Abstract
Background: Increased levels of factor VIII (FVIII) are a prevalent and independent risk factor for deep venous thrombosis (DVT) and are affected by von Willebrand factor (vWF) levels. Design and Methods: ADAMTS13 contributes to vWF levels, and we investigated genetic polymorphisms previously described to be associated with decreased levels of these proteins in 435 patients with DVT (126 M and 309 F; median age 37 years, range 18-68 years) and 580 controls (163 M and 417 F; median age 35 years, range 18-68 years). Subsequently, we investigated the relationship between the genotypes and plasma levels of FVIII, vWF, and DVT risk. Results: Patients with DVT showed higher plasma levels of FVIII:C, FVIII:Ag, and vWF:Ag (
Introduction
Deep venous thrombosis (DVT) is a multifactorial disease, and increased levels of coagulation factor VIII (FVIII) had been demonstrated as risk factor for first 1 and recurrent 2,3 episodes of DVT. The main determinants of FVIII in plasma are the von Willebrand factor (vWF) and the ABO blood group. 4,5 The role of other genetic factors in the determination of FVIII levels is not completely understood. Factors that are shown to significantly influence FVIII levels could potentially represent new risk factors for DVT.
ADAMTS13 is a metalloproteinase responsible for modulation of the molecular size of vWF multimers in plasma. An association between ADAMTS13 activity/antigen levels and vWF antigen has been demonstrated in physiological and pathological conditions characterized by increased risk of thrombosis.
6
More than 80 molecular changes, including polymorphisms, missense mutations, nonsense mutations, splice, frameshift, deletions, and insertions have been described in the
The 4751A>G polymorphism (Y1584C) in exon 28 of the
Brazilians represent a heterogeneous population characterized by the genetic admixture of Europeans, African immigrants, and native indigenous descendents. We recently demonstrated that in this population, increased levels of FVIII and vWF antigen are risk factors for DVT (odds ratio [OR] = 3.0). 21
Here we correlated 4751A>G (Y1584C) polymorphism in the
Materials and Methods
Patients and controls
Our study was designed as a nested case−control study, in which patients with DVT and without any of the classic risk factors for venous thromboembolism were paired with age- and gender-matched healthy controls from the same geographic origin of patients. Between January 1997 and September 2007, 768 consecutive adult patients with a first episode of acute DVT were admitted to the Hematology and Hemotherapy Center of the State University of Campinas in Brazil. All patients with DVT, who were not taking anticoagulants for at least 3 months, were initially eligible for the study. Their medical records were analyzed and those with cancer, chronic liver, renal or inflammatory disease, under medications that could interfere with coagulation or fibrinolysis were not eligible for the study. Blood samples were collected from all remaining patients. After thrombophilia investigation, carriers of natural anticoagulants deficiency (protein C, protein S, and antithrombin), and antiphospholipid antibodies were excluded. Other hereditary thrombophilias such as FV Leiden and FII G20210A mutation were not considered the exclusion criteria for entering the study. We only included patients between ages 18 and 68 years. Deep venous thrombosis was confirmed by imaging tests in all cases. Deep venous thrombosis was considered spontaneous if no external cause of thrombosis was present, such as surgery, immobilization, pregnancy, puerperium, and estrogens intake.
Ethnicity was determined based on ascendance up to the 3 generations and physical characteristics. Healthy adult individuals selected from blood donors were chosen as controls, and they lived in the same geographic area of patients. They were submitted to clinical and laboratory evaluation and the same exclusion criteria were applied to their selection. In addition, individuals with personal or familial history of DVT were excluded from the control group. This study was carried out with the approval of the ethics committee of the State University of Campinas and written informed consent was obtained from all participants.
Laboratory Methods
Factor VIII levels (FVIII:C) were measured by a 1-stage clotting assay with FVIII-deficient plasma (Diamed, Cresser, Switzerland). The FVIII:C tests were performed in duplicate on a automated coagulation analyzer (Koagulab 32-S, Diagnostica Stago, Asnieres-sur-Seine, France). Factor VIII antigen (FVIII:Ag) and vWF antigen (vWF:Ag) levels were measured using enzyme-linked immunosorbent assay (ELISA) with rabbit monoclonal antibodies (Dako, Glostrup, Denmark). C-reactive protein (CRP) levels were determined in the same samples by a nephelometric method (Dade Behring, Marburg, Germany) to exclude the influence of inflammation on FVIII and vWF levels. For individuals with CRP values higher than 1 mg/dL, an additional blood sample was analyzed. Factor VIII and vWF levels were considered only in the presence of normal CRP levels. ABO blood group was determined by agglutination and adsorption-elution test.
The region of vWF containing the 4751A>G polymorphism and the region of the ADAMTS13 containing the 1852C>G polymorphism were amplified by polymerase chain reaction (PCR). The 4751A>G polymorphism in vWF gene was determined using restriction fragment length polymorphism, as previously described. 22 The 1852C>G polymorphism was investigated by single strand conformation polymorphism (SSCP; PhastSystem, Pharmacia Biotech, Sweden) of amplified region of exon 16, including 110 to 115 bp of each junctional intronic region. All abnormal patterns identified were subsequently analyzed by automated sequencing of the amplified product.
Statistical Analysis
Factor VIII and vWF levels were described as median ± standard error. Medians were compared by the Mann-Whitney test. Genotype frequencies in both groups were compared by the Fisher exact test. A
The association between the genetic alterations and the risk for DVT was evaluated by calculation of the OR with 95% confidence interval (CI). All analysis was performed using the R Foundation for Statistical Computing, version 2.9.1.
Results
A total of 435 patients with DVT (126 male and 309 female) with a median age of 37 years (range 18-68 years) were included in the study. The control group consisted of 580 subjects (163 male and 417 female), with a median age of 35 years (range 18 to 68 years). No significant difference of gender, age, and ethnicity between patients and controls was observed (Table 1 ). Venous thrombosis was spontaneous in 163 (37.5%) patients and secondary to an acquired risk factor in 272 (62.5%). Among the latter group, the risk factor for DVT was surgery in 7.0%, immobilization in 9.5%, oral contraceptive use in 20.5%, pregnancy and puerperium in 21.0%, and other risk factors in 4.5%. Heterozygous for FV Leiden or the FII G20210A mutation were found in 27 (6.2%) and 20 (4.6%) patients, respectively. The sites of venous thrombosis were lower limbs (82.5%), retina (5.0%), brain (4.5%), upper extremities (4.0%), splenic-portal circulation (3.5%), and jugular veins (0.5%).
Clinical Characteristics of Patients With DVT and the Control Group
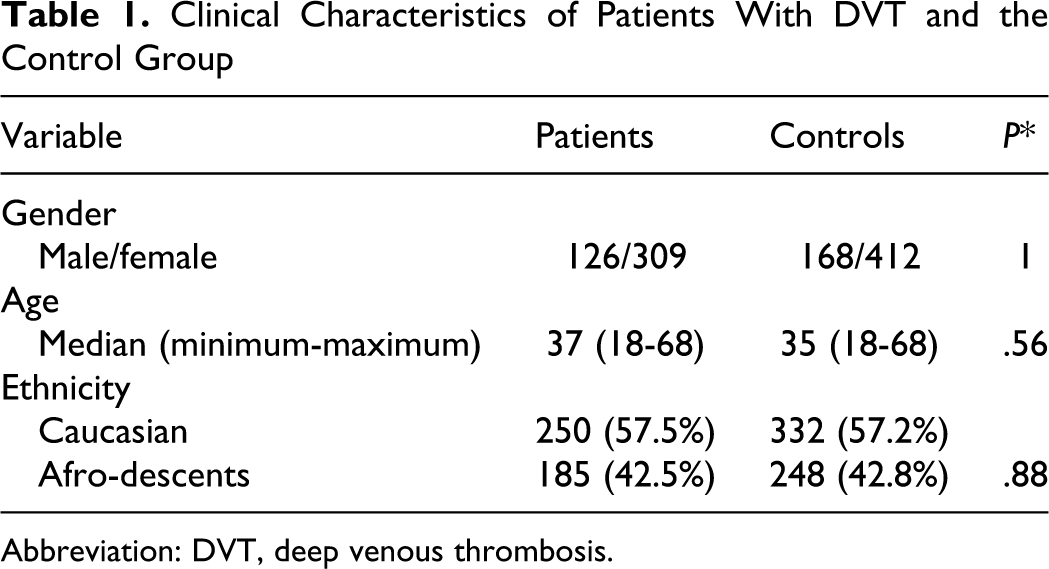
Abbreviation: DVT, deep venous thrombosis.
Patients with DVT had higher plasma levels of FVIII:C (mean 203.7 ±79.3 IU/dL vs 127.0 ±50.9 IU/dL;
Table 2 presents the results of the molecular analysis of our study. Genotype distributions of all molecular alterations were compatible with Hardy-Weinberg equilibrium among controls, suggesting that this population represented an unbiased selection of the general population.
Analysis of Prevalence and Risk for DVT of Molecular Changes Evaluated in this Study a
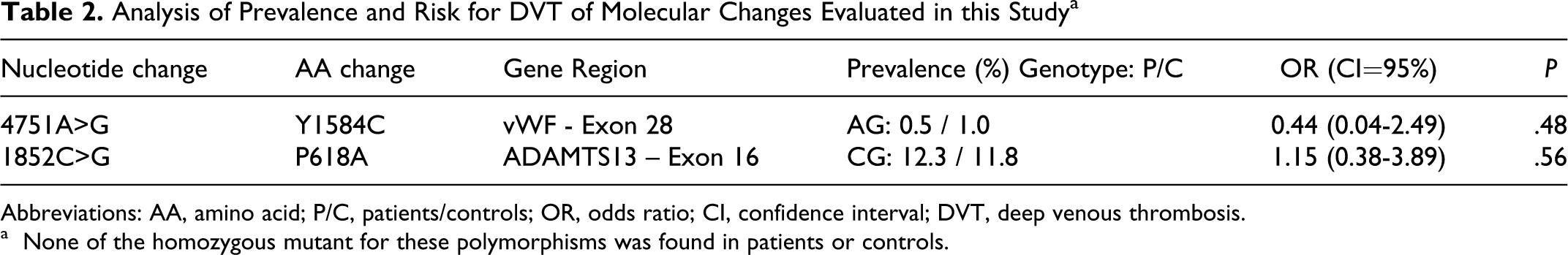
Abbreviations: AA, amino acid; P/C, patients/controls; OR, odds ratio; CI, confidence interval; DVT, deep venous thrombosis.
a
The 4751A>G polymorphism in the
Heterozygosity for the 1852C>G polymorphism in the
Relationship Between the Genotypes and Median Plasma Level of vWF:Ag (IU/dL), FVIII:Ag (IU/dL), and FVIII:C (IU/dL) in Patients and Controls a
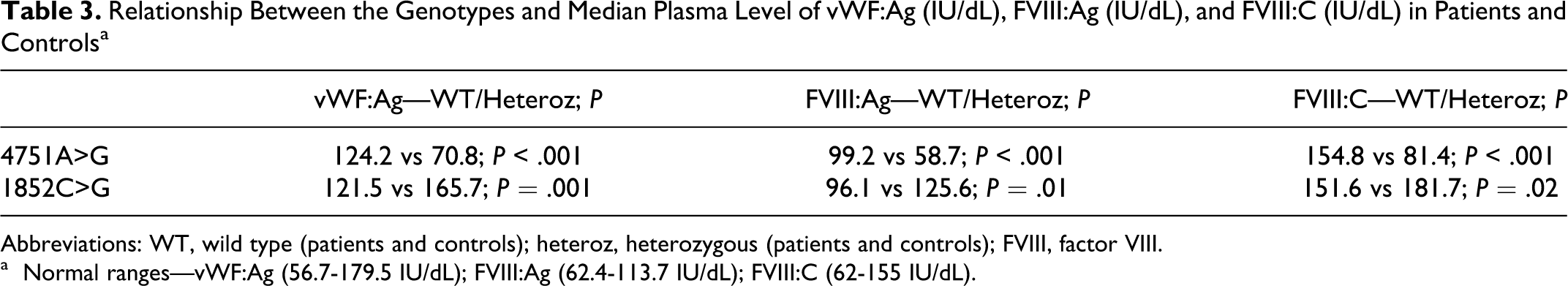
Abbreviations: WT, wild type (patients and controls); heteroz, heterozygous (patients and controls); FVIII, factor VIII.
a Normal ranges—vWF:Ag (56.7-179.5 IU/dL); FVIII:Ag (62.4-113.7 IU/dL); FVIII:C (62-155 IU/dL).
These results were confirmed in the multivariate analysis adjusted for ABO blood group (data not shown).
Sequencing of the SSCP fragments that presented abnormal pattern during determination of 1852C>G polymorphism resulted in the identification of 7 molecular changes in exon 16 and junctional intronic regions of
The distribution of all molecular alterations was not different between patients and controls and was not associated with an increased risk of DVT (see Table 2).
Discussion
Increased FVIII:C levels are dependent on the interaction of genetic and acquired factors. Although some of these factors such as ABO blood group and vWF levels have been defined, the role of other factors such as ADAMTS13 is yet to be determined. Because FVIII:C is a well-established risk factor for DVT, the identification of novel genetic determinants of the individual variability in FVIII:C levels could potentially identify new risk factor for DVT. In recent years, a large number of studies have investigated molecular alterations in the
In our study, we used a highly selected group of patients and controls assisted at the Hematology and Hemotherapy Center of the State University of Campinas, for 10 years. We believe that this population is suitable to help elucidate other genetic determinants of FVIII:C levels and to explore their potential role as a risk factor for DVT. The rationale for excluding patients with DVT who had an underlying risk factor, such as cancer, chronic liver, or kidney disease, was to exclude the role of those diseases on FVIII levels, as this factor is an acute phase protein.
As previously shown by our group, FVIII:C and vWF:Ag levels were significantly increased in our patients with DVT when compared to matched controls. 19 In addition, the concomitant determination of CRP levels when evaluating FVIII:C levels can exclude that the observed changes are secondary to nonspecific inflammation, thus reinforcing the role of FVIII:C as a risk factor for DVT in the population of our study.
Our results confirmed that individuals heterozygous for the 4751A>G polymorphism in the
A large number of studies have investigated the role of polymorphisms and mutations in
Although our study demonstrated a relationship between polymorphisms in
We detected 5 new molecular alterations located in exon 16 and junctional intronic regions of
In conclusion, our study confirms that high plasma levels of FVIII:C, FVIII:Ag, and vWF:Ag are increased in patients with DVT. The 4751A>G polymorphism in the
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
This study was, in part, supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). The author(s) declared that the study sponsor had no involvement in the study design, in the collection, analysis and interpretation of data; in the writing of the manuscript, and in the decision to submit the manuscript for publication.
