Abstract
There is growing research interest into the etiologies of cryptogenic stroke, in particular as it relates to hypercoagulable states. An elevation in serum levels of the procoagulant factor VIII is recognized as one such culprit of occult cerebral infarctions. It is the objective of the present review to summarize the molecular role of factor VIII in thrombogenesis and its clinical use in the diagnosis and prognosis of acute ischemic stroke. We also discuss the utility of screening for serum factor VIII levels among patients at risk for, or those who have experienced, ischemic stroke.
Background
In the 1950s, investigators discovered that patients with prolonged bleeding time could be treated with the replacement of a particular clotting factor. 1 This factor was originally thought to be von Willebrand factor (vWF), but was later characterized as factor VIII (FVIII). 2 Electron microscopy confirmed that the procoagulant FVIII played a primary role in the adhesion of platelets to exposed subendothelium during times of injury, but this did not occur until the 1970s. 3 Produced primarily in liver cells, FVIII circulates through the vasculature in its inactive form and is protected from proteolysis through its noncovalent interaction with vWF. 4,5 When injury occurs, it is activated by thrombin and dissociates from vWF, thereby activating the coagulation cascade. 6 Despite the otherwise “dormant” state, the serum concentration of FVIII directly correlates with the occurrence of de novo and recurrent thrombotic events, 7,8 including acute ischemic stroke. 9 –12 The role of FVIII in the risk and pathogenesis of stroke is the subject of this review.
Although “stroke” comprises various cerebrovascular syndromes, the focus of our review will pertain to ischemic stroke subtypes, and therefore, any mention of stroke will denote an ischemic cerebrovascular event.
Molecular Role of Factor VIII in Thrombosis
Factor VIII, a 280 kDa glycoprotein expressed by the human X chromosome, plays a pivotal role in the propagation of clot formation in both the intrinsic and extrinsic coagulation pathways. 6,13,14 It is stabilized within the plasma by vWF. When bound together, this protein complex has a protective effect on the FVIII antigen (FVIII: ag) and permits appropriate binding and cleavage of FVIII with other components of the coagulation cascade during instances of vessel breakdown and hemorrhage. 6 The FVIII: vWF complex also plays an important role in primary coagulation by mediating platelet attachment to damaged subendothelium. 3,5 Although FVIII is critical for proper coagulation, it has been increasingly shown to be associated with cardiovascular health outcomes when it is transcribed in excess or its activity level is amplified.
The role of FVIII in prothrombotic syndromes, such as myocardial infarction, deep vein thrombosis (DVT), and acute ischemic stroke was originally discovered by investigators who found that the absence of FVIII in hemophilic syndromes was associated with a reduced risk of coronary events. 15 This same group would later demonstrate a strong association between elevated FVIII levels and nonmalignant DVT in a small case–control study. 7 Other investigators described a higher risk of DVT recurrence in patients with elevated FVIII, quantified as a higher relative risk of 8% for every 10 IU/dL elevation in serum FVIII. 8 The relationship between elevated FVIII and other thrombotic disorders such as myocardial infarction, 16 pulmonary embolism, 17 chronic thromboembolic pulmonary hypertension, 18 and death from these disease states, 19 has been well described, however, several of these relationships remain contested. 20 –23
The levels of FVIII appear to increase during the acute phase response, 24,25 implicating its role as a biomarker of systemic inflammation. Within the context of ischemic stroke, elevation of FVIII has been regarded as a result of the inflammation associated with the acute phase of stroke. 10,26 Newer data challenge this oversimplification of FVIII in the acute phase response and implicate an independent correlation between FVIII levels or activity and thrombosis. 27 In this study by O’Donnell et al in patients with thrombotic events, only half of the cases of persistently elevated FVIII levels can be attributed to elevations in vWF. 27 This is highly suggestive that FVIII, and not vWF, remains the driving force of these 2 markers behind thrombotic events. At our center, when analyzing the relationship between FVIII and vWF elevation and ischemic stroke outcomes, we observed that elevation of FVIII was significantly associated with elevated levels of C-reactive protein and erythrocyte sedimentation rate, irrespective of vWF level. 28
There remains no concrete data implicating that acute inflammation induces transcriptional elements or promoters of the FVIII gene, thereby resulting in amplified production of this procoagulant antigen. One particular prospective study that assessed the effect of vitamin E on oxidative stress mechanisms demonstrated that FVIII increases in response to tissue hypoxia, thereby promoting thrombogenesis.
29
Compared to normal oxygen conditions (21% oxygen content), patients in the placebo arm of this trial who experienced significant hypoxia (12% oxygen content) experienced greater increases in FVIII levels (248 vs 171 ng/mL,
Presently, the quantification of FVIII can be made using one of the 2 distinct methods: determination of FVIII activity (FVIII: c) and FVIII: ag levels. Each serves a unique purpose and provides distinct clinical information although both have a positive correlation with each other and with thrombotic disease. 25 FVIII: c is typically measured using a standardized coagulation assay whereas an enzyme-linked immunosorbent assay is typically performed to quantify the amount of FVIII: ag present in sera. In the acute phase response, both FVIII: ag and FVIII: c have been shown to rise, 30 which we speculate may occur in a causal fashion whereby FVIII: ag production increases and effects a subsequent rise in function of the procoagulant. 24 This is could play a role in the risk of stroke and stroke progression, which we have seen in previous investigations. 28
Many studies on FVIII investigate either
Factor VIII and Stroke
Incident Stroke
Although there are some conflicting results, 22,23 it is well established that FVIII levels and/or activity are associated with acute ischemic stroke risk. Historically, the first investigation to relate FVIII with ischemic stroke was published by Gaston et al in 1971. 31 Since then, numerous investigations have been published demonstrating an association between higher FVIII: c or antigen levels and the incidence of stroke. 9,11,12,32,33 In the largest prospective cohort to date, the Atherosclerosis Risk in Communities investigators confirmed the association between elevated FVIII and ischemic stroke. 11 In this study, the investigators followed nearly 15 000 patients without known cardiovascular disease for up to 9 years and characterized 191 ischemic stroke events. The adjusted relative risk for ischemic stroke in patients with the uppermost quartile of FVIII when compared to the lowermost quartile was calculated at 1.93 (95% confidence interval [CI] 1.2-3.1). 11
Stroke Etiology
Much of the data pertaining to FVIII and stroke with respect to etiology involve atherothrombotic strokes or strokes due to large vessel disease.
9,22,34
In one Chinese study, patients with carotid plaque on ultrasonography had a clinically, but not statistically, significantly elevated FVIII: c when compared to healthy controls (1.66 vs 1.43,
Lately, cryptogenic stroke has gained attention as more hypercoagulable states are being related to unclear etiologies of ischemic stroke. In one case–control study of 150 patients by Karttunen et al, the investigators found that patients with cryptogenic stroke had a higher mean FVIII: c when compared to controls (124% vs 112%,
The relationship between cardioembolic stroke and FVIII has been investigated, but is less well described. Data from a Swedish case–control study of 100 patients suggest that elevated FVIII may be associated with stroke in patients with nonvalvular atrial fibrillation.
32
In this investigation by Gustafsson et al, FVIII levels were confirmed using 2 unique clotting assays.
32
In both methods, FVIII was found to be elevated in patients having stroke with nonvalvular atrial fibrillation when compared to either (1) patients with stroke in sinus rhythm or (2) healthy patients (without stroke or atrial fibrillation). Interestingly, these investigators also found that patients with nonvalvular atrial fibrillation who did
Paradoxical embolism, in which a venous thrombus embolizes from the venous circulation to the arterial circulation through an intracardiac shunt (classically a patent foramen ovale), may also be more common in patients with elevated FVIII given their higher risk of DVT. 7,8 This mechanism has been proposed in 1 case–control cohort of patients with cryptogenic stroke, 35 but a causal relationship has yet to be demonstrated in this patient population.
Small vessel disease seems to bear little, if any, relation to FVIII. One study by Knottnerus et al attempted to clarify an association between small vessel disease and FVIII. 36 These investigators found that FVIII: c was not significantly different between patients with isolated lacunes when compared to symptomatic lacunes, as well as extensive white matter lesions (consistent with small vessel ischemic disease) when compared to asymptomatic lacunes who lacked extensive white matter lesions (109% ± 25% vs 113% ± 25% vs 115% ± 26%). 36
Stroke Severity
In our center’s experience, we have been unable to detect an independent association between elevated FVIII: c and baseline stroke severity, as measured by the National Institutes of Health Stroke Scale (NIHSS) score. 10 However, we found that combined elevations of FVIII and vWF were associated with worse neurologic outcome (defined as discharge modified Rankin Scale score > 2). 28 There also appeared to be a clinically significant association between FVIII and vWF levels and inpatient complications (such as infection), neurologic deterioration (defined as an increase in NIHSS score of 2 or more within a 24-hour period 37 ), and recurrent thrombotic events following acute ischemic stroke. 28
Risk of Elevated FVIII
Limited evidence is available currently regarding what characteristics predispose patients to elevated FVIII. The levels of FVIII have some association with the acute phase response, but they have also been independently elevated in patients without other elevations in biomarkers traditionally expressed during acute inflammation. Some studies have suggested a familial relationship 38 and blood type relationship. 39 One genetic study involving over 50 000 patients identified a single nucleotide polymorphism in the ABO locus (rs505922) that was associated with FVIII levels and ultimately risk of stroke. 40 Individuals with non-O blood types (particularly AB blood type) appear to be more likely to have elevated levels of FVIII as compared to those with O-type. There also appears to be an association between elevated FVIII and age and gender, with older patients, blacks, and women at higher risk for this phenomenon. 9,39,41 –43
Pediatric Considerations
Although there are occasional case reports to the contrary, 44 the association between FVIII and incident stroke has not been confirmed in children with stroke. 45,46 This may be due to sample size issues or the differing etiologies of pediatric ischemic stroke.
Table 1 depicts 11 key studies from 1987 to present that have investigated the association between FVIII and acute ischemic stroke.
Summary of Studies Investigating the Association Between Factor VIII and Ischemic Stroke in Adults.a
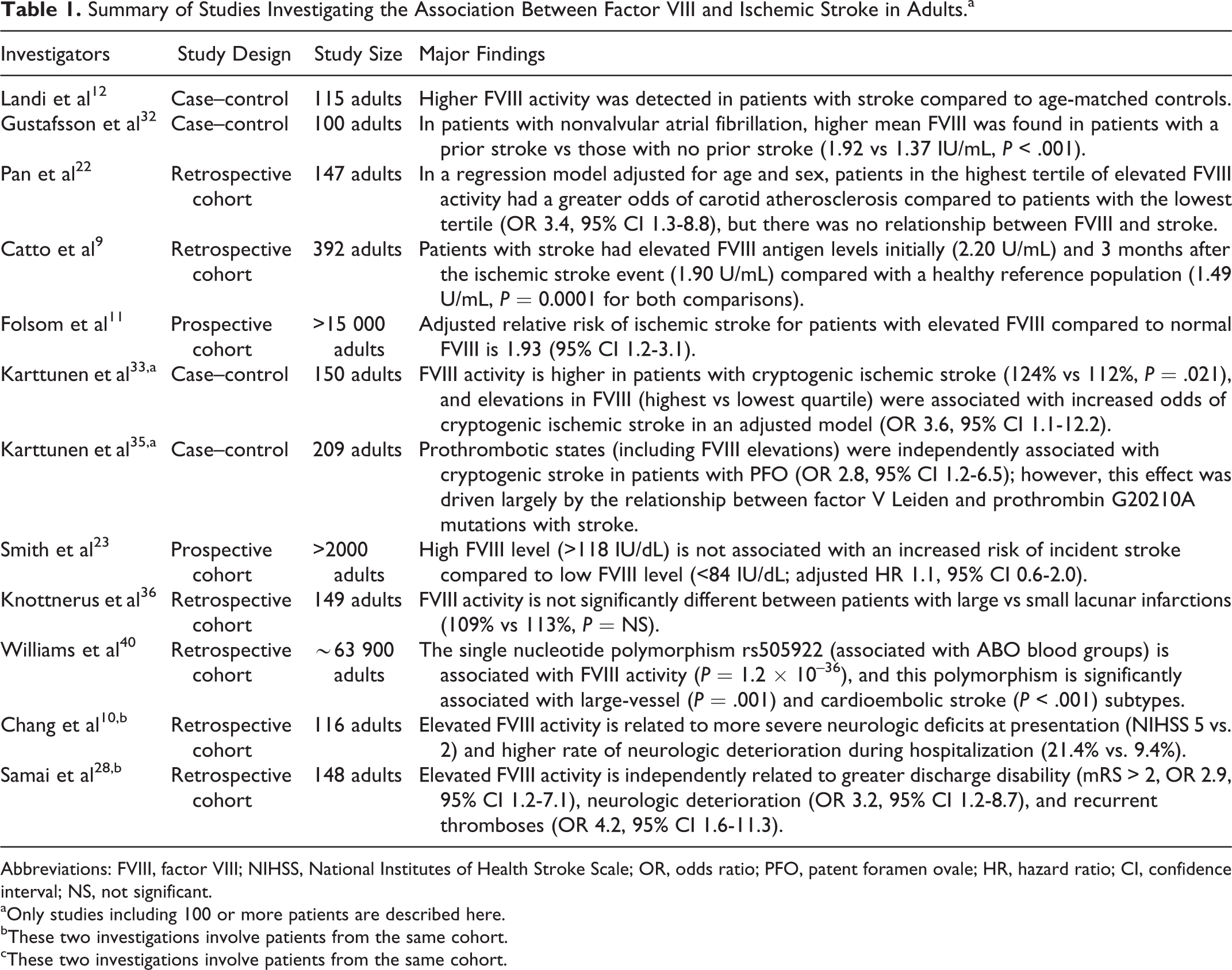
Abbreviations: FVIII, factor VIII; NIHSS, National Institutes of Health Stroke Scale; OR, odds ratio; PFO, patent foramen ovale; HR, hazard ratio; CI, confidence interval; NS, not significant.
aOnly studies including 100 or more patients are described here.
bThese two investigations involve patients from the same cohort.
cThese two investigations involve patients from the same cohort.
Future Directions
Utility of Screening
The association between FVIII and stroke raises the question, should we screen for FVIII levels in patients with stroke, or at least patients at risk of stroke? This has been raised before, 47 but current guidelines do not recommend for or against the serologic testing of FVIII levels for routine stroke evaluation. 48
At the time of publication, the authors of the present article argue against the
Once a patient is found to have elevated FVIII, the clinician may also be left more puzzled than when he or she had initially begun the evaluation—in part due to no known treatment and no known clinical result of such treatment. Presently, we have no guidelines for the treatment of elevated FVIII in humans. Furthermore, we have no data to support that these patients would improve from pharmacologic intervention. We have already demonstrated a higher proportion of neurologic deterioration in patients with elevated FVIII who suffer ischemic stroke, 28 but whether any targeted intervention to reduce FVIII may reduce the risk of neurologic deterioration is unknown.
Conclusions
In conclusion, FVIII levels appear to be associated with both ischemic stroke and outcome following ischemic stroke. This has been demonstrated in stroke of large vessel, cardioembolic, and cryptogenic etiologies. Epidemiologic studies have shown that asymptomatic community dwelling adults with elevated FVIII levels are at twice the risk of ischemic stroke when compared to patients with normal or low FVIII. FVIII is known to be elevated in older patients, blacks, and women. Currently, guidelines do not recommend the routine screening of FVIII in patients with stroke, despite its potential role in prognostication and association with subsequent thrombotic events. Future investigations are called upon to identify whether the measurement of FVIII is justifiable as a screening tool for prognosis or treatment in adult patients who have suffered ischemic stroke.
Footnotes
Author’s Note
This manuscript has not been the subject of any previous oral or poster presentation. Dr. Boehme is supported by NINDS NIH T32 NS007153-31. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NINDS or the NIH. The remaining authors represent no other sources of funding. All authors report no competing financial interests exist.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Boehme is supported by NINDS NIH T32 NS007153-31. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NINDS or the NIH.
