Abstract
Apolipoprotein E (APOE) is a member of the apolipoprotein gene family. APOE is polymorphic with 3 main allelic types: ∊2, ∊3, and ∊4. Certain of these alleles have been associated with higher vascular risk. However, the association of APOE genotypes with retinal biomarkers and risk of retinal stroke is less clear. This study evaluated the role of APOE polymorphisms in retinal vein occlusion (RVO). In the present study, 2-point mutations coding amino acid residues 112 and 158 were amplified using the polymerase chain reaction (PCR) from DNA extracted from Tunisian participants. APOE genotypes were determined by multiplex PCR followed by molecular hybridization. Eighty-eight patients (26 women and 62 men) and 100 age- and gender-matched healthy participants were enrolled. The statistical study revealed a higher frequency of the ∊4 allele in patients as compared to controls (27.3% vs 9%) with a significant association of the ∊4 allele with the disease (P < 10−3, P a < 10−3, odds ratio [OR] = 3.8, 95% confidence interval [CI] = 2.1-6.8). The frequency of the ∊3 allele was significantly lower in the patients with RVO compared to the controls (60.2% vs 82.5%, respectively; P < 10−3, P a < 10−3, OR = 0.32, 95% CI = 0.19-0.53). The ∊3 allele seems to be protective against the disease. There was no association between the APO ∊2 allele and RVO. The association of APOE allele and genotype with RVO requires further investigation in different populations.
Introduction
Human apolipoprotein E (apoE), one of the least abundant plasma apoproteins, is a 34-kDa protein containing 299 amino acid residues that mediates the binding of various lipoprotein receptors, which are crucial for lipid transportation. 1 The APOE gene is located on chromosome 19q13.32. Two single-nucleotide polymorphisms (SNPs) in exon 4 (rs429358—a TGC to CGC substitution at codon 112; rs7412—CGC to TGC substitution at codon 158) give rise to 3 common APOE haplotypes known as the ∊2, ∊3, and ∊4 alleles, which in turn encode 3 different protein isoforms (Apo ∊2, Apo ∊3, and Apo ∊4) composed by cysteine to arginine and arginine to cysteine amino acid changes at positions 112 and 158 (Apo ∊2: Cys-112, Cys-158; Apo ∊3: Cys-112, Arg-158; Apo ∊4: Arg-112, Arg-158). 2,3
apoE is a multifunctional protein that has several roles in metabolic processes and lipid metabolism. 4,5 APOE is one of the most widely studied genes in neurodegenerative and vascular diseases. 2 However, the APOE gene also has similar effects on the small vessels or microvasculature. 6 In the eye, apoE immunoreactivity is localized to Müller cells, Bruch membrane, the retinal pigment epithelium, basal deposits, and drusen. Hence, the retina is a very interesting subject for neurovascular study, and, in fact, retinal vessels provide a unique opportunity to study both systemic and cerebrovascular disease. 7,8
Indeed, retinal vein occlusion (RVO) is one of the major microvascular complications of retina resulting in a progressive severe loss of visual acuity. 9,10 It is an obstruction of the retinal venous system by thrombus formation, and it typically occurs in middle-aged and elderly patients. The condition is uncommon below the age of 40 years, but the incidence increases with age. 11 –13 The pathogenesis of RVO is multifactorial with both local factors and systemic diseases being etiologically important. 14 Possible etiological factors include venous thrombosis and/or endothelial swelling and constriction. Multiple cardiovascular risk factors have also been associated with the condition as well as coagulation abnormalities, changes in blood vessel walls, and systemic conditions such as hypertension, atherosclerosis, and diabetes mellitus. 15,16
We hypothesized that APOE alleles may influence retinal vascular disease at the microvascular level. In the current study, the association of APOE polymorphisms with RVOs disease is investigated in a cohort of middle-aged Tunisians.
Patients and Methods
Participants
The study included 88 consecutive patients with RVO admitted to the Department of Ophthalmology at the Military Hospital of Tunis, from September 2011 to September 2013. All patients underwent a complete ophthalmologic examination. A review of fundus photographs, fluorescein angiograms, and written records of patients with RVO were provided by the participating institutions where the participants were registered, examined, and initially diagnosed. The patients were subjected to a detailed anamnestic investigation for hypertension, dyslipidemia, diabetes, and individual or family history of venous/arterial thrombosis. Baseline ocular history and visual examination findings were recorded from each patient’s initial evaluation. Additionally, the control group consisted of 100 age- and gender-matched healthy participants recruited from the Military Hospital of Tunis and originated from different regions of Tunisia. The study protocol was approved by our ethics committee, and all participants of the study had given their informed written consent.
Genotyping of APOE
Genomic DNA recovered from the peripheral blood leukocytes of patients with DNA extraction kit (QIAamp blood kit; Qiagen GmbH, Hilden, Germany), according to the manufacturer’s protocol, was then amplified through the polymerase chain reaction (PCR). Following PCR, the products underwent a reverse hybridization by the use of the GenoType APOE test that is based on the DNA-STRIP technology (Hain Lifescience GmbH, Nehren, Germany) and permits the molecular genetic characterization of the APOE isoforms ∊2, ∊3, and ∊4. The different target gene sequences are concurrently amplified and biotin labeled in a single amplification reaction. The reaction consists of 0.1 mg of DNA added to 15 mL of preprepared amplification mix that is composed of primers that flank the target sequences and deoxynucleoside triphosphates (dNTPs) in the presence of 1 U Taq polymerase. The PCR cycles were optimized as follows: 2 minutes at 94°C for initial denaturation followed by 35 cycles (15-second denaturation at 94°C, 30-second annealing at 58°C, and 30-second extension at 72°C) and a final extension step at 72°C. The amplification products are denatured and selectively hybridized to a test strip that contains allele-specific oligonucleotide probes (APOE isoforms ∊2, ∊3, and ∊4) immobilized as an array of parallel lines. Bound biotinylated sequences are detected using streptavidin–alkaline phosphatase and color substrate. The amplified fragments were separated gel electrophoresis with 2% agarose (Promega, Madison, Wisconsin) stained with ethidium bromide and visualized under a UV spectrophotometer.
Statistical Study
All analyses related to the case–control study were performed using the Statistical Package for the Social Sciences (version 17; IBM, Armonk, New York). Differences between cases and controls were evaluated using the χ2 test for qualitative variables. In addition, the odds ratio (OR) and 95% confidence intervals (CIs) were calculated. Probability values P < .05 were considered statistically significant. Probability values were adjusted (designated P a) for multiple comparisons of alleles/genotypes using the Holm-Bonferroni procedure.
Results
Gender and age distribution of RVO are presented in Table 1. The cohort consisted of 88 patients with RVO (62 men and 26 women; mean age ± standard deviation [SD] = 54.17 ± 17.70 at the time of study, aged 11-82 years, mean age = 49.79 ± 17.32 at the time of diagnosis) and 100 age- and gender-matched healthy participants. Sixteen (18.18%) patients were diagnosed with central retinal vein occlusion (CRVO), whereas 68 (77.27%) patients had branch retinal vein occlusion (BRVO; Table 1). The patient group had statistically significant risk factors for hypertension (43.18%, P < 10−3, OR = 14.4, 95% CI = 5.34-39.0) and diabetes (43.18%, P < 10−3, OR = 24.6, 95% CI = 7.22-83.6). There were statistically significant differences (P < 10−3) in the hypertension and diabetes risk factors between women and men who participated in the study. This research indicates that smoking may have a marginally protective effect (Table 1).
Ensemble and by Gender: Characteristics of Risk Factors Among Patients With RVO and Controls.a
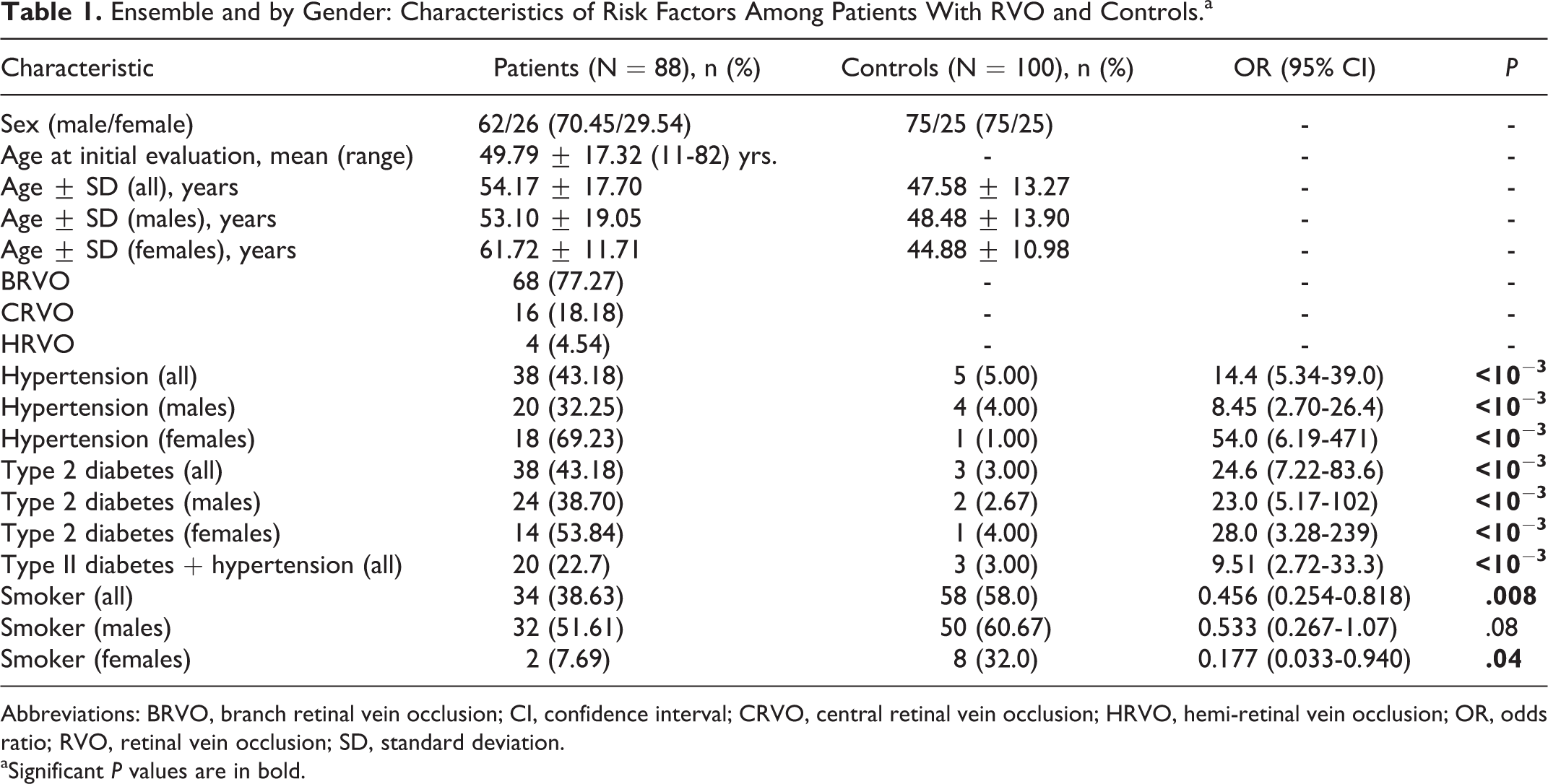
Abbreviations: BRVO, branch retinal vein occlusion; CI, confidence interval; CRVO, central retinal vein occlusion; HRVO, hemi-retinal vein occlusion; OR, odds ratio; RVO, retinal vein occlusion; SD, standard deviation.
aSignificant P values are in bold.
The frequencies of APOE alleles and genotypes in the patients with RVO and the controls are summarized in Tables 2 and 3. The distribution of genotypes of patients versus controls was as follows: ∊3/∊3 (43.2% vs 67%), ∊3/∊4 (29.5% vs 15%), ∊4/∊4 (2.27% vs 1%), ∊2/∊3 (4.5% vs 16%), and ∊2/∊4 (20.5% vs 1%; Table 2). The frequency of the ∊3/∊3 genotype was highest in both the test and control groups, and the statistical analysis of data revealed a strong protective effect (P = .001, P a = .004, OR = 0.37, 95% CI = 0.20-0.71). The difference in the frequencies of the second most common genotype, ∊3/∊4, was also statistically significant between the 2 groups (P = .021, P a = .042, OR = 2.4, 95% CI = 1.1-5.2) and indicates that ∊3/∊4 is a risk factor for RVO. The largest effect size and risk factor were seen with the ∊2/∊4 genotype (P < 10−3, P a < 10−3, OR = 25.5, 95% CI = 3.8-195). Sex differences were observed in the genotype results in the APOE polymorphism for our study population. In fact, the results estimates of males differed significantly only for the ∊2/∊4 genotypes (P a < 10−3; Table 2).
Ensemble and by Gender: Distribution of Apo E Genotypes in Patients With RVO and Controls.
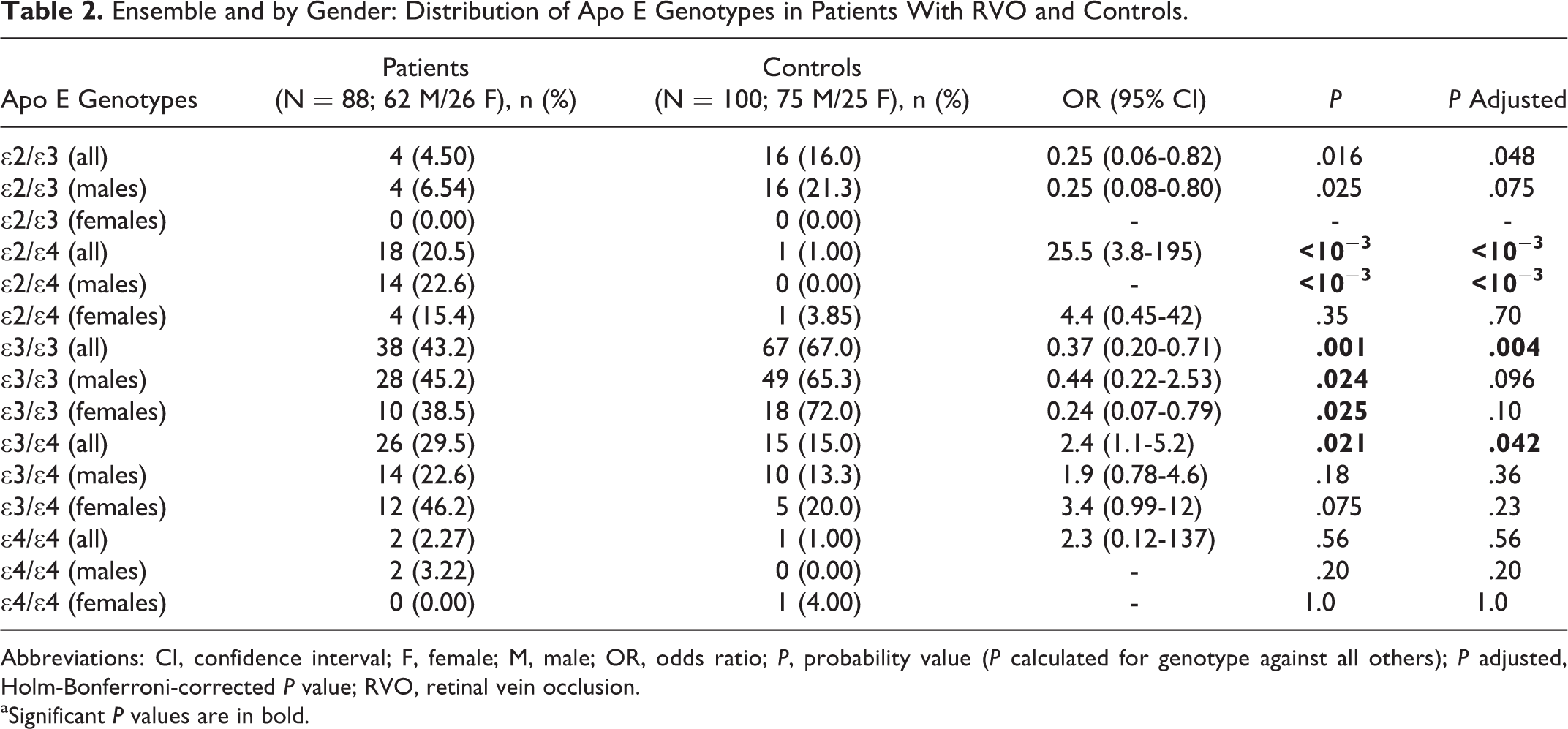
Abbreviations: CI, confidence interval; F, female; M, male; OR, odds ratio; P, probability value (P calculated for genotype against all others); P adjusted, Holm-Bonferroni-corrected P value; RVO, retinal vein occlusion.
aSignificant P values are in bold.
Ensemble and by Gender: Distribution of Apo E Alleles in Patients With RVO and Controls.
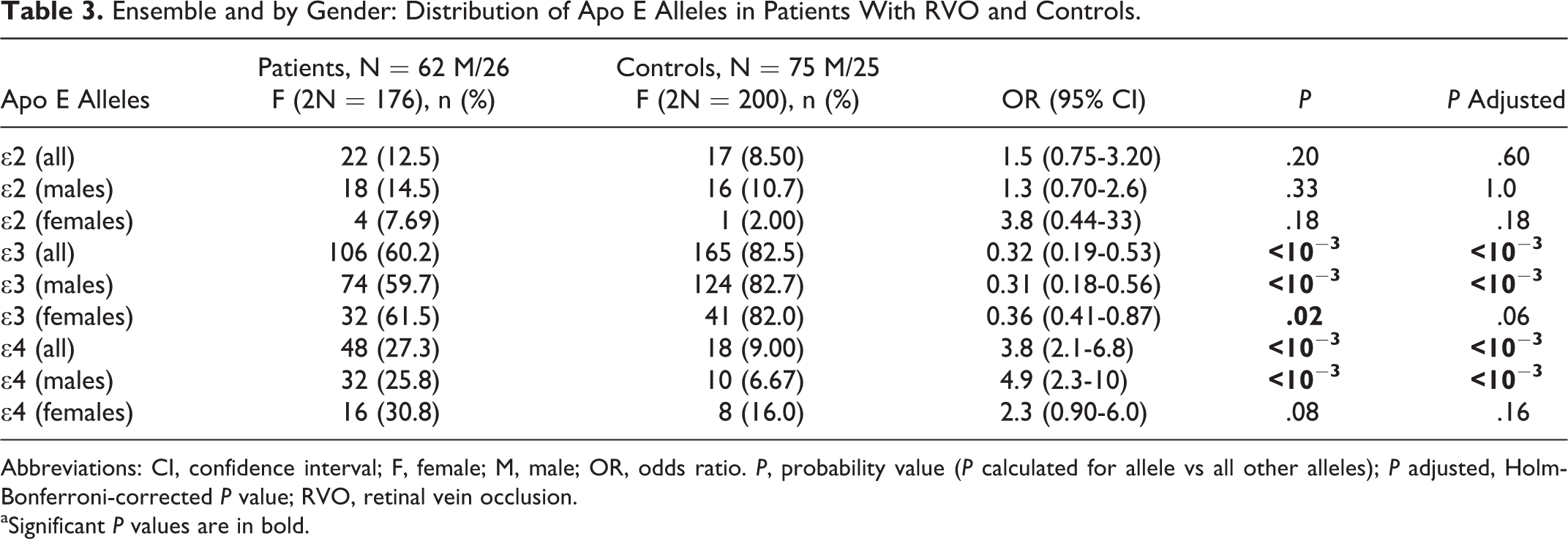
Abbreviations: CI, confidence interval; F, female; M, male; OR, odds ratio. P, probability value (P calculated for allele vs all other alleles); P adjusted, Holm-Bonferroni-corrected P value; RVO, retinal vein occlusion.
aSignificant P values are in bold.
Our study of various APOE alleles also showed variations in patient and control groups. The frequency of the ∊3 allele was significantly lower in the patients with RVO compared to the controls (60.2% vs 82.5%, respectively; P < 10−3, P a < 10−3, OR = 0.32, 95% CI = 0.19-0.53). Conversely, the frequencies of the ∊4 allele was significantly higher in the patients as compared to controls (27.3% vs 9%, respectively; P < 10−3, P a < 10−3, OR = 3.8, 95% CI = 2.1-6.8). The results indicated that the ∊3 and ∊4 alleles are only statistically significant in males compared to females (P a < 10−3 vs P > .05). It should be noted however that the ∊3 allele was statistically significant for females prior to applying the Holm-Bonferroni adjustment. The ∊2 allele frequency was not statistically different between patients and controls (12.5% vs 8.5%, respectively, P = .2). There was also no association by gender found between the ∊2 allele and RVO pathology (Table 3).
Discussion
Diseases of the retinal vascular system may severely interfere with visual function, and occlusion of the retinal vein is a primary cause of visual loss. This vascular disease progresses by 2 major disease mechanisms: alteration to the inner blood–retinal barrier with resulting increased vascular permeability and edema and vessel closure with resulting ischemia leading to the development of abnormal neovascularization. 17
In fact, RVO, obstruction of the retinal venous system by thrombus formation, external compression, or disease of the vein wall, is the second most common retinal vascular disease after diabetic retinopathy. 18 Retinal vein occlusion occurs with equal sex distribution, especially in middle-aged and older individuals with a history of systemic arterial hypertension, diabetes, or generalized atherosclerotic disease. Each of these risk factors is a complex trait, determined also by genetic profiles. 19 –21 The identification of risk factors would be an important step toward the prevention of this potentially disabling disorder. This study attempted to evaluate the potential role of APOE mutations in Tunisian patients with retinal venous obstruction.
Retinal vein occlusion is classified according to the location of the obstruction: CRVO when the obstruction is at the optic nerve head, BRVO when the obstruction involves a branch of the retinal vein, and hemi-RVO when one half of the retina supplied by the vein is affected. 13 Our group of patients with BRVO (n = 68, 77.27%) was greater than the group of patients with CRVO (n = 16, 18.18%). This distribution is not consistent with the results reported in France and highlights the prevalence of CRVO who were present with 73.37%. 22 Another study however showed that the incidence of CRVO is not high. 23 One study in Nepal showed CRVO accounted for 34% of cases and BRVO accounted for 66%. 24 A similar result was found in a Korean study that included data on 203 patients with CRVO (36.4%) and 354 patients with BRVO (63.6%). 25
The age for presentation in this study ranged from 11 years to 82 years. In the Ocular Vascular Unit at the University of Iowa, the age range of patients was between 14 and 92 years in a series of 1108 patients. 26 One significant finding showed that RVO occurred at a younger age in Asian and West Indian participants. 27 The Blue Mountains Eye Study found a prevalence of this pathology of 0.7% in individuals younger than 60 years, 1.2% in individuals aged 60 to 69 years, and 4.6% in individuals older than 80 years. 28
There are many systemic risk factors that predispose to RVO. The most consequential pathologies are those correlated with arterial thrombosis, such as hypertension and diabetes, which are known to affect vascular wall integrity. 29,30 In this study, 43.18% of the patients had hypertension or diabetes mellitus. Research indicates that arterial hypertension has been considered as a predominant risk factor for a majority of cases with RVO. 19 Another result indicated that diabetes mellitus is significantly more common in Asians than white Europeans and was identified in one-third of West Indian patients with RVO. 27 Similarly, a nationwide study of Koreans showed that the prevalence of diabetes and hypertension was generally high in both major subtypes of RVO (CRVO and BRVO). 25 The variability in prevalence rates may be due in part to racial/ethnic differences in study populations. This research indicates that smoking may have a protective effect (Table 1). However, the authors found that it was not a statistically significant risk factor in a study of methylenetetrahydrofolate reductase (MTHFR) polymorphisms and RVO in Tunisian. 31 In contrast, The Beaver Dam Eye Study found that the age-adjusted incidence of retinal branch vein occlusion was strongly associated with current smoking (OR = 4.43, 95% CI = 1.53-12.84) compared with nonsmokers. 32
The present study showed that the APOE polymorphism is a risk factor in patients with RVO. Apolipoprotein E is a member of the apolipoprotein gene family. 33 It is synthesized in the liver and extrahepatic tissues including the brain. It plays an important role in lipid metabolism by acting as a ligand for the low-density lipoprotein receptor to mediate the uptake of apoE-containing lipoproteins by cells and apoE-mediated lipoprotein assembly. 34 A different retinal vascular phenotype occurs in mice with a defect in the very-low-density lipoprotein receptor. In these mice, superficial and deeper retinal blood vessels form as expected, but then ectopic sproutings of small angiomatous growths invade the normally avascular outer retina. Very-low-density lipoprotein receptor interacts with reelin and apoE to guide neurons to their proper cortical laminae during brain development, and it is tempting to speculate that it has a corresponding guidance function for retinal vessels. 35,36
The distribution of APOE genotypes differs among populations. Eichner et al reviewed the frequencies of ∊2, ∊3, and ∊4 alleles in different populations and reported that these range from 0.02 to 0.13 for ∊2, 0.06 to 0.85 for ∊3, and 0.11 to 0.31 for ∊4. 33 Furthermore, wide-range population studies have indicated interethnic variations in APOE polymorphisms. At a global level, higher frequencies of the ∊2 allele were observed in Africa and Oceania (0.099 and 0.083 and 0.111 and 0.052, respectively). Similarly, ∊4 allele averages were higher in Oceania (0.221 and 0.149) and Africa (0.209 and 0.090), whereas Indian and Asian populations showed the highest frequencies of the ∊3 allele. 37
In our study, the 3 heterozygous genotypes (∊2/∊3, ∊2/∊4, and ∊3/∊4) and the homozygous genotype (∊3/∊3) for the APOE polymorphism were associated with increased RVO (P a = .048, P a < 10−3, P a = .042 and P a = .004, respectively). The alleles APO ∊3 and ∊4 were also shown to be associated with an elevated risk of RVO in this study (P < 10−3). To our knowledge, this is the first analysis of published studies that has investigated whether the APOE (∊2/∊3/∊4) polymorphism is associated with the risk of RVO.
In the Atherosclerosis Risk in Communities study, apolipoprotein ∊4 was weakly associated with retinopathy in persons without diabetes but not with other retinal microvascular abnormalities. 6 However, APOE polymorphisms were not predictive of retinal arteriolar or venular diameter. A similar lack of association between APOE polymorphisms and retinal microvascular abnormalities or retinal vessel diameters was also seen in the Cardiovascular Health Study. 38,39 Several clinical studies have also provided contradictory results for the association between age-related macular degeneration (AMD) and different APOE genotypes/alleles. The majority of studies demonstrated the important role of allele ∊4 in this pathology. 40 In a Netherlands study, a decreased risk of AMD was associated with the ∊4 allele. 41 This result was demonstrated in an Austrian and Brazilian study of several AMD associations, including APOE where the ∊4 allele conferred protection. 42,43 Recently, 3 studies assessed the association of the APOE ∊2/∊3/∊4 polymorphism with primary open-angle glaucoma (POAG). Wang et al found that the ∊4/∊4 genotype was a risk factor; however, 2 other studies, including a meta-analysis by Song et al, found that this polymorphism was not associated with POAG susceptibility. 44 –46
In addition, the APOE allele has been associated with various forms of cerebrovascular diseases. 47,48 In particular, several studies have investigated the APOE polymorphism with stroke in different populations. 49 –51 It is a candidate gene for the development of ischemic stroke through its effect on lipid metabolism. 52
However, it was recently suggested that multiple (cis-regulatory) SNPs in and around the APOE locus may contribute to disease susceptibility via alteration of gene expression. In addition to the SNPs used to define the epsilon alleles, 2 SNPs located in the promoter region of APOE have been described (rs449647 and rs405509). Evidence suggests that this region modulates transcriptional activity of APOE. 53
The APOE genetic variations can influence the mechanistic role of apoE in lipid transport and metabolism, resulting in altered circulatory lipoprotein concentrations. These alterations could be beneficial or detrimental in terms of the risk they pose to the development of vascular disease. 51
In conclusion, the current study suggests that the APOE ∊3 and ∊4 alleles were associated with a higher risk of RVO. Future studies in a larger sample will be required to clarify the association of APOE polymorphisms with RVO.
Footnotes
Acknowledgments
The authors would like to thank Dr Christian Winchell for his precious help in correcting this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
