Abstract
The aim of this study was to examine the effect of the traditional oral anticoagulant, warfarin (W), and new anticoagulants, apixaban (A) and rivaroxaban (R), on the level of thrombotic biomarkers in patients with atrial fibrillation (AF). Circulating plasma levels of von Willebrand factor (vWF), prothrombin fragment 1.2 (F1.2), microparticle tissue factor (MP-TF), and plasminogen activator inhibitor (PAI-1) were analyzed as potential markers of clot formation in 30 patients with AF prior to ablation surgery. Patients with AF were divided into 2 groups based on their usage (n = 21) and nonusage (n = 9) of any oral anticoagulant. Furthermore, those on anticoagulants were divided based on their use of newer (R and A, 16) or traditional (W, 4) anticoagulants. A statistical increase (
Introduction
Atrial fibrillation (AF) is currently the most common sustained cardiac arrhythmia encountered in clinical practice and is a major cause of morbidity and mortality among adults in the United States. 1 Atrial fibrillation alone is associated with a 1.5% to 1.9% increase in mortality risk in both men and women across a wide range of ages. 2 The development of AF carries a high risk of stroke which can be further increased based on age and other medical conditions including hypertension, diabetes mellitus, heart failure, and presence of previous stroke. 3 Stroke risk rises in those with AF from 1.5% for those aged 50 to 59 years to 23.5% for those aged 80 to 89 years. 4
Until recently, the only oral anticoagulant agent used in stroke management associated with AF was warfarin. 3,5 Warfarin, a coumarin group drug, prevents the recycling of vitamin K to its active form and thereby blocks the formation of the active forms of prothrombin and factors VII, IX, and X which are involved in both the extrinsic and the common pathways of coagulation. 6,7 Warfarin reduces the risk of stroke by two-thirds. 8 However, warfarin has not been optimally used in patients with AF with an even greater underuse seen in elderly patients who are at higher risk of stroke. 9 New anticoagulants have been introduced that do not require continuous monitoring like warfarin and are more convenient for patients with AF. Rivaroxaban (a direct factor Xa inhibitor 10 ) and apixaban (a highly selective factor Xa inhibitor 11 ) are the most commonly used new oral anticoagulants to prevent the formation of thrombin.
Several biomarkers have been shown to be useful in the identification of a thrombotic process. von Willebrand Factor (vWF) is a known marker of endothelial damage and platelet activation. The vWF antigen and activity have been shown to be associated with the occurrence of ischemic stroke. 12 Prothrombin fragment 1.2 (F1.2) is an index of in vivo thrombin generation and is released during the conversion of prothrombin to thrombin in a 1:1 ratio. 13 Microparticles are vesicular structures derived from platelets, monocytes, endothelial cells, smooth muscle cells, and tumor cells. Only a small fraction of microparticles in the blood express tissue factor (MP-TF). 14 Microparticle tissue factor can trigger blood coagulation. 15 In addition, circulating procoagulant microparticles have been shown to be increased in patients with AF and could indicate a hypercoagulable state causative of atrial thrombus generation. 16 The physiological state of AF produces many mechanisms that contribute to cellular apoptosis and the shedding of procoagulant microparticles within the vasculature. 17 Plasminogen activation inhibitor (PAI-1) is the physiological inhibitor of plasminogen and may contribute to thrombus generation by decreasing the degradation of fibrin. 18 The PAI-1 is stored in platelets, 19 and studies have also shown that platelets produce continuous amounts of active PAI-1. 20
From the standpoint of the mechanisms of anticoagulant actions, warfarin and the newer oral anticoagulants target different sites in the coagulation cascade. The primary aim of this study was to profile the level of previously discussed thrombogenic biomarkers in patients with AF and determine the effect of anticoagulant therapy on the circulating level of these biomarkers in order to obtain initial data for an ongoing study.
Patients and Methods
Blood samples from 30 patients (21 male and 9 female) undergoing ablation surgery for nonvalvular AF at Loyola University Medical Center in Maywood, Illinois, were examined. The patients ranged in age from 18 to 81 years with a mean age of 59.1 years. Patients were consented, and deidentified blood samples were collected prior to the procedure. This study was approved by the Institutional Review Board.
Eligibility Criteria
Patients were 18 years or older with AF who were being treated as an inpatient or outpatient at Loyola University Medical Center. Critically ill patients including patients with acute stroke, acute myocardial infarction, severe heart failure, sepsis, or patients on ventilators were excluded. Females who were pregnant or nursing mothers were excluded, as their clinical status may affect biomarker values.
Methods
Plasma samples from patients with AF was used for this study. Whole blood samples were drawn prior to ablation surgery for AF. Patient whole blood samples were collected into tubes containing 3.2% (0.109 mol/L) sodium citrate. Normal plasma samples from healthy controls were purchased from a commercial laboratory (George King Biomedical, Overland Park, Kansas). Control plasma samples were certified and collected from nonsmoking, drug-free volunteers (ages 19-54 years, mean 33).
Determination of Biomarker Profile
A RANDOX metabolic array assay kit (Randox, London, United Kingdom) was used to profile PAI-1. This allowed quantification of PAI-1 in a single patient sample using a sandwich chemiluminescent immunoassay. Undiluted plasma from 30 patients with AF and 25 normal donors was analyzed on RANDOX evidence investigator using the metabolic biochip according to the manufacturer’s instructions. The biochip was analyzed using software supplied by RANDOX.
Commercially available enzyme-linked immunosorbent assay kits were used to measure levels of vWF (Hyphen BioMed, Nueville-Sur-Oise, France), F1.2 (Siemens, Newark, Delaware), and MP-TF (Hyphen BioMed, Nueville-Sur-Oise, France).
Statistical Analysis
Data were analyzed using Microsoft Excel and Graphpad Prism Software version 6. The results were expressed as mean ± standard deviation. Comparison between groups were evaluated using nonparametric Mann-Whitney
Results
Comparison of Thrombotic Biomarker Levels in the Control and the AF Groups
Figure 1 presents a box plot of thrombotic biomarker levels for the control and AF groups. The circulating plasma levels of vWF and MP-TF were significantly increased in the AF group compared to the control group. The PAI-1, despite a large standard deviation in the control group, also showed a significant increase in the AF group compared to the control group. The F1.2 showed no statistical difference in the AF group compared to the control group (Table 1).
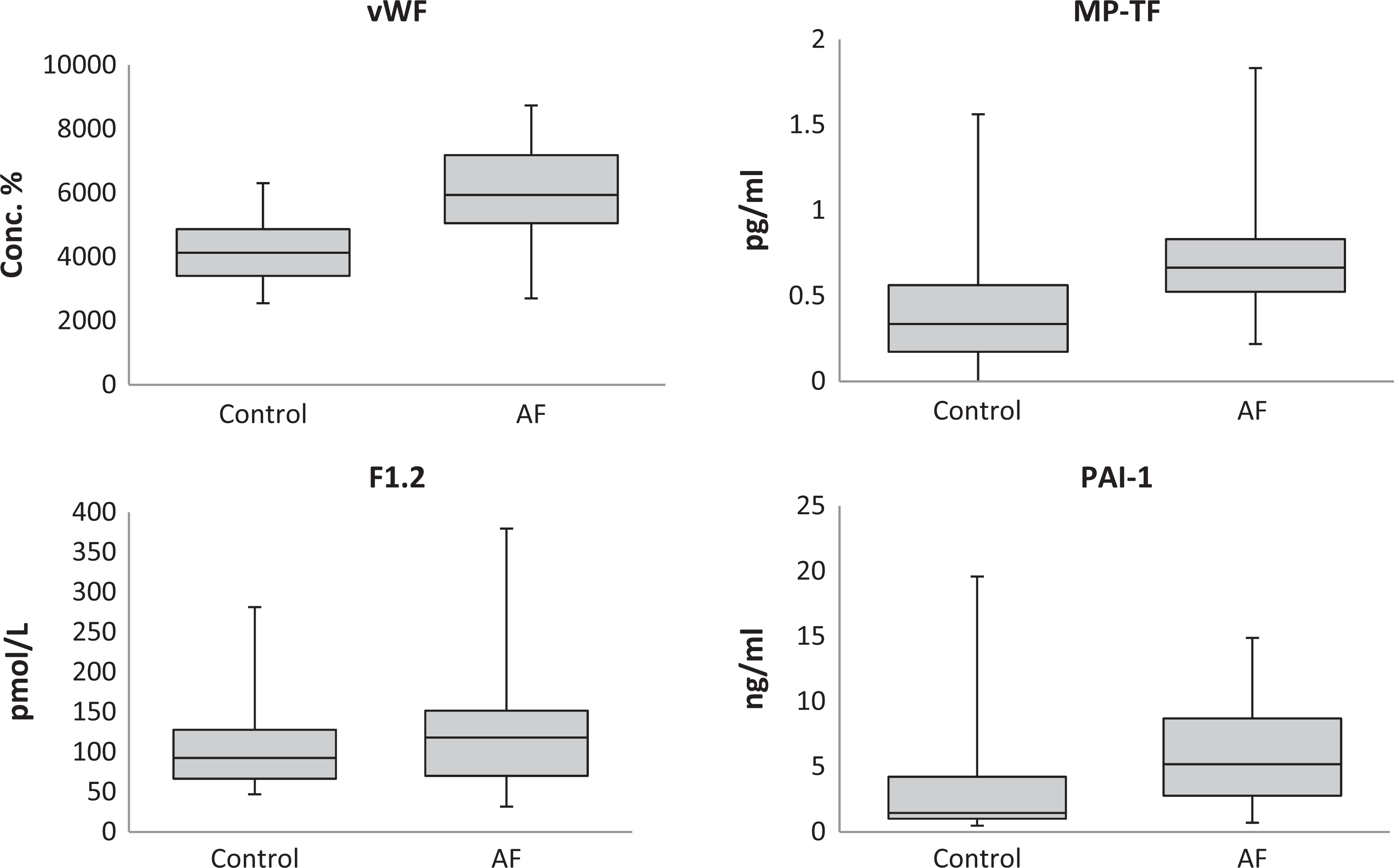
Box plots showing median levels of vWF, F1.2, MP-TF, and PAI-1 measured in control and AF groups. Boxes show interquartile ranges and I bars represent highest and lowest values. AF indicates atrial fibrillation; vWF, von Willebrand Factor; F1.2, prothrombin fragment 1+2; MP-TF, microparticle tissue factor; PAI-1, plasminogen activator inhibitor.
Control and AF Circulating Plasma Levels of Thrombotic Biomarkers vWF, F1.2, MP-TF, and PAI-1.a
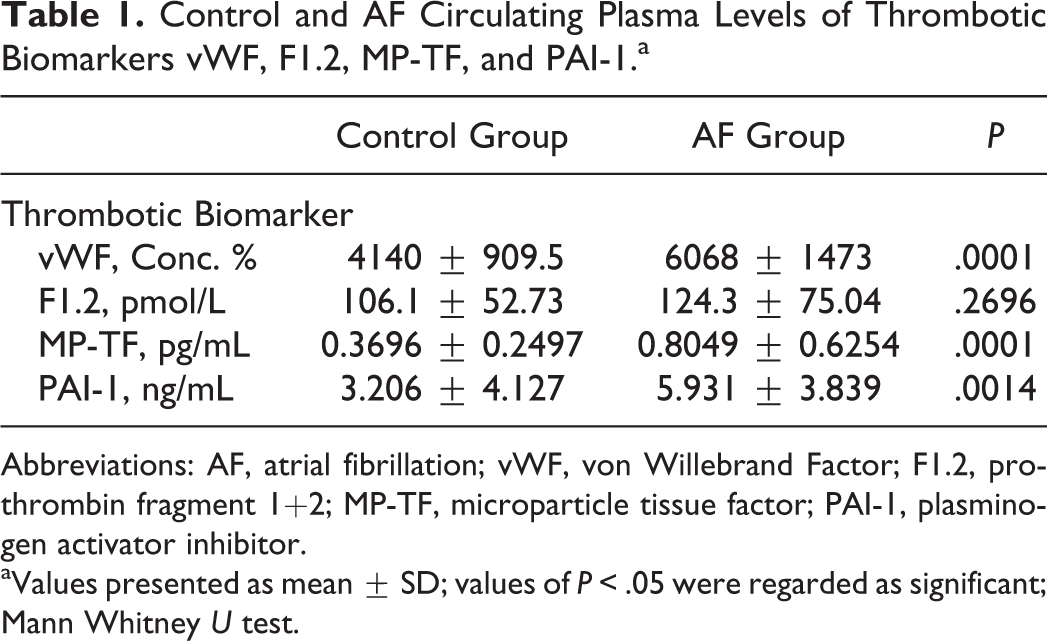
Abbreviations: AF, atrial fibrillation; vWF, von Willebrand Factor; F1.2, prothrombin fragment 1+2; MP-TF, microparticle tissue factor; PAI-1, plasminogen activator inhibitor.
aValues presented as mean ± SD; values of
Percent Change in Thrombotic Biomarkers
The percent changes in the thrombotic biomarker level of the AF group compared to the control group mean are seen in Figure 2. The MP-TF showed the largest percent change from the normal mean (109.7 ± 29.7). The PAI-1 had the second largest percent change (85.0 ± 21.9) followed by vWF (46.6 ± 6.5). The F1.2 had the smallest percent change increase (17.1 ± 12.9) and was the only thrombotic biomarker not statistically increased in the AF group compared to the control group.
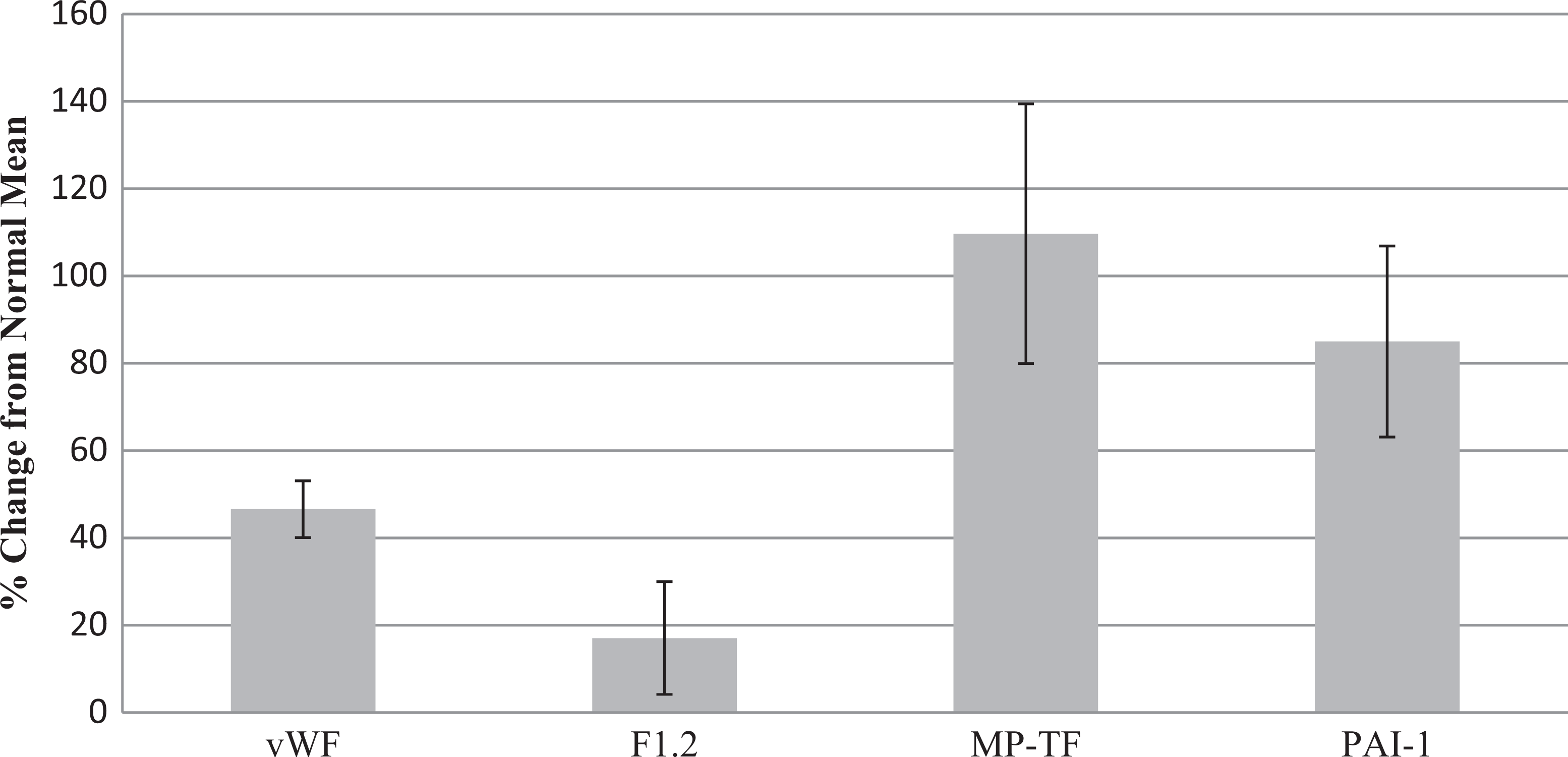
Percent change from normal of vWF, F1.2, MP-TF, and PAI-1 in AF group. AF indicates atrial fibrillation; vWF, von Willebrand Factor; F1.2, prothrombin fragment 1+2; MP-TF, microparticle tissue factor; PAI-1, plasminogen activator inhibitor. Values presented as mean ± standard error; values of
Comparison of the Thrombotic Biomarker Levels Based on Anticoagulant Therapy
The use of anticoagulants was also analyzed to assess the level of thrombogenic biomarkers in patients with AF based on anticoagulant therapy. Patients with AF (n = 30) were divided accordingly into anticoagulant (B, n = 21) and nonanticoagulant (C, n = 9) groups. Furthermore, the anticoagulant group was divided into new anticoagulants (D, rivaroxaban or apixaban, n = 16) or traditional anticoagulants (E, warfarin, n = 4). One patient was anticoagulated with dabigatran and was not included in this comparison.
Comparing patients with AF on anticoagulants (group B) to the control group, a statistical increase was seen in vWF, MP-TF, and PAI-1 in the AF anticoagulant group. Comparing patients with AF not on anticoagulants (group C) to the control group, a statistical increase was seen in F1.2 and PAI-1 in the AF nonanticoagulant group. Comparing patients with AF on anticoagulants (group B) to patients with AF not on anticoagulants (group C), a statistical increase was seen in vWF and MP-TF in patients with AF on anticoagulants. Additionally, comparing patients with AF on new anticoagulant therapy (group D) to patients with AF on traditional anticoagulant therapy (group E), there was no statistical difference in the level of any thrombotic biomarkers (Table 2).
Effect of Anticoagulant Therapy on Circulating Plasma Levels of Thrombotic Biomarkers vWF, F1.2, MP-TF, and PAI-1.a
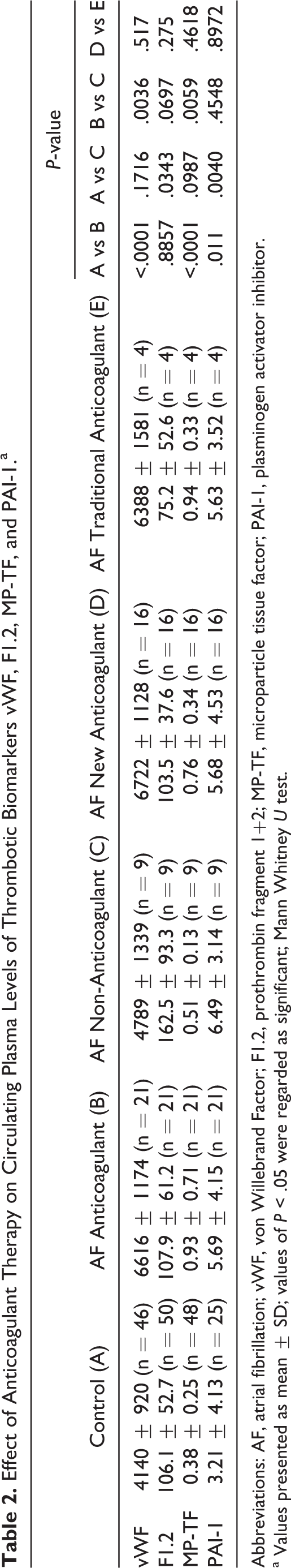
Abbreviations: AF, atrial fibrillation; vWF, von Willebrand Factor; F1.2, prothrombin fragment 1+2; MP-TF, microparticle tissue factor; PAI-1, plasminogen activator inhibitor.
a Values presented as mean ± SD; values of
Discussion
Many patients are assessed using a stroke risk stratification scale (CHADS2, CHADS-VASC, or CHADS) to determine whether anticoagulants should be used to prevent stroke in patients with AF. Of the 30 patients examined in this study, 8 of 9 who were not on anticoagulants had a stroke risk stratification score of 0, whereas 20 of 21 patients who were on anticoagulants had a score of
No difference in the F1.2 levels between the AF and control groups demonstrates that the use of anticoagulants limits the amount of thrombin generation in patients with AF. Previous studies have shown that the use of warfarin in patients with AF decreases levels of F1.2 compared to levels in patients with AF who are not on any anticoagulants.
22
A similar trend was seen in our patients, but the decrease of F1.2 in our anticoagulated patients with AF was not statistically significant (
Additionally, these data suggest that although very effective at maintaining normal F1.2 levels in AF, the newer anticoagulants, rivaroxaban and apixaban, and the traditional anticoagulant, warfarin, still leave additional prothrombotic biomarkers unaffected. These unaffected biomarkers could be the potential target of future drug therapies which could lower the risk of stroke in patients with AF even more than use of newer/traditional anticoagulants alone.
The pathophysiology of AF is complex and multifactorial. Moreover, \ patients with AF are represented by advanced age-group where atherosclerotic changes may contribute to vascular manifestations including thrombogenesis. Regardless of this process, thrombin generation markers such as those studied in this investigation are formed upon the activation processes leading to the conversion of prothrombin to thrombin. Thus, anticoagulants such as the newer anti-Xa agents should decrease the formation of these biomarkers. It is therefore reasonable to consider that beside the primary pathogenesis of AF, atherosclerotic manifestations may also contribute to the formation of these biomarkers of thrombin generation which can be further investigated.
This study represents an interim analysis of the markers of thrombin generation in an ongoing study on patients with nonvalvular AF treated with either warfarin or anti-Xa agents. The study is aimed to understand the role of these biomarkers in the pathogenesis in AF. The initial results of this study have provided useful information regarding the trends, where these thrombin generation biomarkers can be used for risk stratification and other prognostic purposes. However, despite the relatively smaller size of this study, it did allow for a baseline profiling of multiple prothrombotic biomarkers in patients in AF being treated with newer or traditional anticoagulants. Of note, increasing age could also be associated with the increased level of these biomarkers. Our control group included plasma samples from healthy volunteers with a lower age range than that of our study population. It would have been desirable to have a more representative age-matched control group. Based on the limited number of patients and the sample selection at baseline for this ongoing study, at this time there was no clinical correlation analysis carried out on the data generated in our study. Such correlation would be of value in establishing the prognostic value of the thrombin generation biomarkers and should be considered. To our knowledge, no current information has been gathered relating these thrombogenic biomarkers to specific newer and traditional anticoagulant therapies in the same study on AF. Our findings warrant further studies with larger cohort and age-matched controls.
Footnotes
Acknowledgments
The authors gratefully acknowledge the skillful assistance of the nursing staff of the Department of Cardiology at Loyola University Hospital in facilitating this study. We are thankful to Dr Wojcik, the chairperson of the Department of Pathology, for her support. We are also grateful to Dr Gail Hecht for providing the research opportunity to the primary author of this study through the Student Training in Approaches to Research (STAR) summer program. This study was carried out under a T35 grant from the National Heart Lung and Blood Institute of the National Institutes of Health to foster student research at Loyola University Medical Center.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
