence of hypertension in our study and several others highlights the potential biases inherent to retrospective and cross-sectional studies and underscores the need for well-designed prospective studies to determine the true incidence of hypertension in PWHs, which may lie somewhere in between our findings and the findings of others.
Introduction
The availability of safe clotting factor preparations and effective antiviral therapies for chronic hepatitis C virus (HCV) and HIV infection has improved the life expectancy of those born with hemophilia from less than 30 to more than 60 years of age. 1 As a result of the increasing age of the hemophilia population, the role of cardiovascular disease (CVD) and associated risk factors, such as hypertension and other chronic age-related diseases, has become the subject of greater interest.
Recently, several observational studies have assessed the role of hypertension in hemophilia. Two retrospective studies in the United States found an increased prevalence of hypertension in persons with hemophilia (PWHs). 2,3 Similarly, 2 cross-sectional studies from the Netherlands reported comparable findings. 4,5 Several hypotheses have been postulated to explain the etiology for the reported increased prevalence of hypertension in PWHs. 6,7 Hypertension may be detected more often than in the general population, given the frequency and intensity of the ongoing medical care and follow-up necessary for hemophilia. Furthermore, concomitant HCV or HIV infection, chronic kidney disease, kidney bleeding, and clotting factor concentrates are potential causal or contributing factors, but no data are available at this time to support these theories.
Hypertension contributes to nearly 350 000 deaths and costs the nation close to $50 billion each year and may lead to hypertensive heart disease, including heart failure, coronary artery disease and myocardial infarction, and cerebrovascular disease and stroke. 8 The treatment of such complications invariably involves the use of aspirin and/or other antiplatelet agents or anticoagulants, all of which may cause bleeding and are relatively contraindicated in PWHs. Therefore, it is imperative to more fully define the role of hypertension in hemophilia and the potential etiologies responsible for this phenomenon. To our knowledge, the prevalence of hypertension in hemophilia has not been assessed using a large, national database in the United States. We hypothesized that the prevalence of hypertension is increased in PWHs and aimed to compare the prevalence of hypertension and associated risk factors among patients with and without hemophilia using the National Inpatient Sample (NIS).
Methods
This was a cross-sectional analysis using discharge data among adults from the NIS for the most recent 3-year period available, 2009 to 2011. The NIS is a 20-percent sample of discharges from all community hospitals, excluding rehabilitation and long-term, acute-care hospitals, participating in the Healthcare and Utilization Project. It represents more than 95% of the US population. The NIS contains information that is included in a typical discharge abstract, such as primary and secondary diagnoses (new and established), procedures, patient demographics, and length of stay. It is used to make national estimates of health-care utilization, access, charges, quality, and outcomes. 9 The large sample size of this database enables analyses of rare conditions, such as hemophilia.
Discharge weights are applied to unweighted discharge data from the NIS sample to produce discharge-level estimates for all community hospitals in the United States. The NIS hospitals are first stratified according to geographic region, urban/rural location, teaching status, bed size, and ownership. A weight is calculated for each stratum by dividing the number of discharges in that stratum, obtained from American Hospital Association, by the number of NIS discharges in the stratum. Weighted estimates are calculated by uniformly applying stratum weights to the discharges according to the stratum from which the discharge was drawn. 9
Records were identified in the NIS by International Classification of Diseases, Ninth Revision codes for the following: hemophilia, hypertension, atherosclerotic heart disease, myocardial infarction, ischemic stroke, hyperlipidemia, diabetes mellitus, obesity, smoking, renal failure, hematuria, proteinuria, HCV, and HIV (Table 1). 10
ICD-9 Codes.
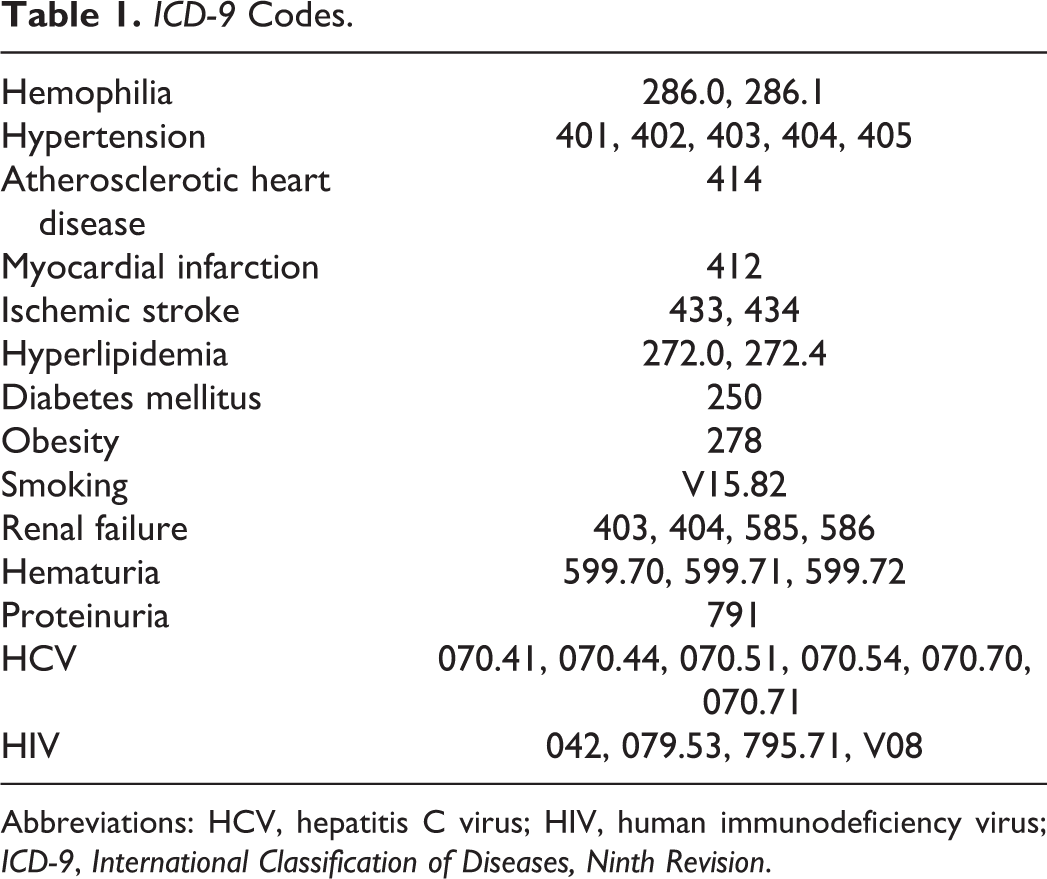
Abbreviations: HCV, hepatitis C virus; HIV, human immunodeficiency virus; ICD-9, International Classification of Diseases, Ninth Revision.
The prevalence of hypertension was estimated among patients with and without hemophilia. Demographics (age and race), length of stay, inpatient mortality, and medical comorbidities (atherosclerotic heart disease, myocardial infarction, ischemic stroke, hyperlipidemia, diabetes mellitus, obesity, smoking, renal failure, hematuria, proteinuria, HCV, and HIV) were compared between patients with and without hemophilia. Comparisons across groups were made for categorical and continuous variables using the Rao-Scott χ2 test and weighted simple linear regression, respectively. Univariate logistic regression was used to estimate the odds of hypertension in patients with hemophilia compared to patients without hemophilia. Similarly, multivariable logistic regression was used to estimate the odds of hypertension in patients with hemophilia compared to patients without hemophilia after adjustment for age, atherosclerotic heart disease, myocardial infarction, hyperlipidemia, diabetes mellitus, obesity, smoking, HCV, and HIV. Clinically relevant covariates were chosen for inclusion in the regression analysis. Among patients with hemophilia with and without hypertension, demographics, length of stay, inpatient mortality, and medical comorbidities were compared.
Results
During the 3-year period from January 1, 2009, to December 31, 2011, there were 3607 hemophilia-related hospital discharges and 8 025 025 nonhemophilia hospital discharges (Table 2). Hypertension was less common in patients with hemophilia than in patients without hemophilia (39.47% vs 56.32%; P < .001; Table 2). Atherosclerotic heart disease, myocardial infarction, ischemic stroke, hyperlipidemia, diabetes mellitus, obesity, smoking, and renal failure were less frequent in the hemophilia group than the nonhemophilia group (Table 2). Further, HCV, HIV, and hematuria were more common in PWHs than persons without hemophilia. In univariate logistic regression analysis, hypertension was less likely in patients with hemophilia than patients without hemophilia (odds ratio [OR]: 0.50; 95% confidence interval [CI]: 0.47-0.54). After fitting a multivariable logistic regression model that adjusts for age, atherosclerotic heart disease, myocardial infarction, hyperlipidemia, diabetes mellitus, obesity, smoking, HCV, and HIV, the odds of CVD remained lower in the hemophilia group than the nonhemophilia group (OR: 0.87; 95% CI: 0.81-0.94; Table 3). Among patients with hemophilia, patients with hypertension were older and more commonly African American than patients without hypertension (Table 4). Atherosclerotic heart disease, myocardial infarction, ischemic stroke, hyperlipidemia, diabetes mellitus, obesity, smoking, and renal failure were more frequent in the hypertension group than the nonhypertension group.
Prevalence of HTN and Associated Comorbidities in Patients With and Without Hemophilia.
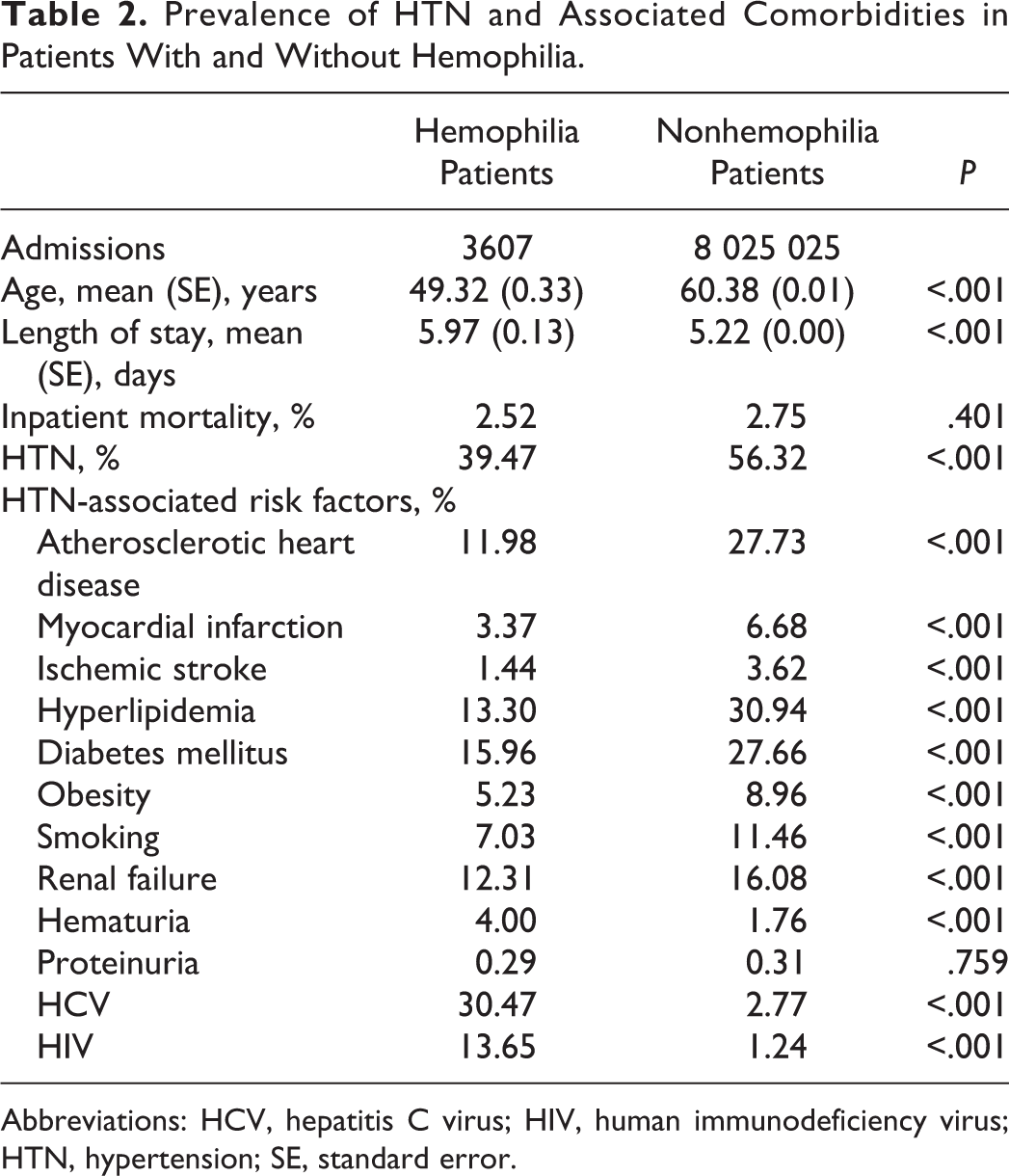
Abbreviations: HCV, hepatitis C virus; HIV, human immunodeficiency virus; HTN, hypertension; SE, standard error.
Odds of HTN Among Patients With Hemophilia.
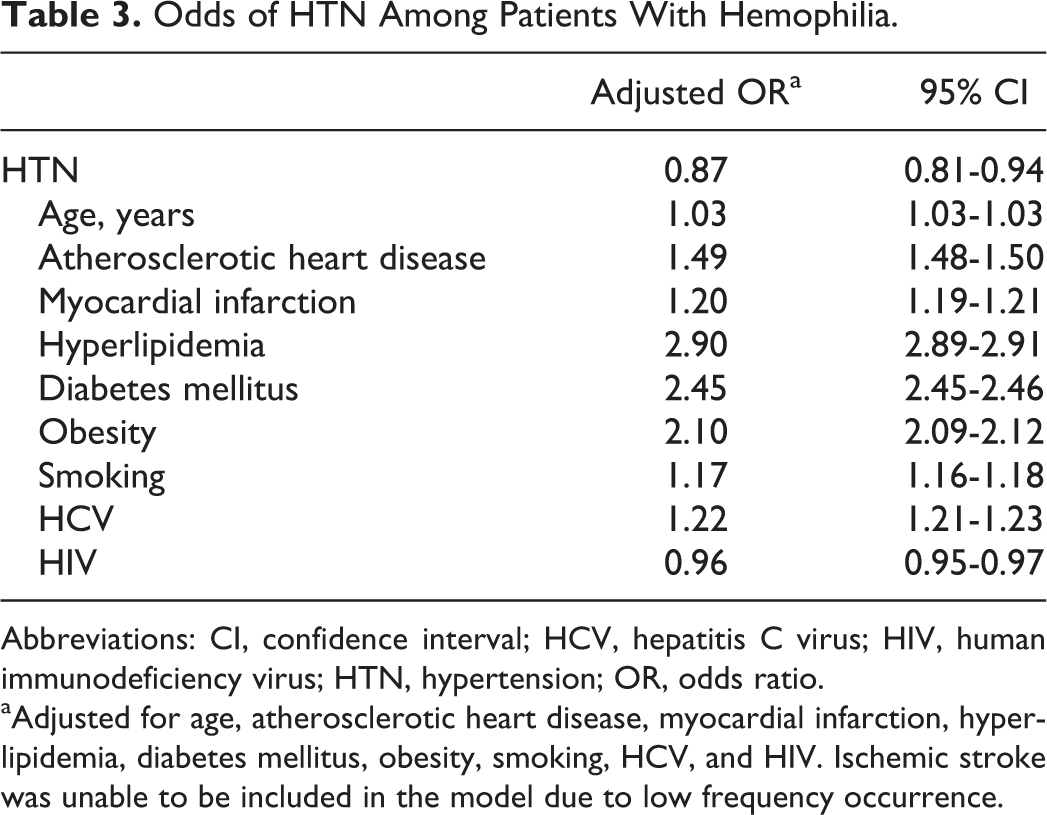
Abbreviations: CI, confidence interval; HCV, hepatitis C virus; HIV, human immunodeficiency virus; HTN, hypertension; OR, odds ratio.
aAdjusted for age, atherosclerotic heart disease, myocardial infarction, hyperlipidemia, diabetes mellitus, obesity, smoking, HCV, and HIV. Ischemic stroke was unable to be included in the model due to low frequency occurrence.
Prevalence of HTN and Associated Comorbidities in Patients With Hemophilia.
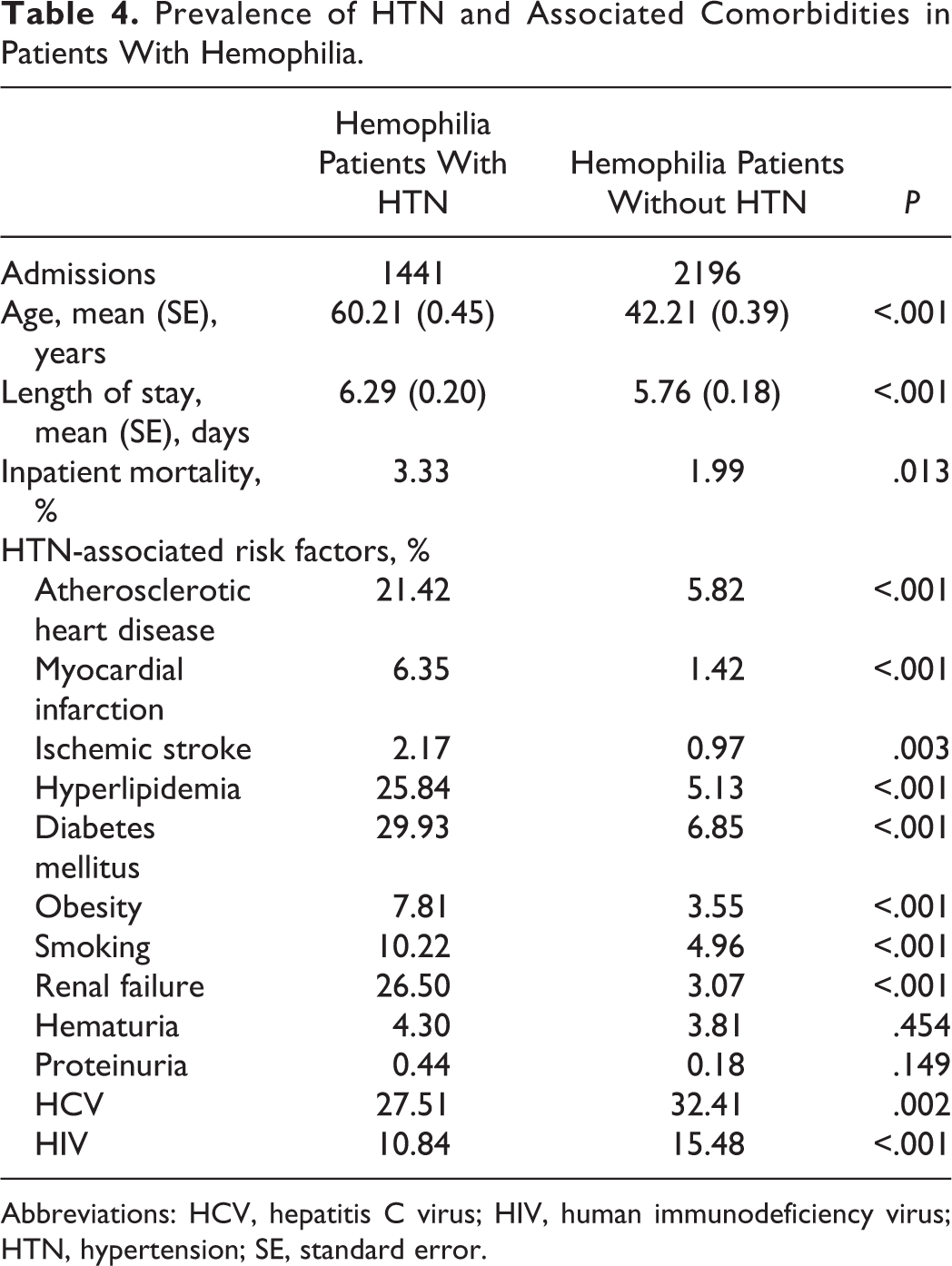
Abbreviations: HCV, hepatitis C virus; HIV, human immunodeficiency virus; HTN, hypertension; SE, standard error.
Discussion
This cross-sectional analysis demonstrates the prevalence of hypertension in PWHs is 17% lower than patients without hemophilia. Moreover, following adjustment for hypertension-associated risk factors, the odds of hypertension in patients with hemophilia is 13% lower than patients without hemophilia. These findings are unexpected given the increased prevalence of hypertension in hemophilia reported in several recent studies (Table 5).
Prevalence of Hypertension in Hemophilia.
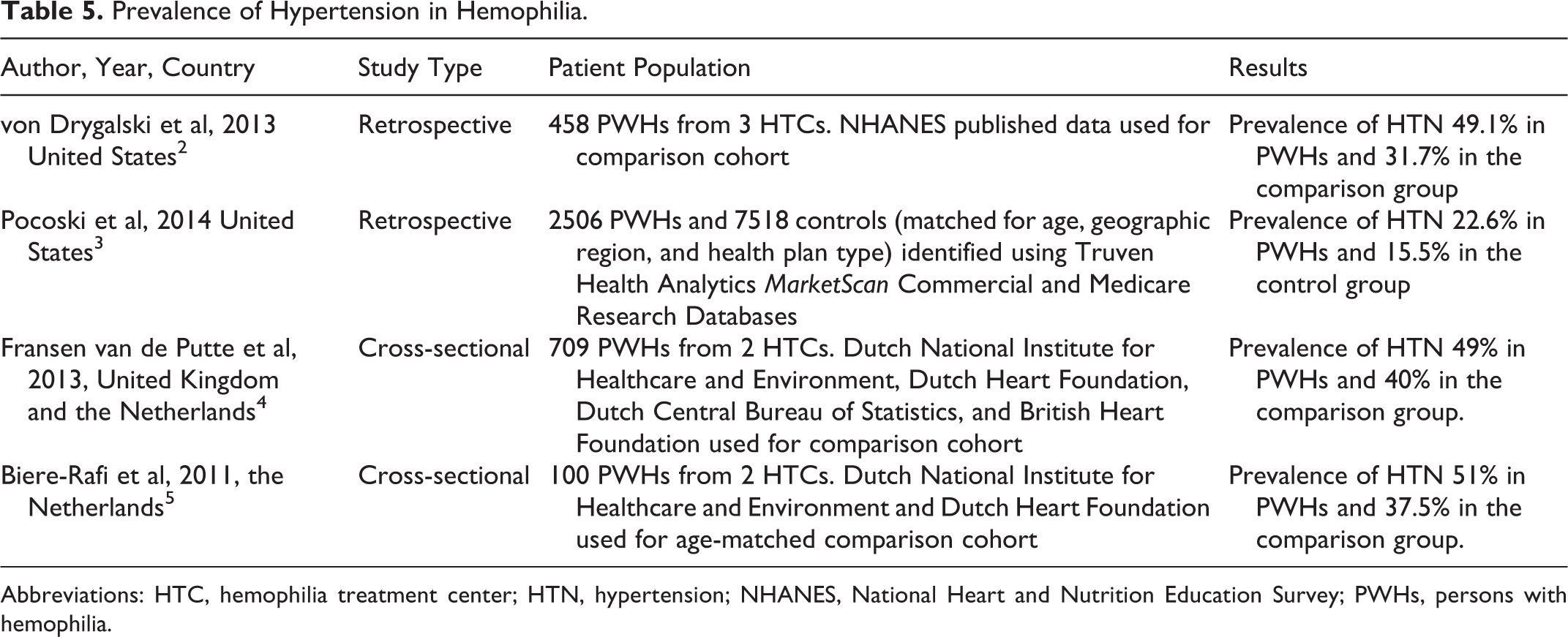
Abbreviations: HTC, hemophilia treatment center; HTN, hypertension; NHANES, National Heart and Nutrition Education Survey; PWHs, persons with hemophilia.
In a retrospective database analysis of 458 PWHs from 3 US hemophilia treatment centers (HTCs) between 2004 and 2012, von Drygalski et al found the prevalence of hypertension was significantly higher in the hemophilia cohort (49.1%) than the general population by the National Health and Nutrition Examination Survey 2 (31.7%). In a second US retrospective database analysis of 2506 PWHs and 7518 age-matched controls using inpatient discharge and outpatient claims diagnoses between 2007 and 2009, Pocoski et al reported an increased prevalence of hypertension (22.6 vs 15.5%) in the hemophilia cohort. 3 Similarly, a European cross-sectional study of a prospective cohort study with 709 PWHs aged 30 years and older at 2 HTCs in the Netherlands and United Kingdom between 2009 and 2011 reported hypertension was more common in PWHs compared with age-matched controls (49 vs 40%). 4 Another European cross-sectional study of 100 PWHs at 2 HTCs in the Netherlands performed by Biere-Rafi et al found hypertension to be more prevalent in the hemophilia cohort compared with age-matched controls with rates of 51.0% and 37.5%. 5 The above studies, including ours, report a wide range of prevalence of hypertension in PWHs between 22.6% and 51.0%. A comparably wide range of values is reported in the control groups, with prevalence of hypertension between 15.5% and 56.3%. These discrepancies are likely the result of differences in study design, which include retrospective and cross-sectional studies and patient population. Patients were selected from distinct geographic regions, United States and Northern Europe, and medical settings, inpatient versus outpatient or a combination of both.
Interestingly, our study found a decreased prevalence of atherosclerotic heart disease, myocardial infarction, and ischemic stroke in patients with hemophilia compared to patients without hemophilia. The prevalence of CVD in hemophilia is uncertain, given conflicting evidence in the literature; however, it appears although PWHs have a similar prevalence of atherosclerosis, cardiovascular events, such as myocardial infarction, and mortality are reduced. 11 –16 It is hypothesized that the inability to generate sufficient amounts of thrombin may account for these findings. 6 It must be pointed out that our study was not designed to determine the prevalence of CVD and our findings do not account for potential confounders, such as diabetes mellitus, obesity, smoking, and so on, which are more common in the nonhemophilia cohort.
This study has a few limitations that warrant discussion. First, discharge diagnoses were used as a means of patient identification, which requires coding accuracy to minimize the potential for misclassification bias. Despite these concerns, a systematic review of studies evaluating the accuracy of hospital discharge data found that the accuracy of collected data is sufficiently robust to support its use in research studies. 17 Second, the NIS uses hospital encounters, that is, admissions, to determine each unit of analysis, rather than patient encounters; therefore, multiple admissions for the same patient cannot be excluded. Third, it is not possible to eliminate the potential for selection bias. Hospitalized patients may not be representative of the general patient population and may be indicative of a subset of chronically ill patients with a higher prevalence of medical comorbidities. This appears to be the case in the nonhemophilia cohort, in which the prevalence of hypertension is 56%, which is much greater than the approximately 33% reported by the National Health and Nutrition Examination Survey. 18 The hemophilia cohort is exposed to the same selection bias; therefore, our findings may be truly representative of a decreased prevalence of hypertension in PWHs. Fourth, it is not possible with the cross-sectional design of the study to determine causality; however, the odds of hypertension remained less in the hemophilia cohort after adjustment for multiple potential confounders. Fifth, it is not possible with the NIS database to answer important questions, such as whether the severity of hemophilia predicts the prevalence of hypertension. This is important as hypertension may be detected more frequently in persons with severe hemophilia since they often require a greater amount of health care. A major strength of our study is the use of a large, population-based database. A major obstacle in performing clinical research in rare diseases, such as hemophilia, is the limited pool of potential patients available for study. Using the NIS, we were able to identify 3607 hemophilia-related discharges and show the prevalence of hypertension is less in PWHs compared with patients without hemophilia. The large size of the NIS database used in our study ensures the ability to detect true differences between groups.
In conclusion, our study is the first to report a decreased prevalence of hypertension among a large cohort of patients with hemophilia in the United States. This is in contrast to the findings of several other recent studies. The discrepancy among the reported prevalence of hypertension in these studies highlights the potential biases inherent to retrospective and cross-sectional studies. These findings underscore the need for well-designed prospective studies to determine the true prevalence of hypertension and related comorbidities, such as CVD, in PWHs, which may lie somewhere in between our findings and the findings of others. This is imperative given the aging hemophilia population, and the clinical dilemmas encountered balancing bleeding and thrombotic risks when these diseases coexist.
Footnotes
Authors’ Note
CS, MA, and MVR designed the research. CS, MA, MVR, JY, and DC performed the research. DC and JY performed the statistical analysis. CS, MA, MVR, JY, and DC analyzed the data and formulated the conclusions. CS, MA, MVR, JY, and DC wrote the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by University of Pittsburgh Cancer Institute.
