Abstract
Venous thromboembolism (VTE) is associated with significant morbidity and mortality. Factors such as the presence of transient risk factors for VTE, risk of bleeding, and location of deep vein thrombosis (DVT) determine the duration of anticoagulation. Extended anticoagulation is offered to patients with unprovoked pulmonary embolism (PE) or proximal DVT and a low risk of bleeding. Anticoagulation for 3 months is advised in patients with provoked DVT or PE, high risk of bleeding, and isolated distal or upper extremity DVT. In patients with unprovoked PE or proximal DVT and a low risk of bleeding, who want to stop anticoagulation after 3 months, further risk stratification is necessary. Clinical scoring system, and thrombophilia testing otherwise not routinely performed, may be considered to measure risk of annual recurrence in such cases. Short-term anticoagulation may be considered in subsegmental PE and superficial vein thrombosis, particularly if patients are at low risk of bleeding and have persistent risk factors for recurrent VTE. In cases of catheter-associated thrombosis, the catheter need not be removed routinely, and the patient may be anticoagulated for 3 months or longer if the catheter is maintained in patients with cancer. Extensive screening for occult cancer in cases of unprovoked VTE is not beneficial. New oral anticoagulants such as apixaban, rivaroxaban, or dabigatran may be preferred to vitamin K antagonists in patients without cancer or renal failure, more so after the development of reversal agents such as idarucizumab and andexanet alfa.
Keywords
Introduction
Venous thromboembolism (VTE), including deep vein thrombosis (DVT) of the extremities or pelvis and pulmonary embolism (PE), is a public health problem associated with significant morbidity and mortality. 1 Approximately 900 000 annual cases of VTE result in 300 000 deaths annually in the United States. 2 Venous thromboembolism results in substantial health-care cost, with approximately 2.5-fold increase in cost compared to inpatients without VTE. 3 Thrombophilia testing alone costs more than USD 500 to 1300 per test and is frequently performed in many cases of VTE, despite recommendations by the American Society of Hematology against the routine testing. Understanding the duration of anticoagulation and the role of thrombophilia testing can be difficult for a busy practitioner. Here, we present a focused review on this topic and provide an algorithm for management of VTE. A detailed discussion of literature is available in the most recent American College of Chest Physicians (ACCP) guidelines. 4
Management of Proximal DVT and Symptomatic PE
The major factors that determine the duration of anticoagulation include the presence of any risk factor for VTE (provoked or unprovoked VTE), degree of risk of bleeding, presence of cancer, and location of DVT (distal or proximal 4 ; Figure 1).
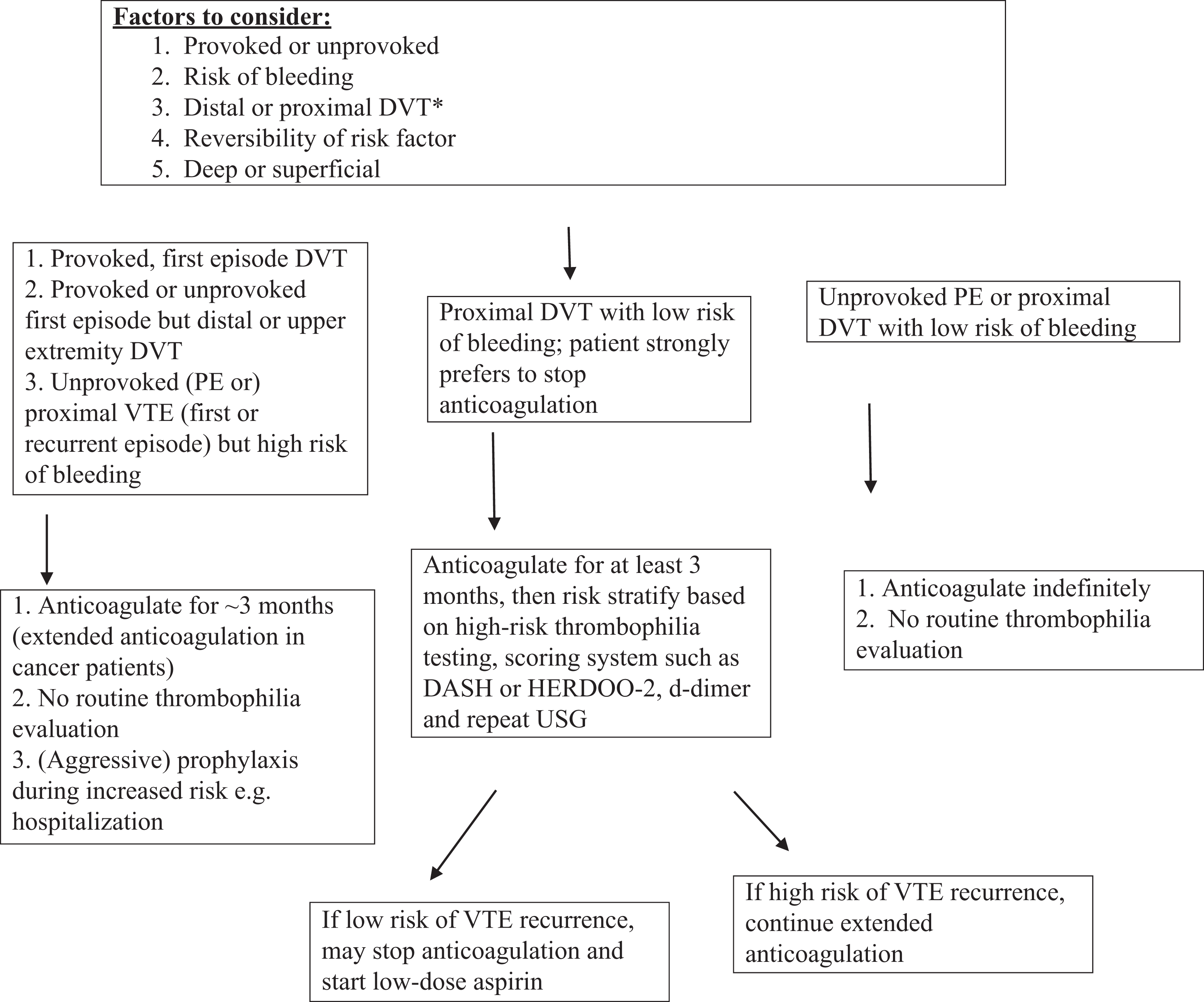
Simplified schema of the management of venous thromboembolism. DASH indicates elevated D-dimer post anticoagulation, age <50 years at initial VTE, male sex, and hormone use in women at initial VTE; DVT, deep vein thrombosis; HERDOO-2, hyperpigmentation, edema or redness of affected leg, elevated d-dimer while on anticoagulation, obesity with body mass index >30, and age ≥65 years; PE, Pulmonary embolism; USG, ultrasonography; VTE, venous thromboembolism. *Superficial femoral and popliteal veins are proximal veins.
Patients with unprovoked PE or proximal DVT with low risk of bleeding should be offered an extended anticoagulation. Anticoagulation for 3 months is advised in provoked DVT or PE, unprovoked PE or DVT in a patient with high risk of bleeding, and in isolated distal or upper extremity DVT. 4 In these patients, routine thrombophilia testing is not necessary and does not change management. 5,6
Further risk stratification is necessary in patients with unprovoked PE or proximal DVT and a low risk of bleeding who want to stop anticoagulation after 3 months. High-risk thrombophilia testing as discussed subsequently may be helpful in such situation; the presence of high-risk thrombophilia may indicate a higher risk of recurrence. Scoring systems such as DASH or HERDOO-2 may be used to measure risk of annual recurrence and guide duration of anticoagulation. DASH stands for elevated
One alternative to extended use of therapeutic anticoagulation may be the use of new oral anticoagulants (eg, apixaban) at a prophylactic dose after 3 months of anticoagulation. In Apixaban after the Initial Management of Pulmonary Embolism and Deep Vein Thrombosis with First-Line Therapy-Extended Treatment (AMPLIFY-EXT) study, extended anticoagulation with apixaban led to reduced risk of recurrent VTE without increasing the rate of major bleeding at both treatment and thromboprophylactic doses. A prophylactic dose of apixaban (2.5 mg twice daily) was associated with similar risk of VTE recurrence (1.7%) and major bleeding (0.2% vs 0.1%) but slightly lower risk of clinically relevant nonmajor bleeding (3% vs 4.2%), compared to the treatment dose (5 mg twice daily). 13 Hence, we offer a prophylactic dose apixaban as an option to our patients.
In patients with unprovoked VTE, who decide to stop anticoagulation, low-dose aspirin should be used in the absence of any contraindication because of a reduction in the risk of recurrent VTE or the rate of major cardiovascular events. 14 –16 In women or younger men without a family history of prostate cancer, vitamin E 600 IU every other day may be an alternative, particularly in patients with factor V Leiden or the prothrombin mutation. 17 However, it is not recommended to check for factor V Leiden or the prothrombin mutation.
Thrombolysis and newer surgical and interventional measures are available to be used in addition to the medical therapy. A study described pharmacomechanical thrombolysis with a rotator thrombolysis device in iliofemoral deep venous thrombosis in 67 patients. 18 Immediate total recanalization was conducted in all patients, the leg diameters returned to normal ranges in the early postoperative period, and hospital mortality or severe complications were not detected. In general, the ACCP guidelines recommend anticoagulant alone over thrombolysis or catheter-assisted thrombus removal including in patients with acute DVT and PE. Thrombolysis is recommended only in select patients such as those with low risk of bleeding and PE associated with hypotension. 4
Management of Other VTE
Any VTE including subsegmental PE, isolated distal DVT, and superficial vein thrombosis may require anticoagulation, particularly if there are persistent risk factors and low risk of bleeding. 19 The 2016 ACCP guideline does not differentiate isolated subsegmental PE from a more proximal PE; however, 2014 European Society of Cardiology recommends that an individualized decision about anticoagulation be made after careful assessment of the risk–benefit ratio. 20 Clinical surveillance may be chosen over anticoagulation in select patients in the absence of symptoms and risk factors for extension. However, a recent study by den Exter et al concluded that patients with subsegmental PE have a high risk of VTE and clinical outcomes similar to that of more proximally located PE. 21 Patients with cancer having subsegmental PE are frequently treated with anticoagulation due to the underlying heightened risk of recurrent VTE. 19,22
The optimal duration of anticoagulation is not clear in patients with subsegmental PE. Based on the ACCP guidelines, the duration of anticoagulation depends on the presence of transient risk factors, presence of cancer, number of VTE, and risk of bleeding. Although there is a possibility that small or asymptomatic PE may resolve without anticoagulation in certain patient population, based on the current guidelines and the lack of studies to justify withholding anticoagulation, the patients are usually treated with anticoagulation for at least 3 months. 19 A careful evaluation of the risks and indications of indefinite anticoagulation must be conducted in all cases. 19
Cancer-related thromboses have a high risk of recurrence and are expected to derive substantial benefit from extended anticoagulant therapy. 23 Anticoagulation may be stopped after 3 months of treatment in provoked VTE if the cancer is thought to have been cured (eg, following surgical resection) or after 6 months of therapy if there is uncertainty about cancer status. 24
In cases of isolated distal DVT, the risk of recurrence is about half that of proximal DVT or PE, thus, anticoagulation is advised for a total of 3 months. 24 Given the lack of extensive studies in such cases, clinical surveillance and serial ultrasound over 2 weeks may be chosen over anticoagulation in select patients with isolated distal DVT in the absence of symptoms and risk factors for extension.
Superficial vein thrombosis may increase the risk of VTE 4- to 6-fold, and when combined with acquired strong thrombotic risk factors, the risk of VTE is 10- to 100-fold. 25 In superficial vein thromboses, 5 cm or more in size, close to deep veins, or associated with underlying diseases such as cancer, fondaparinux 2.5 mg for 6 weeks is approved by the Food and Drug Administration (FDA) for treatment. 26 Fondaparinux consistently decreases the risk of symptomatic extensions of the superficial thrombus from the saphenofemoral junction and subsequent VTE complications. 27
In cases of catheter-associated thrombosis (such as peripherally inserted central catheters, central venous catheters, cardiac pacemakers, and defibrillators), the catheter need not be removed routinely if it is functional and needed, and the patient can be anticoagulated. 28 The ACCP guidelines recommend anticoagulation for 3 months, which may be extended if the catheter is maintained, particularly in patients with cancer. 23
Role of Thrombophilia Testing
High-risk thrombophilia testing may be considered in select patients who are young, have unprovoked VTE, have thrombosis in unusual sites, have a family history of thrombophilia, 29,30 and who are interested in stopping anticoagulation. Thrombophilia evaluation may include testing for antithrombin deficiency (antithrombin activity), protein C deficiency (protein C activity), protein S deficiency (free protein S antigen and activity), and antiphospholipid syndrome. Thrombophilia evaluation should generally be performed by a hematologist with a plan to guide duration of anticoagulation based on the results. Testing may be avoided at the time of diagnosis of VTE or when the patient is on any anticoagulant. Methylenetetrahydrofolate reductase mutation and heterozygous mutation of prothrombin gene mutation or factor V Leiden do not alter management, hence should not be tested routinely. 31,32
The role of thrombophilia testing in pregnant patients with a personal or family history of VTE is controversial. Pregnant patients with prior VTE should receive postpartum VTE prophylaxis in the absence of high risk of bleeding; those with moderate-to-high risk of recurrence should also receive antepartum VTE prophylaxis. Select pregnant patients without a personal history of VTE but with a family history of VTE may benefit from limited thrombophilia testing, where the presence of homozygous factor V Leiden or prothrombin 20210A mutation requires both antepartum and postpartum VTE prophylaxis. However, homozygous factor V Leiden or prothrombin 20210A mutation is rare. Additionally, all other thrombophilias require antepartum vigilance and postpartum prophylaxis, thus may not necessarily alter management. 23
The diagnosis of antiphospholipid syndrome, paroxysmal nocturnal hemoglobinuria, and myelodysplastic syndrome can significantly impact management. Hence, antiphospholipid antibody syndrome testing should be considered in unprovoked VTE in young patients or those with a history of unexplained recurrent abortion. Testing includes lupus anticoagulant, anti-β2-glycoprotein antibody, and anticardiolipin antibody. The diagnosis is made based on the Sapporo criteria. 33 In addition to paroxysmal nocturnal hemoglobinuria and myelodysplastic syndrome, trauma, surgery, intra-abdominal infections, inflammatory bowel disease, pancreatic disease, and cirrhosis are associated with and can cause splanchnic vein thrombosis. Paroxysmal nocturnal hemoglobinuria may be considered in patients with unexplained VTE of sites other than extremity, for example, hepatic vein thrombosis or those with unexplained cytopenia, hemolysis, or iron deficiency. 34 JAK-2V617F mutation, which occurs in patients with myeloproliferative neoplasms disorder, may be tested for patients with unexplained portal vein or mesenteric vein thrombosis and those with elevated blood count. 35
In appropriate setting (eg, critically ill patients), diagnosis such as heparin-induced thrombocytopenia, 36 disseminated intravascular coagulation, 37 thrombotic microangiopathy, 38 catastrophic antiphospholipid antibody syndrome, 39 or thrombotic storm 40 may be considered.
Role of Cancer Screening
Reported incidence of cancer varies widely; older studies had estimated a risk of as high as 20% to 30% in patients with first episode of unprovoked VTE. 41 –47 In a study, the prevalence of an occult cancer in patients with unprovoked VTE was 6% at the time of diagnosis of VTE and increased to 10% after 12 months of diagnosis. 48 More recent studies, however, show a much lower risk, with a cumulative incidence as low as 3.2% over a follow-up of 2.5 years. 49 Although unprovoked VTE may be the first sign of an occult cancer, extensive screening with imaging and tumor markers is not of benefit.
Carrier et al demonstrated that use of imaging studies such as computed tomography (CT) of chest, abdomen, and pelvis did not confer any advantage over the use of limited cancer screening in patients with unprovoked VTE. 50 In another study, screening with 18F-fluorodeoxyglucose positron emission tomography (18F-FDG PET) combined with low-dose CT, used routinely for the diagnosis and staging of various malignant diseases, was not associated with significantly higher rate of cancer diagnosis compared to limited screening. However, the risk of subsequent cancer diagnosis after initial negative screening with 18F-FDG PET/CT was lower than that in patients who had negative initial limited screening. 51 Thus, only age- and sex-appropriate cancer screening should be performed. For example, colonoscopy should be offered to patients older than 50 years. A mammography should be performed in women older than 40 to 50 years of age, and a Papanicolaou smear should be performed in women aged 21 to 65 years. 50 The role of prostate-specific antigen test in men is unclear, given the recommendations of US Preventive Services Task Force against prostate cancer screening. 52
Anticoagulants and Reversal Agents
Commonly utilized anticoagulants for long-term management of VTE include oral anticoagulants such as factor Xa inhibitors (rivaroxaban, apixaban, or edoxaban), direct thrombin inhibitors (dabigatran), vitamin K antagonist (warfarin), low-molecular-weight heparin (LMWH), and fondaparinux. In patients with cancer, pregnant patients, and those with liver disease, LMWH is the preferred agent. Vitamin K antagonist (warfarin) is the preferred agent in patients who require reversal agent and those with chronic kidney disease with creatinine clearance <30 mL/min. Dabigatran and rivaroxaban, but not apixaban, may be associated with an increased risk of gastrointestinal bleeding, thus are less preferred in at-risk patients. In most other patients, new oral anticoagulants such as apixaban, rivaroxaban, or dabigatran may be preferred over vitamin K antagonist. 4 The 4 oral anticoagulants (apixaban, dabigatran, edoxaban, and rivaroxaban) have been shown to be noninferior to standard therapy (warfarin or LMWH). 53 Additionally, oral anticoagulants have exhibited at least similar safety profile compared to standard therapy, with a lower risk of major bleeding with apixaban and rivaroxaban in some studies. 54,55
Reversal agents are discussed in Table 1. 56 In a bleeding patient, platelet and cryoprecipitate transfusion may be required to achieve a platelet count >50 000 to 100 000/µL and fibrinogen level >100 to 150 mg/dL depending on the severity and site of bleeding. The dose of fresh frozen plasma is 10 to 15 mL/kg intravenously (IV; approximately 3-5 units) in most adults; this should increase plasma coagulation factors by ∼15% to 25%. If hemostasis is not achieved, use higher doses and adjust dose based on desired clinical response. 59 Urgent infusion of prothrombin complex concentrate (PCC) may be preferred in major bleeding such as intracranial bleed. 60 Dose of PCC is based on the international normalized ratio (INR) values and ranges from 25 U/kg (maximum dose of 2500 units) for INR of 2 to 4, 35 U/kg (maximum dose 3500 units) for INR of 4 to 6, and 50 U/kg (maximum dose 5000 units) for INR of >6. 61,62 Approximately 1 mg of protamine neutralizes about 100 units of heparin. Maximum dose of protamine is 50 mg. In cases of continuous heparin infusion, consider only the heparin given in the preceding several hours and adjust the protamine dosage depending upon the duration of time since heparin administration. 63 For every 100 units of heparin, 1 to 1.5 mg, 0.5 to 0.75 g, or 0.25 to 0.375 g of protamine is given if the last dose of heparin was given <30 minutes, 30 to 60 minutes or >2 hours, respectively, prior to protamine administration. For subcutaneous heparin, about 1 to 1.5 mg protamine per 100 units of heparin is required. A portion of the dose (eg, 25-50 mg) is given slowly IV followed by the remaining portion as a continuous infusion over 8 to 16 hours (the expected absorption time of the subcutaneous heparin dose). Protamine sulfate only partially reverses enoxaparin. 64,65 For enoxaparin administered <8 hours ago, administer 1 mg protamine for 1 mg enoxaparin. For enoxaparin administered >8 hours ago, administer 0.5 mg protamine for 1 mg enoxaparin. 63 Andexanet alfa is shown to be effective at rapidly reversing anticoagulant activity of therapeutic doses of apixaban and rivaroxaban. 66 Idarucizumab is a humanized monoclonal antibody fragment that binds to dabigatran and its acylglucuronide metabolites and neutralizes the anticoagulant effect within minutes. 67,68 A dose of 5-gm IV (administered as 2 separate 2.5 g doses no more than 15 minutes apart) is FDA approved in cases of life-threatening bleeding or need for emergency surgery in patients on dabigatran. 69
Common Anticoagulants in Clinical Practice and Reversal Agents.

Abbreviations: ECT, Ecarin clotting time; FFP, fresh frozen plasma; HTI, Hemoclot Thrombin Inhibitor assay; INR, international normalized ratio; IV, intravenous; LMWH, low-molecular-weight heparin; PCC, prothrombin complex concentrates; PTT, partial thromboplastin time; SubQ, subcutaneous; TT, thrombin time; UFH, unfractionated heparin.
aThis time period is valid in patients without renal or liver dysfunction. 56
bHemoclot thrombin inhibitor assay and ECT may quantify the effects of dabigatran, however, limitations include the lack of exact reference values, widespread commercial availability, and validation in bleeding patients. 57,58 In emergency situations, the TT and PTT are the most accessible qualitative methods.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Vijaya Bhatt is supported by the 2015-2016 Physician-Scientist Training Program Grant from the College of Medicine, University of Nebraska Medical Center.
