Abstract
Background:
Platelet adhesion is mediated by von Willebrand factor (vWF), and disintegrin-like and metalloprotease domain with thrombospondin type-1 motif, number 13 (ADAMTS13) is a protease that cleaves vWF. A change in the balance between vWF and ADAMTS13 in favor of thrombosis might occur shortly before ischemic cardiovascular (CV) events.
Objective:
To determine whether vWF, ADAMTS13, and the ratio of vWF and ADAMTS13 change during the months preceding an acute CV event.
Design:
Prospective longitudinal observational study.
Setting:
Outpatient.
Patients:
A total of 595 participants with peripheral artery disease (PAD).
Measurements:
Blood samples were obtained every 2 months for up to 3 years and hemostatic factors examined at intervals preceding events.
Results:
Sixty-one participants (cases) experienced events and were matched to 122 PAD controls. During the 2-month interval prior to an event, cases (n = 48) had higher levels of the vWF and ADAMTS13 than controls (n = 95; P = .05), but significance was lost after adjusting for the baseline differences in myocardial infarction, unstable angina, and stroke. During the 10 months prior to an event, median values for vWF and the ratio of vWF and ADAMTS13 were higher in cases than in controls, but the differences were not statistically significant. However, in a subset of 20 patients with complete bimonthly data, there was a trend toward an increase in the ratio in the 10 months prior to a CV event (P = .04).
Conclusion:
In patients with PAD experiencing an ischemic CV event, a significant increase in the ratio of vWF to ADAMTS13 prior to the event could not be confirmed, although there was a weak trend in this direction.
Keywords
Introduction
Myocardial infarction (MI), strokes, and cardiovascular (CV) death might be precipitated by thrombophilic changes in the blood in the weeks leading up to an acute CV event. The several candidate thrombogenic factors include an increase in the von Willebrand factor (vWF) and a loss of its protease, a disintegrin-like and metalloprotease domain with thrombospondin type-1 motif, number 13 (ADAMTS13). 1,2 von Willebrand factor mediates platelet adhesion to injured vessel walls in areas of high shear stress—the branching points where the atheromas typically form. 3 The largest multimers of vWF are the most thrombogenic 4 but are cleaved upon their release from the endothelium by ADAMTS13. 5 A marked decrease in ADAMTS13 activity is associated with recurrent episodes of thrombosis, as exemplified by patients with thrombotic thrombocytopenic purpura. 6 It has been suggested that an imbalance in the levels of plasma vWF and ADAMTS13 might be associated with thrombus formation in patients with atrial fibrillation 7 –9 and stroke. 10
Levels of vWF and ADAMTS13 have been examined in survivors of MI. 11 On multivariable analysis, vWF correlated positively and ADAMTS13 correlated negatively with the risk of MI. Sonneveld et al 12 conducted a prospective study to determine whether vWF, ADAMTS13, or the combination of high vWF and low ADAMTS13 predicted strokes or transient ischemic attacks (TIAs) in 5941 persons aged ≥55 years, without a history of these diseases at study inception. The risk of stroke was significantly increased in those with the lowest quartile of ADAMTS13 activity (hazard ratio, 1.61) and was greatest in participants with the highest levels of vWF and lowest levels of ADAMTS13 (hazard ratio, 1.72). Adding ADAMTS13 to a model that included all traditional risk factors significantly improved stroke prediction (P = .003). The mean time between measurement of ADAMTS13 and the occurrence of stroke was 5.8 years.
The Biomarker Risk Assessment in Vulnerable Outpatients (BRAVO) study was a prospective cohort study of outpatients with peripheral vascular disease (peripheral artery disease [PAD]), which was conducted to assess whether specified biomarkers were temporally associated with new CV events.
13
Blood samples were obtained at 2-month intervals to determine whether circulating levels of biomarkers (
Methods
Methods of the main BRAVO study have been reported. 12 Patients with PAD, as defined by an ankle–brachial index (ABI) <0.90, an ABI > 0.90 with documented evidence of PAD from a noninvasive vascular laboratory, or with prior lower extremity revascularization for PAD, were followed prospectively to identify new CV events. Examinations were conducted at 2-month intervals; at each follow-up, questionnaires were administered to identify hospitalizations since the last study visit, an electrocardiogram was recorded, and blood samples were drawn. Medical records for all hospitalizations reported during follow-up were obtained, reviewed, and adjudicated by study physicians to identify new CV events, using the criteria derived from the Atherosclerosis Risk in Communities and Multi-Ethnic Study of Atherosclerosis (MESA) studies. 14,15 Home visits were made to those unwilling or unable to attend a follow-up clinic visit. If a home visit was not possible, participants were telephoned to obtain information about hospitalizations.
The institutional review boards at Northwestern University and all participating sites approved the protocol. Participants provided written informed consent. Recruitment occurred between September 2009 and September 2012, and participants were followed through January 2013.
Definitions of Cases and Controls
Cases were participants who had a fatal or nonfatal CV event during the study. Cardiovascular death included fatal MI or death due to definite coronary heart disease. 12 Both types of death required the absence of known nonischemic or noncardiac causes of death. Nonfatal events were acute MI, new electrocardiogram findings consistent with MI, hospitalizations for unstable angina, and ischemic strokes.
Two control participants were randomly selected for each case from participants having PAD without a CV event as of the date of the CV event for the corresponding case. Compared to case participants, matched control participants met all of the following criteria: (1) they were within 5 years of age of the case, (2) they were of the same sex as the case, (3) they had a similar number of blood draws leading up to the CV event date as the case, and (4) they were included in the study for at least as long as the case participant.
von Willebrand Factor and ADAMTS13
The concentration of vWF antigen was measured by an immunoturbidimetric method (vWFag, Liatest vWF Antigen kit; Diagnostica Stago, Parsippany, New Jersey). In healthy persons, the mean (SD) was 1.01 (0.26) U/mL, and the coefficient of variation was 7.7%. 16 ADAMTS13 was assayed using a chromogenic enzyme-linked immunoassay (Technozym ADAMTS13 Antigen ELISA; Diapharma, West Chester, Ohio). In our laboratory, the mean (SD) was 0.99 (0.18) μg/mlL, the coefficient of variation was 2.5%, and the mean (SD) ratio of vWF to ADAMTS13 was 1.05 (0.30) in healthy volunteers. The technician performing the assays was blinded as to whether the study samples came from cases or controls.
Statistical Analyses
Because of right skewness of distribution of the hemostatic factors, we summarized the levels at each visit as median and interquartile range and applied log transformation to the levels in subsequent statistical analyses. For vWF, ADAMTS13, and the ratio of vWF to ADAMTS13, we determined among participants who experienced a CV event whether there was a trend toward increasing levels obtained at visits 8 to 10 months, 6 to 8 months, 4 to 6 months, 2 to 4 months, and within 2 months before the event. We used the generalized estimating equation (GEE) regression analyses with log-transformed biomarker levels prior to the CV event from the same individual as the correlated responses and the number of scheduled visits from the measurement to the CV event as the independent variable of interest. Next, we determined whether the levels of vWF and ADAMTS13 and the ratio of vWF to ADAMTS13 at the final visit prior to the CV event were higher in case participants compared to controls, using GEE regression analyses with log-transformed biomarker levels for the case participants and the 2 matched controls as correlated responses within the same cluster and the binary (case vs. control) indicator as the independent variable of interest. We repeated this analysis to adjust for baseline differences in angina, MI, congestive heart failure, and stroke between cases and controls by including corresponding independent variables in the multivariate regression model. Since all of the cases and most of the controls had hypertension, we excluded controls without hypertension from the analysis for adjusting the imbalance in hypertensive history between cases and controls. We also repeated the analyses separately for the subgroups of participants having unstable angina, MI, and ischemic stroke as the CV event. Finally, we separately analyzed data from participants who had not missed any visits during the 10 months preceding an event so that the same participants were included in the analysis for each visit.
Role of the Funding Source
The funding source played no role in data collection, analyses, or manuscript writing.
Results
A total of 595 participants were recruited. During a mean follow-up of 18.7 ± 9.8 months (range: 0-40), 61 (10.3%) participants had CV events. The events included MI (n = 26), hospitalization for unstable angina (n = 15), acute ischemic strokes (n = 11), cardiac deaths (n = 8), and resuscitated cardiac arrest (n = 1). One hundred and twenty-two participants having PAD without events (controls) were matched by age, sex, length of follow-up, and number of blood specimens to the 61 case participants. The baseline characteristics of the cases and controls have been reported previously 17 and are shown in Table 1. Case participants walked fewer blocks and had a higher prevalence of angina, MI, congestive heart failure, stroke, and hypertension than the controls. Because 1 death was possibly, but not definitely, due to coronary heart disease, the hemostatic factors were omitted from further analysis for that participant .
Baseline Characteristics of the Cases and Controls.
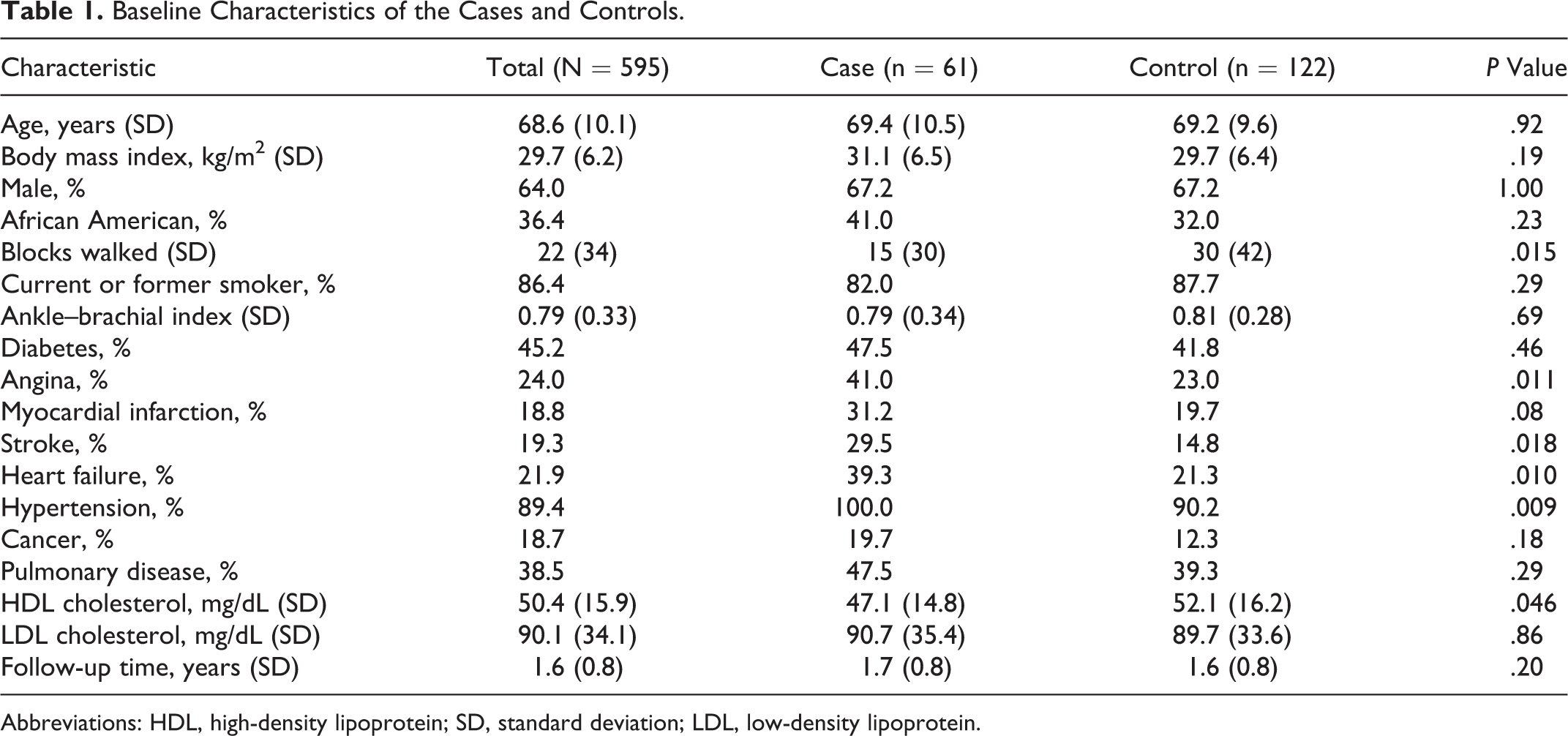
Abbreviations: HDL, high-density lipoprotein; SD, standard deviation; LDL, low-density lipoprotein.
Forty-eight cases had data available for the 2-month interval prior to an event; their vWF to ADAMTS13 ratios were higher than the 94 controls (1.61 vs 1.55, P = .05), but the significance was lost after adjusting for a baseline history of angina, MI, congestive heart failure, and stroke (Table 2). Among participants with CV events, there were no significant differences in the hemostatic factors based on the type of event, whether MI or unstable angina was used as the reference value. However, those having an MI had significantly higher levels of vWF and the ratio of vWF to ADAMTS13 than matched controls (Table 3). Most cases had data about additional visits during the 10 months prior to an event; their serial levels of vWF and ADAMTS13 and the ratio of vWF and ADAMTS13 are shown in Figures 1 to 3. Although the median levels of vWF and the ratio of vWF to ADAMTS13 were consistently higher in cases than in matched controls, the differences at each time point were not statistically significant.
von Willebrand factor (U/mL), ADAMTS13 (µg/mL), and the Ratio of vWF to ADAMTS13 in Cases and Controls With Peripheral Artery Disease Within 2 Months of a Cardiovascular Event (Median and Interquartile Range).
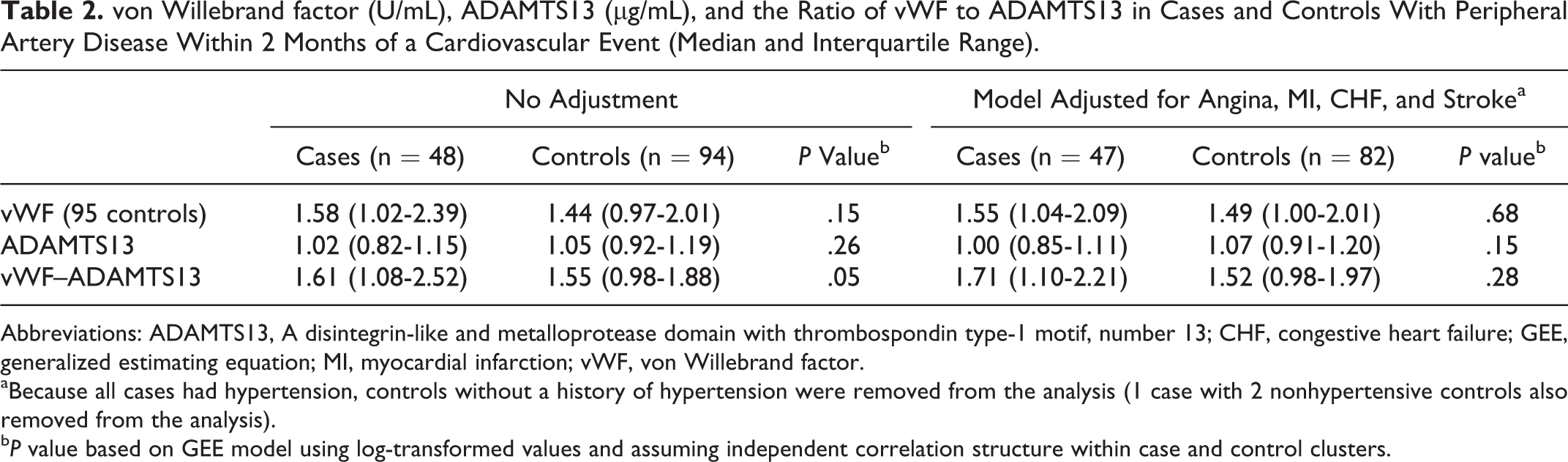
Abbreviations: ADAMTS13, A disintegrin-like and metalloprotease domain with thrombospondin type-1 motif, number 13; CHF, congestive heart failure; GEE, generalized estimating equation; MI, myocardial infarction; vWF, von Willebrand factor.
aBecause all cases had hypertension, controls without a history of hypertension were removed from the analysis (1 case with 2 nonhypertensive controls also removed from the analysis).
b P value based on GEE model using log-transformed values and assuming independent correlation structure within case and control clusters.
von Willebrand Factor (U/mL), ADAMTS13 (µg/mL), and the Ratio of vWF to ADAMTS13 in Cases Within 2 Months of a Cardiovascular Event (Median and Interquartile Range).
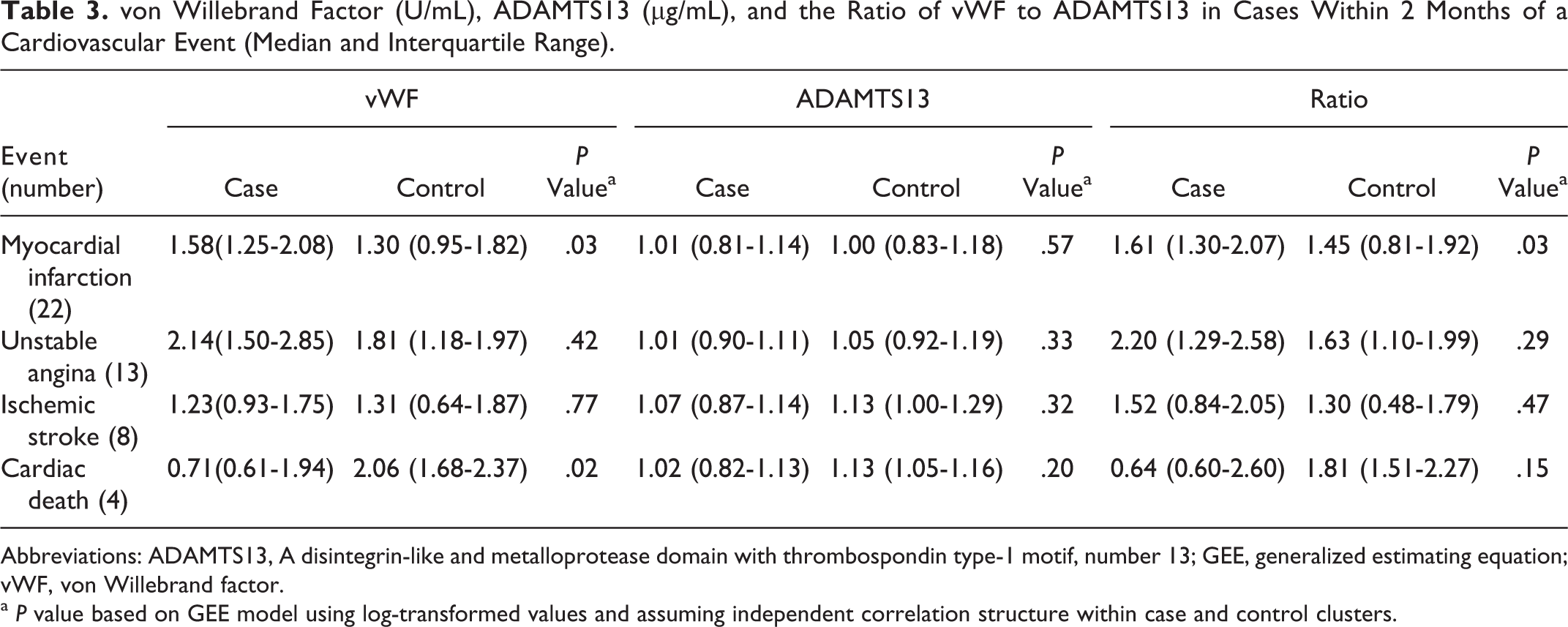
Abbreviations: ADAMTS13, A disintegrin-like and metalloprotease domain with thrombospondin type-1 motif, number 13; GEE, generalized estimating equation; vWF, von Willebrand factor.
a P value based on GEE model using log-transformed values and assuming independent correlation structure within case and control clusters.
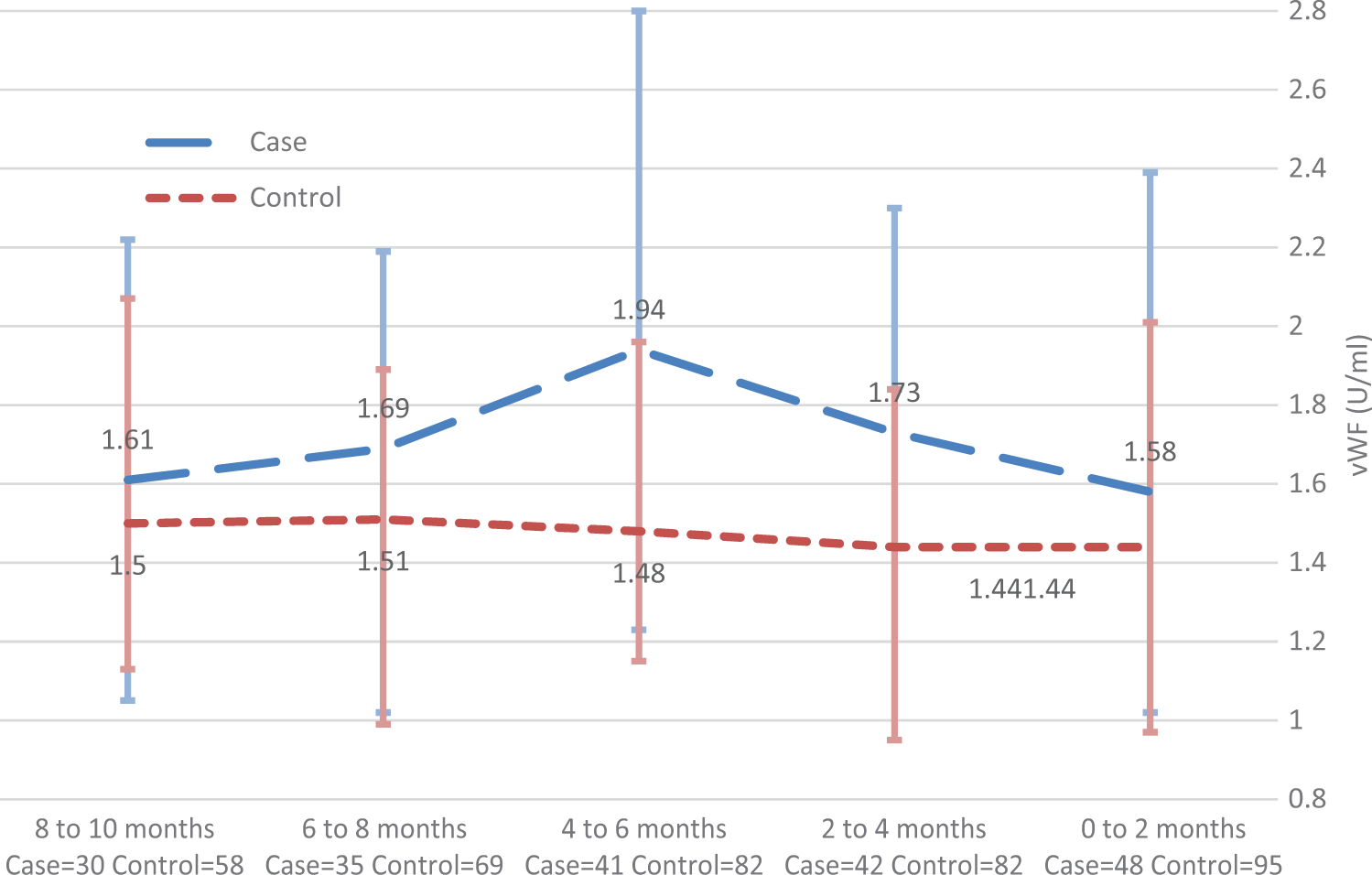
von Willebrand factor (vWF; U/mL), median and interquartile range for cases and controls (all available data for individual visits).
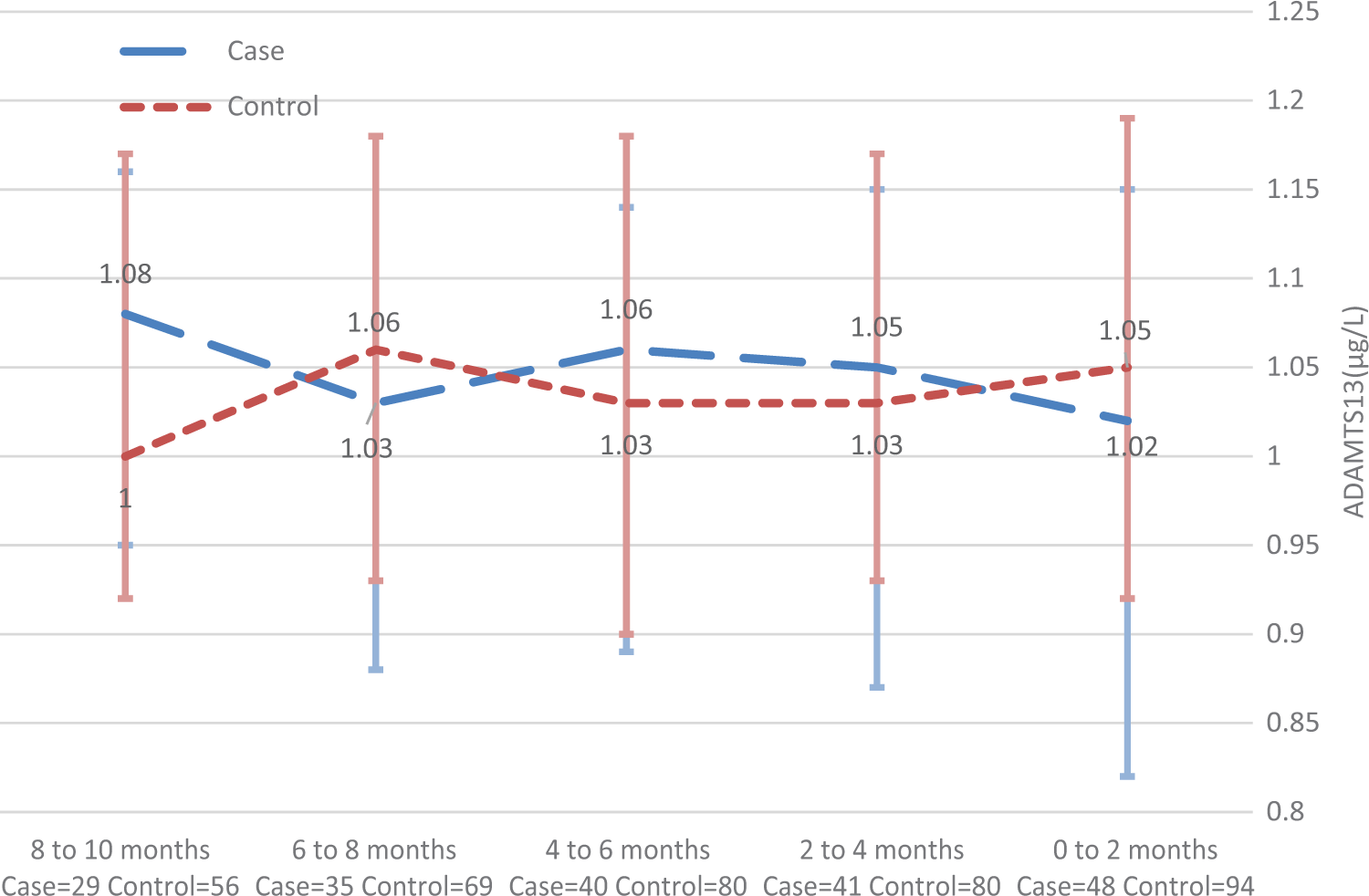
A disintegrin-like and metalloprotease domain with thrombospondin type-1 motif, number 13 (ADAMTS13; µg/mL), median and interquartile range for cases and controls (all available data for individual visits).
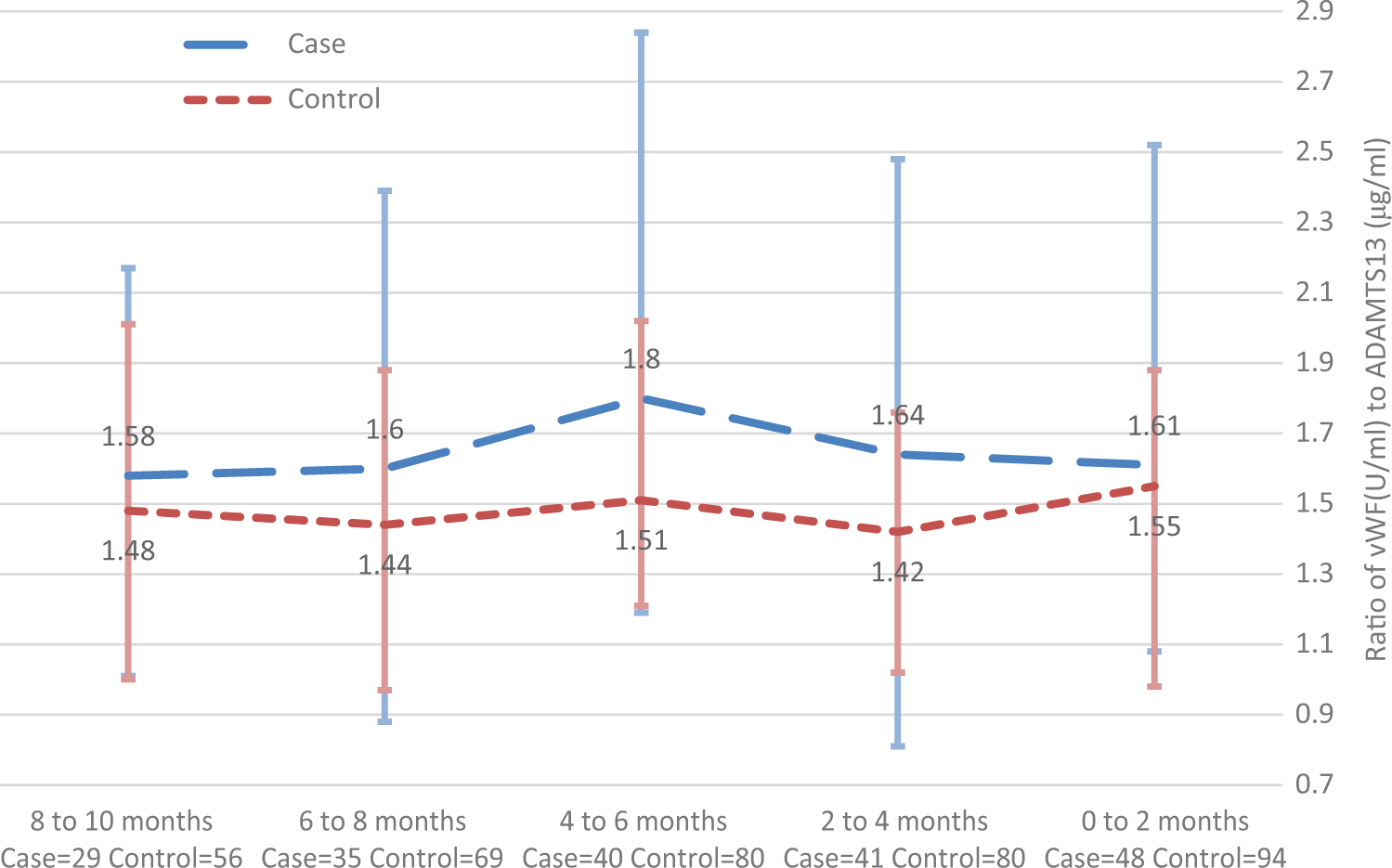
The ratio of von Willebrand factor (vWF; U/mL) to a disintegrin-like and metalloprotease domain with thrombospondin type-1 motif, number 13 (ADAMTS13; μg/mL), median and interquartile range for cases and controls (all available data for individual visits).
To determine whether there was a trend toward higher levels of vWF in the 10 months immediately preceding a CV event, an analysis was performed in those participants (20 of the 61 with events) who had data for every 2 months of that period. The analysis showed that the levels of vWF gradually rose, but there was little change in ADAMTS13 (Table 4). This resulted in an increase in the ratio of vWF to ADAMTS13 and a trend toward a higher ratio in the months immediately preceding the CV event (P for trend, .04).
von Willebrand Factor (U/mL), ADAMTS13 (µg/mL), and the Ratio of vWF and ADAMTS13 in Cases at Bimonthly Intervals Prior to a Cardiovascular Event (Median and Interquartile Range).

Abbreviations: ADAMTS13, A disintegrin-like and metalloprotease domain with thrombospondin type-1 motif, number 13; GEE, generalized estimating equation; vWF, von Willebrand factor.
a P trend values based on GEE model using log-transformed values and assuming an independent correlation structure for visits by the same participants.
Discussion
In this observational study, we measured vWF and ADAMTS13 in the months immediately preceding the onset of stroke, MI, and other CV events. We observed that the median levels of ADAMTS13 were similar in cases and controls (Figure 2). The median levels of vWF and the ratio of vWF to ADAMTS13 were consistently higher in cases than in matched controls (Figures 1 and 3), but the differences at each time point were not statistically significant except for the ratio of vWF to ADAMTS13 2 months prior to an event. While this ratio was higher in cases than in controls (P = .05), the difference in ratios became nonsignificant after adjusting for the baseline prevalence of vascular events in the 2 groups. We did observe a trend toward a higher ratio of vWF to ADAMTS13 in the months immediately preceding the CV event (P for trend = .04).
The risk of CV disease is higher with gene variants that increase the levels of vWF and lower with variants that decrease the levels of vWF. 18,19 Elevated levels of vWF have been observed in individuals with objectively proven atherosclerosis 20 as well as in patients experiencing sudden cardiac death, nonsudden cardiac death, and nonfatal MI. 21 Significant associations of vWF with ischemic stroke have also been reported. 22,23 More recently, the focus has been on ADAMTS13, a protease that cleaves the most thrombogenic vWF multimers. These multimers are released from endothelial cell Weibel-Palade bodies and increase the concentration of platelets along the vessel wall through an interaction with platelet glycoprotein 1bα (GP1bα). 24 Widespread microvascular thrombosis with severe deficiencies of ADAMTS13 occurs in patients with thrombotic thrombocytopenic purpura, and MI has been associated with ADAMTS13 levels below the fifth percentile. 25 The association of MI with low levels of ADAMTS13 and high concentrations of vWF is independent of each other. 11
Temporal association of vWF and ADAMTS13 with cerebrovascular events has been examined in 2 studies. McCabe et al 26 observed moderately reduced ADAMTS13 activity and elevated vWF antigen within 4 weeks after the onset of TIAs or stroke, and Sonneveld et al 12 found that individuals with ADAMTS13 levels in the lowest quartile at baseline had an increased risk of stroke in the next 5 to 10 years. Ours is the first study to prospectively examine vWF and ADAMTS13 bimonthly immediately preceding stroke and other CV events.
Our study has limitations. First, our sample size was reduced because we did not have complete data for a subset of participants with PAD, especiallly those who were most ill and least likely to participate in a study visit. Such individuals might possibly be expected to have the most profound perturbation of hemostatic factors, and their inclusion might have lent additional support to our hypotheses. Second, we didn’t ask participants not to smoke prior to phlebotomy. Smoking shortly before blood draws might have acutely affected the levels of vWF and ADAMTS13. 27 Third, because we performed antigen rather than activity assays, we could not assess the functional impact of the raised vWF to ADAMTS13 ratios we found. Third, this observational study cannot determine the specific contribution of vWF and ADAMTS13 to the CV event; for example, another factor might have increased the vWF to ADAMTS13 ratio as well as triggered the vascular occlusions. Finally, we studied participants with PAD because they are a high-risk population with high rates of CV events, so the results might not be generalizable to other patient populations.
In summary, patients with PAD experiencing an ischemic CV event had higher ratios of vWF to ADAMTS13 than controls, but these differences were not significant after adjusting for the baseline vascular disorders. In a subset of 20 patients with complete bimonthly data, there was a trend toward an increase in the ratio during the 10 months prior to a CV event (P = .04).
Footnotes
Authors’ Note
D. Green wrote the first draft, edited the manuscript for important scientific content, supervised the laboratory assays, and participated in data analyses and interpretation. L. Tian led the statistical analyses and interpretation, and Y. Liao performed the analyses and assisted with the interpretation of the data. P. Greenland participated in the statistical analyses and interpretation and provided important intellectual content to the manuscript. K. Liu, M. Kibbe, R. Tracy, S. Shah, J. T. Wilkins, and M. D. Huffman assisted with study oversight, participant recruitment, and data collection and interpretation. D. Lloyd-Jones assisted with obtaining funding, adjudication of ischemic heart disease events, data interpretation, and manuscript development. M. M. McDermott originated the study concept and design, obtained funding, supervised the data collection and analysis, and revised the manuscript. All authors approved of the final version.
Acknowledgment
We thank Loyda Vida for expert technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This trial was supported by grant R01 HL089619 from the National Heart, Lung, and Blood Institute.
