Abstract
In 2 double-blind studies, ambulatory patients with objectively proven, disseminated metastatic breast carcinoma (TOPIC-1) or stage III/IV non–small-cell lung carcinoma (TOPIC-2) were randomized to certoparin 3000 IU or placebo subcutaneously once daily, for 6 months. Primary efficacy outcome was objectively confirmed symptomatic or asymptomatic venous thromboembolism (VTE). Safety outcomes included bleeding (major and minor), and thrombocytopenia. TOPIC-1 was halted after an interim analysis. Venous thromboembolism occurrence was not different between treatment groups in TOPIC-1 (4% treated with certoparin, 7 of 174 vs 4% receiving placebo, 7 of 177, odds ratio [OR] 1.02; 95% confidence interval [CI] 0.30-3.48) and in TOPIC-2 (4.5%, 12 of 268) vs 8.3%, 22 of 264, respectively, OR 0.52; CI 0.23-1.12). Mortality was not different between groups. A post hoc analysis showed certoparin significantly reduced VTE in stage IV lung carcinoma (3.5% vs 10.2%; P = .032) without increased bleeding. In conclusion, thrombosis risk and prophylactic benefit was highest in stage IV lung carcinoma patients.
Keywords
Introduction
Venous thromboembolism (VTE) is an unwanted complication for cancer patients. 1 At the extreme, fatal pulmonary embolism (PE) may end life prematurely, 2 but even the occurrence of a distal deep vein thrombosis (DVT) impairs the quality of life and complicates management. 3 The pathogenesis of VTE in cancer is complex, involving multiple interactions between tumor and components of the hemostasis system. 4 The development of a systemic hypercoagulable state is considered a key pathogenic feature and contributes to the processes of primary tumor growth, neoangiogenesis, and metastasis. 5 Cancer location and clinical stage affect VTE risk,6,7 along with chemotherapy per se, 7 and certain types of chemotherapy in particular (eg, thalidomide or lenalidomide). 8 Metastatic cancer carries a high risk of thrombosis, 1 with an approximately 20-fold increased risk in patients with metastatic disease compared with those without metastases. 9
In patients undergoing surgery for cancer, thrombosis risk is well characterized and is regarded as high 10 ; short-term prophylaxis with low-molecular-weight heparin (LMWH) reduces the risk of VTE, 11 and long-term prophylaxis minimizes the risk further, 12 with no significant increase in bleeding. There remains little data on ambulatory patients receiving chemotherapy, in terms of both the risk of VTE and the need for prevention measures. Only 1 randomized study has assessed primary thromboprophylaxis in stage IV breast cancer patients receiving multiagent chemotherapy. 13 The study found a 4% thrombosis rate in the placebo group and an 85% relative risk reduction with warfarin treatment. More recently, the PROTECHT study reported that the LMWH nadroparin reduced the incidence of VTE in ambulatory patients with metastatic or locally advanced lung, gastrointestinal, pancreatic, breast, ovarian, or head and neck cancer who were receiving chemotherapy. 14 In light of this, clinical guidelines suggest that further clinical trials are required before recommendations can be made for prophylaxis in ambulatory patients receiving chemotherapy for cancer. 15
We performed 2 multicenter, randomized, placebo-controlled, blinded studies to confirm the elevated risk of VTE in patients with metastatic breast cancer (TOPIC-1) or inoperable, late-stage non–small-cell lung cancer (TOPIC-2) and to determine whether extended LMWH prophylaxis is an effective and safe intervention.
Materials and Methods
Patients
Adult patients with objectively proven, disseminated metastatic breast carcinoma, receiving first- or second-line chemotherapy were eligible for inclusion in TOPIC-1. Adult patients with objectively proven, inoperable disseminated primary non–small cell lung carcinoma of stage III or IV receiving standard first- or second-line chemotherapy were eligible for inclusion in TOPIC-2. The presence of metastatic disease had to have been confirmed by combinations of chest X-ray, scintigraphy, computerized tomography (CT), ultrasound, magnetic resonance imaging, and fine needle aspiration biopsy. Radiotherapy was allowed in both studies and any hormonal treatment in TOPIC-1.
Patients were excluded from TOPIC-1 if they had inflammatory breast cancer or were receiving anthracycline monotherapy or gemcitabine (monotherapy or in combination). Patients were excluded from TOPIC-2 if they had small-cell lung carcinoma, brain metastases, hemoptysis of ≥grade 2, or a Karnofsky index <70. Patients were excluded from either study for the following reasons: bedridden; previous VTE diagnosis; current heparin or oral anticoagulant therapy; long-term aspirin or other current antiplatelet drugs; active gastrointestinal bleeding; hemorrhagic stroke; hereditary bleeding disorder; thrombocytopenia (platelets <75 000/mL); partial thromboplastin time >2 times upper limit of normal (ULN); known hypersensitivity to heparin; severe diabetic retinopathy; creatinine >2 times ULN; osteoporotic fracture; myocardial infarction in the preceding 6 months; and participation in a clinical trial with an experimental drug in the preceding 4 weeks.
Study Design
These were randomized, double-blind, adaptive group-sequential, placebo-controlled trials conducted at 39 centers in Germany, Czech Republic, Ukraine, Romania, and Belarus. Patients were randomly assigned to placebo or certoparin sodium (Mono Embolex, Novartis GmbH, Nürnberg, Germany) using a computer-generated randomization list. Patients were allocated to the lowest available randomization number available for each study center. Randomization numbers were allocated sequentially as patients were enrolled at each center. Only the external statistician from the Safety Committee had access to the randomization codes. Randomization was block-stratified according to treatment with hormone-based chemotherapy. Treatment was continued for 6 months. Study drug was supplied as a pre-filled 3-mL multidose pen. An injection volume of 0.3 mL containing 3000 IU certoparin, or isotonic saline, was administered once daily for 6 months. Patients, or their carer, were instructed on injection technique. At baseline, a full assessment of blood chemistry was conducted. During the 6-month treatment period, patients were seen in clinic at weeks 2, 4, 8, 12 16, 20, and 24. All clinic visits included compression ultrasound for DVT (except week 2), assessment of any VTE symptoms, recording of any adverse event, such as bleeding episodes, provision of study drug, and a review of concomitant medications and blood biochemistry. In addition to the assessments listed above, visits at weeks 12 and 24 included detailed physical examination and assessment of the tumor, including staging and determination of any skeletal complications using bidimensional chest X-ray, CT, nuclear spin tomography, bone scan, and/or abdominal ultrasound as necessary.
Study protocols were approved by local ethics committees and the study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients. Efficacy outcomes were validated by a blinded, independent Central Thrombosis Evaluation Team; safety end points were validated by a Data Safety Monitoring Committee consisting of 2 clinicians (blinded to treatment) and an independent statistician with access to the treatment assignments.
Study Outcomes
The primary efficacy outcome was the first occurrence of objectively confirmed VTE during the 6-month treatment period to include symptomatic or asymptomatic DVT (proximal or distal) confirmed by venography and/or ultrasonography; symptomatic PE confirmed by CT ventilation–perfusion scintigraphy, or shown at autopsy; thrombosis of the jugular or subclavian veins confirmed by ultrasonography; and superficial thrombophlebitis (if heparin-based treatment was required). Secondary efficacy parameters were symptomatic VTE, the overall thrombosis rate (to include arterial thrombotic events, superficial thrombophlebitis, and central line thrombosis) and objectively verified skeletal events (to include all fractures, spinal cord compression, and requirement for surgery to treat fractures or for bone irradiation). Patient survival was a prespecified end point if the studies continued after interim analysis and superiority was proven. Two post hoc analyses were conducted: a comparison of mortality at study end and a stratified analysis of the primary end point of TOPIC-2 according to tumor stage.
Safety outcomes were primarily heparin-related adverse events—bleeding, thrombocytopenia, and osteoporotic fractures. Major bleeding was defined as bleeding that was fatal, retroperitoneal, intracranial, requiring transfusion of ≥2 units of packed red cells, or associated with a drop in hemoglobin of ≥20 g/L. Minor bleeding events were those that did not meet major bleeding criteria. Thrombocytopenia was defined as a fall in platelet count below 100 000/mL, or of >50% from baseline. The possibility of heparin-induced thrombocytopenia (HIT) was investigated with laboratory assays to detect heparin-dependent immunoglobulin G (IgG) antibodies in cases for whom clinical suspicion was warranted (platelet count fall >50% from baseline and the thrombocytopenia arose ≥5 days after heparin initiation). Patients underwent bone scans at baseline and at week 16 and 20 to assess the occurrence of osteoporotic fractures.
Statistical Analysis
The preliminary sample size estimate for each study was enrollment of 626 patients to give the study a statistical power of 80% to detect a 33% risk reduction in VTE (30% with placebo and 20% with certoparin), with a 1-sided significance level of .025. The sample size calculation for each study was based on an adaptive design with an interim analysis, because there was uncertainty about the expected event rates. Interim analysis was scheduled after 260 patients had been enrolled to give a statistical power of 67% to detect a 50% risk reduction in the primary end point (30% with placebo and 15% with certoparin) with a 1-sided significance level of .01. A 1-sided significance test was deemed appropriate for interim analyses because it was assumed that there would be a reduction in VTE events with certoparin treatment compared with placebo. P values <.01 would result in the corresponding null hypothesis that there is no difference between the treatments to be rejected and the study would be halted; P values >.12 would also result in study termination (for practical reasons, the total sample size of each study had to be limited to a maximum of about 800 patients and a P value >.12 would have required enrollment of >500 patients after interim analysis to prove superiority). A mortality comparison would only be conducted if certoparin was proven superior to placebo in the reduction of VTE.
Patients who received at least 1 dose of study medication were included in the safety analyses. Efficacy analyses were performed on the intention-to-treat population. Between-group efficacy and safety comparisons are presented as odds ratios (ORs) with 95% confidence intervals (CIs) and superiority was tested using Fisher exact test.
Results
Patient Characteristics
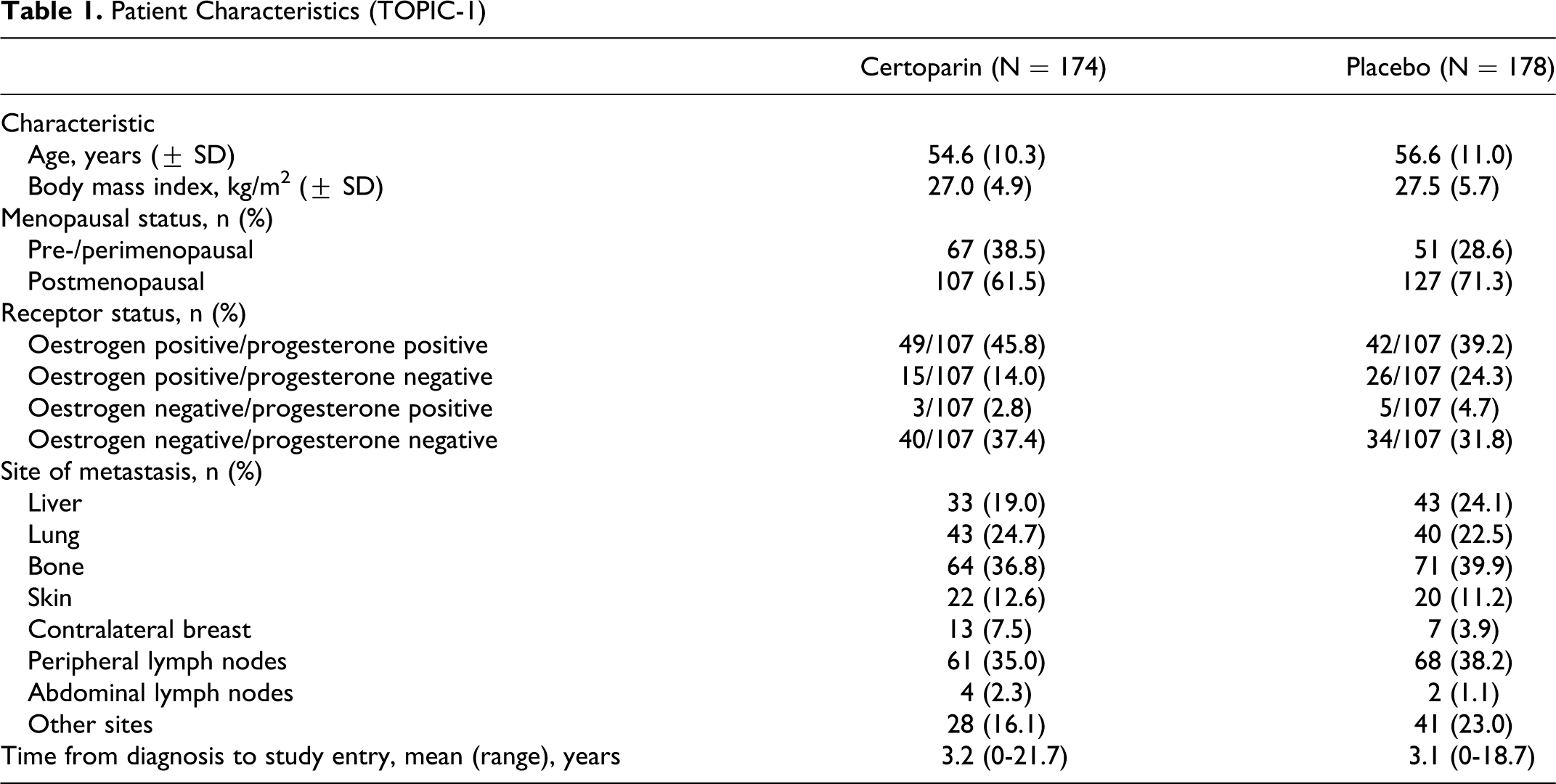
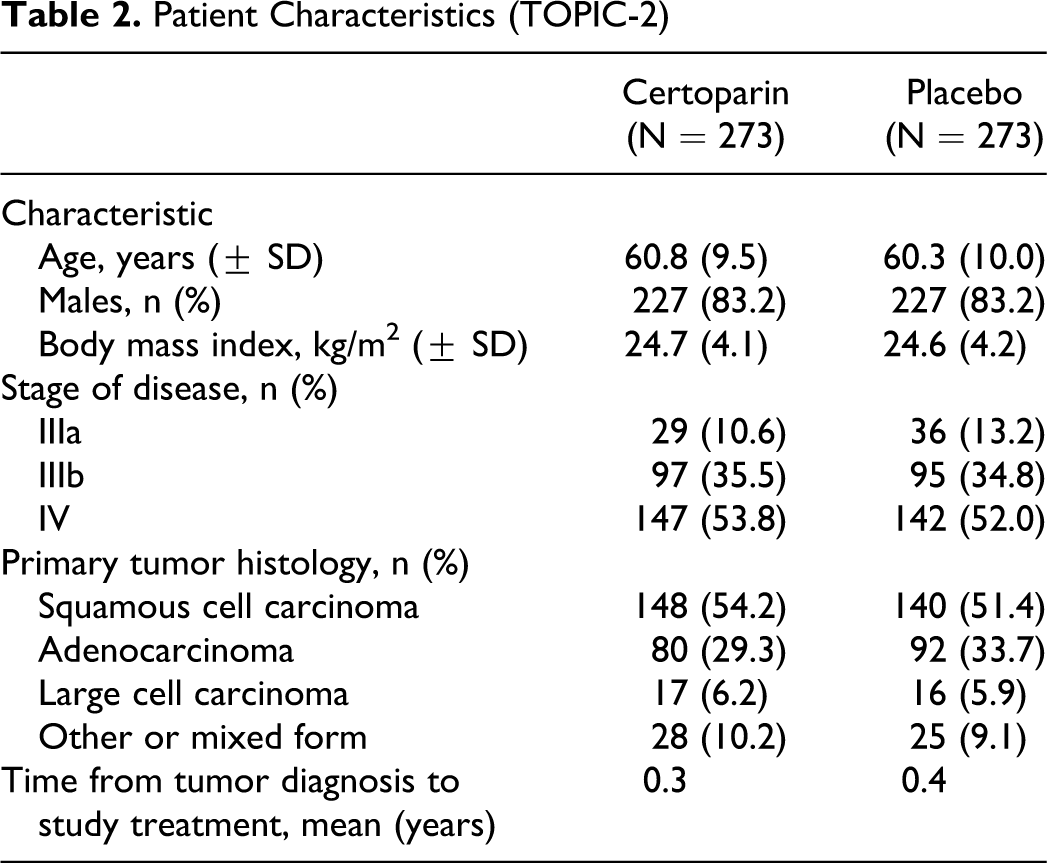
Between April 1999 and November 2004, 900 patients were recruited to the TOPIC trials (Figure 1). The predefined interim analysis of TOPIC-1 failed to show superiority of certoparin over placebo; there was a short delay before study termination, at which point 353 patients had been randomized. The intention-to-treat populations comprised 351 (TOPIC-1) and 532 (TOPIC-2) patients (Figure 1); in total, 15 patients were excluded because a post-baseline thrombosis screening was not conducted or because the patient was diagnosed with thrombosis on baseline screening. Treatment groups in both studies were reasonably well matched with regard to baseline and tumor characteristics (Tables 1 and 2).
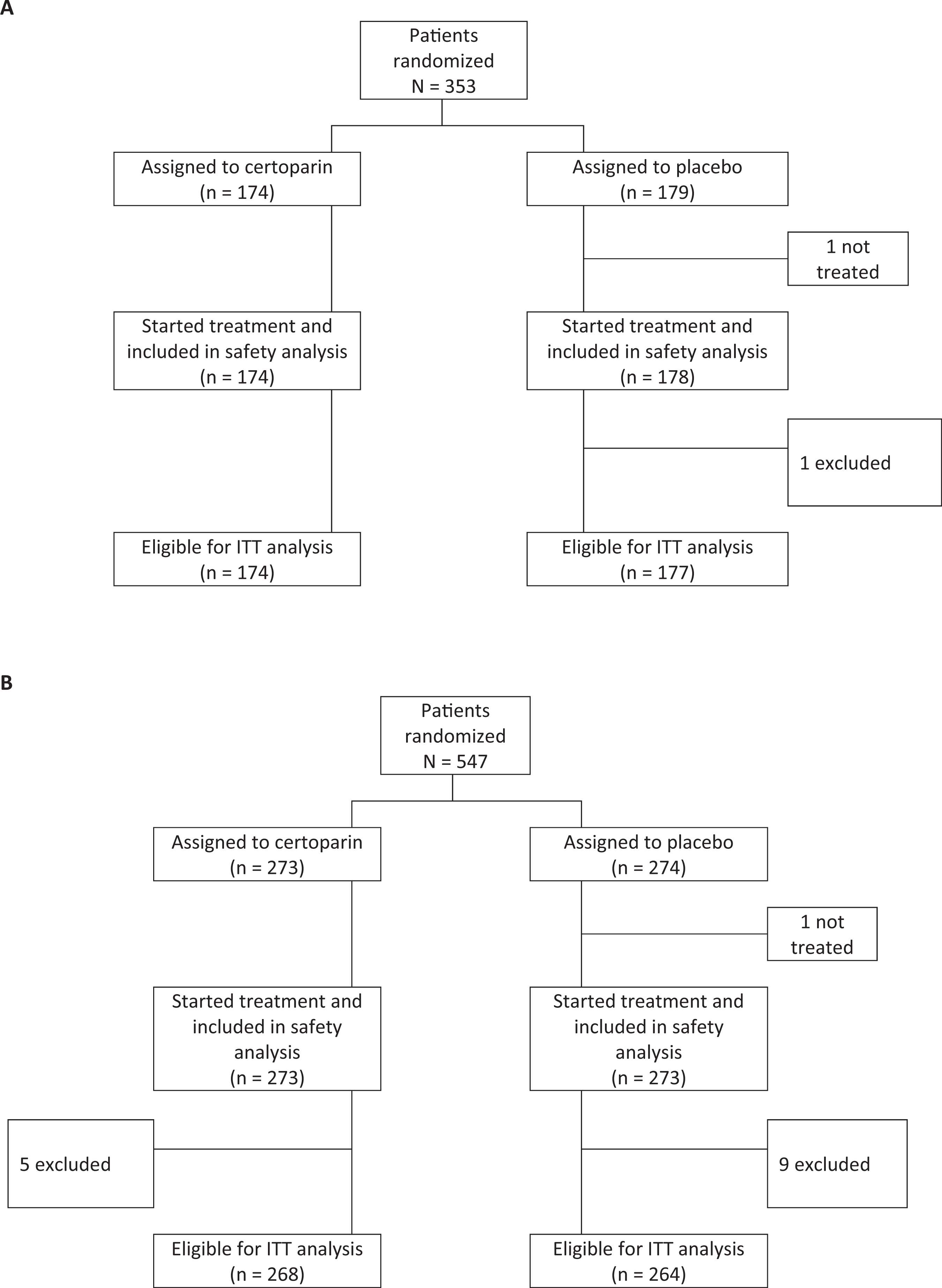
(A) Flowchart of TOPIC-1 and (B) flowchart of TOPIC-2.
Patient Characteristics (TOPIC-1)
Patient Characteristics (TOPIC-2)
Efficacy Outcomes
In TOPIC-1, there was no difference in the occurrence of VTE between the certoparin-treated patients and the placebo group (4.0% [7 of 174] vs 4.0% [7 of 177], respectively; OR 1.02, 95% CI 0.30-3.48, P = 1.000). In TOPIC-2, there was no difference in the occurrence of VTE between the certoparin-treated patients and the placebo group (4.5% [12 of 268] vs 8.3% [22 of 264], respectively; OR 0.52; 95% CI 0.23–1.12, P = .078). Secondary efficacy outcomes are shown in Table 3.
Efficacy Outcomes
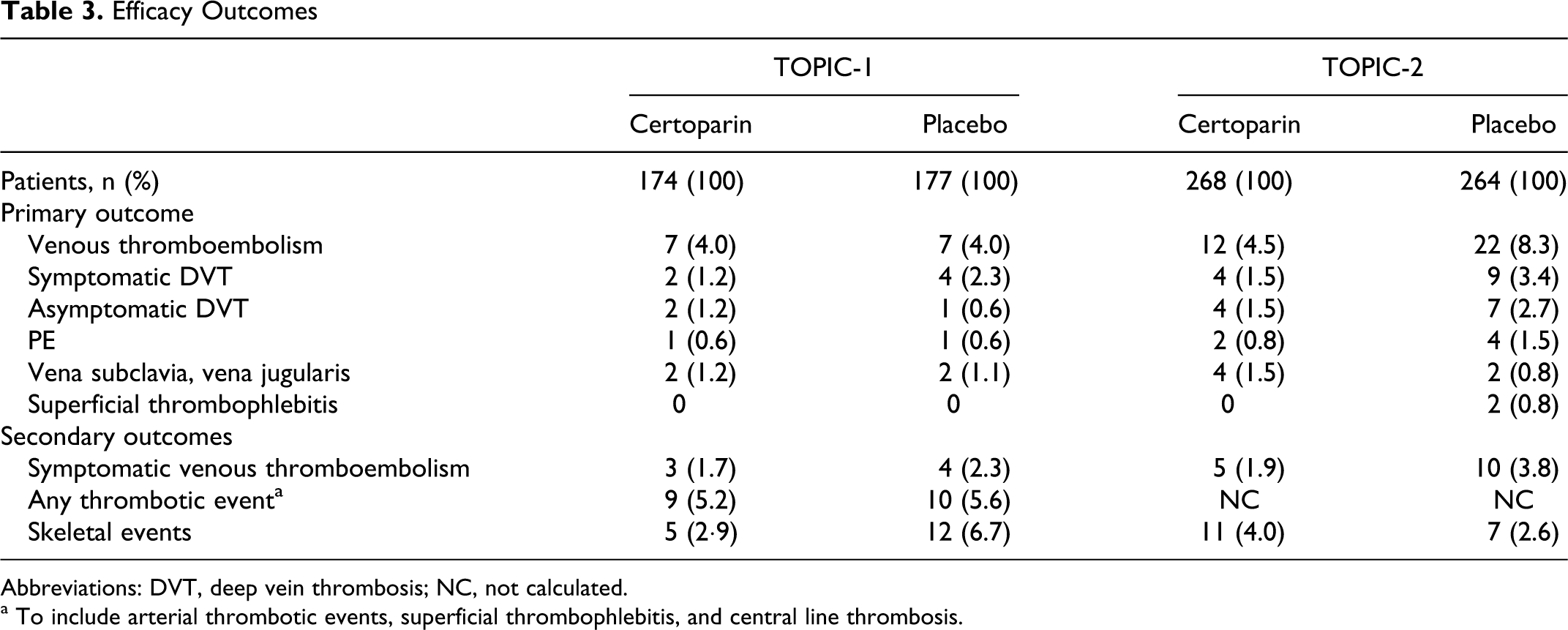
Abbreviations: DVT, deep vein thrombosis; NC, not calculated.
a To include arterial thrombotic events, superficial thrombophlebitis, and central line thrombosis.
For stage III lung cancer, the event rates were not different between the certoparin and placebo groups (5.7% vs 6.4%, respectively); but in stage IV lung cancer patients, the incidence of VTE was significantly reduced with certoparin treatment (3.5% [5 of 144] vs 10.2% [14 of 139]; OR 0.32, 95% CI 0.09-0.98; P = .032).
Overall mortality during the 6-month treatment period in the safety population of TOPIC-1 was 8.6% (15 of 174) with certoparin and 6.7% (12 of 178) with placebo; and in TOPIC-2 was 20.2% (55 of 273) with certoparin and 21.6% (59 of 273) with placebo.
Safety Events
Bleeding events and thrombocytopenia are presented in Table 4. There were no fatal bleeding events and no events were adjudicated to be related to certoparin treatment. There were no cases of laboratory-confirmed HIT described. In 1 case (TOPIC-2), a fracture was related to osteoporosis.
Bleeding Events and Thrombocytopenia During Study Period
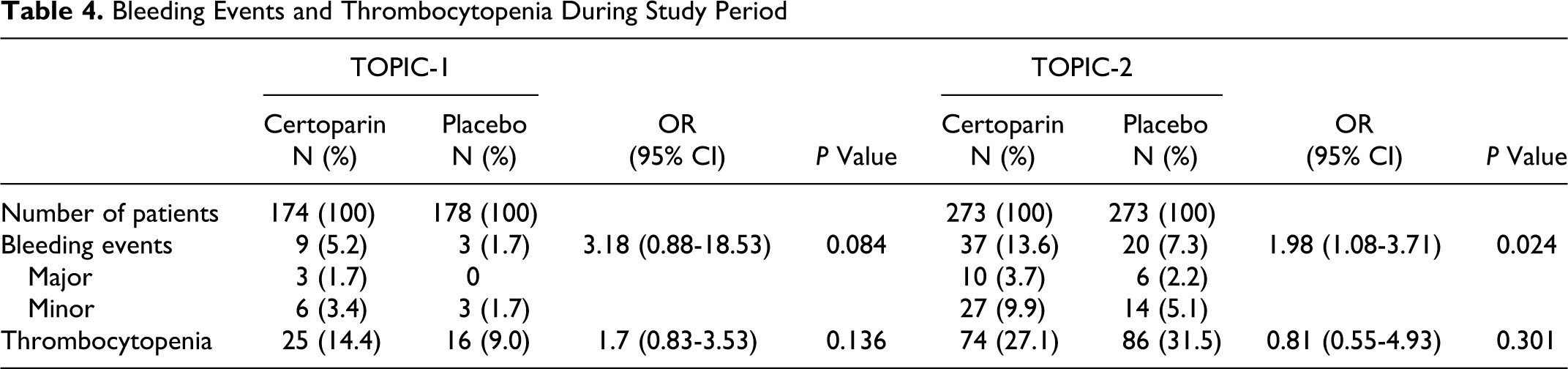
In stage III lung cancer patients, the incidence of bleeding was significantly increased with certoparin treatment (16.7% vs 4.6%; OR 4.17; 95% CI 1.55-13.03, P = .002). There was no difference in bleeding in stage IV patients (10.9% vs 9.9%, OR 1.12; 95% CI 0.52-2.38, P = .848).
Discussion
In these trials, the rate of VTE was substantially lower than anticipated, with few events in patients with metastatic breast cancer or inoperable, late-stage, non–small-cell lung cancer; as a consequence, TOPIC-1 was terminated prematurely. In TOPIC-1, the rate of VTE in both the placebo and certoparin groups was similar to that found in the placebo group of the warfarin prophylaxis study in stage IV breast cancer (4.4%) 13 and also to a case–control study that described an OR of 3.5 (95% CI 1.7–7.6) for VTE in patients with breast cancer compared with those without cancer. 9 The lack of risk reduction with certoparin is in contrast to the large reduction seen with warfarin. 13 The similarities between the rate of VTE with certoparin and placebo in this trial, and with the placebo arm of the warfarin trial, suggest that the certoparin dose used in TOPIC-1 may have been too low to provide effective prophylaxis in these patients with breast cancer. The findings of TOPIC-1 may have been influenced by the extended time frame between diagnosis and study treatment initiation (mean 3.2 years), given the view that patients with cancer have a greatly increased risk of thrombosis in the first few months after cancer diagnosis. 9 However, a lower than expected rate of thrombosis was also observed in PROTECHT, although LMWH prophylaxis did significantly reduce the rate of VTE compared with placebo (2.0% vs 3.9%; P = .02). 14 Most of the thromboembolic events occurred in patients with lung or gastrointestinal cancers, with the rate being particularly low in patients with breast and ovarian cancer.
The patients enrolled in TOPIC-2 were newly diagnosed with cancer, with a mean time between diagnosis and study treatment initiation of 0.3 years. Thus, the timing of the prophylaxis would appear to have been optimal to give patients with lung cancer the best hope of benefiting from LMWH. The risk reduction seen in TOPIC-2 was not statistically significant, but our study was underpowered because higher rates of VTE were used to calculate sample size. A post hoc combined analysis of lung cancer patients from PROTECHT and TOPIC-2 demonstrated a relative risk of symptomatic VTE events of 0.58 (95% CI 0.28-1.06) in favor of LMWH, and significant reduction in the relative risk of all VTE events (symptomatic and asymptomatic) in favor of LMWH (0.54 [95% CI 0.31-0.95]). 16 Our post hoc findings in stage IV patients, in whom thrombosis risk and prophylactic benefit was highest, suggest this patient group is worthy of further investigation in adequately powered clinical trials.
The incidence of major bleeding in TOPIC-1 was lower than the previous warfarin study, 13 but there were markedly more bleeding events in the patients with lung cancer and significantly more events in the LMWH treatment arm. These findings differ from the PROTECHT study, which found major bleeding in only 1% of patients with lung cancer. 14 In comparison, the definition of major bleeding was similar between PROTECHT and TOPIC-2, the end points were reviewed in both studies by independent panels, but the histological type of lung cancer differed somewhat between the 2 studies, 16 with small-cell lung cancer excluded from TOPIC-2. A possible explanation for the different bleeding findings may relate to more cases of thrombocytopenia in TOPIC-2, reported in 27% to 31% of patients.
There is interest in the potential survival benefits of anticoagulation in patients with cancer 17 and the biological affect of heparins on tumors, including experimental evidence from various animal models that supports the ability of heparin to attenuate metastasis. 18 Previously, certoparin was shown to improve survival following breast and pelvic cancer surgery. 19 In the TOPIC studies, a survival analysis was prespecified only if certoparin was proven superior to placebo in the prevention of VTE. The data presented which show no difference in survival versus placebo are post hoc and underpowered because of the early termination of TOPIC-1 and therefore contribute little to this research.
The validity of our findings is strengthened by the randomized treatment assignment, the double-blind, placebo-controlled designs, and the objective confirmation of any VTE event. A potential weakness is that detailed assessments of the natural history of the cancer were not undertaken, and the exact chemotherapy regimens employed before and after study entry are not available. This will be of more importance to define in the future as new, experimental agents currently being introduced into oncology may be associated with increased risk of VTE in certain tumor types.20,21 Low-molecular weight heparin was self-administered for an extended period of time, and patient compliance was based only on the counting of syringes prescribed; no pharmacokinetic measurements, such as anti-Xa, were made and so firm compliance data are unavailable. An unavoidable limitation is that these trials were designed and conducted in the late 1990s and early 2000s, when less was known about the pathophysiology of thrombosis in cancer and the thrombogenicity of chemotherapy regimens than is known now. If these trials were to be conducted again, it is likely that the inclusion criteria would be narrowed to include patients with more advanced disease and those receiving more thrombogenic chemotherapy regimens. Data from similar trials conducted recently have shown that selection of a tightly defined patient group with a high risk of thromboembolic events can demonstrate a substantial benefit from LMWH therapy22,23; whereas, inclusion of a diverse group of tumor types and chemotherapies is unlikely to show benefit. 14
In conclusion, VTE was less frequent in patients with advanced, disseminated cancer than anticipated, particularly so for patients with inoperable, late-stage, non–small-cell lung cancer, although symptomatic VTE rates were relatively high. Six months of treatment with prophylactic dose certoparin did not significantly reduce the rates of VTE in ambulatory breast cancer patients or lung cancer patients treated with a number of chemotherapy regimens. Patients with stage IV lung cancer may benefit from extended prophylaxis because a significant reduction in VTE, with no increase in bleeding events, was demonstrated in a post hoc analysis. Trials in more tightly defined groups of ambulatory cancer patients receiving chemotherapy at high risk of VTE should be conducted to establish the value of thromboprophylaxis.
Footnotes
Acknowledgments
We are indebted to the investigators for their help in recruiting patients into the study. Dr Aidan McManus (Edge Medical Communications) provided writing and editorial assistance in the preparation of this manuscript, funded by Novartis Pharma GmbH.
These data were presented in part at the XXth Congress of the International Society on Thrombosis and Haemostasis, August 6-12, 2005, in Sydney, Australia.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The TOPIC studies were supported by an unrestricted grant from Novartis Pharma GmbH, Germany. The investigators remained in control of the study database. Interpretation of data and preparation of the manuscript were undertaken by the investigators, and fulfilled a requirement within the protocol.
