Abstract
Background:
Plasma levels of estimated whole blood viscosity (eWBV) have been increased by endothelial inflammation. Because there were no consistent data for assessing the eWBV levels for prediction of cardiovascular event (CVE) in patients with chronic kidney disease (CKD). We aimed to investigate the relationship between plasma eWBV levels and CVEs in patients with CKD.
Materials and Methods:
We conducted a prospective, cross-sectional, long-term follow-up study, assessing the relationship between plasma eWBV levels and CVE (either fatal or nonfatal) in patients with newly diagnosed CKD. We also evaluated estimated glomerular filtration rate (eGFR), pentraxin 3 (PTX3), high-sensitivity C-reactive protein (hsCRP), and flow-mediated dilatation (FMD).
Results:
Study patients were divided into 2 groups: patients with CVE and patients without CVE. The eWBV levels were higher in patients with CVE. Additionally, PTX3 and hsCRP were higher, and FMD and eGFR were lower in patients with CVE compared to those without CVE. According to the Cox regression analysis, WBV, plasma asymmetric dimethylarginine levels, FMD, hsCRP, eGFR, systolic blood pressure, calcium, and history of diabetes were independent predictors of CVEs in patients with CKD. Kaplan Meier survival curves were generated to establish the impact of the WBV on the cumulative survival of the cohort. Patients with eWBV values higher than 5.2 centipoise (cP) had lower survival rates when compared to patients with eWBV values lower than 5.2 cP (log rank = 4.49 df = 1 P = .034).
Conclusion:
In conclusion, plasma eWBV levels may increase the presence of lower eGFR and affect CVE in patients with CKD independent of classical and unconventional risk factors.
Introduction
Chronic kidney disease (CKD) is known to be associated with high rate of cardiovascular diseases (CVDs), because of them, it is also a higher mortality rate compared to that of general population in the worldwide. 1 It has also much more than higher cardiovascular event (CVE). 2 Endothelial dysfunction (ED) is the early pathophysiological stage in the advancement of atherosclerotic process that causes the CVD manifestations. 2 The pathophysiologic atherosclerosis process is a complex multifactorial features; one of the crucial of them; blood flow induced shear stress may cause the ED. Thus, shear stress, particularly the blood flow pressure, plays an important role in the pathophysiology of the atherosclerotic plaque.
Estimated whole blood viscosity (eWBV) is known to be positively correlated with shear stress and is therefore a useful method. 3 Recent studies displayed that eWBV levels play a major role in atherosclerosis. Estimated whole blood viscosity is found to be related to carotid thickness. Also, it is believed that rheologic factors are concerned about the subclinical step of atherosclerosis. 4,5 Indeed, there is increasing evidence that eWBV levels may be a predictor for risk of CVD. 6 Furthermore, eWBV is increased in many chronic illnesses such as hypertension, metabolic syndrome, diabetes mellitus (DM), CVD, and stroke. 7 –9 Additionally, several large prospective studies have shown the important link between eWBV and the risk of primary CVE. 10,11
Cardiovascular diseases beginning with shear stress are the major cause of mortality in patients with CKD. From that point of view, the aim of the current study is to investigate the associations between WBV and differ markers as predictor of CVE and is also planned to assess the relationship between plasma WBV and CVE in patients with CKD.
Materials and Methods
Study Population
The study was designed to determine the risk factors for CVE in patients with newly diagnosed CKD and also the relationship between eWBV and CVE. The study was certified by the local ethics committee. All the individuals signed the written informed consent form. A total of 775 patients who were referred to the Nephrology Department of the Gulhane Military Medical Academy, Ankara, Turkey, with the diagnosis or prediagnosis of renal failure from January 2008 to July 2014 enrolled in the study. All patients had CKD which was diagnosed in accordance with estimated glomerular filtration rate (eGFR) and presence of renal damage that was described by National Kidney Foundation, Kidney Disease Outcomes Quality Initiative(K/DQOI) Guidelines. 12 In all, 417 patients (aged 51 ± 12 years old) were included in the study.
Measurement of Blood Pressure
Blood pressure (BP) measurements were performed from the left arm on the brachial artery using sphygmomanometer. The BP values were recorded as systolic BP (SBP) and diastolic BP (DBP) with a mean of at least 3 consecutive measurements following a 15-minute resting period. Hypertension was described as mean SBP ≥140 mm Hg or mean DBP≥ 90 mm Hg or using any antihypertensive medication.
Estimation of eGFR
Severity of CKD was classified according to the eGFR which was calculated using the following Modification of Diet in Renal Disease formula: GFR = 186 × Pcr-1.154 × age-0.203 × 1.212 (for black) × 0.742 (for female). 13 The results were categorized as stage 1 to 5 as described by the K/DOQI.
All the individuals were followed up for a long time using time-to-event analysis by the time occurrence of fatal or nonfatal CVEs. We included all of the major cardiovascular outcomes including fatal and nonfatal (eg, myocardial infarction, death, and stroke). Follow-up data were obtained from hospital records or through patient interviews (in person or by telephone), their families, or patient’s primary care physicians. The diagnosis of peripheral vascular disease (PVD) and aortic aneurysm was made by physical examination (including ankle brachial index), abdominal ultrasound, and duplex ultrasonography as required.
Exclusion Criteria
We excluded from the study 140 patients who were taking medication such as erythropoietin (n = 62), active D vitamin (n = 55), or supplemental vitamin pills (n = 23) because of the probable effects on endothelial functions. Additionally, patients with acute infections and those who are unwilling to participate were excluded from the study (n = 71). One hundred and forty seven patients who could not be followed up due to lost contact or transfer to other dialysis unit (n = 105), those with viral hepatitis (n = 7) PVD or vasculitis (n = 14), or withdrew consent (n = 21) were excluded from the study population. The diagnosis of PVD and aortic aneurysm was made by physical examination (including ankle brachial index), abdominal ultrasound, and duplex ultrasonography (as required.)
Blood Specimens and Laboratory Analysis
Blood samples were obtained intravenously following 12-hour fasting in the morning. We tested the levels of plasma glucose, triglycerides (TG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), albumin, total protein, and so on from those venous blood samples. We analyzed TC, TG, and HDL-C by enzymatic colorimetric method with Olympus AU 600 auto-analyzer using reagents from Olympus Diagnostics, GmbH (Hamburg, Germany). Low-density lipoprotein cholesterol was measured by Friedewald formula. 14 Serum levels of total calcium were tested by the Cresolphtalein Complexone method using Menagent Calcium 60-second kits (Menarini Diagnostics, Florence, Italy). We also analyzed the serum phosphorus levels via the ammonia molybdate complex method using Menagent Phosphofix kits (Menarini Diagnostics). Intact parathormone (PTH) was tested by immunoradiometric assay (IRMA) IRMA, using a commercial kit (Immulite Intact PTH; Diagnostic Product Corporation, Los Angeles, California) with a sensitivity of 1 pg/mL.
Measurement of high-sensitivity C-reactive protein (hsCRP) was done by the dilution of serum samples with a ratio of 1:101 via diluent solution. Calibrators, kit controls, and serum samples were all added on each micro-well with an incubation period of 30 minutes. Following 3 washing steps, 100 µL of enzyme conjugate (peroxidase labeled anti-CRP) was added on each micro-well and incubated at room temperature in dark for 15 minutes. The reaction was stopped with a stop solution, and photometric measurement was performed at a wavelength of 450 nm . The serum concentration was calculated as mg/L with a graph that was made by noting the absorbance levels of the calibrators.
The serum basal insulin value was measured by coated tube method (Diagnostic Product Corporation). Insulin resistance score, homeostasis model of assessment-estimated insulin resistance (HOMA-IR), was measured by the following formula 15 : HOMA-IR = fasting plasma glucose (mg/dL) × immunoreactive insulin (IRI; µIU/mL)/405.
The levels of pentraxin 3 (PTX3) were measured from frozen serum using a commercial enzyme-linked immunosorbent assay kit (Quantakine DPTX 30; R&D Systems Inc, Minneapolis, Minnesota) according to the manufacturer’s instructions.
Serum levels of asymmetric dimethylarginine (ADMA) were measured using high-performance liquid chromatography. 16 In brief, to 1 mL serum, a mixture of 1 mL serum enriched by 20 mg of 5-sulfosalisilic acid (5-SSA) was added and left in an ice-bath for 10 minutes. The precipitated protein was removed by centrifugation at 2000g for 10 minutes. Supernatant of 10 μL which was filtered through a 0.2-µm filter was mixed with 100 µL of derivatization reagent (prepared by dissolving 10 mg o-phtaldialdehyde in 0.5 mL of methanol, 2 mL of 0.4 mmol/L borate buffer (pH 10.0), and 30 µL of 2-mercaptoethanol) and then injected into the chromatographic system. Separation of ADMA was achieved with a 150 × 4 mm I.D. Nova-pak C18 column with a particle size of 5 µm (Waters, Millipore, Milford, Massachusetts) using 50 mmol/L sodium acetate (pH 6.8), methanol, and tetrahydrofurane as mobile phase (A, 82:17:1; B, 22:77:1) at a flow rate of 1.0 mL/min. The areas of peaks detected by the fluorescent detector (Ex: 338 nm; Em: 425 nm) were used as quantification. The variability of the method was less than 7%, and the detection limit of the assay was 0.01 µmol/L.
Flow-mediated dilatation (FMD) reflects the nitric oxide (NO) bioavailability to the endothelium and is well determined by the serum ADMA concentration. 17 The FMD (endothelium-dependent vasodilatation) of the brachial artery was assessed noninvasively, using high-resolution ultrasonography. 18 The method for the vascular assessment met the criteria that were mentioned by the International Brachial Artery Reactivity Task Force. 19 Measurements were made by a single observer using an ATL 5000 ultrasound system (Advanced Technology Laboratories Inc., Bothell, Washington) with a 12-Mhz probe. Following the patients resting at supine position for at least 15 minutes, the patient’s arm was gently immobilized in the extended position. At least 3 appropriate measurements of end-diastolic brachial artery diameter were made from single 2-dimensional frames from the brachial artery 2 to 4 cm above the antecubital fossa. A pneumatic tourniquet was inflated to 200 mm Hg until the radial pulse could not be palpable. After 5 minutes, the cuff was deflated. Following 60 seconds of deflation, flow measurements were carried out. Maximum diameter of FMD dilatation was determined as the average of at least 3 consecutive measurements. The FMD was then calculated as the change in diameter as percentage compared to baseline resting diameters. All the images were digitally recorded and analyzed by radiologists blinded to project.
Whole Blood Viscosity Estimation
Estimated whole blood viscosity at high shear stress was determined from various possible pairs of hematocrit and total proteins, arithmetically, according to validated formula, WBV (208 s−1) = 0.12 × HCT + 0.17(TP − 2.07). 20
Urinary analysis
The 24-hour urine collection was performed 3 times, and the average of three 24-hour proteinuria measurements was taken as the representative of each participant’s 24-hour protein excretion rate.
Statistical Analysis
Normality of numerical variables was tested by Kolmogorov-Smirnov test. Numerical data with normal distribution were presented as mean ± standard deviation (SD), while data with skewed distribution were presented median (interquartile range). Numerical data with normal distribution was analyzed by independent samples t test, while data with skewed distribution were analyzed by Mann-Whitney U test. Categorical variables were presented as percentages and analyzed by chi-square test. P values <.05 were determined for the statistical significance.
Correlation analysis of numerical or categorical variables was performed by Pearson and Spearman correlation tests, respectively. Multivariate COX regression analysis with stepwise model was used to determine the predictors for cardiovascular outcomes. Survival and time-to-event analysis of cardiovascular outcomes (using a composite of fatal and nonfatal events) was done using Cox proportional hazards model, including adjustment for potential confounding factors. Data are presented in the form of hazard ratios (HR) and 95% confidence intervals (CIs). All the statistical analyses were performed using SPSS 17.0 (SPSS Inc, Chicago, Illinois).
Results
Baseline demographic and clinical parameters of study patients are given in Table 1. Of the patients, 187 were on antihypertensive medications and statins (40 patients were treated with calcium channel antagonists, 9 with beta-blocker agents, 8 with alpha blockers, 66 with angiotensin-converting enzyme [ACE] inhibitors, 45 with ACE angiotensin receptor blocker, 13 with loop diuretics, and 6 with statins). At the time of evaluation, 110 patients were on antidiabetic therapy (37 patients were treated with oral antidiabetic agents and 63 with insulin). As soon as diabetic nephropathy was diagnosed, all patients taking oral antidiabetic medications were transitioned to insulin. Eighty-six (21%) of these patients had a history of CVD as defined by medical history and/or clinical findings at the time of enrollment. Of these 86 patients, 16 had had cerebrovascular accidents (stroke), 53 had CVD (acute myocardial infarction, unstable angina pectoris, or had undergone coronary artery bypass surgery or percutaneous coronary intervention), 14 had a history of peripheral ischemic atherosclerotic vascular disease; and 3 patients had a history of an aortic aneurysm. Smoking habits were recorded as follows: While 187 patients were former or current smokers, 230 were nonsmokers.
Baseline Demographic and Clinical Characteristics of the Study Participants.a
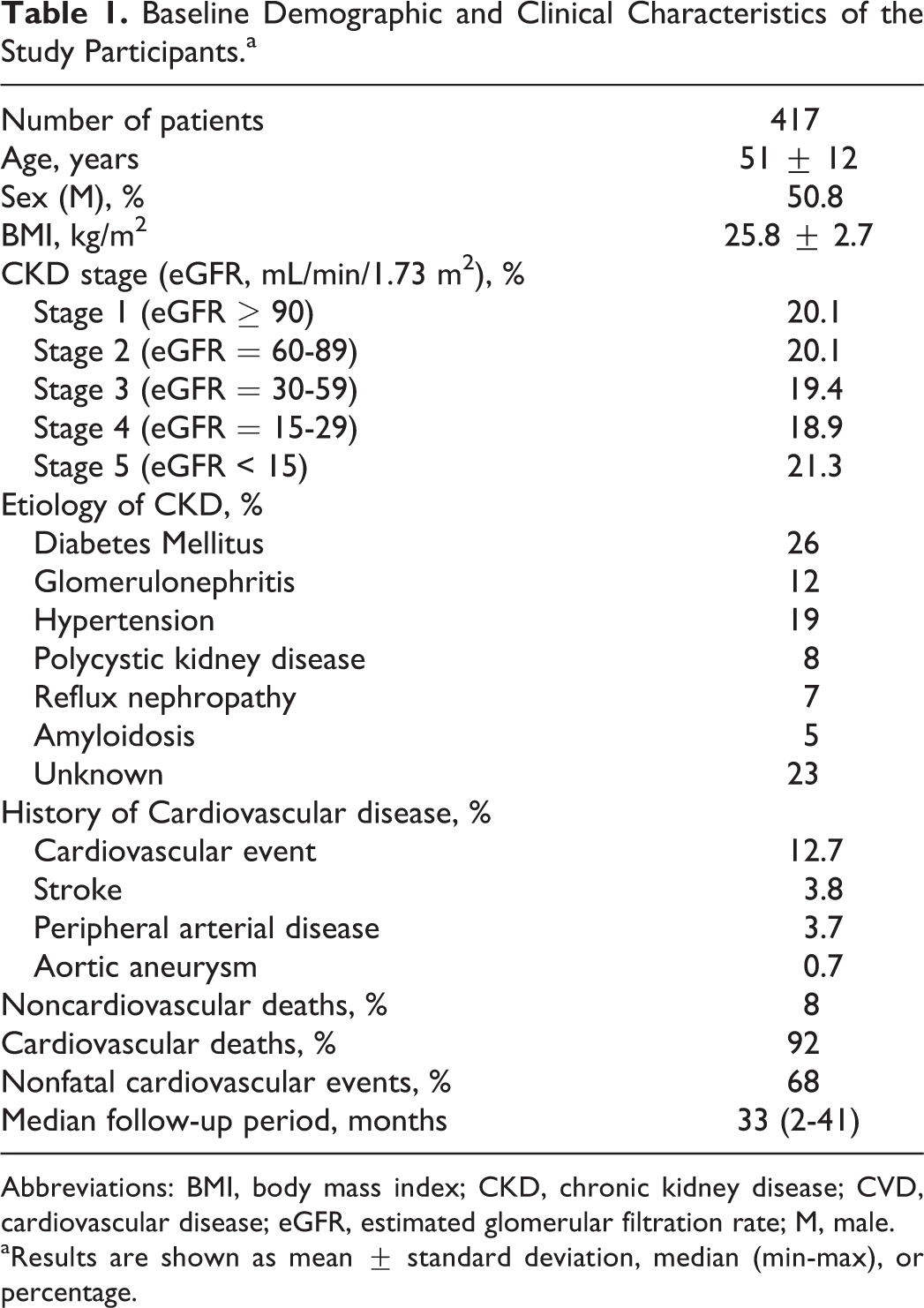
Abbreviations: BMI, body mass index; CKD, chronic kidney disease; CVD, cardiovascular disease; eGFR, estimated glomerular filtration rate; M, male.
aResults are shown as mean ± standard deviation, median (min-max), or percentage.
Study patients divided into 2 groups as patients with CVE and patients without CVE. Cardiovascular outcomes were determined from the day of examination onward, with a median follow-up period of 33 (range 2 to 44) months. Fifty-two patients died, 48 of which was due to cardiovascular causes, 2 due to malignancy, and 2 due to infection. Cardiovascular mortality (n = 48) was defined as death due to coronary heart disease (n = 27), sudden death (n = 3), stroke (n = 14), or complicated peripheral vascular disease (n = 4). In addition to the 48 cardiovascular deaths, 103 nonfatal CVEs were registered during the follow-up as follows: stroke (n = 26); myocardial infarction (n = 58); peripheral vascular disease (n = 12) and aortic aneurysm (n = 7).
Comparison of baseline clinical and demographical characteristics of the 2 study groups was presented in Table 2. On the other hand, vascular and biochemical analyses of the 2 groups were shown in Table 3. The eWBV levels were higher in the patients with CVE compared to patients without CVE. Additionally, markers of inflammation (PTX3 and hsCRP) were higher and FMD and eGFR were lower in CVE compared to without CVE.
Comparison of Baseline Clinical and Demographical Characteristics of the 2 Study Groups.
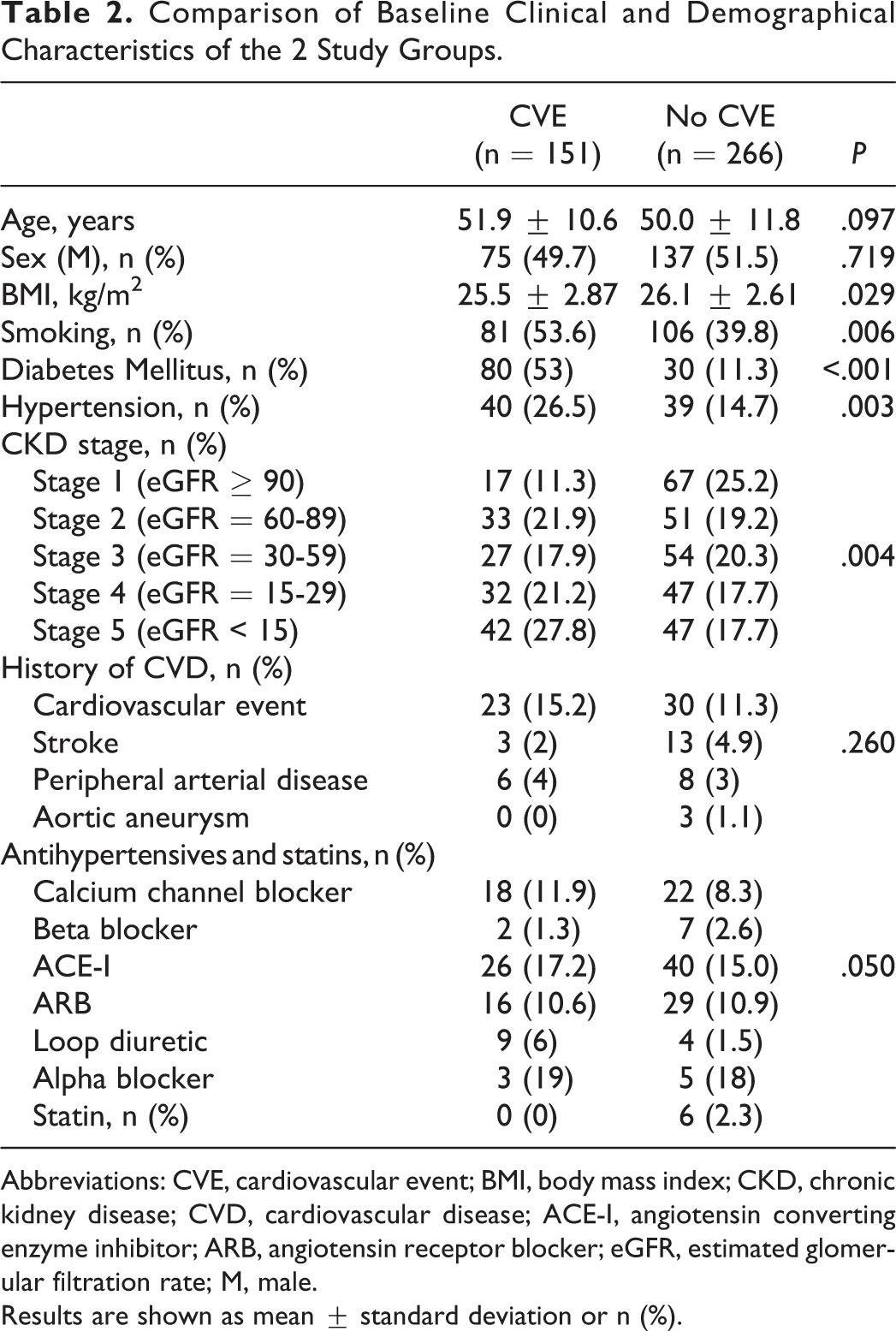
Abbreviations: CVE, cardiovascular event; BMI, body mass index; CKD, chronic kidney disease; CVD, cardiovascular disease; ACE-I, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; eGFR, estimated glomerular filtration rate; M, male.
Results are shown as mean ± standard deviation or n (%).
Comparison of Biochemical and Vascular Analyses of the 2 Study Groups.a
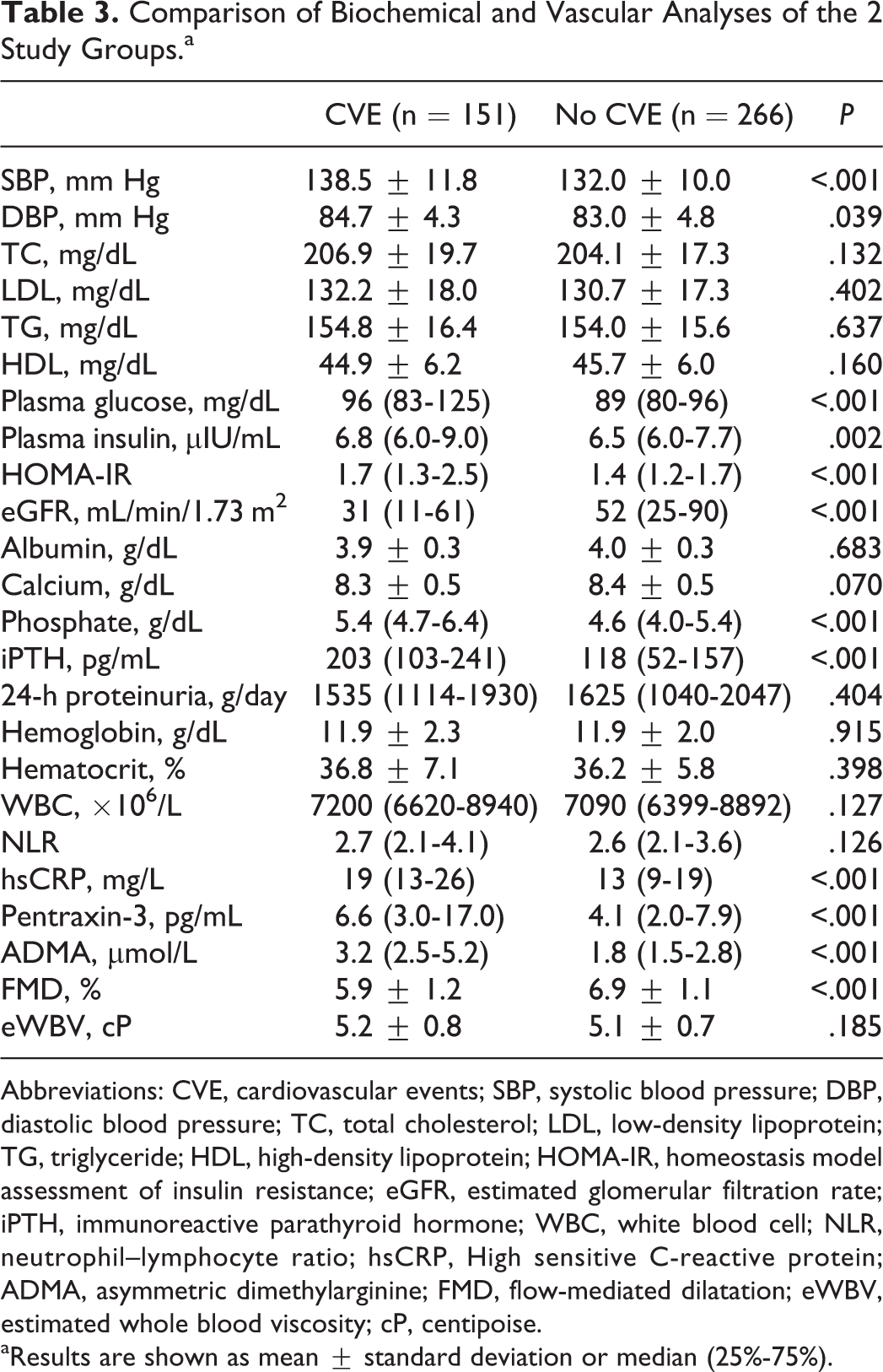
Abbreviations: CVE, cardiovascular events; SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; LDL, low-density lipoprotein; TG, triglyceride; HDL, high-density lipoprotein; HOMA-IR, homeostasis model assessment of insulin resistance; eGFR, estimated glomerular filtration rate; iPTH, immunoreactive parathyroid hormone; WBC, white blood cell; NLR, neutrophil–lymphocyte ratio; hsCRP, High sensitive C-reactive protein; ADMA, asymmetric dimethylarginine; FMD, flow-mediated dilatation; eWBV, estimated whole blood viscosity; cP, centipoise.
aResults are shown as mean ± standard deviation or median (25%-75%).
We performed the COX regression analysis so as to establish the covariates that were independent predictors of cardiovascular outcomes (Table 4). The eWBV (HR: 1.31 [1.04-1.65]; P = .02), the plasma ADMA levels (HR: 1.31 [1.13-1.52]; P < .001), FMD (HR: 0.79 [0.67-0.94]; P = .006), hsCRP (HR: 1.03 [1.011.05]; P = .03), eGFR (HR: 0.99 [0.98-0.99]; P < .001), SBP (HR: 1.02 [1.011.04]; P = .001), calcium (HR: 1.58 [1.13-2.20]; P = .008), and the history of diabetes (HR: 4.24 [2.97-6.05]; P < .001) are independent predictors of CVEs in patients with CKD.
Multivariate Cox Regression Analysis Predicting for Cardiovascular Events (Fatal and Nonfatal).
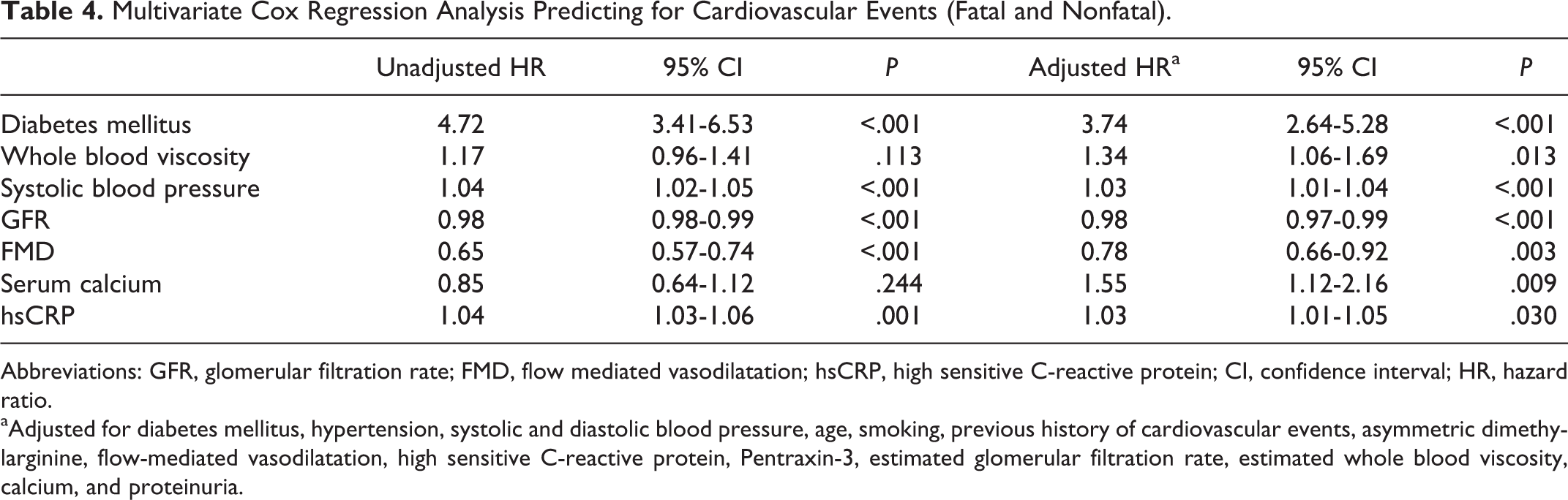
Abbreviations: GFR, glomerular filtration rate; FMD, flow mediated vasodilatation; hsCRP, high sensitive C-reactive protein; CI, confidence interval; HR, hazard ratio.
aAdjusted for diabetes mellitus, hypertension, systolic and diastolic blood pressure, age, smoking, previous history of cardiovascular events, asymmetric dimethylarginine, flow-mediated vasodilatation, high sensitive C-reactive protein, Pentraxin-3, estimated glomerular filtration rate, estimated whole blood viscosity, calcium, and proteinuria.
Estimated whole blood viscosity values of the 2 study groups in according to CKD stages are presented in Figure 1. The eWBV of patients with CVEs were found to be higher than in patients without CVEs in all CKD stages except stage 4 (For stage 1 P < .001, for stage 2 P = .006, for stage 3 P = .04, for stage 4 P = .639, for stage 5 P < .001, respectively; Figure 1). Kaplan Meier survival curves were generated to establish the impact of the eWBV on the cumulative survival of the cohort (Figure 2). As shown in survival curve, patients with eWBV values higher than 5.2 cP had lower survival rates as compared to patients with eWBV values lower than 5.2 cP (log rank = 4.49 df = 1 P = .034).
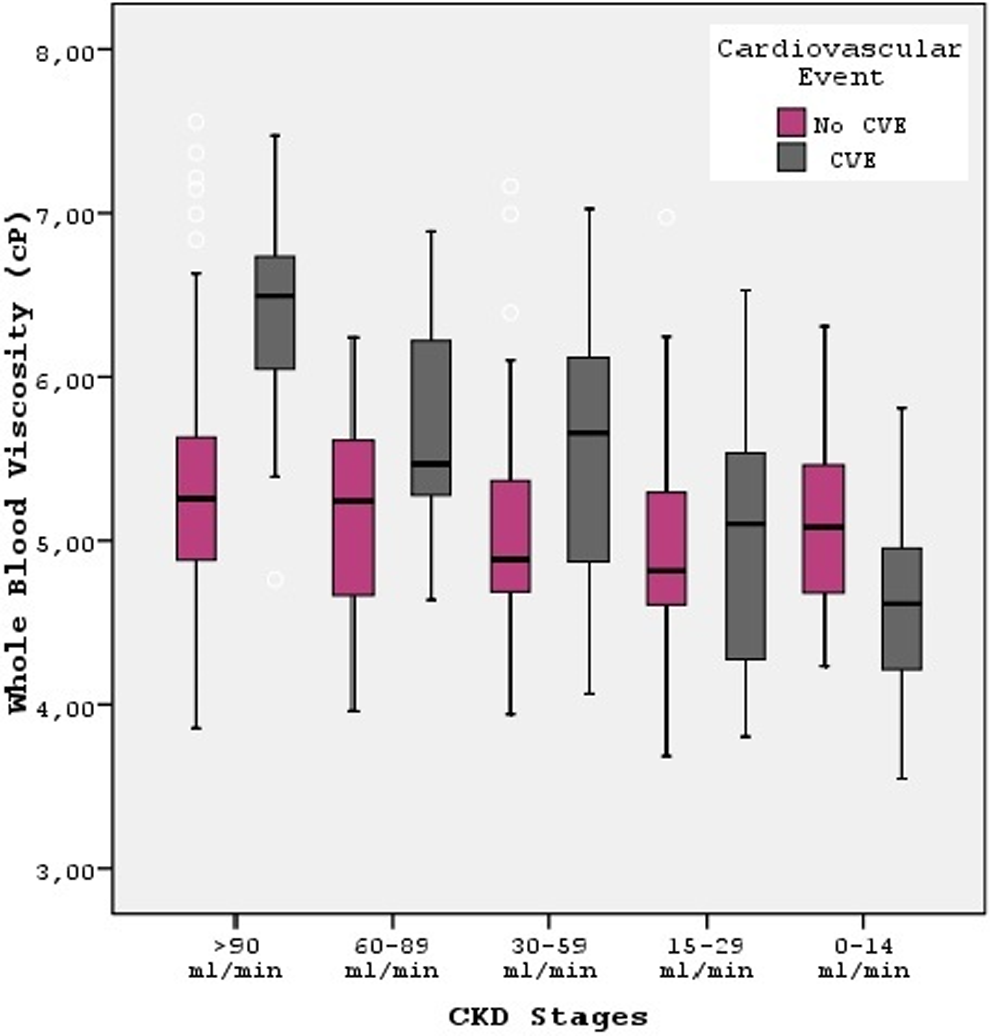
Box plots showing estimated whole blood viscosity values of both study groups according to chronic kidney disease (CKD) stages. Estimated whole blood viscosity values of the patients with CVEs were found to be higher than those of the patients without CVEs in all CKD stages except for stage 4. (For stage 1 P < .001, for stage 2 P = .006, for stage 3 P = .004, for stage 4 P = .639, and for stage 5 P < .001, respectively).
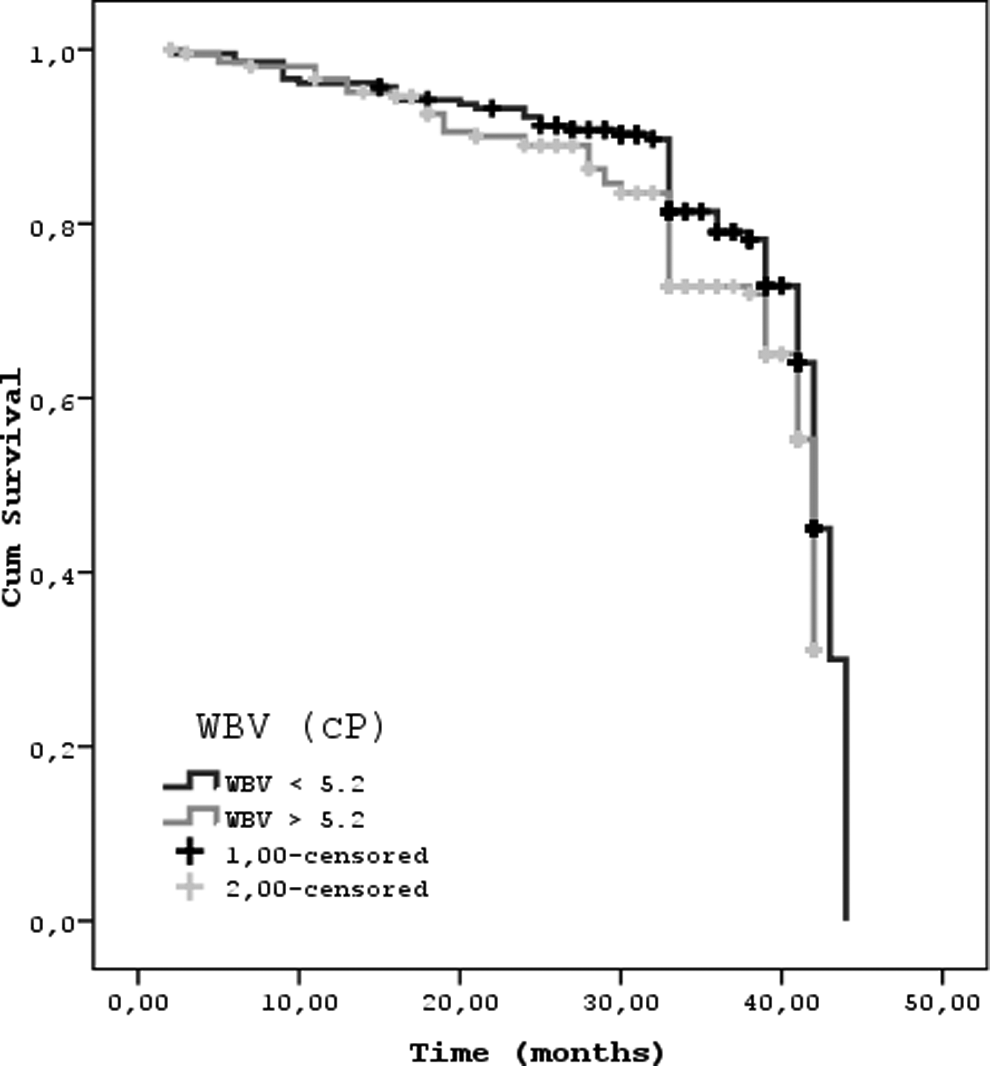
The cumulative survival graphic of patients with chronic kidney disease (CKD) according to estimated whole blood viscosity (log rank = 4.49, df = 1, P = .034).
Discussion
In this study, we aimed to investigate the endothelial function and circulating level of the potential predictor of CVE, eWBV, in patients with CKD and found that eWBV levels were independent predictors of CVE in the population with CKD.
Chronic renal disease is independently associated with the development of CVD. 21 Vascular inflammation is one of the major conditions for developing of CVD, and patients with CKD indicate a subset with an increased risk of CVD. As we know, previous studies have shown the relation between CVE and widely used inflammatory markers such as FMD and hsCRP. As it has been reported, FMD, the well-established noninvasive method used to evaluate the endothelial function, 22 and hsCRP, a well-known surrogate indicator of CVD, have been concerned about their capacity to predict future CVE, in both non-CKD and CKD populations. Our study reported similar results as in previous study that observed the increased hsCRP and reduced FMD in patients with CKD. 23 However, although, in addition to these well-known markers, some novel inflammatory markers such as eWBV have been investigated in these populations, 24 data on patients having CKD with CVEs are limited.
Vascular regular physiology includes the arrangement of vascular conditions such as inhibition of proliferation, thrombosis, and inflammation of the vessel wall. Vascular ED has a major role in the increased risk of CVD. Shear stress identified that disturbed or oscillatory flows near arterial bifurcations, branch ostia, and curvatures are related to atheroma formation. The characteristics of the blood, that is, eWBV, hemoglobin concentration, and hematocrit, predominantly affect blood velocity and pattern and, as a result, the structure and function of the vessels. 3 Also, it affected endothelial function by the release of many mediators. Elevated eWBV levels are strongly considered as the initial indicator of atherosclerosis and development of vascular disease in high-risk patients. 25 Metabolic disturbances may affect rheological features in terms of both direct and indirect effects. The direct one is mediated by exchanges in erythrocyte membrane ingredients and plasma mediators, even though indirect effects are composed of oxidative stress and inflammation likely as a result of ED. The eWBV is vigorously associated with the severity of the metabolic syndrome. 26 In a recently published study, the authors demonstrated that the severity of obesity is measured by body mass index (BMI) and was significantly associated with eWBV and red blood cells aggregation when abdominal obesity as evaluated with the waist to hip ratio was also related to hematocrit. In a previous study, it was observed that obesity is related to increased red cell aggregation, and abdominal fat leads to higher blood viscosity due to a rise in hematocrit, and that overall adiposity as evaluated with the BMI is associated with higher eWBV values and red cell rigidity. 27 Both hyperlipidemia and high viscosity have importantly accelerated the development of atherosclerosis in synergistic condition. 28 Some previous studies reported that eWBV is strongly associated with LDL and TG levels and inversely correlated with HDL levels. Evidence obtained from previous clinical studies supported that there was a strong association between nonalcoholic fatty liver disease (NAFLD) and metabolic syndrome. A number of studies showed that NAFLD is associated with an increased risk of CVD and atherosclerosis such as carotid artery disease and endothelial dysfunction. Hong-yan Zhao et al 29 reported that increased eWBV was positively correlated with the risk of prevalent NAFLD in adults.
In addition, being part of the vascular shear stress directly influences the arterial pressure, and higher eWBV may also be a result of increased BP caused by increased filtration pressure, leading to hemoconcentration or direct disturbances of rheological variables. 25 Also, DM, a well-established traditional risk factor for CVD, is considered a CVD equivalent and it is well known that DM primarily results from insulin resistance in many body cells. The pathophysiology of insulin resistance (IR) is sophisticated, and it is considered that IR may cause multiple disturbances in signal transmission in hepatic, muscle, and adipose tissue. 30 Blood viscosity is negatively associated with flow and may also be associated with flow-related IR. As long as the blood osmolarity increases because of increased glucose level, the capillary permeability increases, thus increasing hematocrit and subsequently the eWBV. 20
Increased blood or plasma viscosity has been demonstrated in almost all conditions related to accelerated atherosclerosis. High plasma eWBV levels were displayed to cause an elevated risk of acute myocardial infarction (AMI) in patients with unstable angina pectoris. 31 Furthermore, an association of an early elevation in plasma viscosity during AMI and reinfarction or death was observed. Compared to patients with stable angina pectoris and healthy individuals, an increase in plasma viscosity was displayed in patients with severe unstable angina pectoris. In addition, in a previous study, the authors showed increased plasma eWBV levels in patients with stable angina pectoris even without coronary artery diseases. 31
The changes in eWBV levels have been showed in patients with cerebrovascular diseases. Plasma eWBV levels are elevated in patients with internal carotid atherosclerosis, independent of the presence of risk factors for CVD. 32 The presence of silent cerebral infarction (SCI) that results from the transient ischemia attack or symptomatic stroke is accompanied with CVD. The eWBV is a new marker for SCI independent of traditional CVD risk factors. Concordantly, individuals with SCI had higher eWBV levels than those without SCI. 33
Mechanical associations of blood and vessels shown by eWBV have a major role in the emission of endothelium-derived mediators and subsequent vascular remodeling. 34 This primarily may cause the increased blood viscosity, especially in small arterioles and capillary beds. Indeed, increased eWBV in the microcirculation may change by reducing microvascular flow affecting capillary perfusion and reducing tissue oxygen delivery. 35 Hemorheologic stressful conditions affecting the microcirculation importantly may link to tissue and organ damage, and it also increases endothelium shear stress promoting the release of potent vasoconstrictive mediators related to accelerate the ED in patients with CKD. 35 Hemorheological changes linked to elevated eWBV in capillary beds may contribute to aggravate silent tissue hypoxemia and precipitate CVE in patients with CKD. 34 In association with eWBV, this abnormality is related to arterial outward remodeling, increased arterial stiffness, and reduced FMD. Concordantly, we showed low FMD values in adverse clinical outcomes in patients with CKD. We also demonstrated that FMD values are independent predictors of cardiovascular outcomes in patients with CKD. Oxidative stress can induce red cell rigidity and hemolysis, which in turn can cause increased blood viscosity. 31 The plasma level of ADMA, as known inhibitor of nitric oxide synthase, is related to oxidative stress, which has been associated with endothelial cell dysfunction and atherosclerosis in the general population. The ADMA is also consistently associated with CVE and clinical adverse outcomes in patients with CKD. 36,37 In the present study, concordant with those data, we showed higher ADMA values in patients with CVE compared to those without CVE. Furthermore, PTX3 is a novel immunoinflammatory indicator that has been described to be associated with atherosclerotic risk factors. 38 Also, higher PTX3 concentrations were associated with the ED, and they impacted the predictability of cardiovascular outcomes in patients with CKD. 2 In addition to them, we displayed higher PTX3 values in CVE compared to no CVE in patients with CKD. Further, evidence of oxidative stress and a concomitant demonstration of any of the effective vascular events including increase in eWBV are necessary to establish oxidative damage. Chronic kidney disease is a complex disease that leads to a high prevalence of vascular complications. Impairment in the microvascular blood flow beginning at the endothelial cell contributes to these conditions. 21 Finally, eWBV levels have strongly and independently predicted the cardiovascular outcomes.
Moreover, there are some limitations of present study. First, the eWBV cannot be directly calculated, and there are also missing data on the other components of eWBV, such as erythrocyte rigidity and aggregability. Nonetheless, the equation used in this study has been confirmed in several previous studies, and WBV is commonly described by hematocrit and plasma protein values. 20,25
In conclusion, we demonstrated in patients with CKD that eWBV levels were higher in the CVE group compared to the group without CVE. More importantly, we found that high eWBV levels would independently predict cardiovascular outcomes in patients with CKD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
