Abstract
This study aimed to investigate the significance of the prethrombotic state (PTS) and 4 plasma markers in predicting perisurgical adverse cardiac events in patients with coronary heart disease (CHD) undergoing abdominal surgery. Perioperative adverse effects were recorded in 128 consecutive patients with CHD undergoing elective abdominal surgery. Plasma
Introduction
The prethrombotic state (PTS), also known as the hypercoagulable state, refers to a pathological condition characterized by an imbalance in coagulation and anticoagulation factors.
1
Previous studies have shown that patients with coronary heart disease (CHD) usually develop PTS
2,3
and that perioperative adverse cardiac events are closely related to abnormalities in coagulation function (especially the PTS). However, little attention has been paid to this relationship in clinical practice, and to date, few clinical studies have investigated the correlation between PTS and perioperative adverse cardiac events. The present study aimed to investigate the significance of 4 plasma markers of PTS (
Materials and Methods
Selection of Patients
The present study was approved by the ethics committee of the First Affiliated Hospital of Xinjiang Medical University and informed consent was obtained from each patient before the study. A total of 128 consecutive patients with CHD (stable angina or old myocardial infarction) who received abdominal surgery in our hospital were recruited from October 2010 to August 2011.
Inclusion Criteria
Inclusion criteria included the following: age 61 to 75 years; elective abdominal surgery with moderate to high risk (general abdominal surgery, urinary surgery or gynecological surgery) and a surgery time of 90 to 180 minutes; CHD diagnosed according to the World Health Organization criteria for CHD; stable angina or old myocardial infarction (6 months); cardiac function completely evaluated preoperatively as New York Heart Association class I-II; prothrombin time, presurgery activated partial thromboplastin time, thrombin time, fibrinogen, and platelet count in the normal range; and informed consent was obtained before surgery
Exclusion Criteria
Exclusion criteria were the following: emergency surgery or low-risk surgery; concomitant severe infection; dysfunction of liver, spleen, kidney, or lung; malignancies; concomitant congenital heart disease, cardiomyopathy, rheumatic heart disease, pulmonary heart disease, severe heart failure, or severe arrhythmia; unstable angina or myocardial infarction within the previous 6 months; hematological disease; intraoperative blood loss exceeding 20% of blood volume; inability (psychiatric disorders, consciousness disturbance, mental retardation) to cooperate with clinicians; and blood-borne infectious diseases (hepatitis B, hepatitis C, syphilis, and AIDS).
Data Collection
Levels of
Primary variables were the 4 markers for determination of PTS,
Sample Collection and Measurement
The EDTA-K2 anticoagulated whole blood (2.0 mL) was collected for the measurement of P-selectin. Sodium citrate (1:9) anticoagulated whole blood (5 mL) was collected for the measurement of plasma
End Point Events: Adverse Cardiac Events
Adverse cardiac events included myocardial ischemia, myocardial infarction, arrhythmia, congestive heart failure, cardiac death, and so on. The criteria for the diagnosis of individual adverse cardiac events were (1) myocardial ischemia: horizontal or down-sloping ST-segment depression of ≥1 mm or elevation of ≥ 2 mm at 80 milliseconds after the J point; (2) myocardial infarction: precordial pain continued for 30 minutes, and Q wave, depression, or arched elevation of ST segment and cTnI >3.1 g/L; (3) arrhythmia: evident symptoms of atrial fibrillation, paroxysmal supraventricular tachycardia, or premature ventricular contraction affecting hemodynamics or requiring pharmacotherapy; (4) congestive heart failure: shortness of breath, jugular vein distention, gallop rhythm, and pulmonary edema on chest x-ray; (5) cardiac death: death from cardiac events including myocardial infarction, heart failure, arrhythmia, and so on. These events were used as a single cumulative end point.
Statistical Analysis
Continuous data are presented as median with interquartile range; categorical data are presented as count (percentage). Differences between those with and without adverse cardiac events for continuous and categorical data were tested with the nonparametric Mann-Whitney test and Fisher exact test, respectively. Changes in
Results
Participants
During the period from October 2010 to August 2011, 138 patients fitting the inclusion and exclusion criteria were prospectively enrolled in the study. The follow-up period was 30 days; 10 patients who were lost to follow-up were excluded from the analysis. The remaining 128 patients included 96 (75.0%) males and 32 (25.0%) females, with a median age of 64. In all, 29 (22.7%) of these patients experienced 1 or more adverse cardiac events during or after the surgery.
Association Between Patient Characteristics and Adverse Cardiac Events
The patient group that experienced adverse cardiac events was older (67.0 vs 64.0 years, P = .022) and had a higher percentage of individuals with nonHan Chinese ethnicity (51.7% vs 29.3%, P = .044) than the group without adverse cardiac events. Patients who had adverse cardiac events also had a higher body mass index (24.2 vs 22.7 kg/m2, P < .001) were more likely to be obese (27.6% vs 7.1%, P = .006) and were more likely to have a history of diabetes mellitus (37.9% vs 8.1%, P < .001) and a history of coronary stenting or coronary artery bypass graft surgery (48.3% vs 16.2%, P = .001). In addition, the duration of surgery in those with adverse cardiac events was significantly longer than in those without adverse cardiac events (150 vs 120 minutes, P < .001; Table 1).
Summary of Characteristics of Patients With and Without Adverse Cardiac Events.a
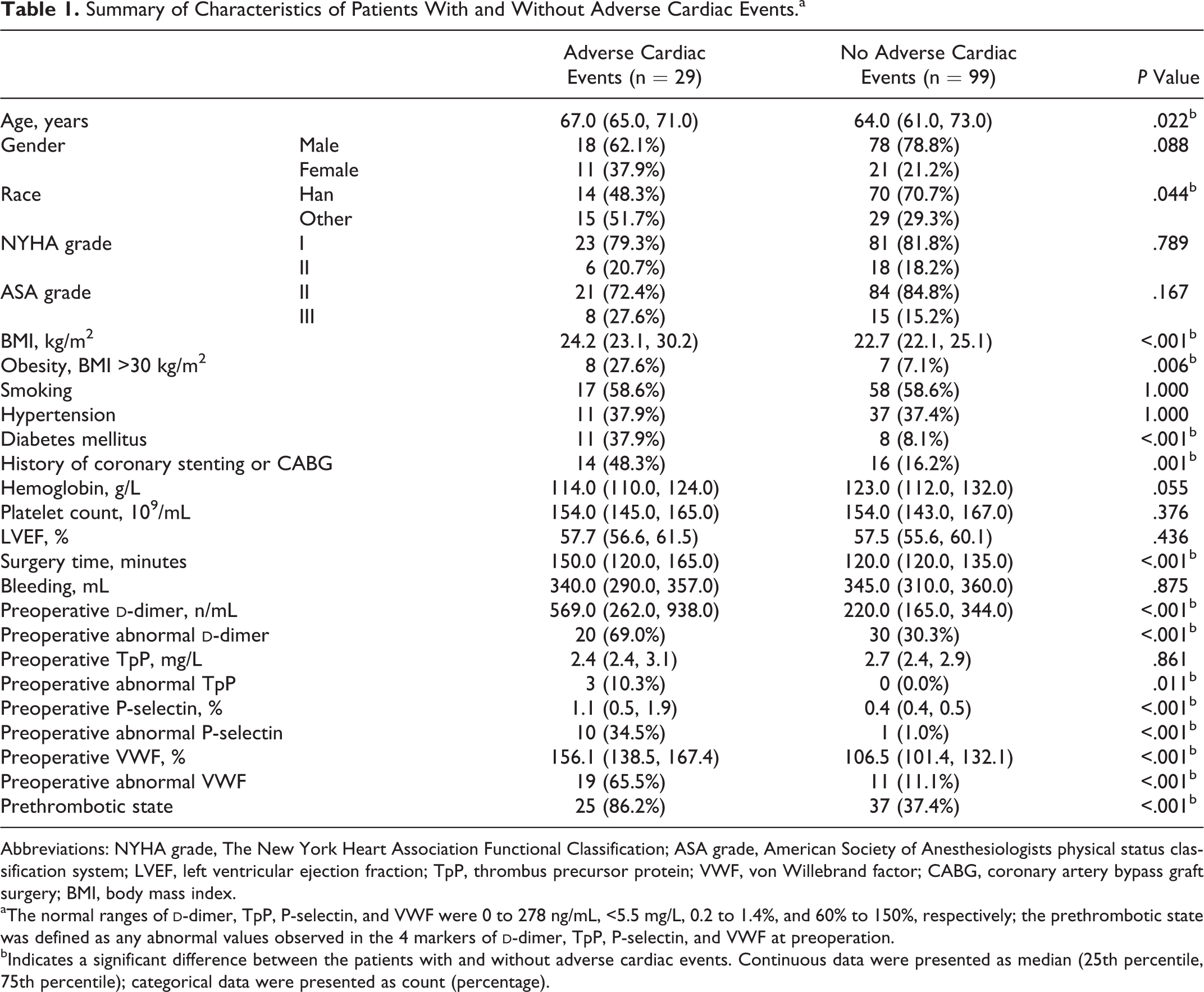
Abbreviations: NYHA grade, The New York Heart Association Functional Classification; ASA grade, American Society of Anesthesiologists physical status classification system; LVEF, left ventricular ejection fraction; TpP, thrombus precursor protein; VWF, von Willebrand factor; CABG, coronary artery bypass graft surgery; BMI, body mass index.
aThe normal ranges of
bIndicates a significant difference between the patients with and without adverse cardiac events. Continuous data were presented as median (25th percentile, 75th percentile); categorical data were presented as count (percentage).
Association Between PTS and Adverse Cardiac Events
The preoperative levels of the
Risk Factors For Adverse Cardiac Events
In the univariate analysis, race, obesity, diabetes mellitus, heart disease, surgery time, and abnormal preoperation
In multivariate model I, 6 variables were included (gender, diabetes mellitus, surgery time, and preoperation
In multivariate model II, 5 variables were included (gender, obesity, diabetes mellitus, surgery time, and PTS). After controlling for the other 4 variables, females had a significantly higher risk of incurring adverse cardiac events than males (OR = 40.68, P = .002); patients with the history of diabetes mellitus were more likely to incur adverse cardiac events (OR = 402.80, P < .001); the risk of occurrence of adverse cardiac events was increased every minute by increase in the surgery time (OR = 1.09, P < .001); and those with PTS were more likely to experience adverse cardiac events (OR = 64.32, P < .001; Table 2).
Summary Risk Factors for Adverse Cardiac Events.a
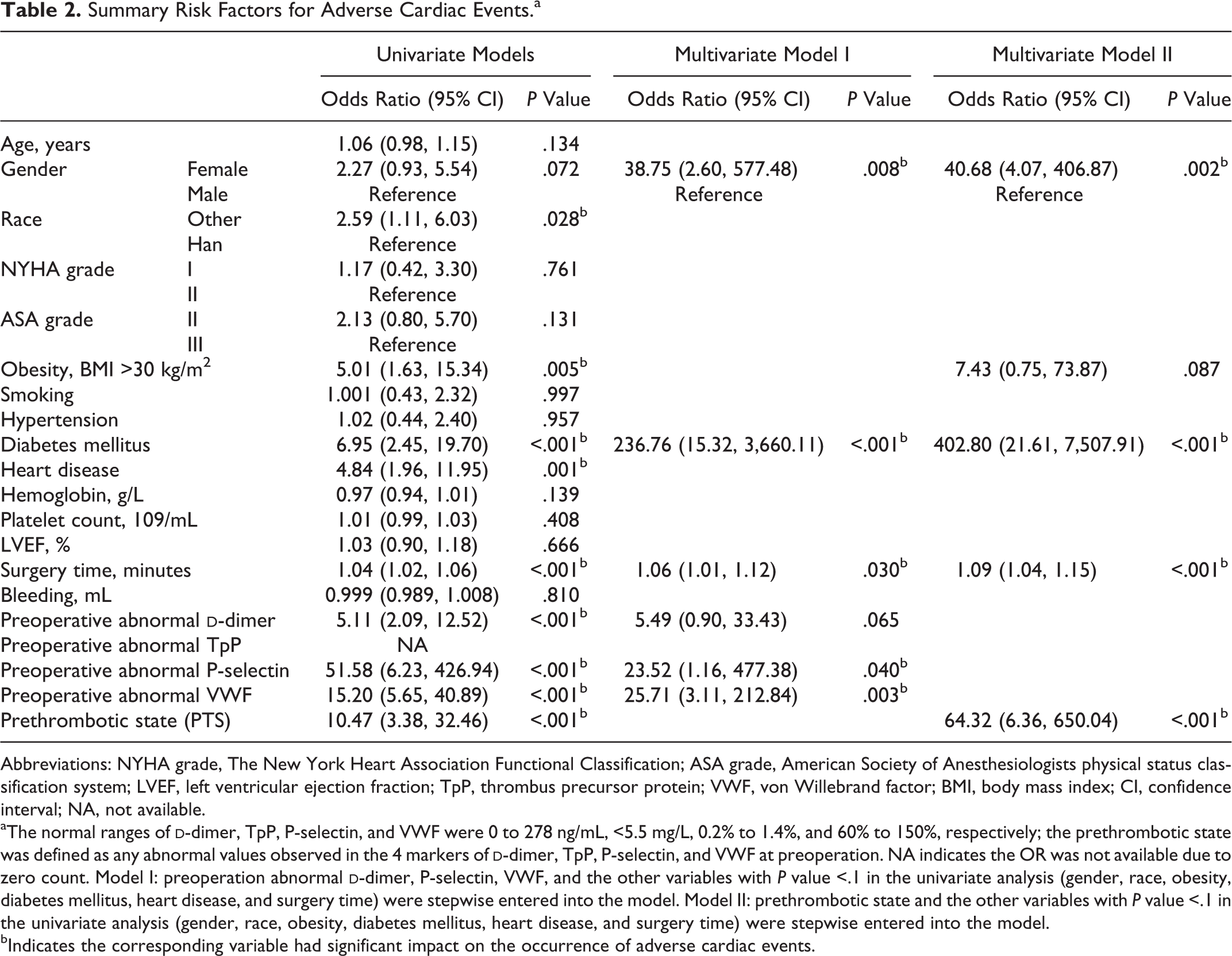
Abbreviations: NYHA grade, The New York Heart Association Functional Classification; ASA grade, American Society of Anesthesiologists physical status classification system; LVEF, left ventricular ejection fraction; TpP, thrombus precursor protein; VWF, von Willebrand factor; BMI, body mass index; CI, confidence interval; NA, not available.
aThe normal ranges of
bIndicates the corresponding variable had significant impact on the occurrence of adverse cardiac events.
Trends of PTS-Related Markers in the Perioperative Period
d
-Dimer
In the adverse cardiac event group,
TpP
In adverse cardiac event group, TpP levels were significantly increased from 2.37 mg/L preoperatively to 3.19 mg/L 24 hours after the operation and were significantly higher than the 2.59 mg/L in those without adverse cardiac events. In those without adverse cardiac events, no significant change was observed in TpP in the 4 time points of the perioperative period.
P-selectin
A significant increase in P-selectin with time was observed in both the groups (P < .001), and a higher slope for this increase was seen in the adverse cardiac event group. In the adverse event group, P-selectin levels were significantly increased from 1.1% at preoperation to 1.8%, 2.1%, and 2.4% in 24, 48, and 72 hours after the operation, respectively. P-selectin levels in those with adverse cardiac events were significantly higher than in those without adverse cardiac events in the 4 time points of the perioperative period (P < .001).
VWF
A smooth increase in VWF with time was also observed in both the groups. The VWF levels in those with adverse cardiac events were significantly higher than in those without adverse cardiac events at each of the 4 time points of perioperative period (P < .001; Figure 1).
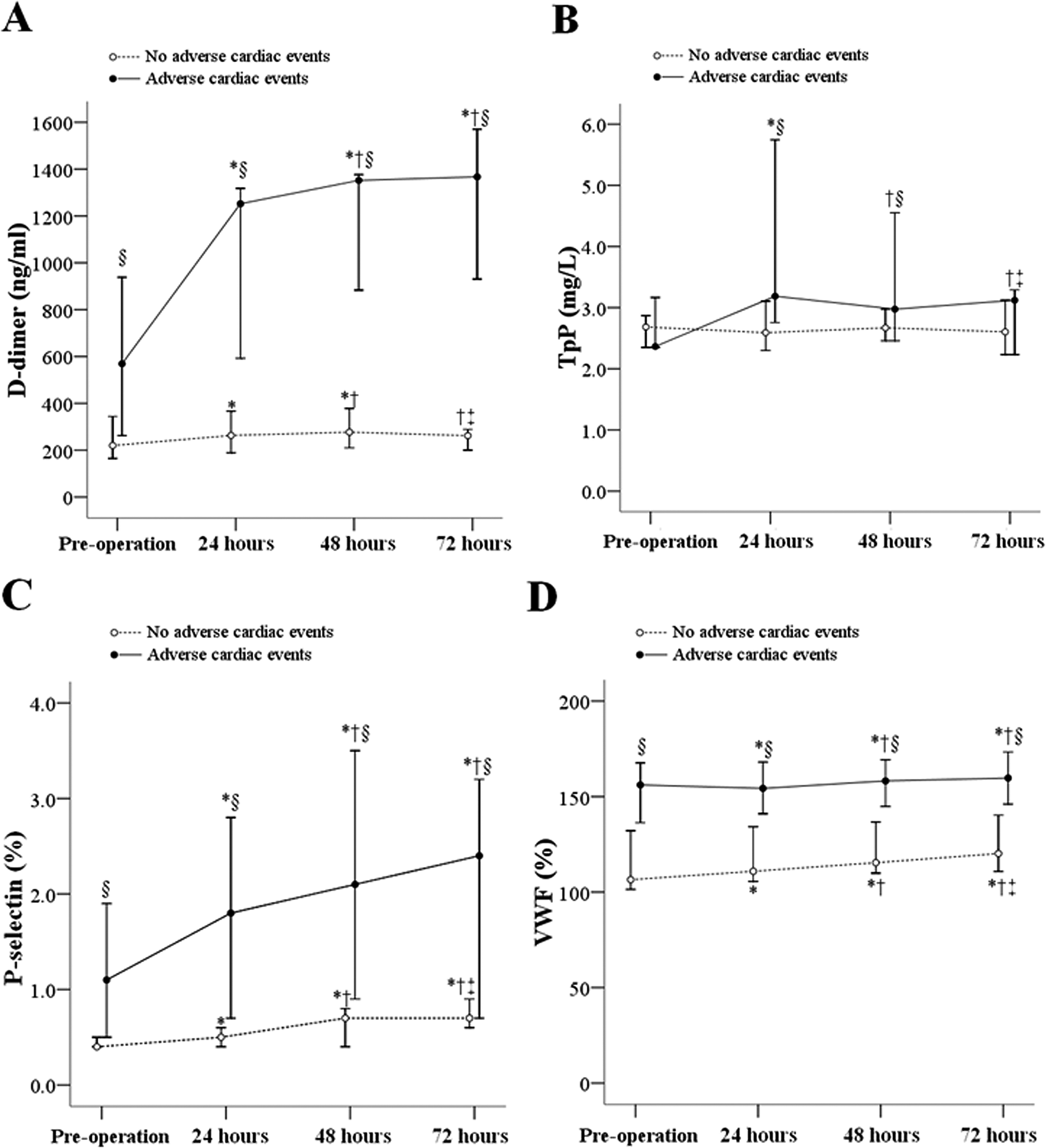
Summary for the trends of
Discussion
A number of studies have shown that PTS plays a crucial role in the development of atherosclerosis and ischemic heart disease.
2,3
In the present study, preoperative TpP, P-selectin,
Plasma
Presurgery elevation of
The incidence of adverse cardiac events in our study was dramatically higher than that previously reported in patients with CHD receiving noncardiac surgery (1%-5%). In addition, 86.2% of the patients developed adverse cardiac events had PTS, which was significantly higher than 37.4% in nonadverse cardiac event participants (P < .001). This suggests PTS is highly correlated with adverse cardiac events and that the markers for PTS are positively associated with adverse cardiac events. Thus, we speculate that PTS in patients with coronary atherosclerosis may be an important precipitating cause of adverse cardiac events.
To date, in the American College of Cardiology/American heart Association Guideline (2007) for patients with CHD receiving noncardiac surgery 11 and in the European Guideline (2009), 12 PTS is not addressed. Our results showed that PTS is an independent risk factor for adverse cardiac events and is closely related to perioperative adverse cardiac events. Multivariate logistic regression analysis showed PTS to have an OR of 64.32 (P < .001) for adverse cardiac events. Thus, PTS may be an important trigger factor and should be considered in the preoperative evaluation.
This study has several limitations. It was a single, not a multicenter study. The PTS is a pathological condition involving many factors, and we made no investigation of its etiology. And there are many markers for PTS, but in this study we focused only on the 4 representative markers. In our multivariate analysis, in addition to PTS, female gender and diabetes were robust and independent predictors of perioperative cardiac events. One unanswered question is the respective sensitivity and selectivity of the 3 predictors and what improvement in prediction of risk could be obtained by using them together.
In conclusion, preoperative PTS is an independent risk factor for perioperative events in patients with CHD undergoing noncardiac surgery, and its clinical use in preoperative evaluation might enable better regulation of postoperative coagulation state and decrease the incidence of perioperative adverse cardiac effects. Our findings may provide evidence supporting the use of evaluation of risk factors in prediction of perioperative adverse cardiac events and for regulation of the perioperative coagulation state.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: key mandatory plan of project of supporting Xinxiang with science and technology (No: 200991126).
